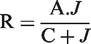Abstract
When conducting controlled laboratory studies with non-traditional laboratory animals it is important that methods for determining body weight stability are reliable. This helps ensure the health and welfare of animals when they are maintained during periods of free feeding or food restriction. This study compared different methods for determining body weight stability in six common brushtail possums (Trichosurus vulpecula) maintained on a free-feeding diet under laboratory conditions. A criterion of five consecutive weighings with less than ±2.5% change across days and no more than two consecutive days of weight loss or weight gain was judged to be the most suitable criteria for determining stability. It is important to study non-traditional animals, especially endangered or pest species, under controlled laboratory conditions and to have robust methods for establishing body weight stability.
One of the most important considerations when keeping laboratory animals is the control of physiological variables, such as body weight. 1 There is no agreed method, however, on how to control body weight in non-traditional laboratory animals that does not compromise health, welfare, 2 or the natural feeding behaviour of the animals. 3 No universally accepted standard exists on how to assess when body weights are stable during periods of free feeding or food restriction. This is particularly important for non-traditional laboratory animals with large individual differences, in contrast to those bred specifically for laboratory use. 4
In many experimental protocols restricting food intake or availability to some percentage of an animal’s free-feeding body weight and/or usual food consumption is used to establish a state of hunger to maintain motivation for responding during experiments.3,5–8 The free-feeding weight established prior to food restriction is typically the average of some number of days, 7 and ideally weights will have been judged as stable over that time. A variety of methods have been used to determine ‘stability’. The most basic method is a visual inspection of graphed weight data. 6 Statistical methods involving averaging across animals,7,9 and percentage change across weighings or specified periods of time for individual animals, 10 have been used.
The brushtail possum, Trichosurus vulpecula, is an arboreal omnivore from Australia which was introduced to New Zealand in the 1900s. 11 In New Zealand they consume native and exotic flora and fauna, predate and compete with native animals, and have a negative impact on agriculture.11,12 For these reasons, it is important to maintain captive populations of possums so that research can be undertaken to better understand this animal. There is no existing standard, however, for determining a stable or optimum body weight in the possum in captivity.
Possums are an example of a species where individuals fluctuate in body weight. Wild-caught possums in Australia range from 1350–3500 g for males and 1300–3150 g for females. 13 They are generally heavier in New Zealand (e.g. males 1400–3900 g, females 1400–3300 g).14–16 Measurements of possums in the wild are of limited use in estimating optimum body weight because their weight is influenced by health, location, food availability, climate and population density.17,18
Studies of newly captured possums have claimed that possums’ body weights took over 20 weeks to appear stable in shared enclosures, 19 and approximately six weeks when housed individually. 20 Neither study explained how body weight stability was determined except for visual inspection of graphed body weights.
In the current study we compared three different ways (visual inspection of graphs, a mathematical model and percentage change in body weight) of establishing the stability of free-feeding body weight in possums. The aim was to determine which method was most reliable and feasible for use in the laboratory. Six brushtail possums (two female and four male) aged between 2–6 years were wild-caught and had been housed in captivity for 3–8 years. Possums were kept in custom-built individual wire-netting cages with a nest box on top under a 12:12 h reversed dark/light cycle (School of Psychology, The University of Waikato, Hamilton, New Zealand). Cleaning and maintenance occurred during the light rotation. The possums had constant access to water. Possums received a minimum of 200 g of food (dock [Rumex obtusifolius], apple, and pellets [Camtech Manufacturing Ltd., Te Rapa, New Zealand]) at 10:30 h each day. At 08:00 h each morning, the leftover food was weighed. The amount of food offered was increased the next day if less than 50 g was left by the possums and decreased if more than 100 g was left. The experiment was conducted over 60 days.
To facilitate weighing, the possums were trained to move from their home cage into a metal box for a food reward. The box was weighed and the possums returned to their home cage. Possums were weighed every four days to minimize stress.
Visual inspection of body weight data revealed all six possums gained weight during free feeding with very short bouts of apparent ‘stability’ (Figure 1). A second method using visual inspection involved averaging body weights over three weighing days,
9
but again this did not show stability as weights continued to increase over time (Figure 1).
Body weight (every four days in grams) for the 60-day free-feeding regime for all possums. Open triangles are weights averaged over three weighing days. VAC = variance accounted for; SE = standard error.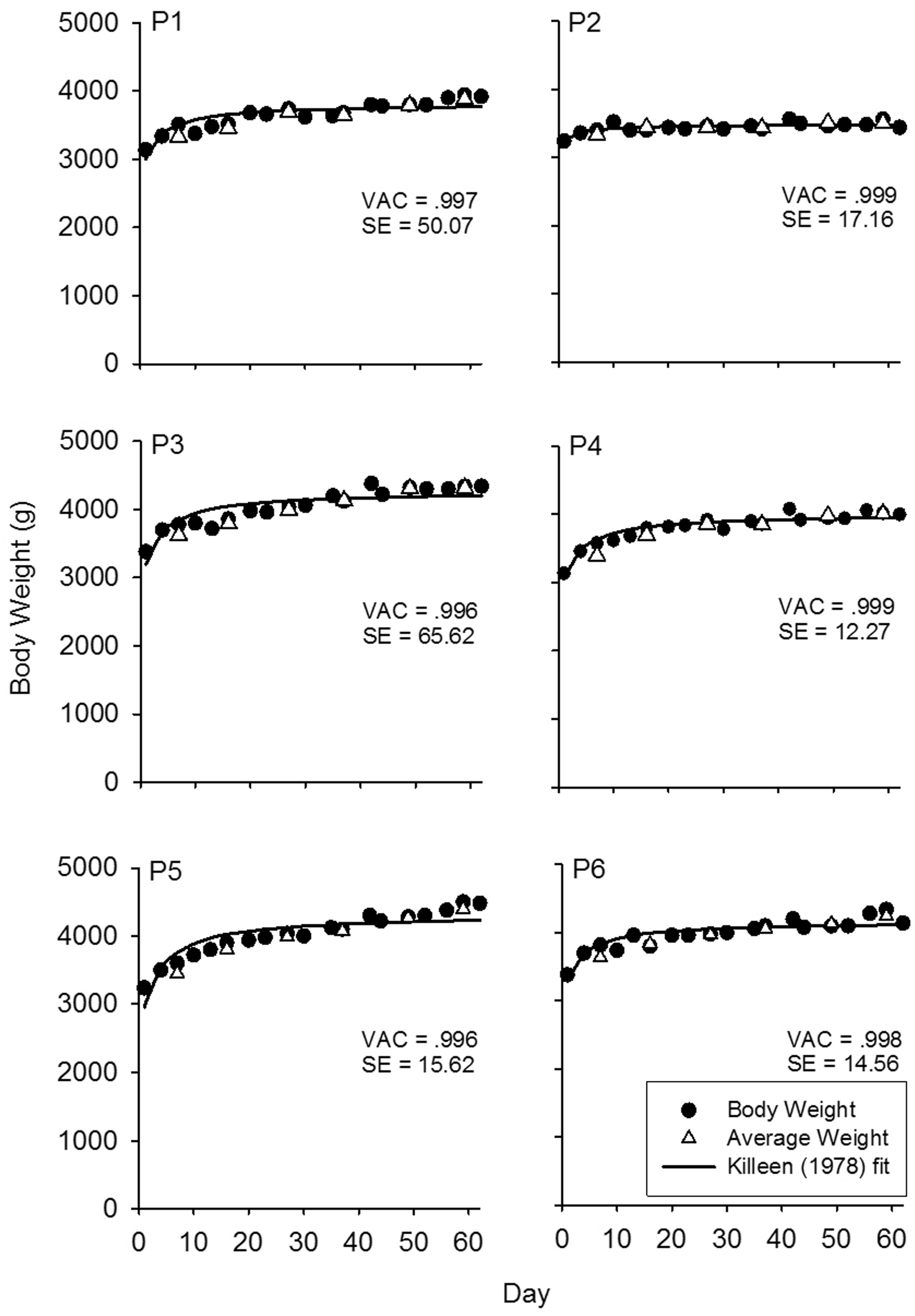
A mathematical model previously used to assess stability in response rate curves was applied to the body weight data in an attempt to identify the asymptote or point of stability.
21
The equation has two parameters (J = number of days and C = a time constant), R is the predicted body weight and dependent variable, A is the asymptote:
Non-linear least squares regression was used to determine the best fits of the equation to the data and are presented as the solid lines in Figure 1; they approximate the body weights well (the average variance accounted for was 99.8%). The model predicted that 99% of body weight stability will be reached after 12 days. Visual inspection of the data suggests, however, that there were still upward trends in weight after the asymptote was reached.
Others have used the percentage change in weight, showing no more than a 3% difference in individual body weights.
10
We applied a criterion to our data that body weight could not vary more than ±2.5% from the previous weight. Five percent total variability was chosen as it is a commonly used increment range in studies requiring animals to be food deprived. Inspection of the data indicated that it was necessary to add another component to this criterion to avoid increasing or decreasing trends in body weight (Figure 1). Thus, for stability to be established there had to be fewer than three consecutive increases or decreases in body weight identified by a visual inspection of Figure 2. The dark bands show periods of stability within ±2.5% of the previous weight and fewer than three consecutive weight gains or losses. Using this percentage change criteria, the body weights of P1, P2, and P6 were stable after 10–12 weighing sessions and P3 showed a longer period of stability in body weight by the end of the free-feeding period (60 days). P4 and P5 showed short periods of stability during the 60 days.
The percentage change between days of body weights of six possums. The shaded bands show periods of stability according to the percentage change criterion of ±2.5%.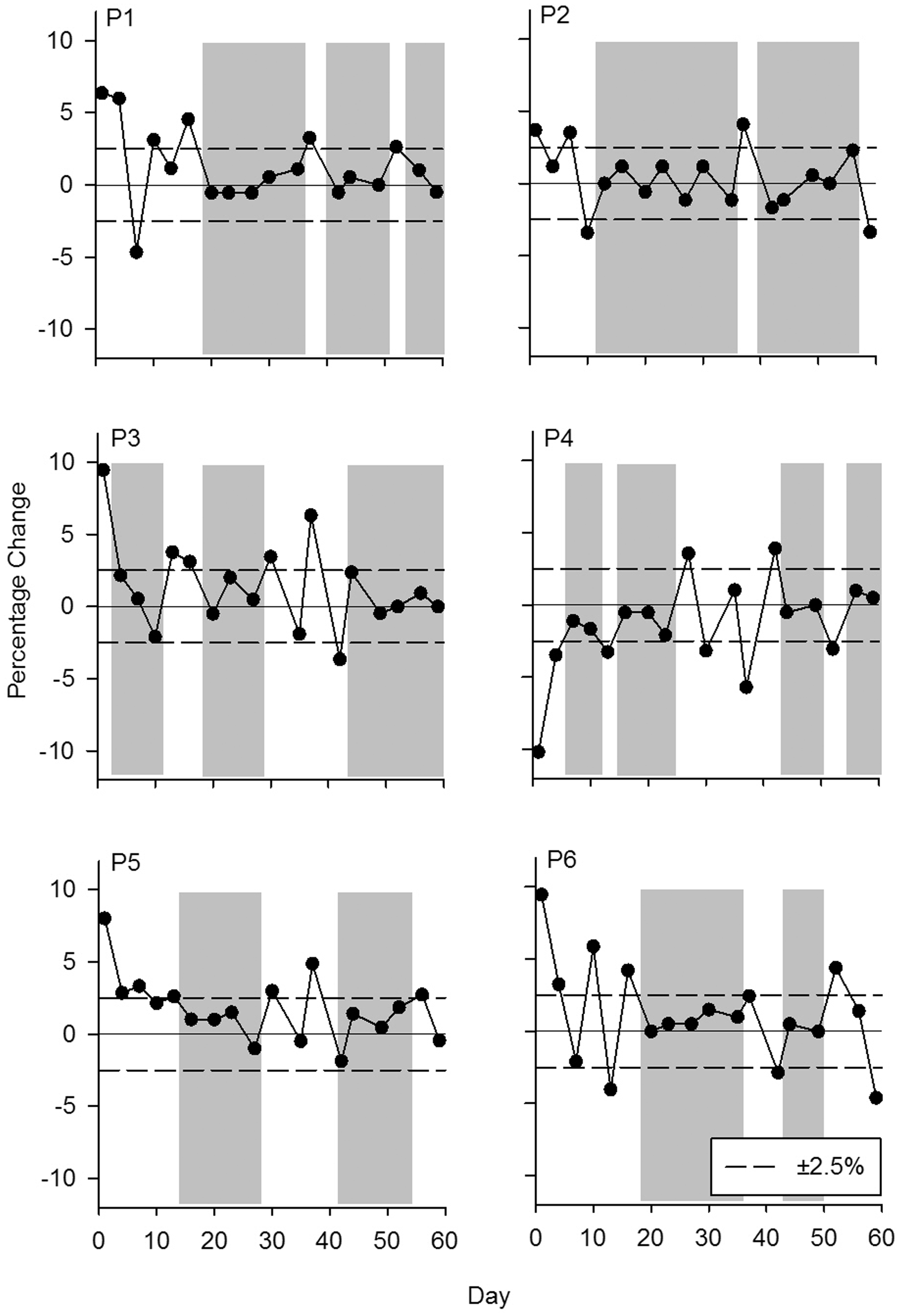
The body weights of four possums during the 60-day period showed periods of stability according to the mathematical and visual criteria; however, the criterion of ±2.5% may have been too conservative for two possums as there were fewer than five consecutive stable weighings within the free-feeding period. Another consideration for body weight to be considered stable is the minimum number of days that meet the criterion. Some possums showed longer periods of stability than others therefore a minimum period might be required to ensure there are no increasing or decreasing trends in body weight. If the tolerance was widened to ±3% then bouts of stability would have been longer for all animals. For healthy adult possums settled in a laboratory environment when provided ad libitum access to food it will require between 20 and 60 days to reach a stable weight as determined by both visual and percentage variation stability criteria.
When non-traditional animals are used in laboratory studies, weight ranges, food intake and body condition scores are considered when determining stability criteria.22,23 In addition, body weights of these animals should only be measured against the same individuals’ data rather than against a ‘standard’. Visual inspection of the data can reveal trends over long periods of time even when those increases are relatively small (e.g. see Figure 1), and consecutive gains and losses near zero percent indicate stable body weight. 10 Reliance cannot be placed solely on the percentage change in body weight to assess stability, however, as minimal percentage changes between weighing sessions may hide continually increasing body weights. The mathematical model underestimated the number of weighing sessions required to reach stability as determined by either visual inspection or percentage variation stability.
We recommend using more than one method to determine the stability of an animal’s body weight. A combination of visual inspection and percentage change analysis provided a practical and robust method for determining stable body weight which could be used in a wide range of non-traditional laboratory animals. Further research comparing and verifying these methods with traditional laboratory species, such as rats or mice could validate a general ‘standard practice’ for assessing stable body weights in captive and laboratory animals. It could also provide an opportunity for determining how best to establish body weight criteria across the range of species used in animal research.
Footnotes
Acknowledgements
This research was supported by a University of Waikato Doctoral Scholarship to the first author. The authors would like to thank Ian Cronin, Kathleen Doolan, Rachael Lockhart, Derek Riley, and Amanda Brown for their assistance in running the experiment. Ethics approval was obtained from the University of Waikato Animal Ethics Committee (Protocol 865).
Conflict of interest
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.