Abstract
It is still unclear whether a high-intensity interval exercise (HIIE) protocol is effective for promoting anti-inflammatory and anti-atherogenic responses. The objective of this study was to analyze the acute effects of HIIE on inflammatory and metabolic responses in obese and overweight girls and boys. Initially, the body composition of 19 sedentary volunteers was evaluated. The participants completed an interval experimental run on a treadmill (2:1 min at 95% maximum velocity, totaling ~3 km) and blood samples were collected at rest and immediately after the exercise protocol. Serum was analyzed for interleukins 6 and 10 (IL-6 and IL-10), tumor necrosis factor-α (TNF-α), cortisol, glucose, triacylglycerol (TG), non-esterified fatty acid (NEFA), plasminogen activator inhibitor-1 (PAI-1), and the lipoprotein profile. Under baseline conditions, the groups of boys and girls exhibited differences in the anthropometry measures for fat-free mass (p = 0.011), gynoid fat (p = 0.002), visceral fat (p = 0.004), and glucose (p = 0.031). After the exercise session, cholesterol, glucose, NEFA, PAI-1, IL-10, and TNF-α did not demonstrate effects for time, group, or interaction under the different conditions. Cortisol (F = 9.018; p = 0.008; partial η2 = 0.347), TG (F = 25.189; p < 0.0001; partial η2 = 0.597), and IL-6 levels (F = 6.543; p = 0.020; partial η2 = 0.278) showed an effect for time. In conclusion, our results demonstrated that HIIE promoted an immune-endocrine response, increasing cortisol and IL-6 levels immediately after the exercise protocol in overweight and obese girls.
Introduction
Excess fat accumulation in adipose tissue is directly related to the metabolic disruptions generated by obesity, as the adipose tissue depots are characterized as an endocrine organ due to their involvement in important immunometabolic processes, producing numerous cytokines with a pro-inflammatory character such as leptin, elevations in C-reactive protein (CRP), and plasminogen activator inhibitor 1 (PAI-1), and a reduction in adiponectin, which is an important anti-inflammatory adipokine. 1 This prolonged imbalance in pro-/anti-inflammatory adipokines results in a chronic systemic inflammatory state, associated with an increased incidence of metabolic syndrome in youth, leading to increases in cardiovascular risks. 2
High-intensity interval exercise (HIIE) is a time-efficient strategy to improve both aerobic and anaerobic fitness, as well as promoting adaptations that result in enhanced skeletal muscle fat oxidation capacity and improved glucose tolerance. 3 Adipokines, such as leptin and adiponectin, are biologically active proteins that are significantly correlated with the body mass index (BMI) 4 and associated with insulin resistance. HIIE was demonstrated to significantly decrease plasma leptin concentration and increase plasma adiponectin concentration. 5 Moreover, interleukin 6 (IL-6) often displays hormone-like characteristics that affect homeostatic processes. Therefore, part of the protective effect from exercise training is related to changes in immune function with production and release of inflammatory markers, such as IL-6, which often displays hormone-like characteristics that affect homeostatic processes. 6 In addition, IL-6 increases the production of anti-inflammatory cytokines such as interleukin-10 (IL-10) and interleukin 1 receptor antagonist (IL-1ra) and reduces tumor necrosis factor alpha (TNF-α), 7 acting as a protective factor against chronic low-grade inflammation, as well as in the treatment of obesity and its risk factors.
It is believed that the inflammatory and metabolic benefits of HIIE are directly associated with the stress caused by this exercise protocol. 8 Previously, HIIE has been shown to be perceived as being similar or less hard than the continuous exercise in obese individuals. 9 Moreover, HIIE is a time-efficient strategy to improve both aerobic and anaerobic fitness. 5 In view of this, this study aimed to analyze and compare the acute effects of HIIE on inflammatory and metabolic responses in overweight and obese girls and boys.
Materials and methods
Subjects
This research was approved by the Research Ethics Committee. Potential volunteers contacted the group staff and were invited to attend the Laboratory at the University, with their parents or legal guardians, for the initial screening. After explaining the study, signed consent was obtained from the parents or legal guardians.
The inclusion criteria utilized for the initial triage were as follows: (a) between 11 and 17 years, (b) obese or overweight according to age- and sex-specific BMI criteria, (c) the absence of any major diseases and clinical indications that prevent physical activity, and (d) not practicing systematic exercise.
Experimental design
In total, 24 adolescents attended the initial screening and agreed to participate. Of them, four adolescents did not achieve the HIIE protocol and one adolescent refused the blood collection after testing. Thus, the final analyses were performed with 19 obese and overweight adolescents of both sexes (12 boys and 7 girls).
The adolescents were instructed to follow some recommendations before performing any exercise protocols: not to perform physical exercise for 48 h prior to the test and not to undertake any kind of diet, only fasting 3 h before the commencement of exercise. The investigators were not blind to the study aims and were aware of all the objectives.
Body composition assessment
Body mass (BM) was measured to be 0.1 kg with an electronic scale (Filizzola PL 150; Filizzola® Ltd, Brazil). Body height and trunk length were measured with a fixed wall-mounted stadiometer (Sanny®, São Paulo, Brazil) with an accuracy of 0.1 cm and a maximum extension of 2.20 m. BMI criteria were determined as follows: body mass (kg)/height (m²). The body composition measurement was performed by dual-energy X-ray absorptiometry (DEXA; Lunar DPX-NT; General Electric Healthcare, Little Chalfont, Buckinghamshire, UK). The following variables were measured: fat-free mass (FFM), fat mass (FM), trunk fat mass (TFM), and percentage of fat mass (%FM).
Intra-abdominal adiposity tissue (IAAT) and subcutaneous adiposity tissue (SAT) were measured through an ultrasound examination of the upper abdomen. SAT was measured as the distance between the outer skin and abdominal rectus muscle, and IAAT was defined as the distance between the inner wall of the abdominal cavity and the anterior wall of the aorta. All adolescents complied with the recommendation to fast for at least 3 h prior to the evaluation. The examination was performed by a single qualified radiologist, using a TOSHIBA Eccocee scanner with a 3.7-MHz convex transducer (Tokyo, Japan).
Exercise protocol
During the first day, maximum stress tests on a treadmill (Inbrasport ATL 2000, Porto Alegre, Brazil) were performed to measure the peak speed of each adolescent. Before the test, the adolescents performed a 5-min warm-up running on a treadmill at a velocity of 5 km/h. The test started at a velocity of 3.0 km/h and an inclination of 1%. The velocity was increased by 0.5 km/h each minute. The peak speed was determined as the highest velocity reached in the test. When the participants were not able to conclude the 1-min stage, the speed was expressed according to the time of permanence in the final stage, determined as follows: Vmax = velocity of penultimate stage + ((time, in seconds, remained in the final stage multiplied by 1 km/h)/60 s).
Seventy-two hours after the maximum stress test, the participants underwent an HIIE session. All participants were evaluated in an ambient with controlled temperature and in the afternoon, in order to offer the same circadian conditions to all participants. Before the test, the adolescents performed a 5-min warm-up walking on a treadmill at a velocity of 5 km/h. The HIIE session consisted of 10 running sets of 2 min at 95% of peak speed plus a passive rest of 1 min between sets. The exercise protocol lasted for a total of 34 min and had a mean volume of ~3.3 km (exercise protocol plus warm-up). Heart rate was monitored throughout the exercise protocol using a heart rate monitor, and the ratings of perceived exertion (RPE) were logged on a Borg scale before and after the 10 sprints.
Blood samples and analyzes
Blood samples were collected at rest (baseline) and immediately after the exercise protocol (end). The blood samples (15 mL) were placed in two 5-mL vacutainer tubes (Becton Dickinson, BD, Juiz de Fora, MG, Brazil) containing ethylenediaminetetraacetic acid (EDTA) for plasma separation and one 5-mL dry vacutainer tube for serum separation. The tubes were centrifuged at 3000 r/min for 12 min at 4°C just after blood collection, and the plasma and serum samples stored at –20°C until analysis.
Total cholesterol, triacylglycerol (TG), and glucose were assessed using commercial kits (Labtest®, São Paulo, Brazil). Non-esterified fatty acid (NEFA) was assessed through the colorimetric method using a commercial kit (Wako Diagnostics, Suite Mountain View, CA, USA). Cytokines (IL6, IL-10, and TNF-α) were assessed using enzyme-linked immunosorbent assay (ELISA) commercial kits (Affymetrix/eBioscience, Ambriex S/A, São Paulo, Brazil). PAI-1 was measured using an assay kit from R&D Systems (R&D Systems, Abingdon, UK), and cortisol was assessed using a commercial kit (Alkor-Bio©, St. Petersburg, Russia). All kits were suitable for human specimens according to the manufacturer’s recommendations.
Statistical analysis
Data normality was verified using the Shapiro–Wilk test, and descriptive data are shown as mean and standard deviation. The two-way analysis of variance (ANOVA) with repeated measures was used to compare the differences in metabolic variables and inflammatory markers between groups (girls and boys) at baseline and post exercise. The sphericity was confirmed according to Mauchly’s W test and Greenhouse–Geisser correction. The statistical significance was set at 5% for all analyses, and the calculations were conducted using SPSS, version 17.0 (SPSS Inc., Chicago, IL, USA).
Results
Following the descriptive statistics analysis, when comparing the groups of boys and girls at baseline, it was possible to observe differences in the variables FFM (kg; boys: 45.22 ± 5.57, girls: 37.43 ± 5.94; p = 0.011), gynoid fat (%; boys: 50.04 ± 2.39, girls: 54.89 ± 3.58; p = 0.002), visceral fat (cm; boys: 3.85 ± 1.30, girls: 2.07 ± 0.61; p = 0.004), and glucose (boys: 107.33 ± 6.80, girls 98.09 ± 10.40; p = 0.031).
When comparing the behavior of Heart Rate (HR) and RPE during the session of HIIE, it was possible to observe effects of time (F = 72.146; p < 0.0001; partial η2 = 0.809) and group (F = 13.793; p = 0.002; partial η2 = 0.448) for HR, and only an effect of time (F = 19.166; p < 0.0001; partial η2 = 0 .530) for RPE.
For cholesterol, glucose, NEFA, PAI-1, IL-10, and TNF-α, there were no effects for time, group, or interaction, while for cortisol (F = 9.018; p = 0.008; partial η2 = 0.347), TG (F = 25.189; p < 0.0001; partial η2 = 0.597), and IL-6 (F = 6.543; p = 0.020; partial η2 = 0.278), there were effects for time (Table 1).
Metabolic and inflammatory responses at baseline and post-HIIE in obese and overweight adolescents.
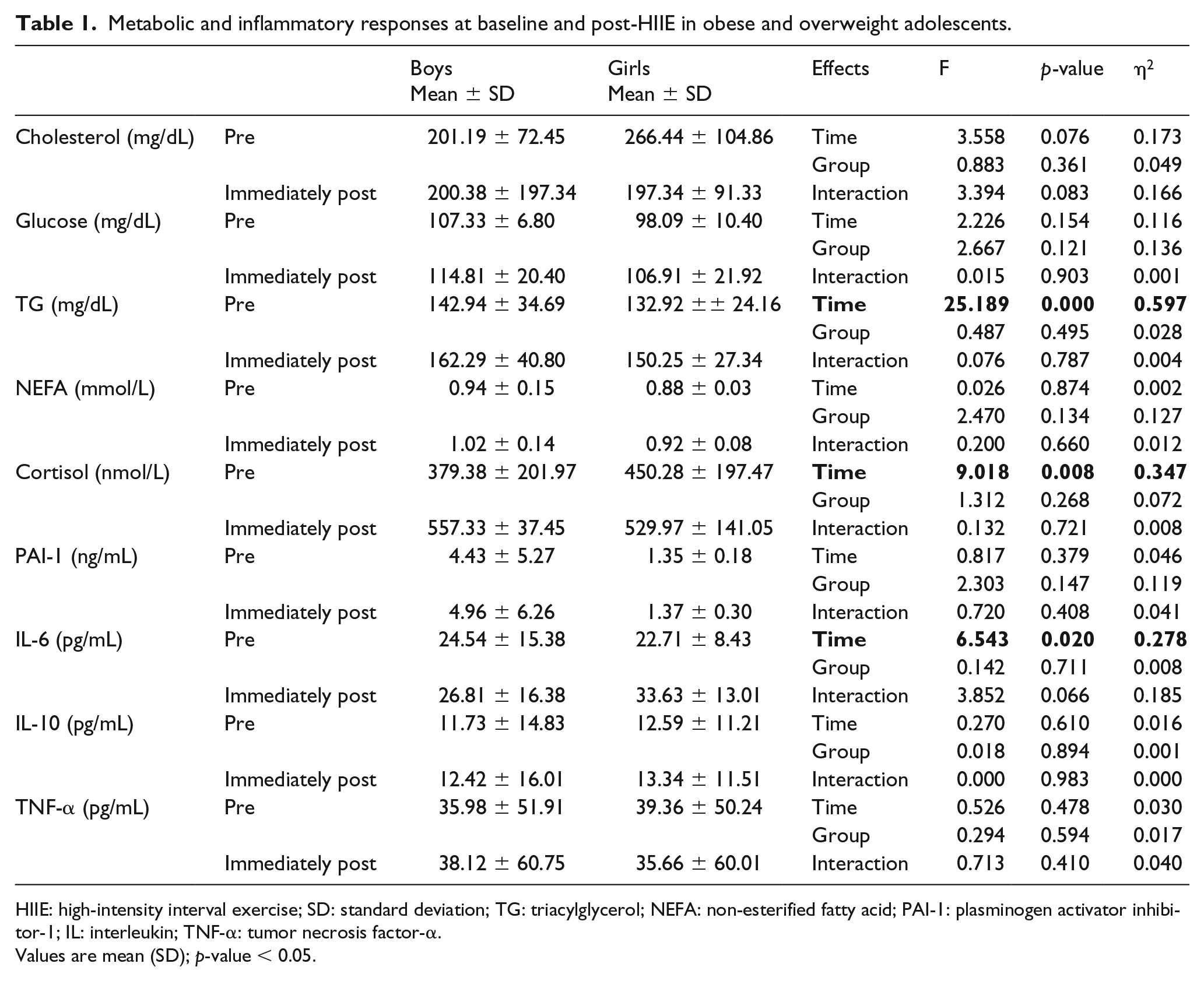
HIIE: high-intensity interval exercise; SD: standard deviation; TG: triacylglycerol; NEFA: non-esterified fatty acid; PAI-1: plasminogen activator inhibitor-1; IL: interleukin; TNF-α: tumor necrosis factor-α.
Values are mean (SD); p-value < 0.05.
When analyzing the effect of HIIE on IL-6 according to sex, a difference was found pre and immediately after for girls (p = 0.040), but not for boys (p = 0.615) (Figure 1).
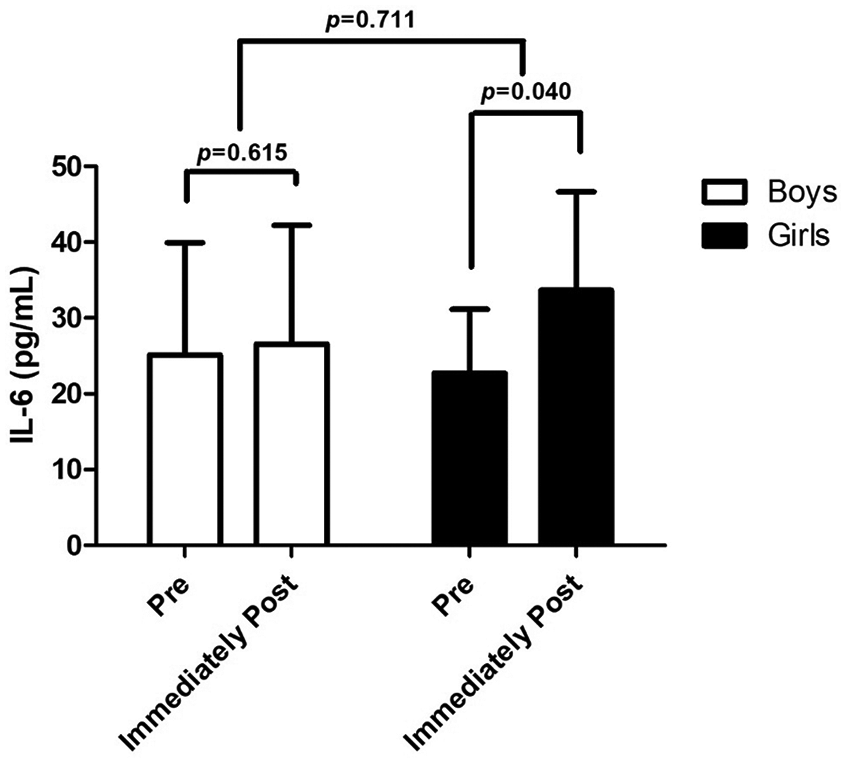
IL-6 pre- and immediately post-HIIE in obese and overweight boys and girls.
Discussion
In this study, an increase in cortisol and TG levels was observed after the HIIE protocol, associated with increased release of IL-6 in girls. Baseline data demonstrated that boys have higher values for visceral fat, FFM, and glucose levels under baseline conditions than girls.
Body fat distribution is an important metabolic and cardiovascular risk factor. It has been demonstrated that accumulated fat in subcutaneous and visceral deposits correlates with obesity-associated diseases and mortality more than peripheral deposits. 10 However, for girls, FM, gynoid fat, and cholesterol levels were higher when compared with boys. Taken together, our data suggest that our boy volunteers are more prone to development of a low-grade inflammatory condition.
HIIE promoted alterations in the metabolic profile of obese adolescents, observed by immediately increasing circulating TG levels in response to a single bout of HIIE and maintenance of glucose levels. These alterations may be related to mobilization via adipose tissue and reestablished fatty acid stores, principally in skeletal muscle. 11 These positive changes are crucial during childhood and may prevent obesity complications.
In addition, higher cortisol levels were observed after HIIE for both sexes as well as higher IL-6 levels only for girls. Thus, we suggest that the high rate of glycolysis during the HIIE protocol was effective for enhancing the production of IL-6 and cortisol, possibly favoring the metabolic process to maintain contractile activity; recent study supports this comment. 12 Further studies are needed to better understand the differences between boys and girls, principally regarding the IL-6 response.
Therefore, the limitation of this study was the absence of data collection in the recovery period to follow the kinetics of cytokine and hormone activity, which prevented the detection of a possible increase in IL-10.
In this study, we observed that higher cortisol and TG levels in the HIIE session, together with increased release of IL-6 (IL-6 only in girls), can exert a potential effect on the lipolysis process in obese adolescents of both sexes, providing a starting point for a discussion on understanding the mechanisms by which exercise influences metabolism and anti-inflammatory effects, leading to restoration of endocrine-metabolic functions.
Footnotes
Acknowledgements
The authors would like to thank FAPESP for their support (2013/25310-2). Paula Alves Monteiro was supported by PNPD (Programa Nacional de Pós Doutorado of Coordenação de Aperfeiçoamento de Pessoal de Nível Superior - Brazil (CAPES)).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: The author(s) received financial support for publication of this article from Sao Paulo Research ~ Foundation (FAPESP, Brazil), under grant n 15/26148-0.
