Abstract
Light is the principal synchronizing environmental factor for the biological clock. Light quantity (intensity), and light quality (type of light source) can have different effects. The aim of this study was to determine the effects of the type of light experienced from the time of birth on mouse growth, circadian behaviour and stress levels. We raised pigmented and albino mice under 24 h light–dark cycles of either fluorescent or white light-emitting diode (LED) light source during the suckling stage, and the animals were then exposed to various light environments after weaning and their growth rate, locomotor activity and plasma corticosterone concentration were measured. We found that the type of light the animals were exposed to did not affect the animals’ growth rates or stress levels. However, we observed significant effects on the expression of the locomotor activity rhythm under low contrast light–dark cycles in pigmented mice, and under constant light in both albino and pigmented mice. These results highlight the importance of environmental light quality (light source) on circadian behavioural rhythms, and the need for close monitoring of light environments in animal facilities.
Mammals have developed an autonomous endogenous clock in their brain to coordinate and anticipate their physiology and behaviour to the daily circadian changes in light and darkness in the environment, induced by the earth’s rotation. The principal circadian pacemaker is located in the brain suprachiasmatic nucleus (SCN). 1 Light is the most important synchronizing environmental factor for the circadian clock. Photic information is detected by the photoreceptors in the retina and is then transmitted, via the retinohypothalamic tract, to the SCN. The SCN will use this information to synchronize (entrain) the animal’s circadian rhythms to the environment. Thus light has the ability to influence physiology and behaviour in animals including humans.
In addition to the classical photoreceptor rods and cones, which collect and process light to generate an image of the world, there is a third type of photoreceptor, the intrinsically photosensitive retinal ganglion cells (ipRGCs), which are key to measuring environmental irradiance, and which then modulate physiological responses to light including photoentrainment. 2 Studies in transgenic rd/rd cl mice, whose retinas lack rods and cones, have shown that these animals are able to regulate their circadian rhythms with the same sensitivity as fully sighted animals, 3 and have thus demonstrated that ipRGCs play an important role in the regulation of the circadian system and photoentrainment. ipRGCs are particularly sensitive to blue light, as they contain the photopigment melanopsin, which has a peak sensitivity of almost 480 nm. 4 Therefore, light photoreception is not only sensitive to irradiance levels (melanopsin phototransduction is only engaged at moderate to high irradiance 5 ), but also has spectral sensitivity, defined by the spectral efficiency of the photopigments expressed within rods, cones and ipRGCs. 6 This means that environmental light with higher irradiance and shorter visible wavelength range will have a more pronounced effect on ipRGCs and consequently, on the circadian system. Therefore, light source will play an important role in experimental animals exposed to artificial lighting (e.g. fluorescent or light-emitting diode [LED]), which has a spectral distribution different to that of natural sunlight.
The circadian system of mammals is immature at birth and they finish their maturation postnatally. In the womb and at birth, the pup’s circadian rhythms are synchronized to the environment through various maternal signals. 7 In rats and mice, clock gene rhythms start to be affected by the photoperiod at postnatal day 10, coinciding with the time when most connections to and from the SCN, including the retinohypothalamic tract, have reached adult patterns with eyes open, and thus the significance of maternal influence starts decreasing in parallel with an increasing effect of external photic stimuli by around the second postnatal week. 8 Interestingly, there is a difference in the rate of development of the various photoreceptors: the development of ipRGCs in rodents starts from around the time of birth, long before rods and cones develop, 8 which suggests that a particular environmental light type which targets ipRGCs may exert more pronounced effects during development than a light type which targets rods and cones.
The aim of this study was to determine the effects of light type experienced from birth on circadian rhythm function. We used both albino and pigmented mice, as albino mice are more sensitive to environmental light damage9,10 and may thus have different sensitivities to light type compared with pigmented mice. Postnatal light environment can affect growth rates and stress levels in rodents,11,12 and exposure of adult mice to altered light–dark cycles or constant light increases corticosterone concentrations, and anxiogenic and depressive-like behaviours.13–16 Therefore, we also tested the effects of postnatal light types on growth rates, and plasma corticosterone concentrations in mice kept in different light types (fluorescent and LED) and under different light environments (light–dark cycles, constant dark and constant light).
Animals, materials and methods
Animals and experimental design
Four pregnant female mice (two C57BL/6 J and two CD1) raised in our colony were kept in a fluorescent light environment, and five additional pregnant female mice (three C57BL/JB and two CD1) in a LED light environment under 12:12 h light–dark cycles (LD, L: 28 µW/cm2, D: 0 µW/cm2) until the day of delivery (see details on light sources below). During the suckling stage – from the day of birth (postnatal day 0, P0) until weaning on P23 – pups were kept with their dams in their respective light environments.
On the day of weaning (P23), pups were moved to individual cages. A total of 28 female pups were used: six LED-raised C57BL/6 J mice, nine LED-raised CD1 mice, four fluorescent-raised C57BL/6 J mice and nine fluorescent-raised CD1 mice. Mice were exposed to four successive experimental stages (Figure 1): stage 1 (LDm, P23–P37, LD cycles of medium-light intensity [28 µW/cm2]), stage 2 (DD, P38–P53, constant darkness), stage 3 (LDl, P54–P92, LD cycles of low-light intensity [2.8 µW/cm2]), and stage 4 (LL, P93–P114, constant light of low-light intensity [2.8 µW/cm2]). During stages 1 to 4, all the mice were kept in the same light type (fluorescent or LED) that they had been raised in during the suckling stage.
Experimental design. The animals were raised under 24 h LD cycles of either fluorescent or white light-emitting diode (LED) light during the suckling stage. They were then exposed to various light environments from weaning, of the same light type than that experienced during the suckling stage. LD: 24 h light–dark cycle (12:12 h); DD: constant dark; LL: constant light; l: low-light intensity (2.8 µW/cm2); m: medium-light intensity (28 µW/cm2); P: postnatal day.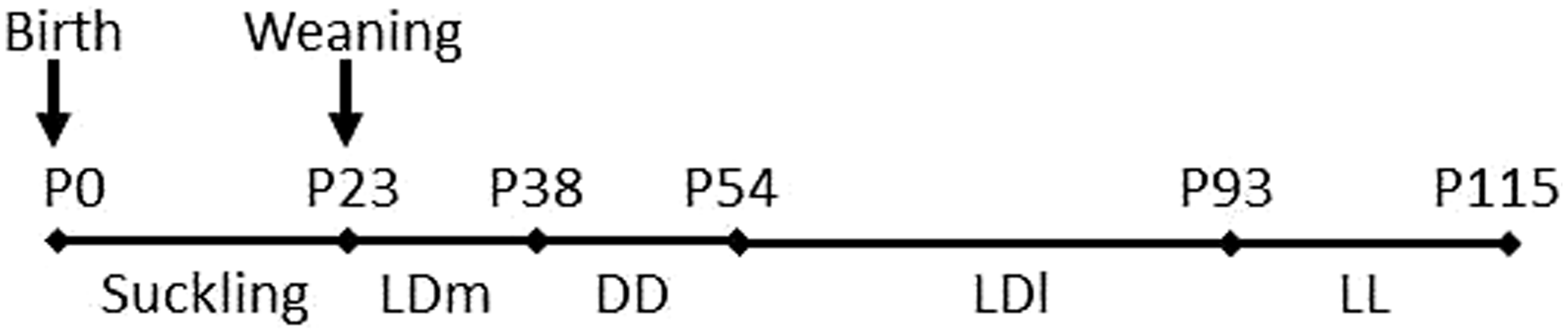
Locomotor activity was recorded continually throughout the experiment by means of infrared activity meters placed outside the animals’ cages. Data were stored in a computer in 15 min bins, and analysed thereafter. At the end of each experimental stage, a blood sample was taken from each mouse to analyse its corticosterone plasma concentration. Its body weight was measured once a week from weaning until the end of the experiment. Throughout the experiment, the mice were maintained under a controlled ambient temperature of 21 ± 2℃ and relative humidity of 60 ± 5%, with water and food (B&K Universal, Hull, UK) ad libitum. At the end of the experiment, the mice were culled by cervical dislocation. All experimental procedures were conducted in accordance with the United Kingdom Animals (Scientific Procedures) Act 1986.
Light sources
Throughout the experiment, the animals were kept in light-tight ventilated cabinets (Tecniplast, Buguggiate, Italy) with either a white fluorescent (FH14W/840 HE; Osram, Milan, Italy) or cold white LED (OSM1P/30; VLM, Hilden, Germany) light source. Although the light intensity changed during the experiment (see above), it was always the same in both the fluorescent and LED cabinets at each stage of the experiment.
Light spectral distribution in each cabinet was measured before the start of the experiment by means of a spectrometer (Ocean Optics, Dunedin, FL, USA) to confirm the differential spectral distribution between the fluorescent and LED light sources (Figure 2).
Spectral distribution of the fluorescent and light-emitting diode (LED) light sources used in this experiment.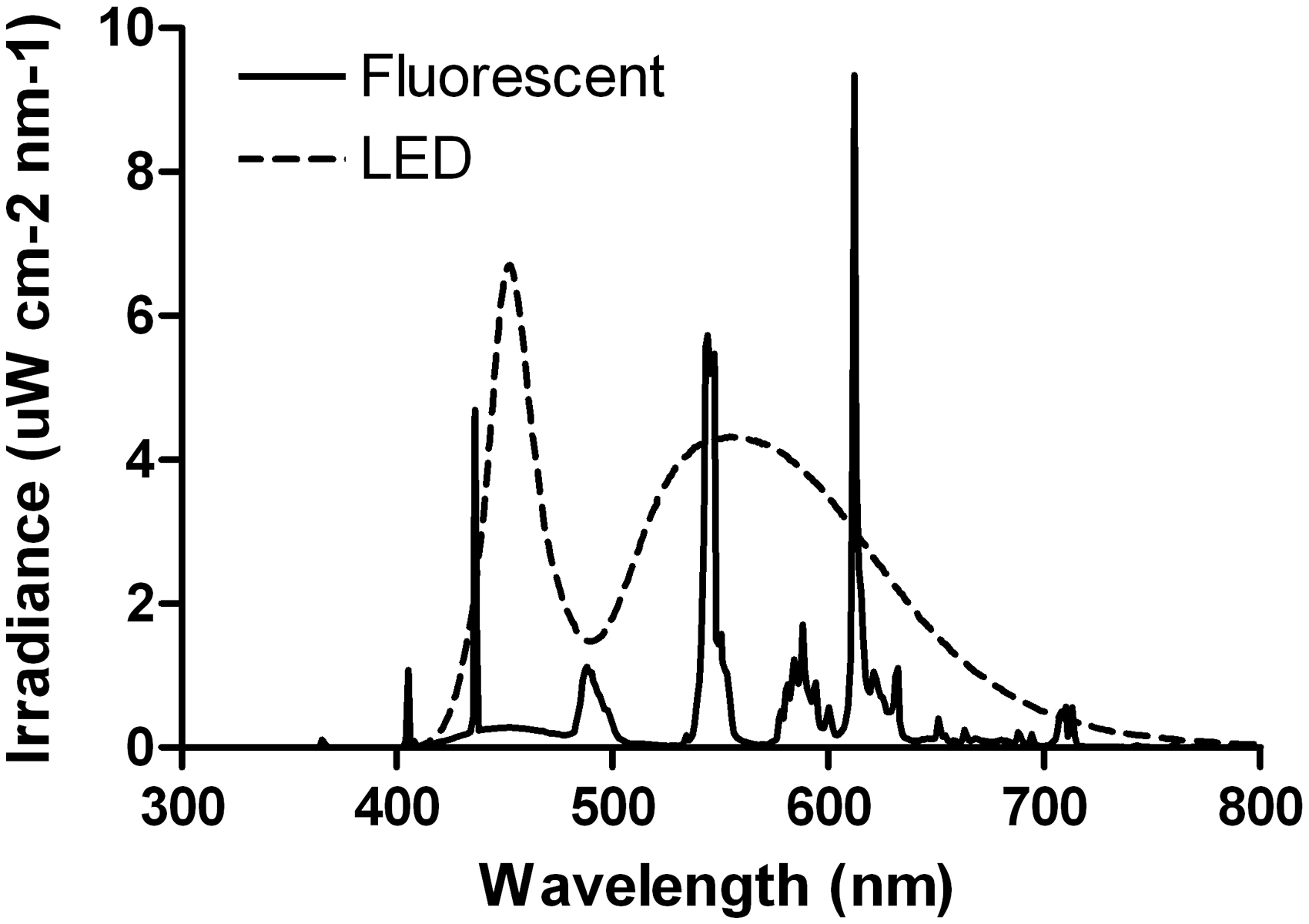
Behavioural analysis
The chronobiology software package El Temps (A Díez-Noguera, Universitat de Barcelona, Barcelona, Spain) was used to calculate several variables of the motor activity rhythm. The period of the motor activity rhythm was calculated using the X2-periodogram. The percentage of variance (PV) explained by the highest peak obtained in the periodogram was used as an indicator of the stability of the circadian rhythm of locomotor activity. We used the mean wave form of daily activity to study the characteristics of the alpha or active phase. Alpha was defined as the period of time during the daily cycle in which most of the activity levels were above the median. The duration of the alpha phase in relation to the duration of the daily cycle (%) was compared between groups. The activity levels during the active phase in relation to the total daily activity levels (%) were also calculated.
Corticosterone analysis
At the end of each experimental stage, a blood sample from the tail vein was taken from each mouse to analyse its plasma corticosterone concentration. To account for potential influences of the oestrus phase on corticosterone levels, blood sampling was performed at the trough of the corticosterone circadian rhythm (circadian time 2 [CT2] or zeitgeber time 2 [ZT2], which corresponds to 10 h before the onset of activity at CT/ZT12), as the plasma corticosterone concentration at this time point is similar throughout the various phases of the oestrous cycle in females, and similar to that in males.
17
Blood samples were mixed with 3 µL of 5 nM EDTA (Sigma-Aldrich, Gillingham, UK), spun at 2656
Data and statistical analysis
For all experimental stages, ‘light type’ refers to the type of light source (fluorescent versus LED) in which the mice were raised in during the suckling period. This light type corresponds with the same light type the animals were subsequently exposed to in the experimental stages LDm, LDl and LL.
SYSTAT version 10 (SPSS Inc, Chicago, IL, USA) was used to analyse all data. A general linear model was used to analyse locomotor activity rhythms and corticosterone concentrations. The independent variables were the light type during the suckling stage (fluorescent or LED) and the experimental stage (LDm, DD, LDl, LL); interactions between the independent variables were also included in the model. Two models were run, one for each mouse strain (CD1 and C57BL/6 J). An analysis of variance (ANOVA) of repeated measures was used to test the effect of postnatal light type on growth rate. When a statistically significant difference was encountered, a subsequent Bonferroni post hoc test was applied. Significance was set at P < 0.05 for all statistical tests.
Results
In this experiment, pups were either raised under fluorescent or LED light–dark environments throughout the suckling period, and then kept under various light environments of the same light type until the end of the experiment (see Figure 1 for experimental design). Figure 2 shows the spectral distribution of the fluorescent and LED light sources used.
Type of light effects on body weight
Body weight was examined weekly from weaning until the end of the experiment. We found that, both in C57BL/6 J and CD1mice, the types of light the animals were kept in had no effect on their body weights or growth rates (Figure 3).
Growth rates. Body weight increase throughout the experiment of C57BL/6 J (a) and CD1 (b) mice kept in either fluorescent or white light-emitting diode (LED) light from birth.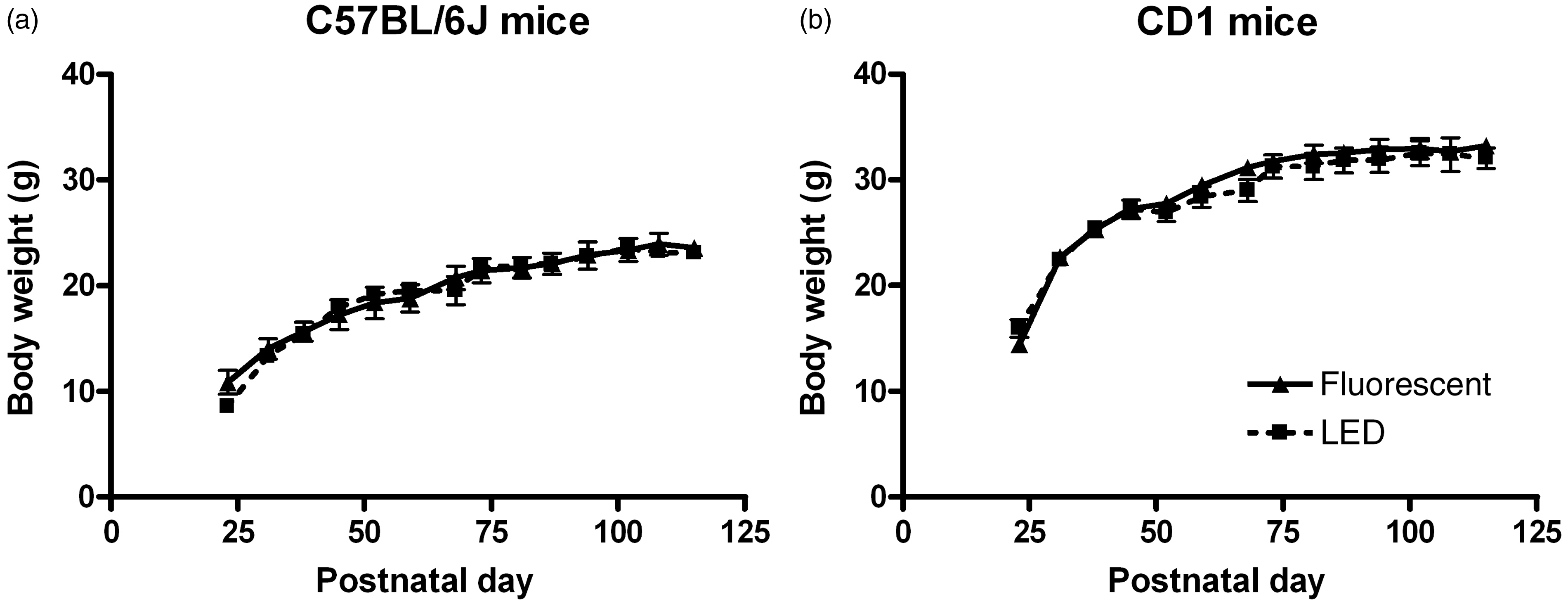
Type of light effects on behaviour
Locomotor activity was recorded constantly throughout the experiment (Figure 4). Four key aspects of the circadian rhythm of locomotor activity were examined: period, strength (as indicated by the PV of the highest peak in the periodogram), and duration and amount of locomotor activity performed during the active (alpha) phase of the rhythm.
Locomotor activity rhythm. Representative actograms of C57BL/6 J (a, c) and CD1 (b, d) mice kept in either fluorescent (a, b) or white light-emitting diode (LED) (c, d) light from birth. The actograms show the animals’ locomotor activity rhythm throughout the various experimental stages. LD: 24 h light–dark cycle (12:12 h); DD: constant dark; LL: constant light; l: low-light intensity (2.8 µW/cm2); m: medium-light intensity (28 µW/cm2).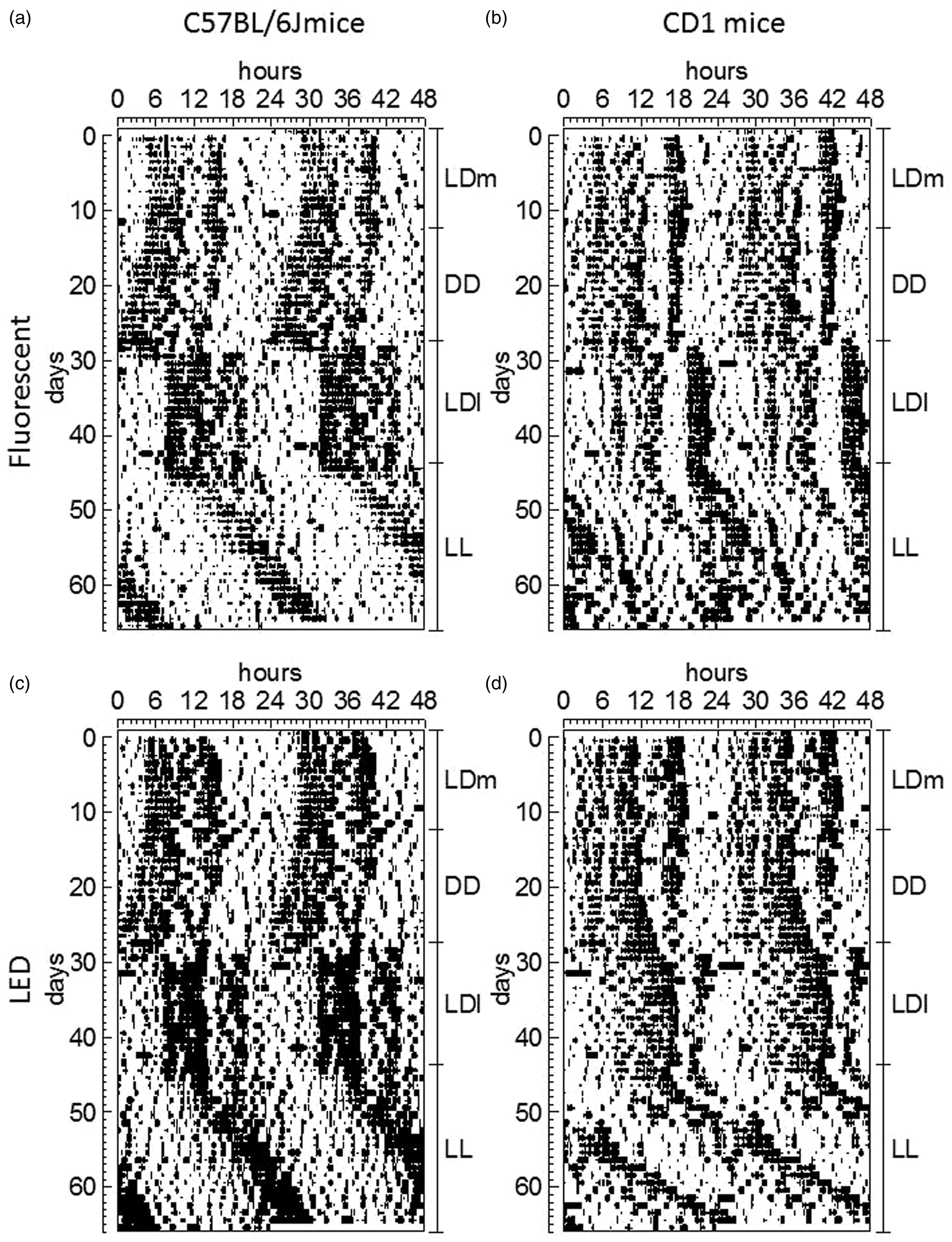
In C57BL/6 J mice, the experimental stage had a statistically significant effect on the period (P < 0.001, Figure 5a) and PV (P < 0.001, Figure 5c) of the locomotor activity rhythm, and also on activity duration (P = 0.010, Figure 5e), but not on activity levels (Figure 5g). We found by post hoc analysis that the period in the LL stage was longer than all the other stages (P < 0.001), and that the period in the DD stage was shorter than all the other stages (P < 0.001). The strength of the rhythm or PV was higher in the LDl stage compared with all the other stages (P < 0.01 versus DD, LL; P < 0.05 versus LDm). The duration of the active phase was significantly shortened in the LL stage compared with the LDl and DD stages (P < 0.05). Regarding the effects of type of light, we found a significant interaction between the experimental stage and the light type, with mice kept in LED light showing longer periods than mice kept in fluorescent light, but only in the LL stage (P < 0.001, Figure 5a). We also found that, in the LDl stage, mice kept under LED light had stronger rhythms (higher PV values) than those kept under fluorescent light (P < 0.05, Figure 5c).
Locomotor activity rhythm analysis. Main circadian rhythm characteristics in each experimental stage in C57BL/6 J and CD1 mice raised under a fluorescent or light-emitting diode (LED) light during the suckling stage. (a, b) Period. (c, d) Percentage of variance (PV). (e, f) Duration of daily active phase. (g, h) Activity levels during the active phase. Data presented as mean ± standard error (n = 4–10). #P < 0.05, ##P < 0.01, ###P < 0.001 Fluorescent versus LED; *P < 0.05, **P < 0.01, ***P < 0.001. LD: 24 h light–dark cycle (12:12 h); DD: constant dark; LL: constant light; l: low-light intensity (2.8 µW/cm2); m: medium-light intensity (28 µW/cm2).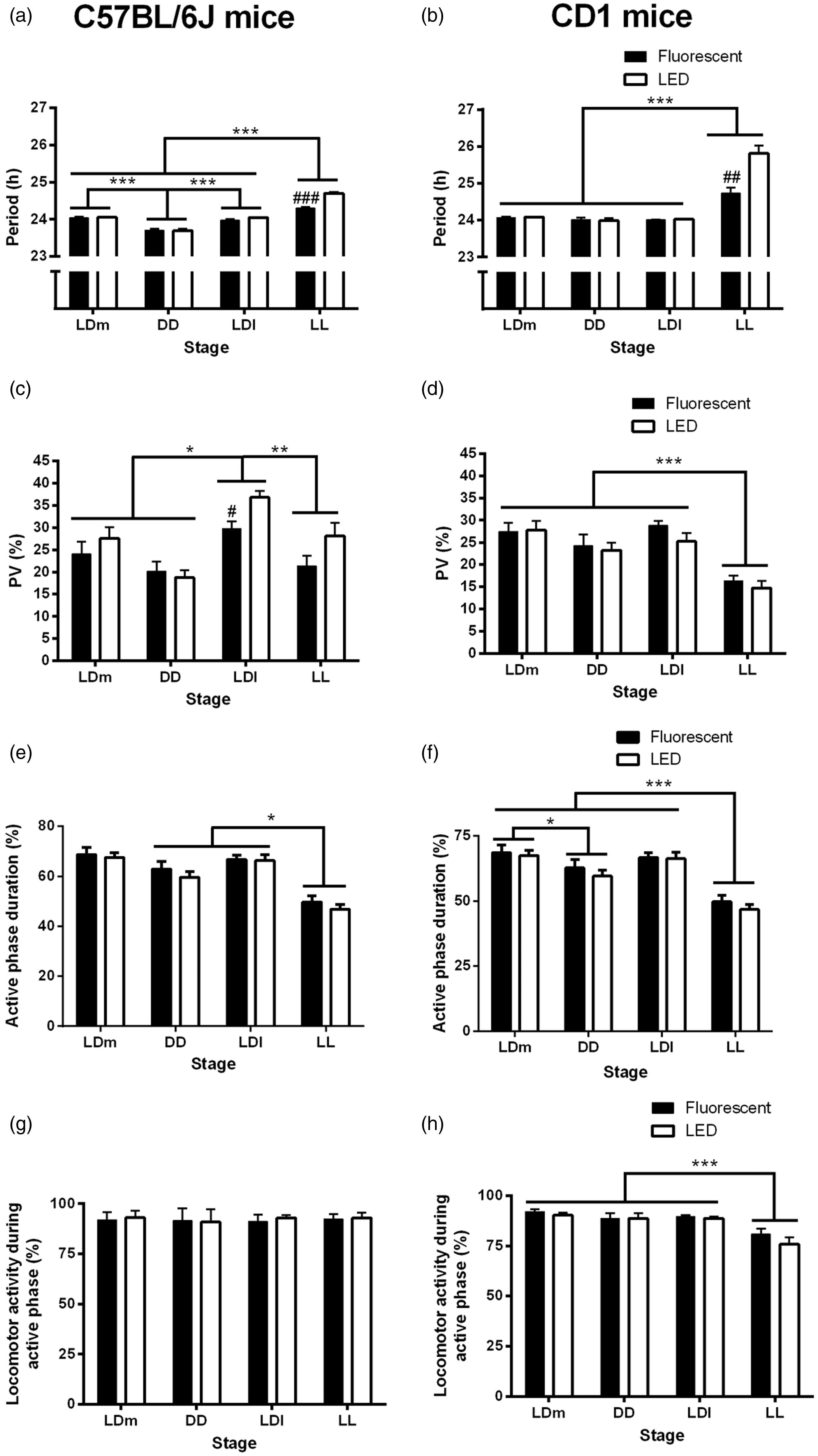
In CD1 mice, we found a significant effect of the experimental stage on period (P < 0.001, Figure 5b). Post hoc analysis showed that the free-running period in the LL stage was significantly longer than that of all the other stages (P < 0.001). PV also depended on the experimental stage (P < 0.001, Figure 5d). Specifically, we found that PV was lower in the LL stage compared with all the other stages (P < 0.001). The duration of the active phase was also shorter in the LL stage than in all the other experimental stages (P < 0.001, Figure 5f), and also shorter in the DD stage compared with the LDm stage (P < 0.05). The amount of activity performed during the alpha phase also depended on the experimental stage (P < 0.001, Figure 5h), with lower activity levels in the LL stage compared with all the other stages (P < 0.001). In addition, we found that the type of light the animals were kept in had statistically significant effects on the period of the locomotor activity rhythm in the LL stage, so that the free-running period was longer in the mice kept in LED light than in those kept in fluorescent light (P < 0.01, Figure 5b).
Type of light effects on stress levels
We determined stress levels by examining corticosterone plasma concentrations at the end of each experimental stage. In the C57BL/6 J mice, we found that corticosterone plasma concentration was independent of the experimental stage and type of light experienced (Figure 6a). In the CD1 mice, the experimental stage had a statistically significant effect on the plasma corticosterone concentration (P < 0.01, Figure 6b), however significance was lost when running the post hoc test, which suggests that the difference between stages is weak. The type of light the mice were kept in had no significant effect on plasma corticosterone concentration (Figure 6b).
Plasma corticosterone concentrations. Plasma corticosterone concentration in each experimental stage, in (a) C57BL/6 J mice and (b) CD1 mice kept under a fluorescent or light-emitting diode (LED) light from birth. Data presented as mean ± standard error (n = 4–10). LD: 24 h light–dark cycle (12:12 h); DD: constant dark; LL: constant light; l: low-light intensity (2.8 µW/cm2); m: medium-light intensity (28 µW/cm2).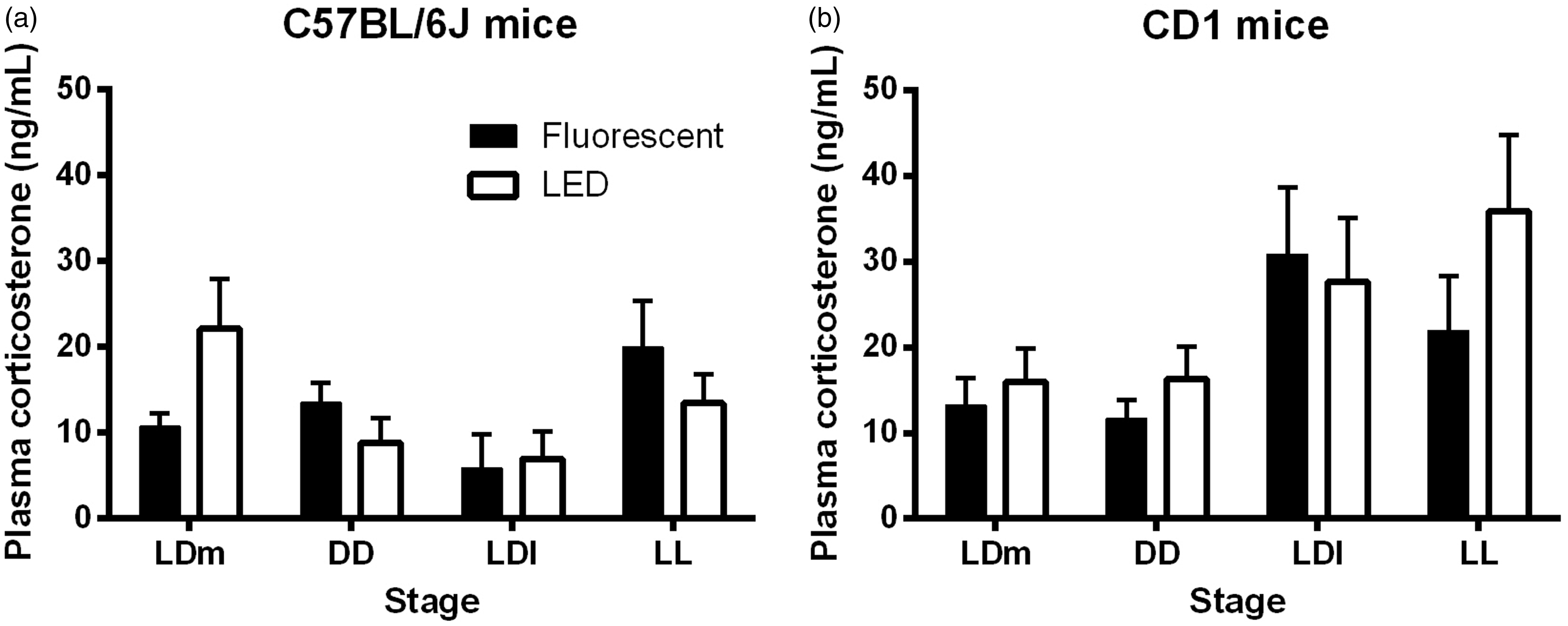
Discussion
We investigated the effect of light type experienced from birth on circadian behaviour, growth rates and stress levels in albino and pigmented mice. We found no effects of postnatal light type on growth rates or stress levels under different light environments; however we found significant effects on circadian behaviour in both CD1 and C57BL/6 J mice.
We found no effects of light type experienced during postnatal development on body weight after weaning. This is in contrast to Borniger et al., 11 who found that female mice raised under LD cycles with dim light at night had significantly lower body mass than control mice exposed to LD cycles with darkness at night after weaning (postnatal weeks 5 and 7), but no differences were observed later on. The main difference between the two experiments is that while our animals were exposed to LD cycles with light during the day and darkness during the night, the mice in the study by Borniger et al. were not. These results suggest that continuous illumination during postnatal development may slow down the initial growth rate in newborn pups, but alternating LD cycles of either fluorescent or LED light and darkness do not.
Studies raising rodent litters under extreme light conditions during the postnatal period (i.e. LL, DD) have shown that postnatal light environments (and not maternal circadian rhythms) have a long-term effect on the future expression of the pup’s circadian rhythm. 18 Here, all the litters were exposed to normal LD cycles with medium-light intensity during pregnancy and the suckling stage, and thus they were all synchronized to a 24 h rhythm, suggesting that no major differences in maternal circadian rhythms occurred between the groups.
As expected, 19 exposure of CD1 mice to LL significantly lengthened their free-running period (tau) and weakened their circadian rhythms of locomotor activity. We also found LL induced negative masking in albino mice, 20 as indicated by the significant reduction in activity duration and activity levels in the LL stage compared with the other experimental stages. Interestingly, we found that the type of light experienced during the suckling stage particularly targeted the locomotor activity rhythm in LL. Indeed, in the LL stage, the tau was longer in LED-raised compared with fluorescent-raised mice. According to Aschoff’s rule, tau in LL lengthens with increased light intensity, 21 and since all the animals were exposed to the same light intensity, the results suggest that CD1 LED-raised mice perceived a higher light intensity than CD1 fluorescent-raised mice. This could be explained by the fact that white LED light contains more blue light than fluorescent light (Figure 2), and since ipRGCs sensitivity peaks around 480 nm (blue light 4 ), the signal received by ipRGCs and transmitted to the SCN may be stronger under LED light compared with fluorescent light.
In C57BL/6 J mice, we also found a tau lengthening when the animals were exposed to LL. Similarly to CD1 mice, the tau of the locomotor activity rhythm of the mice exposed to LED light in the LL stage was longer than that of the mice kept in fluorescent light. In addition, we found that the LED-raised mice showed stronger rhythms than the fluorescent-raised mice in the LDl stage. These results suggest: (a) mice kept in LED light may perceive higher light intensities, and (b) a better ability of pigmented LED-raised mice to entrain to low contrast LD cycles. Again, these results could be due to the fact that white LED light has a higher proportion of blue light in its composition than fluorescent light (Figure 2), thus giving a stronger signal initially to the ipRGCs, and ultimately the SCN.
The tau length of an animal exposed to constant darkness is species-specific, 22 and thus it is not surprising that the period in DD differs between C57BL/6 J and CD1 mice. Inter-strain differences in the tau in DD have been associated with differences in responses to environmental light, 23 which could explain the slight differences found in the expression of the circadian rhythm of locomotor activity in LL and LDl between C57BL/6 J and CD1 mice. Nevertheless, no effects were found in either mouse strain resulting from the light type the animal was exposed to from birth on the tau in DD, which suggests that light type does not affect the intrinsic functioning of the circadian pacemaker.
Changes in the amount of light a rodent is exposed to during postnatal development (the photoperiod) have been shown to have long-term effects on plasma corticosterone concentrations and anxiety-like behaviour later in life.11,12 In this study we examined the effects of light type on stress levels by determining plasma corticosterone concentrations at the end of each experimental stage. The entire procedure of blood collection, from opening the cabinet housing the mouse cages to putting the blood on ice took 3–4 min, thereby avoiding stress-induced increases in corticosterone.24,25 This is demonstrated by the finding that corticosterone measurements at ZT/CT2 were between 10–40 ng/mL in this study, which is in line with the published literature, and significantly lower than a stress-induced plasma corticosterone concentration (>1000 ng/mL).25,26 We found no significant changes in overall corticosterone plasma concentrations due to light type in both C57BL/6 J and CD1 mice. In addition, we found no significant differences in plasma corticosterone concentrations due to the experimental stage. This is interesting, as constant light exposure has been shown to increase overall plasma corticosterone concentrations. 13 However this study was performed in rats. In mice, six weeks of LL exposure has been shown to actually decrease plasma corticosterone concentrations, 15 possibly due to the down-regulation of the hypothalamic–pituitary–adrenal axis (HPA). 27 Thus it is possible that two weeks of LL exposure in our study was not a long enough period of time to induce depression of the HPA axis. Taken together, these results suggest no major changes in the stress responses due to various light environments and light sources in both mouse strains in our experimental conditions.
Due to lack of pigment, albino retinas are more sensitive than pigmented retinas to damage by environmental light received either during postnatal development 28 or in adulthood.9,10 We thus hypothesized that albino CD1 mice would be more sensitive than C57BL/6 J mice to the effects of light type in this experiment. The finding that the growth rates and stress levels in both mouse strains were unaffected by light type or experimental stage, and that the free-running period of the locomotor activity rhythm in the LL stage was longer in the LED-kept mice of both strains, suggest similar effects of light type on albino and pigmented mice. However, the finding that pigmented mice kept under LED light have stronger rhythms than those kept in fluorescent light in a low-contrast light–dark cycle (LDl stage), suggests that pigmented mouse retinas may in fact be more sensitive to spectral changes in light input.
Both the amount of light and the timing of the light exposure received during the suckling period have been previously shown to have long-term consequences on the expression of circadian behavioural rhythms in rodents.28–30 In this study we have found that circadian behavioural rhythms of both albino and pigmented mice are affected by the light type they are exposed to from birth. The circadian system appears to be particularly sensitive to light type under low contrast LD cycles and under constant light. Additional experiments are required to further elucidate the role of type of light on the programming of the developing circadian system. Nevertheless, we did not find that the type of light had any significant effects on growth rates or stress levels in the LD and DD stages, or in response to LL exposure. These results highlight the importance of environmental light quality and quantity on circadian behavioural rhythms, and the need for close monitoring of light environments in animal facilities.
Footnotes
Conflict of interest
None declared.
Funding
This study was funded by Tecniplast SpA (Buguggiate, VA, Italy).
