Abstract
Chemotherapy-induced neuropathic pain is one of the major problems for cancer patients. Although paclitaxel and cisplatin are widely used in women, most laboratory studies of chemotherapy-induced neuropathic pain have been conducted on male animals. The current study examined the gender differences in chemotherapy-induced neuropathic pain in mice. Neuropathic pain was induced by intraperitoneal injection of paclitaxel (2 mg/kg) for five consecutive days and cisplatin (1 mg/kg) for seven consecutive days. Cold allodynia was evaluated by measuring the paw withdrawal frequency and duration of paw licking in mice; however, mechanical allodynia was assessed by von Frey filaments. Neuropathic pain began to manifest after a few days (P < 0.001). Cold allodynia was more robust in female mice (P < 0.001) treated with paclitaxel, while no differences were observed between the two genders in the manifestation of paclitaxel-induced mechanical allodynia. Interestingly, no gender differences were observed in cisplatin-induced cold and mechanical allodynia tests. In conclusion, gender differences play a major role in neuropathic pain induced by paclitaxel. The differences between male and female animals should be considered in future studies and the findings should be generalized to humans with caution.
It is believed that gender differences exist in the pathology of chronic and neuropathic pain. 1 Various clinical studies have proposed that women are more sensitive to pain than men.2,3 Furthermore, animal studies have demonstrated that female rodents show a lower threshold for pain in different models; however, the underlying mechanisms are not well understood. 4 Chemotherapy-induced neuropathic pain is one of the major side-effects in cancer treatment using taxane and platinum complexes. 5 The pain is characterized by numbness, tingling, burning sensation and cold allodynia. 6 Neuropathic pain may impair the patient’s quality of life and compromise a patient’s ability to tolerate chemotherapy. 7 The risk of neuropathy is proportional to the dose and duration of administration of chemotherapeutic agents.8,9 There is no established approach to the prevention and treatment of chemotherapy-induced nerve damage. 10
Paclitaxel and cisplatin cause significant neuropathic pain. These are major drugs for the treatment of breast and ovarian cancers and are widely used infemale patients. 8 Different animal models are available to investigate the effectiveness of these treatments.11,12 Rats and mice are frequently used to evaluate mechanical and cold allodynia tests. 13 These tests are predominantly performed on male animals. Sometimes researchers ignore the role of gender in experiments whereas others believe that the female hormonal cycle can cause problematic variations in their findings. 14
However, the relation between levels of chemotherapy-induced neuropathic pain and gender differences has not yet been studied in mice. In this study, we investigate the role of gender in paclitaxel- and cisplatin-induced neuropathic pain.
Materials and methods
Animals
NMRI mice (Pasteur institute, Tehran, Iran) weighing 20–30 g were used. The animals were housed under a 12 h light/dark cycle with free access to food and water, and were assigned to different treatment groups (n = 6–8 in each group). All experiments were conducted in accordance with the guidelines for the care of laboratory animals of the Ethics Committee of Isfahan University of Medical Sciences.
Drugs and drug administration
Cisplatin and paclitaxel were purchased from Sigma–Aldrich Inc (St Louis, MO, USA). Cisplatin was freshly dissolved in sterile saline, and paclitaxel was dissolved in ethanol–cremophor–saline (5:5:90, v/v/v). Male and female mice received paclitaxel (2 mg/kg) intraperitoneally for five consecutive days with a cumulative dose of 10 mg/kg, or vehicle. 15 Cisplatin (1 mg/kg) was intraperitoneally administered to male and female mice for seven consecutive days. A control group received saline. 16
Behavioral assay
The mice were placed on top of an aluminum mesh table and allowed to adjust to the situation for approximately 15 min. Briefly, for the assessment of cold allodynia, acetone was applied three times via a needle and syringe to the plantar surface of each hind paw with 30 s intervals. 17 The time spent licking the paw was recorded with a stopwatch. The frequency of licking was calculated and expressed as a percentage using the following formula (number of trial attending the hind paw/total number of trial)×100. For assessment of mechanical allodynia, von Frey filaments (Stoelting, Wood Dale, IL, USA) ranging from 0.16 to 6 g were used as previously described. 18 Bending force was applied to the plantar skin of the right hind paw, and each application was held for 6 s, using the up–down method to determine threshold sensitivity.
Data analysis
Data are presented as mean ± standard deviation, and were compared by one-way analysis of variance (ANOVA) followed by Fisher LSD post hoc test for multiple comparisons. Time-course analysis of behavioral data was compared by repeated measures ANOVA for each experimental group.
Results
Effects of paclitaxel on cold allodynia
Paw withdrawal frequency and duration of paw licking increased in both genders after treatment with paclitaxel (2 mg/kg, for 5 consecutive days). The control group did not show any changes in nociceptive response. Also paw withdrawal frequency and duration of paw licking were more robust in female mice compared with male mice or the control group (P < 0.001). Interestingly, pain was manifested sooner in the female mice than in the male mice (Figures 1a and 1b).
(a) Time course of paw withdrawal frequency after paclitaxel treatment. Paclitaxel (2 mg/kg) was injected for five consecutive days, and the control group received vehicle. A significant gender difference was observed for paclitaxel-induced cold allodynia (**P < 0.01, ***P < 0.001 when compared with the control group, and ##P < 0.01, ###P < 0.001 when compared with the male group). (b) Duration of paw licking after paclitaxel treatment. Paclitaxel (2 mg/kg) was injected for five consecutive days, and the control group received vehicle. A significant gender difference was observed for paclitaxel-induced cold allodynia (**P < 0.01, ***P < 0.001 when compared with the control group, and ##P < 0.01, ###P < 0.001 when compared with the male group. *P < 0.05, **P < 0.01, ***P < 0.001 when compared with the vehicle group, and ##P < 0.01, ###P < 0.001 when compared with the male group).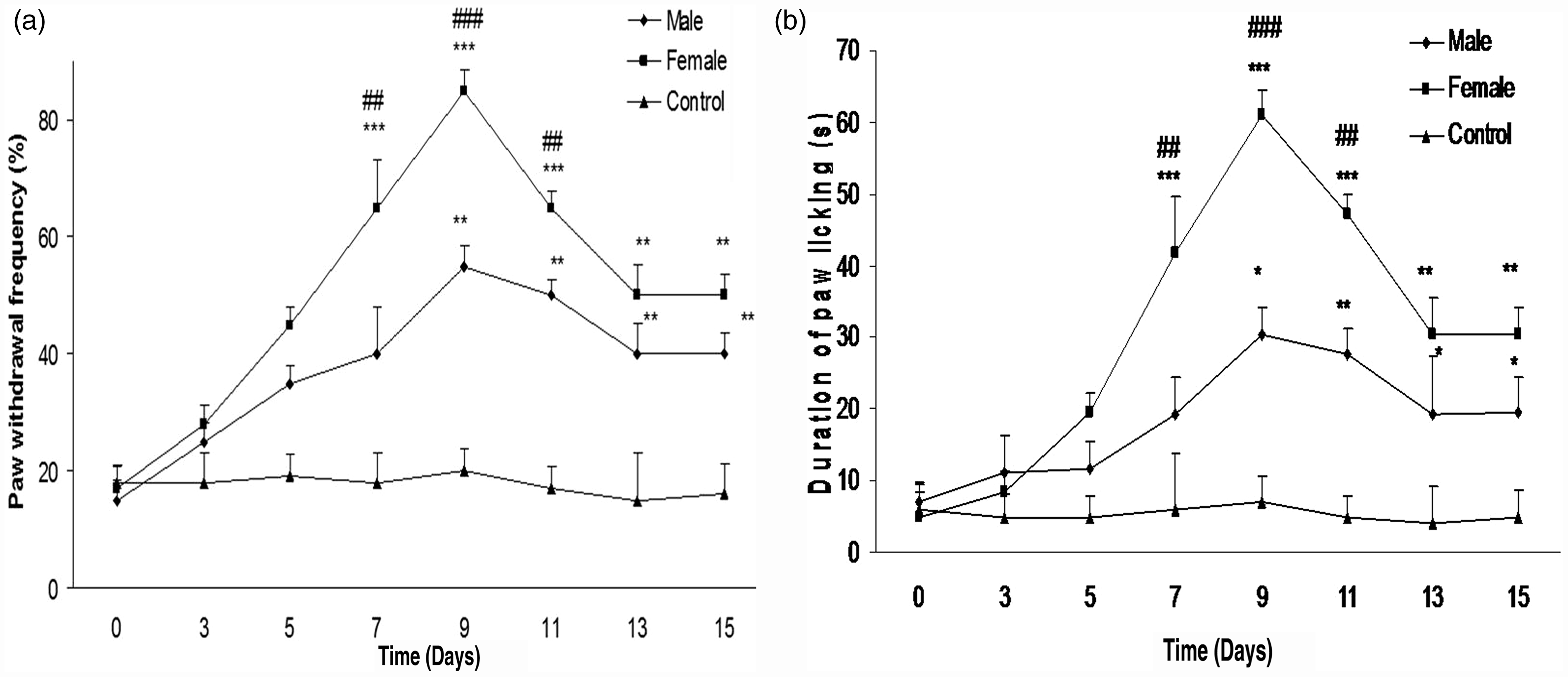
Effects of paclitaxel on mechanical allodynia
We evaluated withdrawal response to mechanical stimulation using von Frey filaments. Paclitaxel (2 mg/kg, for 5 consecutive days) led to a significant reduction in paw withdrawal threshold in male and female mice (P < 0.01) (Figure 2). However, we observed no significant differences in responses between male and female mice during this period of time.
Paw withdrawal threshold after paclitaxel treatment. Paclitaxel (2 mg/kg) was injected for five consecutive days, and the control group received vehicle. No significant gender difference was observed for paclitaxel-induced mechanical allodynia (**P < 0.01 when compared with the control group).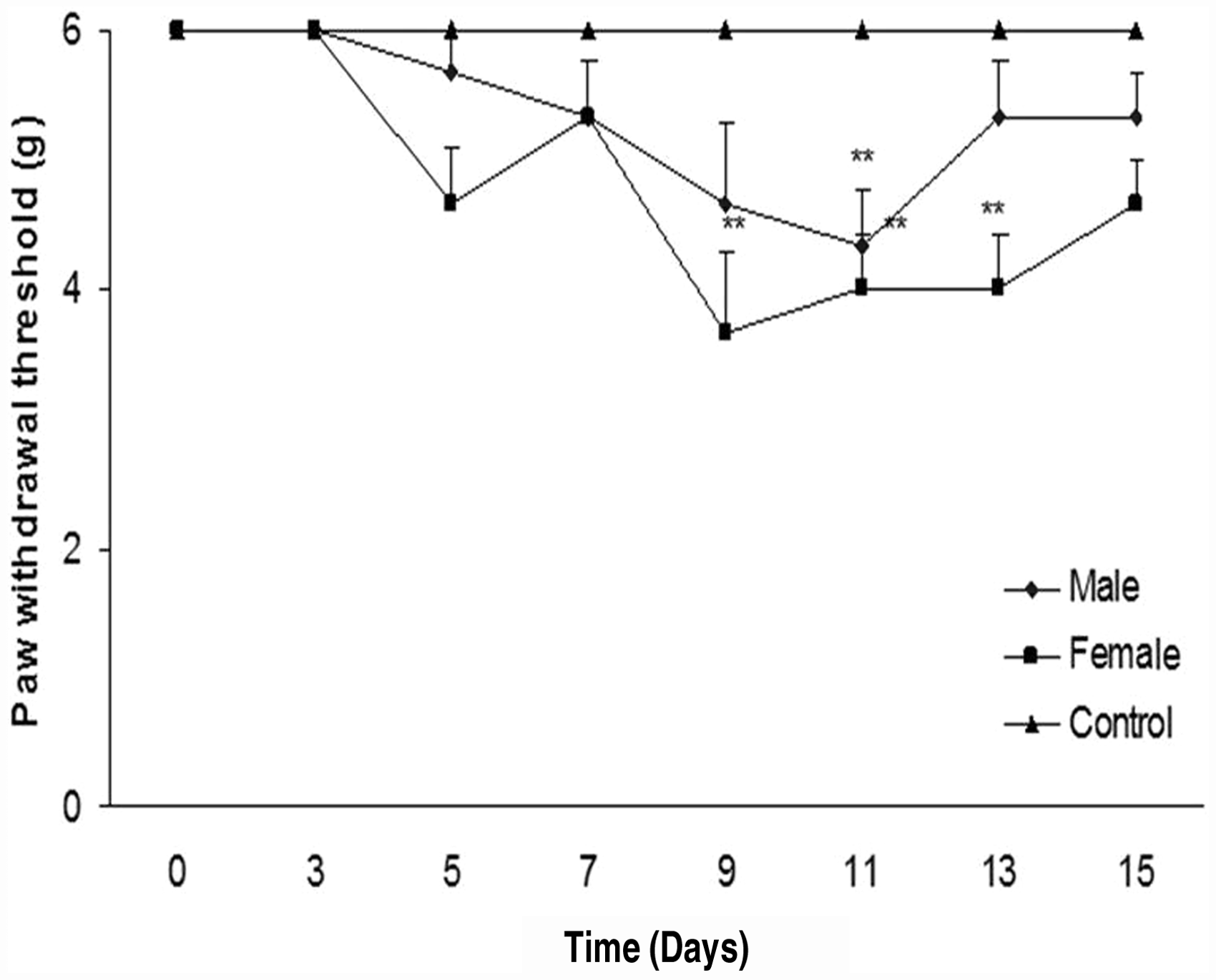
Effects of cisplatin on cold allodynia
Administration of cisplatin (1 mg/kg) for seven days evoked cold allodynia in treatment groups (P < 0.001). Both paw withdrawal frequency and duration of paw licking increased in male and female mice. However, no significant differences were observed between the genders (Figures 3a and b).
(a) Paw withdrawal threshold frequency after cisplatin treatment. Cisplatin (1 mg/kg) was injected for seven consecutive days, and the control group received vehicle. No significant gender difference was observed for cisplatin-induced cold allodynia (**P < 0.01, ***P < 0.001 when compared with the control group). (b) Duration of paw licking after cisplatin treatment. Cisplatin (1 mg/kg) was injected for seven consecutive days, and the control group received vehicle. No significant gender difference was observed for cisplatin-induced cold allodynia (***P < 0.001 when compared with the control group).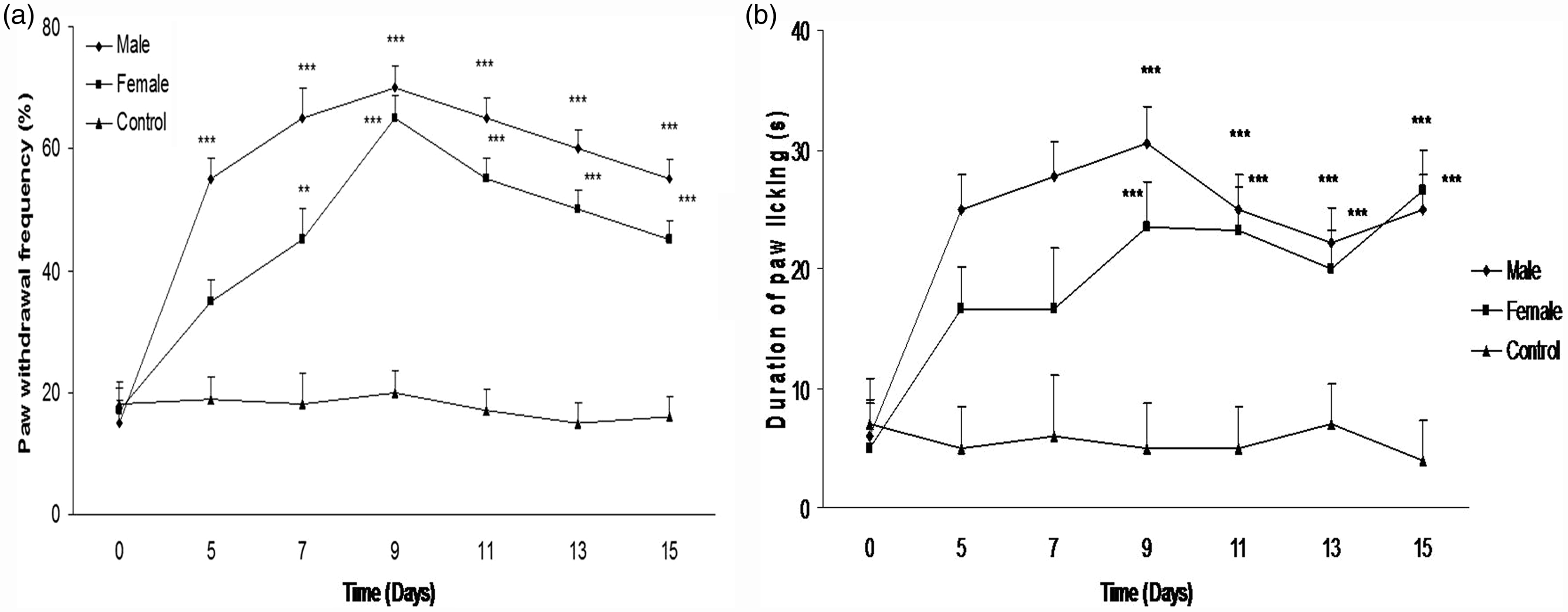
Effects of cisplatin on mechanical allodynia
As shown in Figure 4, a statistically significant difference was observed in the manifestation of mechanical sensitivity between cisplatin-treated mice and the control group (P < 0.01), while no differences were observed between the genders (Figure 4).
Paw withdrawal threshold frequency after cisplatin treatment. Cisplatin (1 mg/kg) was injected for seven consecutive days, and the control group received vehicle. No significant gender difference was observed for cisplatin-induced mechanical allodynia (**P < 0.01 when compared with the control group).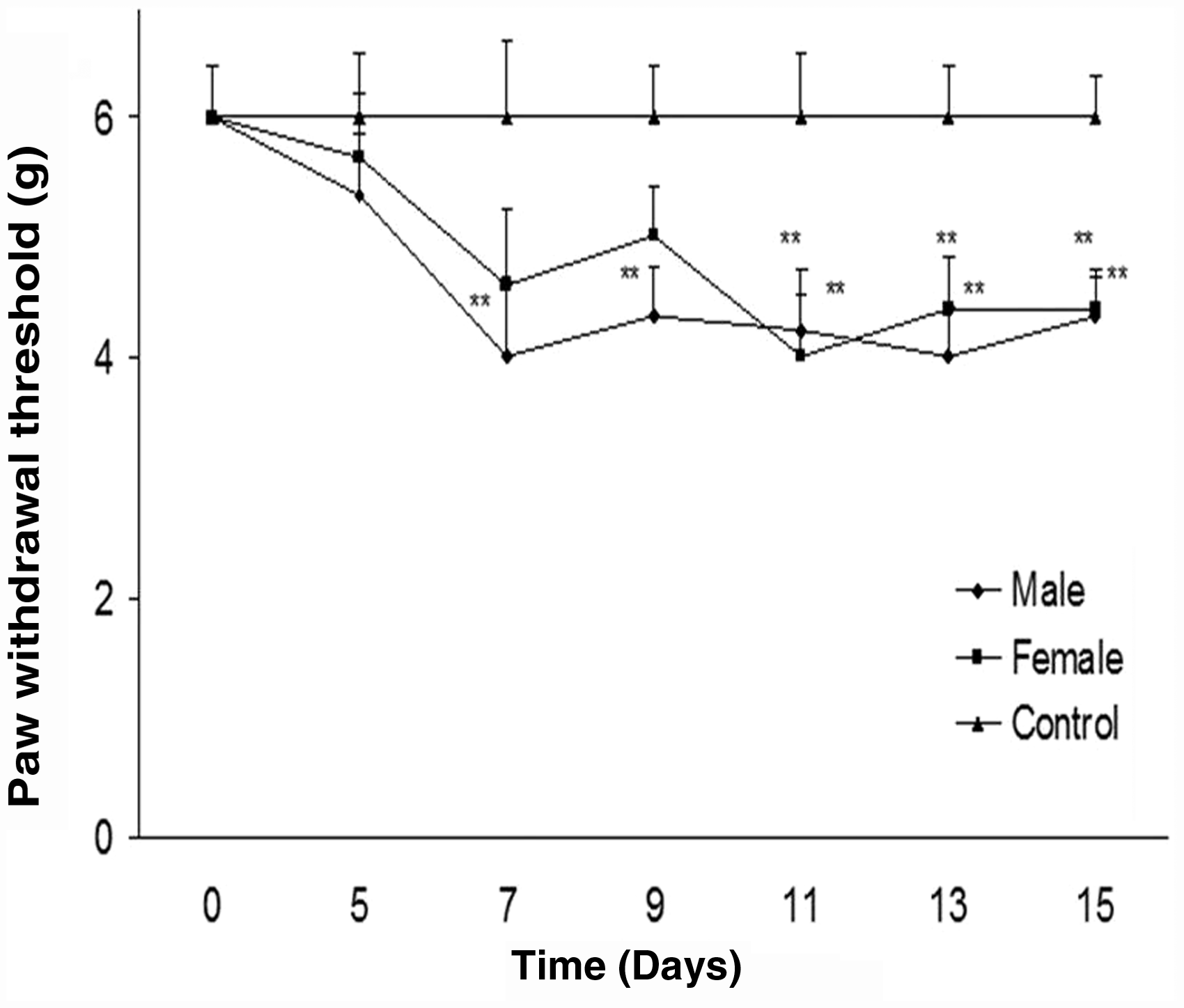
Discussion
The aim of this study was to evaluate the effect of gender differences on levels of chemotherapy-induced neuropathic pain in mice. Paclitaxel and cisplatin are frequently used for the treatment of breast and ovarian cancers; however, the vast majority of preclinical studies to identify the proper treatment for neuropathic pain have been conducted using male mice. In this study we sought to determine whether male mice are good candidates for these models of pain. We showed that paclitaxel-induced cold allodynia was more robust in female mice. Interestingly, no gender differences were observed for paclitaxel-induced mechanical allodynia- and cisplatin-induced neuropathic pain.
Paclitaxel is used as a chemotherapeutic agent with different types of cancers such as breast, cervical and ovarian cancers. It enhances microtubule polymerization, 19 but the mechanisms by which paclitaxel induces neuropathic pain have not yet been demonstrated. 7 Paclitaxel induces degeneration of axonal microtubules but has no effects on the axonal cytoskeleton. 20 We observed no differences in paclitaxel-induced mechanical allodynia between male and female mice; and this finding is consistent with a study which also showed no gender differences in paclitaxel-induced mechanical allodynia in rats. 21 However, gender differences in cold allodynia were not consistent with previous studies, possibly suggesting different mechanisms are involved. The differences between mice and rats have already been noted during other behavioral studies. For example, periorbital and forepaw thresholds, but not hind paw thresholds, are reduced in mice with craniotomy in the migraine model. In contrast to mice, these effects are not observed in rats. 22 It is believed that mouse species are more susceptible than rats to pain and stress. 23 Behavioral differences also exist in response to stressors, and significant differences have been noted among species and even strains within species. 24
Cisplatin is widely used for the treatment of urinary, bladder, and ovarian cancers. 25 It has been reported that sensory perception and sensory nerve conduction are reduced following treatment with cisplatin. 26 Nerve biopsy evaluations have revealed loss of myelinated fibers. 27 Analysis of dorsal root ganglion in animals has shown atrophy of neuronal soma and nucleus. 28 Sexual dimorphism exists in peripheral nerve abnormalities. A study has demonstrated that mortality and weight loss are more robust in male mice treated with cispaltin. 29 In the present study, we observed no differences between male and female mice in cisplatin-induced neuropathic pain. However, one other study has found faster regeneration and functional recovery in female animals. 30
Vincristine induces neuropathic pain which is more severe in females than in males, 31 suggesting that the impact of gender difference depends on the type of tests and models. A large body of evidence has suggested that endogenous sex steroid plays a major role in mediating sex differences in nociception. Pain modulation is more effective during the ovulatory phase than during the early follicular and mid-luteal phases. 32 There are many possible mechanisms by which sex hormones may affect pain. For example, serotonergic and noradrenergic neurons in the nucleus raphe magnus and locus coeruleus express estrogen and progesterone receptors and interact with sex hormones, which leads to the release of opioids in the spinal cord. 33 Interaction with N-methyl-D-aspartate (NMDA) receptors in the spinal cord/brainstem is another mechanism; NMDA receptor antagonists reduce the activity of neurons in trigeminal subnucleus caudalis in an estrogen-dependent manner. 34
Experimental hyperalgesia models have shown significant gender differences; however, not all pain animal models show robust gender differences.35,36 Clinically, pain is more pronounced in women, but few pain conditions are predominant in men, including post traumatic headaches and abdominal migraines. 37 These studies suggest that different mechanisms and risk factors may be involved in each gender. 38
In conclusion, our study demonstrated that there was no significant gender difference in a mouse model of cisplatin-induced neuropathic pain; however, a gender difference was observed in paclitaxel-induced cold, but not mechanical, allodynia.
It may therefore not be advisable to evaluate different analgesics in male animals, as there are many differences between genders and among different models. Human studies should also be performed and the results should be compared with animal studies to determine the best model of pain for evaluating different drugs.
Footnotes
Acknowledgment
The authors gratefully thank Mr Adel Vaseghi at British Colombia Institute of Technology, Vancouver, Canada.
Declaration of conflicting interest
None.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
