Abstract
Pigs are used for long-term biomedical experiments requiring repeated injections, infusions and collections of blood samples. Thus, it is necessary for vascular catheters to be indwelling to avoid undue stress to the animals and the use of restraints. We propose a refined model of percutaneous insertion of long-term central venous catheters to minimize the surgical trauma and postoperative complications associated with catheter insertion. Different sizes of needles (18 Ga versus 21 Ga) for initial puncture of the veins were compared. In conventional pigs weighing less than 30 kg, catheter insertion may be facilitated by using a microintroducer set with a 21 Ga needle. In pigs weighing 50 kg, a standard 18 Ga needle may be preferable.
The pig is the most commonly used large animal species for animal experimentation in the European Union. In 2011, more than 77,000 pigs were used, namely more than four times as many pigs as dogs. 1 Pigs have many advantages over other large animal species, such as their physiological and anatomical similarities to humans and their worldwide availability. 2 Biomedical research often requires access to the venous system for injections, infusions and blood sampling. Although peripheral venipuncture in pigs is feasible, in long-term experiments it imposes several problems. Venipuncture requires constraints and inflicts a certain level of discomfort on the animals. It also induces stress in the animals, which may in turn affect outcome of experiments. 3 Also, repeated venipuncture takes time and may sometimes be difficult to achieve, potentially causing problems for studies where timing or repeated samplings are of the essence.
To overcome the drawbacks of repeated venipuncture, different techniques of long-term venous cannulation have been developed in pigs. Subcutaneously implanted vascular access ports have been used. 4 Their most obvious drawback is the need to use restraints for the placement of a Huber needle whenever the port is being used. 4 To avoid this problem, externalized catheters may be used. 5 Recently, a surgical model of chronic venous cannulation has been proposed. 6 In humans, however, a surgical cutdown technique involving externalized venous access would be considered a last resort, as it would leave a potentially painful wound with an obvious risk of infection. Instead, insertion of externalized long-term vascular catheters in humans is usually achieved by percutaneous insertion of the catheter using the Seldinger technique. 7 The catheter is then subcutaneously tunneled to a separate exit point. This approach minimizes the risk of infection as well as of surgical trauma. It also allows for patency of the cannulated vessel, which is ligated in surgical cutdown approaches. 6
Percutaneous insertion of vascular catheters using the Seldinger technique involves several steps, including the introduction of a guide wire in the vessel of choice. In our experience, introduction of the guide wire may be time-consuming and can prove challenging, even for experienced surgeons. Once the wire is correctly placed in the vessel, the other steps can be easily performed in sequence.
To facilitate repeated injections and blood sampling in conscious pigs without the need for restraint, we developed a new model of percutaneously inserted long-term central venous catheters in line with human standards of care. We also sought to investigate the optimal method of venipuncture and guide wire placement in conventional pigs of different sizes.
Animals
Four 9-week-old, female Yorkshire × Swedish Landrace pigs, included in a pharmacokinetic (PK) study, were used after ethical permission was granted by the Animal Ethics Committee in Umeå, Sweden. The animals were received from Forslundagymnasiet, an agricultural upper secondary school with commercial breeding in Umeå, Sweden. After arrival at the experimental facility, the animals were allowed a period of acclimatization of up to eight days during which the handlers socialized daily with them. The pigs were individually kept in 8 m2 pens throughout the experiment. The pens allowed tactile, visual, and auditory contact through cage bars, and straw was used as bedding. The animals were fed twice daily (Sluka, a commercially available pig diet without antimicrobials from Granngården, Uppsala, Sweden) and had free access to water. Photoperiods were 12:12 h light:dark, and room temperature was kept at 20 ± 1℃.
Placement of externalized long-term central venous catheters (9.5 Fr double lumen; Lifecath®, Vygon, Ecouen, France) was performed at the beginning of the PK study, when the animals were 10 weeks old with body weights ranging from 24 to 30 kg. After completion of the intended four-week PK study, the same animals were used for the comparison of venipuncture techniques. The average daily weight gain (DWG) during this period was 732 g.
Six female Yorkshire × Swedish Landrace (25 to 32 kg) included in another study on mechanical ventilation (with separate ethical permission from the Animal Ethics Committee in Umeå) were used to verify feasibility of the method and to time guide wire placement using a microintroducer set. The animals were purchased from the same breeder and were kept as described above, though they were housed in pairs.
Materials and methods
Insertion of long-term catheters
All four animals in the PK study were equipped with long-term venous catheters (9.5 Fr double lumen catheter; Lifecath®). The procedure was performed under general anesthesia by two different surgeons using different animals. For premedication, ketamine 10 mg/kg (Ketalar® 10 mg/mL; Pfizer AB, Sollentuna, Sweden), xylazine 2.2 mg/kg (Rompun® vet 20 mg/mL; Bayer Animal Health, Copenhagen, Denmark) and atropine 0.05 mg/kg (Atropin® Mylan 0.5 mg/mL; Mylan AB, Stockholm, Sweden) was injected intramuscularly. Premedication was administered with the animal in its pen. When sedation occurred, the animal was transferred to a surgical theatre. A single dose of 8 mg/kg cefovecin (Convenia® 80 mg/mL; Orion Pharma Animal Health, Sollentuna, Sweden) was administered intramuscularly as a prophylactic antibiotic. An auricular vein was cannulated and anesthesia was induced by an intravenous bolus dose of 2 mg/kg propofol (Propofol-Lipuro® 10 mg/mL; B Braun Medical AB, Skene, Sweden), followed by infusion of 6–10 mg/kg/h propofol and 0.2–0.4 µg/kg/min remifentanil (Ultiva®, GlaxoSmithKline AB, Solna, Sweden). The animals were orotracheally intubated and mechanically ventilated. Two milliliters per kilogram per hour of Ringer’s acetate (Ringer-Acetat® Baxter Viaflo; Baxter Medical AB, Kista, Sweden) was administered as a maintenance fluid. Twenty milliliters of bupivacaine with adrenaline (Marcain Adrenalin® 2.5 mg/mL + 5 µg/mL; AstraZeneca AB, Södertälje, Sweden) was infiltrated subcutaneously in relevant areas for postoperative analgesia.
With the animal lying in a lateral position and with the table tilted 10° head down, the distance from the right atrium to the intended access point in the external jugular vein was estimated. A sterile operating field on the side of the neck was then created. All the following procedures were carried out with meticulous attention to sterile conditions. Venipuncture was done percutaneously using a previously described landmark technique.
8
Briefly, a needle was inserted just lateral to the sternocephalicus muscle in the mid portion of the neck at an angle approximately 30° caudal to the skin (Figure 1). A 10 mL syringe was used for continuous gentle aspiration to ensure swift identification of the vessel once it was punctured.
The needle is inserted just laterally to the left sternocephalic muscle and angled caudally. Gentle aspiration with a syringe half-filled with normal saline helps detection of intravenous placement of the needle. For anatomical clarity, the picture is not taken under sterile conditions and the animal is placed in dorsal recumbency.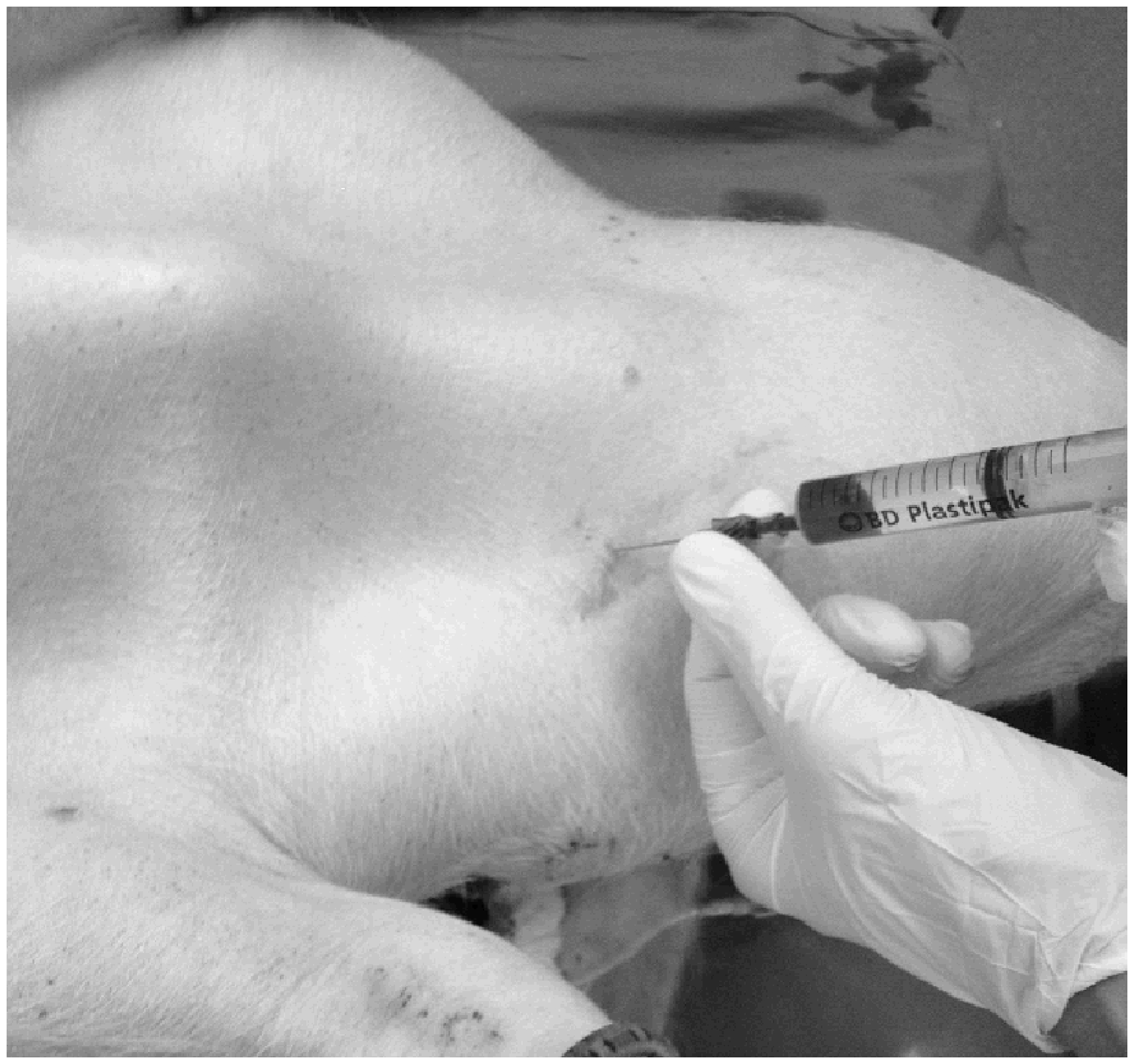
In two animals, the vein was primarily punctured with an 18 Ga needle included in the catheter set (Lifecath®). The standard Seldinger guide wire (0.97 mm diameter, Lifecath®) was then inserted and the needle was removed. A 4–5 mm incision in the skin was made adjacent to the guide wire.
In the two subsequent animals, a 21 Ga needle from a microintroducer set (Micropuncture Introducer Set®; Cook Medical, Bloomington, IN, USA) was used to puncture the jugular vein. Venipuncture was performed using the technique described above. When the 21 Ga needle was placed in the vessel, a 0.46 mm guide wire, supplied in the microintroducer set, was inserted and the needle was subsequently removed. A 4–5 mm incision in the skin was made adjacent to the guide wire, and a 4 Fr dilator, also supplied in the set, was inserted over the guide wire. The guide wire and the inner cannula of the dilator were then removed, and the standard 0.97 mm guide wire was inserted. In all the animals, proper placement of the 0.97 mm guide wire in the cranial vena cava was verified by fluoroscopy.
A 4–5 mm skin incision was then made adjacent to the dorsal midline in the middle part of the neck. Using a tunnelator, included in the catheter set, the 9.5 Fr catheter (Lifecath®) was tunneled from the dorsal incision to the guide wire as described in the manufacturer’s manual. Care was taken to place the cuff in the subcutaneous tunnel created. The catheter was then cut to fit the estimated distance to the right atrium. The large bore dilator from the catheter set was then advanced over the guide wire. The guide wire and the dilator’s inner stylet were removed and the catheter was introduced through the remaining introducer sheath, which was subsequently removed.
The skin at the puncture site was closed with a single non-absorbable suture (Dermalon® 2-0; Covidien, Dublin, Ireland) and covered with an occlusive dressing (Tegaderm®; 3 M, St Paul, MN, USA). The catheter’s wings were sutured with two single 2-0 sutures. No suture was needed for the incision at the catheter’s exit point. It was, however, covered with dressings (Tegaderm®) cut to fit around the catheter. Both lumens were filled with heparin solution (Heparin LEO® 100 units/mL; LEO Pharma AB, Malmö, Sweden). A protective cover was made from a 20 × 20 cm piece of cotton cloth. In the middle of the cloth, a 10 cm incision was made where a 10 cm piece of Velcro was attached to both sides with staples. The cover was then placed over the catheter’s exit point and secured to the skin using single sutures (Dermalon® 2-0) evenly spaced (approximately 1 cm apart) around the cover. Care was taken to fold the cloth slightly between each suture to allow for growth of the animal. After the procedure, anesthesia was discontinued and the animals were extubated. Supplemental oxygen was flowed over the animal’s snout over the first few minutes after extubation to maintain an arterial saturation of oxygen above 96%. The animals were then allowed to recover in their pens for 3–7 days before inclusion in the intended PK study. All injections and samplings during the study were followed by instillation of heparin solution (100 units/mL) of the used lumen.
Comparison of venipuncture techniques in pigs weighing 47–50 kg
After completion of the four-week PK study, and immediately before euthanasia, the animals were again anesthetized according to the protocol described above and used for comparison between guide wire placement methods in larger animals. A 0.97 mm guide wire was repeatedly placed percutaneously in the left and right external jugular veins using either the standard 18 Ga needle (Vygon) or a microintroducer set (Cook Medical) with a 21 Ga needle. Venipuncture was performed using the landmark technique as described in the previous section. The animal was placed in a lateral position with the vein to be punctured facing up. Before puncture of the contralateral vein, the animals were turned to the opposite side. The standard needle and the microintroducer technique were both used in sequence on all vessels in a randomly chosen order. The time from initial puncture of the skin to proper placement of the 0.97 mm guide wire was noted for each technique. Both left and right external jugular veins were punctured with subsequent insertion of the guide wire in all the animals but one. In that animal, the right external jugular vein could not be identified with either needle. The experiment was ended by euthanasia with injection of potassium chloride intravenously.
Time to guide wire placement with a microintroducer set in pigs weighing 25–32 kg
The animals were anesthetized and orotracheally intubated as described above. Lying in dorsal recumbency, the external jugular vein was punctured with the 21 Ga needle, using the landmark technique as described in the previous section. Placement of the 0.97 mm guide wire was facilitated by the microintroducer set as described above. The time from initial puncture of the skin to proper placement of the guide wire was noted. The guide wire was then used for insertion of a short-term central venous catheter. General anesthesia, as described in the previous section, was administered until euthanasia with potassium chloride. A single surgeon performed all procedures in this and the preceding series. The animals were part of an experiment on mechanical ventilation.
Statistics
The statistical analyses were performed using SPSS version 22 (IBM, Armonk, NY, USA). Testing for differences between measurements between groups was performed using a Mann–Whitney U-test. A P value less than 0.05 was considered to be significant.
Results
Long-term catheters
All four animals participated in the study for four weeks and each catheter was used for three injections of study drug as well as for blood sampling on 43 different occasions. The double lumens allowed for injection of the study drug through one lumen and collection of blood samples through the other. All catheters remained functional throughout the study period. The wounds and the catheter exit points were inspected daily. Throughout the experiment, no clinical signs of infection or thrombosis were detected. Use of one catheter lumen was discontinued and that lumen was kept clamped during the last week, as it was accidentally rendered unsterile on the inside after an injection.
Catheter insertion in pigs weighing 24–30 kg
Ultrasound guided placement of the guide wire with primary vessel puncture using an 18 Ga needle proved to be difficult and time-consuming. In both animals this procedure was used and it took more than one hour to complete. The vessel was easily punctured with adequate backflow of blood. Insertion of the 0.97 mm guide wire through the 18 Ga needle was, however, extremely difficult. Once the guide wire was in place, the catheter was tunneled and correctly placed in less than 10 min. In the two animals the microintroducer set was used, the 0.97 mm guide wire was correctly placed in less than 15 min and the whole procedure from initial skin puncture to correctly placed catheter was completed in approximately half an hour.
Time to guide wire placement in 50 kg pigs
With the microintroducer set, time to correct placement of the 0.97 mm guide wire (n = 7) was on average 128 s (range 38 to 275 s). When the 18 Ga needle was used to puncture the vessel (n = 7), the average time to guide wire placement was 69 s (range 38 to 98 s). There were no statistically significant differences between the groups (P = 0.128).
Time to guide wire placement with microintroducer set in pigs weighing 25–32 kg
The average time to 0.97 mm guide wire placement (n = 6) was 6.5 min (range 3 to 15 min). All the animals were successfully cannulated.
Discussion
We present a minimally invasive method for chronic venous cannulation of pigs using a simple and reliable standard technique routinely used in humans. The Seldinger technique of percutaneous access for vascular catheterization was first described 1953. 9 With minor modifications, it can easily be adopted for insertion of tunneled venous catheters. Operators trained in placement of tunneled venous catheters in humans can easily adopt the technique in pigs. The catheters may be used for both injections and blood sampling for a prolonged time without the need for restraints.
The percutaneous approach minimizes damage to tissues adjacent to the cannulated vessel as well as to the vessel itself, 10 thereby virtually obliterating the need for postoperative analgesics except for a local anesthetic administered during the procedure. Compared with an open surgical procedure, the minimally invasive approach of percutaneous venous access also allows for shorter recovery times. The lack of surgical trauma should also minimize the inflammatory response with minimized release of cytokine mediators. Further, the procedure reduces the risk of infection and unnecessary use of antibiotics and discomfort to the animal.
Our group has previously experienced problems with intravenous placement of the Seldinger guide wire using a relatively large bore needle in juvenile pigs. The standard guide wire will not pass through a smaller needle. However, the microintroducer set allows for primary vessel puncture using a small needle, followed by insertion of a guide wire through the small diameter needle and subsequent placement of the standard guide wire and later the large bore introducer. In juvenile pigs, a microintroducer set facilitates easy access to the external jugular vein, whereas in larger animals, it produces an unnecessary extra step in the process. In larger animals, a standard needle may be used directly to place the guide wire correctly.
Refinement
Our proposed model of venous access focuses on minimizing trauma and stress to the animals. The procedure is performed under general anesthesia and is limited to percutaneous access with needles, dilators and a tunnelator. The chosen exit point on the animal’s neck assures a minimal impact on the animal’s post procedure well-being. The choice of an externalized catheter allows for repeated vascular access without restraints or induction of pain. The anesthetic method was chosen for its speedy induction and extremely short recovery time. Typically, the animals were ambulating unassisted within 30 min of extubation.
Reduction
Reliable long-term venous access allows for longitudinal studies in included animals, thereby potentially reducing the number of animals needed.
Footnotes
Acknowledgement
This work was supported by the Medical Faculty, Umeå University and the Council of Västerbotten, Sweden.
