Abstract
Acetaminophen (APAP), a widely used analgesic and antipyretic drug, has the potential to cause lethal hepatotoxicity. Mice are widely used for developing murine models of APAP-induced hepatotoxicity, and many researchers have used these models for APAP-related studies including the fields of biology, pharmacology and toxicology. Although drug-induced hepatotoxicity is dependent on a number of factors (species, gender and age), very few studies have investigated the effect of aging on APAP hepatotoxicity. In this study, we evaluated the effect of age on APAP-induced hepatotoxicity in different weekly-aged mice to establish a model of APAP-induced hepatotoxicity that is an accurate reflection of general experimental conditions. Male ICR mice 4, 6, 8, 10 and 12 weeks old were given APAP intraperitoneally, and mortality, hepatic damage and the plasma concentration of APAP metabolites were evaluated. It was found that younger male ICR mice were relatively resistant to hepatotoxicity induced by intraperitoneal APAP administration. In addition, the APAP-glucuronide concentration in plasma remained essentially the same among the differently-aged mice, while APAP-sulfate levels were dramatically decreased in an age-dependent manner. Thus, it is recommended that mice of the same ages be used in studies related to APAP-induced hepatotoxixity. These results provide evidence in support of not only the age-related changes in susceptibility to APAP-derived hepatotoxicity in mice but also in developing mouse models for APAP-related studies.
Acetaminophen (APAP), N-acetyl-p-aminophenol, is an effective analgesic and antipyretic drug that is widely used as a prescription drug as well as an over-the-counter drug. Although APAP is a first-line analgesic therapy for non-malignant pain, caution must be taken regarding an overdose due to its potential for causing lethal hepatotoxicity. In fact, previous systematic investigations conducted by the World Health Organization (WHO) Collaborating Center for International Drug Monitoring reported that APAP is one of the most common liver injury induced-drugs associated with fatalities. 1 At therapeutic doses, the majority of APAP is metabolized in the liver to non-toxic metabolites, namely APAP-sulfate or APAP-glucuronide, and excreted into the bile and urine. When, however, a toxic dose is taken, sulfation and the glucuronidation pathway become saturated, and excess APAP is then oxidized by cytochrome P450 (CYP) to N-acetyl-p-benzoquinone imine (NAPQI), a toxic metabolite of APAP. Although NAPQI combines with hepatic glutathione (GSH) and is neutralized, NAPQI directly interacts with cellular macromolecules along with the depletion of hepatic GSH, which results in hepatic injury. 2
APAP-induced animal models, such as mice and rats, have contributed to the great progress made in studies of APAP-related issues, such as pharmacotherapy and toxicity. However, several points must be carefully considered when creating APAP-induced animal models, because drug-induced hepatotoxicity is dependent on a number of factors, such as species, gender and age. In the case of APAP, Davis et al. reported that species differences of APAP-induced hepatotoxicity exist and these are more severe in mice than in rats. 3 Furthermore, experimental research has demonstrated that APAP-induced hepatotoxicity appears to be gender-dependent in mice and rats: male murines are more susceptible to APAP-induced hepatotoxicity than are females.4–7 Thus, the male mouse model is one of the most extensively used laboratory animal species in APAP-related toxicity studies.8–10 Despite the massive experimental literature related to the effect of these factors (species and gender) on APAP-induced hepatotoxicity, very few studies have investigated the effect of aging on APAP hepatotoxicity. Previously, Beierschmitt et al. investigated the age dependency of hepatotoxicity in male CD-1 mice after oral APAP administration at a dose of 600 mg/kg, and the results clearly showed that older mice developed more severe hepatotoxicity than younger mice. 11 However, this study did not reflect experimental murine models of hepatotoxicity induced by APAP administration at subtoxic and toxic doses, because most APAP-related studies have generally involved the use of intraperitoneal APAP administration rather than oral APAP administration. In addition, Esteban et al. have reported that intraperitoneal APAP administration appears to be more adequate for hepatotoxicity studies in mice. 12
Therefore, this study aimed to investigate the influence of weekly-age on the APAP-induced hepatotoxicity, and to establish a model of APAP-induced hepatotoxicity that is an accurate reflection of general experimental conditions. To this purpose, different weekly-aged male mice (4, 6, 8, 10 and 12 weeks old) were intraperitoneally injected with APAP.
Materials and methods
Reagents
APAP-sulfate and APAP-glucuronide were purchased from Sigma-Aldrich (St Louis, MO, USA). Perchloric acid and Transaminase CII Test ‘Wako’ was purchased from Wako Pure Chemical Industries, Ltd (Osaka, Japan). Other chemical materials were purchased from Nakalai Tesque, Inc (Kyoto, Japan) if not otherwise specified. All chemicals were of the highest commercial grades available.
Animals
Male ICR mice of various ages (3, 5, 7, 9 and 11 weeks old) were purchased from Kyudo Co, Ltd (Saga, Japan), and were acclimatized for one week. All animal experiments were performed according to the guidelines, principles, and procedures for the care anduse of laboratory animals of Sojo University. Allanimals were maintained under conventional housing conditions, with food and water available ad libitum in a temperature-controlled room with a 12 hlight/dark cycle for one week before the experiments. The mice were fasted for 12 h before being used in experiments.
Influence of age on mortality derived by APAP toxicity
The groups according to age in weeks were intraperitoneally injected with APAP dissolved in saline (12 mg/mL) at a dose of 300 mg/kg (n = 20) or 600 mg/kg (n = 10) between 21:00 and 22:00 and their survival was observed for 24 h after injection.
Influence of age on APAP hepatotoxicity
Ten mice from each group received 300 mg/kg APAP dissolved in saline (12 mg/mL, intraperitoneally) between 21:00 and 22:00. All surviving mice were sacrificed, and blood samples were collected 12 h after the administration of APAP. Each blood sample was centrifuged at 3000
The assessment of APAP metabolites
Within each group, mice of various ages (n = 5) were given APAP (300 mg/kg, intraperitoneally) between 21:00 and 22:00 as described above and blood samples were collected from the inferior vena cava one hour after APAP injection. The APAP-sulfate and APAP-glucuronide concentrations in plasma were analyzed as previously described
14
with minor modifications. In short, 10 µL of plasma was added to 20 µL of 6% perchloric acid and the resulting solution was vortexed. The mixture was centrifuged at 17,000
Data analysis
All data are expressed as means ± SD. The means for groups were compared by using analysis of variance (ANOVA) followed by Student’s t-test. A probability value of P < 0.05 was considered to be significant.
Results
Effect of age on mortality after APAP treatment in mice of different ages
Changes in survival after treatment with APAP (300 mg/kg, intraperitoneally) in mice as a function of age were examined. As shown in Figure 1a, 50% of the 12-week-old mice died within 12 h after APAP treatment. On the other hand, more than 90% of the mice of other ages survived during this period of observation. Furthermore, mice of different ages were also challenged with a lethal dose of APAP (600 mg/kg, intraperitoneally). As a result, the mice started to die around 6 h after the APAP treatment, and all of the 12-week-old mice died within 20 h after the APAP treatment. Interestingly, the survival rate at 24 h after APAP treatment declined as the age of the mice advanced (Figure 1b).
The influence of age on survival rate after intraperitoneal acetaminophen (APAP) injection at a dose of 300 mg/kg (a) and 600 mg/kg (b) in mice of different ages. The survival rate after intraperitoneal APAP injection between 21:00 and 22:00 are shown. Each value shows the percentage for (a) 20 mice and (b) 10 mice.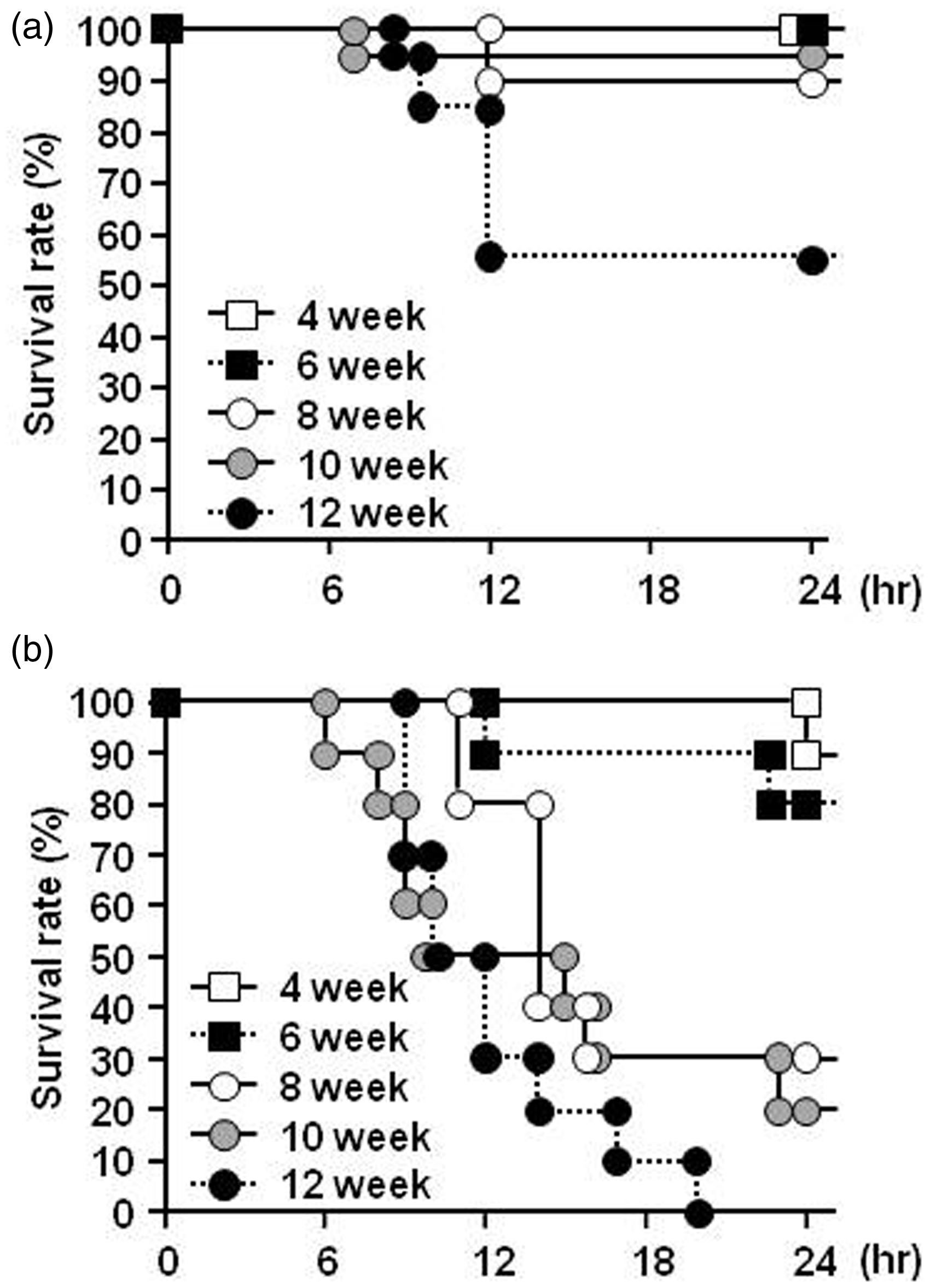
Effect of age on APAP-induced hepatic injury in mice of different ages
Figures 2a and 2b show serum GOT and GPT levels 12 h after the APAP treatment. As a result, serum GOT and GPT levels increased in an age-dependent manner. Accompanied by increasing GOT and GPT in serum, the amount of total hepatic GSH, which has long been known to function as a defensive and protective agent from excess reactive oxygen species, especially a critical line of defense against APAP-induced hepatic injury,
15
decreased in an age-dependent manner (Figure 2c).
Biochemical parameters: (a) aspartate aminotransferase (GOT), (b) alanine aminotransferase (GPT) in serum and (c) total hepatic glutathione (GSH) after an intraperitoneal acetaminophen (APAP) injection at a dose of 300 mg/kg in mice of different ages. All surviving mice were sacrificed, and blood samples and liver samples were collected 12 h after intraperitoneal APAP administration. Each value is the mean with SD for 10 mice (4 and 6 week old), 8 mice (8 week old), 9 mice (10 week old) and 5 mice (12 week old). **P < 0.01 vs. 4 week, #P < 0.05 vs. 6 week, ##P < 0.01 vs. 6 week, †P < 0.05 vs. 8 week, ††P < 0.01 vs. 8 week.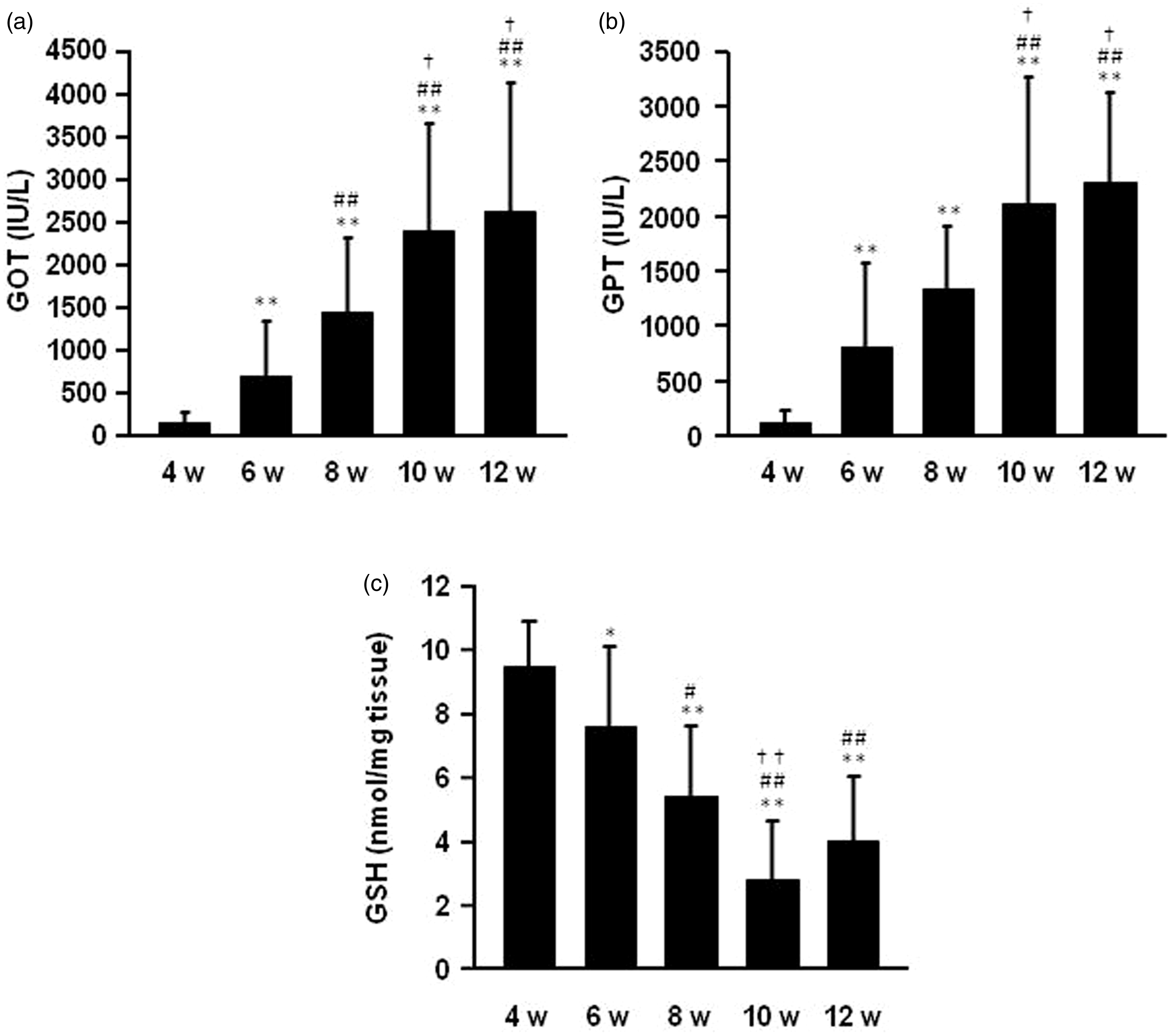
A histological evaluation of the livers was also carried out. The results of H&E staining of liver sections indicated that the administration of APAP induced morphologically observable and severe hepatic damage, including necrosis within the centrilobular region of the livers, and this effect was age-dependent (Figure 3a). In addition, the area of hepatic injury increased with increasing age of the mice, reaching approximately 40% of the area of vision in 12-week-old mice (Figure 3b).
(a) light micrographs of livers stained with hematoxylin and eosin (H&E) and (b) hepatic injury area in mice of different ages treated by intraperitoneal acetaminophen (APAP) injection at a dose of 300 mg/kg. All surviving mice were sacrificed, and liver samples were collected 12 h after intraperitoneal APAP administration. Hepatic injury was assessed using a conventional epifluorescent microscope. Each value is the mean with SD for 10 mice (4 and 6 week old), 8 mice (8 week old), 9 mice (10 week old) and 5 mice (12 week old). *P < 0.05 vs. 4 week, **P < 0.01 vs. 4 week, ##P < 0.01 vs. 6 week, ††P < 0.01 vs. 8 week.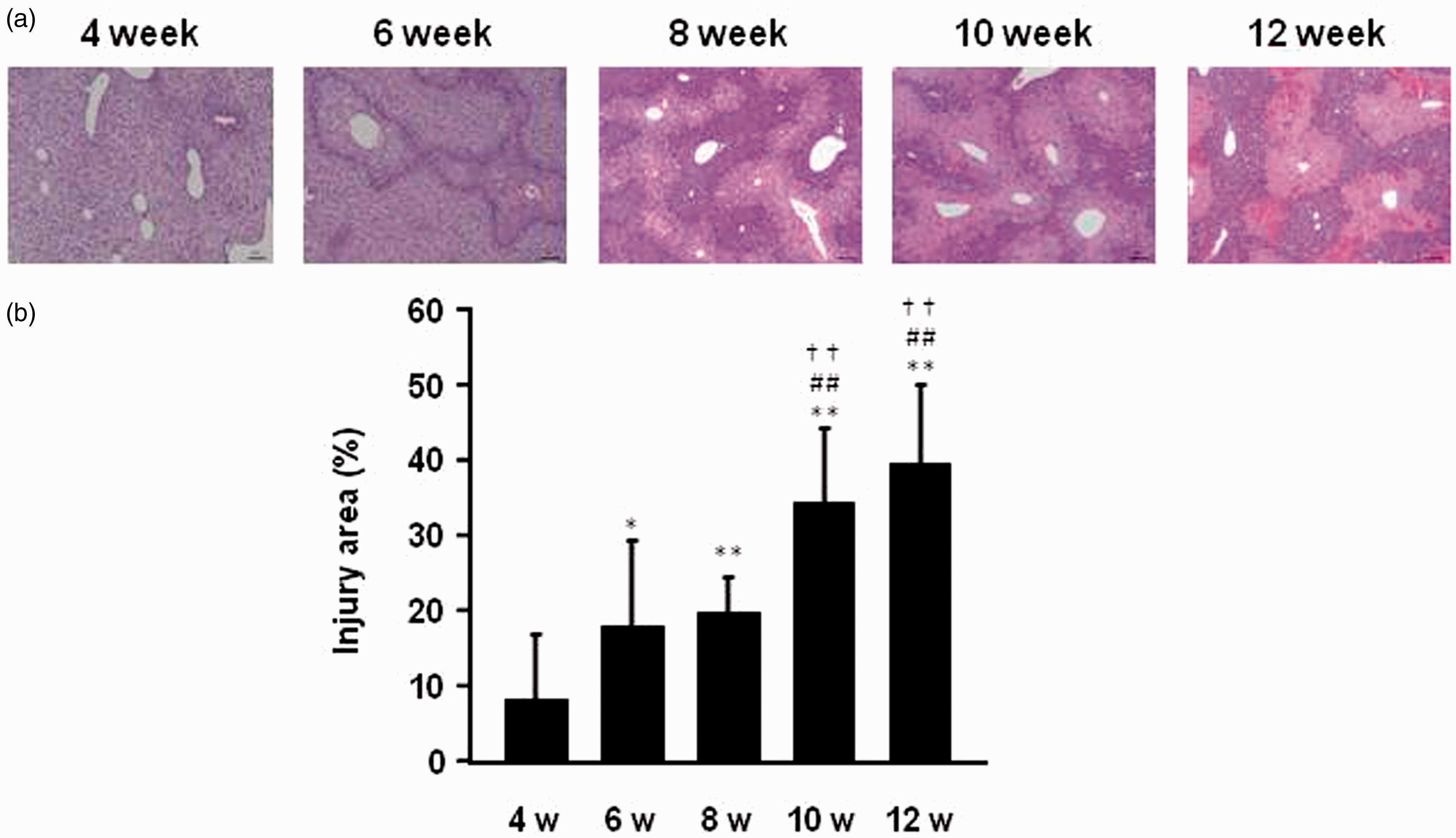
APAP-sulfate and APAP-glucuronide concentration in plasma
Finally, the concentration of APAP metabolites, APAP-sulfate and APAP-glucuronide in the plasma in each group of mice one hour after an intraperitoneal APAP administration was investigated. As a result, the APAP-glucuronide concentration in plasma remained essentially the same among the differently-aged mice one hour after the administration of APAP at a dose of 300 mg/kg (Figure 4a). On the other hand, APAP-sulfate levels were dramatically decreased in an age-dependent manner (Figure 4b).
The plasma concentration of (a) acetaminophen-glucuronide (APAP-Glc) and (b) acetaminophen-sulfate (APAP-Sul) after intraperitoneal APAP injection at a dose of 300 mg/kg in mice of different ages. Blood samples were collected from the inferior vena cava one hour after intraperitoneal APAP injection. The APAP-sulfate and APAP-glucuronide in plasma were analyzed by HPLC as described in the ‘Materials and methods’ section. Each value is the mean with SD for 5 mice. **P < 0.01 vs. 4 week, ##P < 0.01 vs. 6 week, ††P < 0.01 vs. 8 week.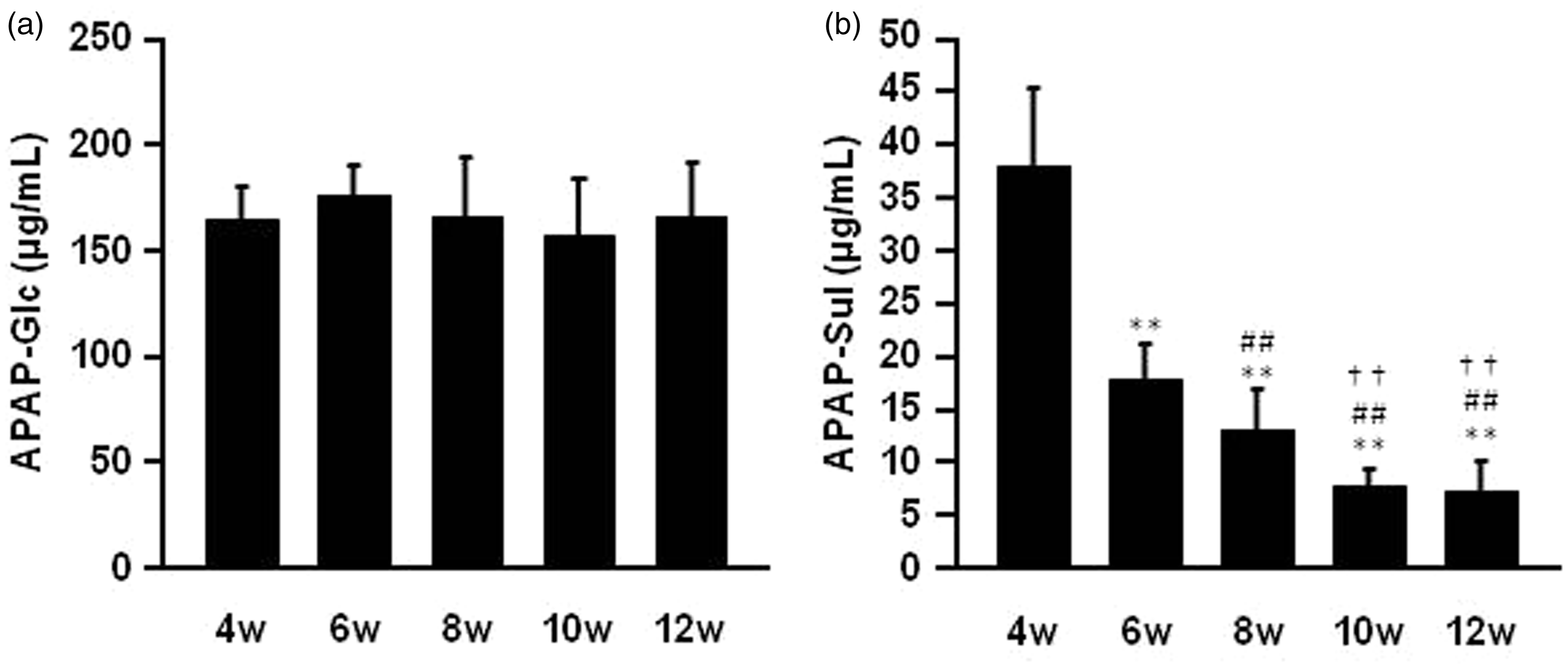
Discussion
The findings reported here indicate that male younger ICR mice are relatively resistant to hepatotoxicity induced by intraperitoneal APAP administration, and that serious hepatotoxicity and increased mortality occurs with advancing age (Figures 1, 2 and 3). There have been other reports of age-related changes in susceptibility to APAP-induced hepatotoxicity in mice and rats. Hart and Timbrell investigated the effect of the oral administration of APAP on hepatotoxicity in litters of immature male BALB/c mice (3–35 days old), and the findings showed that neonate mice were resistant to APAP hepatotoxicity. 16 In addition to mice, a similar age dependency of hepatotoxicity after an intraperitoneal APAP administration in rats was reported.7,17 Taken together with the results obtained from this and previous studies, both an oral or intraperitoneal APAP overdose would be expected to cause more serious toxicity in older animals than in younger ones.
Aging is associated with physiological changes including blood flow, body weight, hepatic mass, body water and fat volume. 2 Especially, an age-related reduction in liver size is known to occur in older animals including humans,18,19 which is of concern, since dosing mice of different ages on a per kilogram of total body weight basis could result in an increased loading dose of APAP to the livers of the older mice. In fact, we examined the age-related changes in the ratio of liver weight to body weight, which showed that the liver mass per body weight was significantly less in 10 and 12-week-old mice compared with mice of other ages (data not shown). Therefore, one possible contributing factor to the age-related alteration in sensitivity to APAP-induced hepatotoxicity in this study is the decrease in liver mass per body weight due to advancing age.
Another possible contributing factor to an age-related alteration in sensitivity to APAP-induced hepatotoxicity might be the decrease of age-related changes in the hepatic metabolism, including Phase I and II drug metabolizing enzymes. The majority of APAP is metabolically converted into non-toxic metabolites in the liver via Phase II metabolism to glucuronide and sulfate derivatives, and excess APAP is oxidized to NAPQI, which is a toxic metabolite of APAP, via the Phase I drug metabolizing enzyme, CYP2E1. Furthermore, NAPQI is neutralized by Phase II conjugation with cysteine by hepatic GSH. In this study, the plasma concentration of APAP-sulfate one hour after intraperitoneal APAP administration was dramatically decreased with advancing age (Figure 4b), while the APAP-glucuronide concentration in plasma remained essentially constant among the various groups of mice. Reilly et al. investigated the modulation of hepatic gene expression of mice that had been intraperitoneally treated with APAP at a dose of 300 mg/kg using microarray technology, resulted in the altered expression of numerous genes within the liver, including metabolism, 6 h after the APAP treatment. 20 Clearly, further studies in this area focusing on Phase I and II metabolizing protein expression and activity related to APAP metabolism as a function of age will be needed to completely understand these possible variables.
Furthermore, Adamson and Harman have previously reported age-related changes of susceptibility to hepatotoxicity induced by APAP using cultured hepatocytes isolated from postnatal or adult mice. 21 Our findings are in agreement with these previous in vitro findings showing that cultured hepatocytes isolated from young animals are more resistant to hepatotoxicity than those isolated from animals at an older age. Interestingly, they also realized that GSH peroxidase, GSH reductase and superoxide dismutase, which are involved with protection against reactive oxidative stress, were all higher in the younger mice than older mice. Therefore, age-dependent alterations of susceptibility to hepatotoxicity induced by APAP may be involved in the difference of the intracellular antioxidant potency between younger and older mice.
Although our results demonstrated that APAP-induced hepatic injury developed more seriously in older mice, some limitations must also be taken into account. Some of the research groups normally used ICR mice in their APAP-related research,22–24 but other groups routinely used mice of different strains, such as C57BL/6 and BALB/c. As mentioned in the Introduction, drug-induced hepatotoxicity is dependent on a number of factors, such as species, gender and age. Therefore, the degree of hepatic damage may be different among mice of different strains. This would be a subject of further study. Another point of note in the present study is that we could not provide conclusive data on the mechanisms whereby aging is associated with increasing APAP-induced hepatotoxicity. It was recently reported that several important factors are at play that can have an effect on APAP-induced hepatotoxicity. Some studies have suggested that the activity of glutamate-cysteine ligase, a rate-limiting enzyme in GSH synthesis, is associated with sex differences in APAP-induced hepatotoxicity in C57BL/6 and CD-1 mice.6,25,26 In addition, Mohar et al. have reported that the gender-specific adduction of peroxiredoxin-6 is an important factor underlying the sensitivity of male C57BL/6 mice to APAP-induced hepatotoxicity. 5 Furthermore, recent studies have shown that inflammation- or immune-related mechanism is involved in the progression of APAP-related hepatotoxicity.27–29 Although these factors were not taken into consideration in the present study, they might contribute to the age-related changes in susceptibility to APAP-derived hepatotoxicity in mice.
In conclusion, male mice are widely used for developing murine models of hepatotoxicity induced by intraperitoneal APAP administration at subtoxic and toxic doses, and many researchers have used this model for APAP-related studies including the fields of biology, pharmacology and toxicology. Some of these studies used mice of different ages in their research. In this study, hepatotoxicity and fatality induced by the intraperitoneal administration of APAP were more prevalent in older ICR mice than younger animals. Thus, it is recommended that mice of the same ages be used in studies related to APAP-induced hepatotoxicity and that strict standardized operation protocols (SOPs) in animal experimentation should be established. Furthermore, Adamson and Harman have shown that the cultured hepatocytes isolated from young mice are less susceptible to the toxic effects not only of APAP but also of other hepatotoxins, such as furosemide, iodoacetic acid and t-butylhydroperoxide. 21 Therefore, it is likely that younger mice are relatively resistant to hepatotoxicity induced not only by APAP but also by other hepatotoxins. The findings reported here not only provide further evidence in support of the age-related changes in susceptibility to APAP-derived hepatotoxicity in mice but also help in the development of mouse models for APAP-related studies.
Footnotes
Acknowledgements
Skillful technical assistance by Mr Hiroki Maesaki is appreciated.
Funding
This work was supported by a research grant from Sojo University.
Declaration of conflicting interests
No conflicts of interest were declared.
