Abstract
The effect of acetaminophen on sulfamethazine
Keywords
Introduction
Acetylation is a major route of biotransformation and detoxification for many drugs, contributing a significant role in drug metabolism and metabolites production that are more readily excreted than the parent drug. The enzymes involved in phase-II biotransformation reactions are transferases and their products are conjugates. In humans, N
N-acetyltransferase 2 (NAT2) (EC 2.3.1.5) is an
The drugs containing aromatic amines or hydrazine groups are converted to aromatic amides and hydrazides, respectively. 6 Inactivation of intracellular NATs by exogenous or endogenous chemical agents could impair the key detoxification pathways and result in enhanced cell toxicity and carcinogenesis. 1 Work on NATs has proved that various ethnic populations may have different metabolic potentials towards a specific drug 7 which is an important implication for clinical trials regarding the modern concept of individualized therapy. 8
Sulfamethazine (SMZ) syn. sulfadimidine is a sulfonamide, a drug of choice for a range of bacterial infections in humans and other animals. SMZ is inactivated by conversion to its
NAT1 is located in almost every tissue examined,
14
whereas NAT2 is mainly expressed in the liver
3
and gut.
15
The irreversible inhibition and inactivation of biotransformation enzymes like NATs by xenobiotics and their metabolites is of toxicological and clinical significance.
1
The toxicological potential and clinical consequences associated with
Acetaminophen (Paracetamol,
Materials and methods
The research work was designed to study the effect of acetaminophen on sulfamethazine acetylation by human NAT2 in healthy male volunteers. Acetylation of sulfamethazine was determined in blood and urine samples after oral administration of both drugs.
Reagents and chemicals
The drug sulfamethazine (Powder) and standards of sulfamethazine and acetaminophen were purchased from Sigma-Aldrich, USA. Acetaminophen (Panadol) manufactured by Glaxo Smith Kline, Pakistan Limited, was purchased from the market. All chemicals and solvents used were of HPLC grade high purity. Deionized double distilled water was obtained from Adventec (GS-590 distillery & CPW-200), Japan from Central High Tech Lab, University of Agriculture Faisalabad. Drug-free plasma was obtained from Allied Hospital, Faisalabad.
Study design
In the first phase of the study, the volunteers were given an oral dose of sulfamethazine 500 mg alone and the concentration of SMZ and acetyl sulfamethazine (AcSMZ) was determined in blood and urine samples by HPLC. These volunteers were given a washout period of 10 days for the next phase of the study. After 10 days the same selected volunteers were again administered 500 mg sulfamethazine along with 1000 mg acetaminophen, and again blood and urine samples were analyzed for concentration of SMZ and AcSMZ. The ratio of AcSMZ to SMZ was calculated and used to determine phenotype of individuals.
Volunteers
Male volunteers for this study were students (aged 21–29 years) from the University of Agriculture, Faisalabad, Pakistan. Each volunteer was apprised of the design of the study, dosing and sampling protocols, and a prior written consent was taken by him. The volunteers were kept fasting for 12 hrs and blank samples of blood and urine were collected. Following this, the volunteers were allowed to take a sulfamethazine 500 mg capsule orally. All enrolled volunteers were non-smokers with no serious illness. The age, body weight, height, body temperature, and blood pressure of each volunteer was recorded and given in Table 1.
Demographic data of volunteers enrolled (n = 19) in the study.

Sample collection
Before drug administration, blank blood and urine samples were taken. The total urine sample of each volunteer was collected up to 6 hrs after drug administration. The urine samples were stored and kept frozen at −20°C till further analysis. Six hours after drug administration, approximately 5 mL of blood was collected in a heparinized plastic centrifuge tube. The blood samples were centrifuged at 3000 rpm for 15 min and plasma was separated and stored at −20°C till further analysis.
Screening for HCV antibodies
Volunteers were screened for anti-HCV antibodies through immuno-chromatographic (ICT) rapid strip method 19 by ACON Laboratories, Inc. (USA) to rule out any potential risk during sample handling and in other laboratory procedures. All volunteers were found to be negative except one with a borderline result who was excluded and referred to the clinical laboratory for confirmation.
Analytical procedure
Quantitative analysis of sulfamethazine and its acetyl metabolite in plasma and urine samples was performed through HPLC method by Irshaid et al. 20
Equipment and instrumentation
HPLC consisted of a Shimadzu CBM-20A (Communication Bus Module) system controller, UV-Visible SPD M20A detector, LC-20 AT pump with FCU-10AL VP flow controller valve and DGU 20A degasser was used. Separation was achieved at ambient temperature using a Thermo Scientific C18 HPLC column (4.6 × 1.5 mm), with pore size 5 µm. The chromatographic data were collected and analyzed using LC solution software.
Chromatographic conditions
The analysis of sulfamethazine was performed by using an isocratic mode. The mobile phase consisted of a mixture of 0.06M phosphate buffer (pH 6.0) acetonitrile and methanol in proportion of 200:70:30(v/v) respectively. The pH was adjusted to 5.9 by 2N HCl and 1N NaOH. The mobile phase was filtered under vacuum by using micro-filter of cellulose acetate (Sartorius Ag. 37070 Gottingen, Germany) with a pore size of 0.45 µm and diameter of 47 nm. Then mobile phase was sonicated (ELMA Sonicator, Germany) for 15 min. The UV detector was set at 254 nm. Flow rate was maintained at 1 mL/min having a run time of 10 min.
The average retention time for SMZ and AcSMZ in plasma was 3.9 and 4.5 min and in urine was 3.8 and 4.4 min, respectively. A representative chromatogram of plasma drug concentration is shown in Figure 1.

Representative chromatogram of plasma sulfamethazine (SMZ) and acetyl sulfamethazine (AcSMZ) with representative peaks at 3.9 and 4.5 min, respectively.
Statistical calculations
The data obtained from both phases of study were analyzed using paired t-test and the respective
Results
Under the chromatographic conditions described, plasma and urine concentrations of SMZ and AcSMZ were determined in 19 (n = 19) healthy Pakistani men from Faisalabad, after oral administration of 500 mg sulfamethazine in two separate phases. The acetylation capacity of human NAT2 without and along with acetaminophen co-administration was evaluated to study the
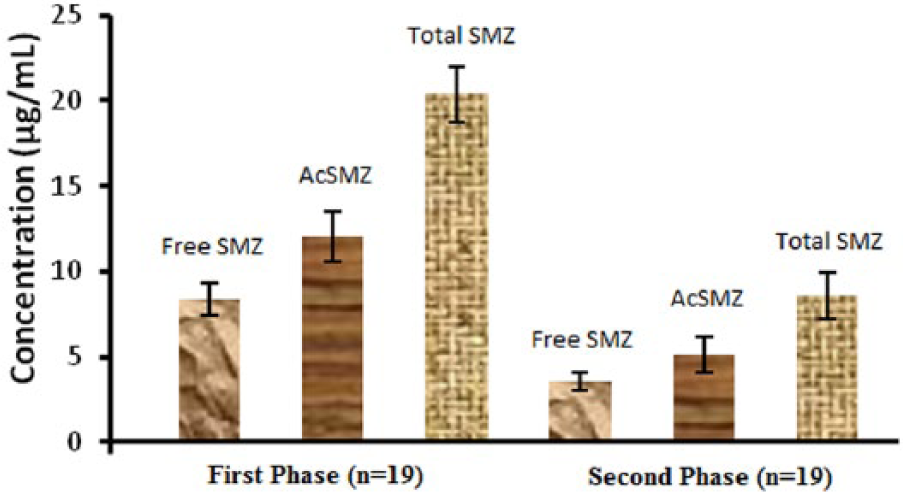
Comparative diagrammatic representation of Mean ± SE value of plasma-free, acetyl, and total sulfamethazine concentration for the first and second phases of the study in male volunteers.
In the first phase of the study, the mean ± SEM plasma acetyl and free sulfamethazine concentrations were 12.01 ± 1.50 and 8.357 ± 0.984 µg mL–1 in the range of 0.754–23.854 and 0.239–17.321 µg mL–1, respectively. The mean ± SEM value of total sulfamethazine was 20.364 ± 1.59 µg mL–1 in the range of 0.993–30.588 µg mL–1. The mean ± SEM value of relative acetylation was 58.404 ± 4.61 µg mL–1 in the range of 17.886-78.195 µg mL–1.
The frequency distribution histogram of the first phase plasma is shown in Figure 3. The histogram shows an apparent trimodal distribution with two antimodes at acetylation ratio of 0.42 and 1.32. The respective slow, intermediate and fast acetylator phenotype was established by using the plasma acetyl sulfamethazine (AcSMZ) to sulfamethazine ratio (AcSMZ/SMZ). Slow acetylators had acetylation ratio <0.42, intermediate acetylators >0.43 <1.01 and rapid acetylators >1.011 at 6 h after an oral dose of SMZ (500 mg). Four out of 19 subjects had acetylation ratio <0.42 and were classified as slow acetylators. Thus the frequency of slow acetylators among the selected subjects was found 21.05% based on plasma.

The frequency distribution histogram for male volunteers (n = 19) on the basis of acetylation ratio (plasma) in the first phase of the study.
The frequency distribution of volunteers based on urine acetylation ratio in the second phase of study is also represented in Figure 4. Comparative drug concentrations of free, acetyl and total sulfamethazine in urine samples collected in both phases of the study is represented diagrammatically in Figure 5.

The frequency distribution histogram for male volunteers (n = 19) on the basis of acetylation ratio (urine) in the second phase of the study.

Comparative diagrammatic representation of mean ± SE value of urine free, acetyl, and total sulfamethazine concentration for the first and second phases of the study in male volunteers.
Discussion
Inter-individual and inter-ethnic differences in drug metabolism occur frequently. These can influence both therapeutic and toxic drug responses and the acetylation is the best established example of genetic polymorphism in drug metabolizing capacity. 21
From the study of Rothen et al.
18
acetaminophen inhibits NAT2 competitively both
Sulfamethazine is termed as polymorphic substrates as the acetylation of other compounds does not vary among individuals, e.g. p-aminobenzoic acid and termed as monomorphic substrate. 23 Slow acetylators of the polymorphic NAT2 suffer more often from side effects of NAT substrates 18 and are usually associated with risk for developing cancer like acute lymphoblastic leukemia in early age. 24
The obtained statistics of our study conclude that acetaminophen significantly (
Acetaminophen and sulfamethazine drug interaction, their molecular bases, renal excretion, and urinary clearance should be studied to develop the concordance between plasma and urine acetylation status.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was funded by the Higher Education Commission (HEC), Pakistan.
