Abstract
Over the years, autologous platelet-rich plasma (PRP) has been used to great advantage in the healing of acute and chronic wounds. However, it is impossible to use in some situations so alternative PRPs need to be considered, such as homologous or heterologous PRPs. In order to evaluate the effects of PRP in the form of autologous, heterologous and homologous gels on in vivo cutaneous wound healing, 18 animals divided into three groups of six, were used for the study, with another six animals used as PRP donors. For the heterologous group an adult, mixed breed dog was used to obtain the PRP. The wounds were induced using an 8 mm punch. The left side was treated with NaCl 0.9%® (A) and a PRP gel was applied (GA = autologous; GHE = heterologous and GHO = homologous) on the right side (B). The wounds were evaluated for a period of 17 days. On the 17th day biopsies were taken for histopathological assessment of the wounds. The percentage of contraction was evident in side B (treated), which was confirmed in the microscopic analysis of the histological sections. Thus, it was concluded that PRP, regardless of the source, improves and accelerates the healing process, demonstrating its therapeutic potential on cutaneous lesions and its use in patients with impaired wound healing.
Wound healing is a complex biological process involving cellular and chemical responses, with the initial purpose of reorganizing the extracellular matrix components with the objective of rapid wound closure, in addition to the obtaining of a functional and aesthetically satisfactory scar.1,2 Although there is no epidemiological data on this in Brazil, alterations in skin integrity affect the quality of life of sufferers and constitute a serious public health problem as they have physical, psychological and social impacts, coupled with high economic costs. 3
Studies of platelet-rich plasma (PRP) focus on the evaluation of their growth factors, which are important in the repair of wounds during healing and are released through platelet degranulation. These granules, when in contact with their respective nuclear receptors, stimulate angiogenesis, replication of tissues, healing, and growth of new organic structures.4,5
PRP can be obtained using a simple method, with a good cost/benefit ratio, through which high concentrations of growth factors are acquired. 6 To present regenerative properties and the desired therapeutic effects, concentrations should be greater than 300,000 platelets/µL. However, some authors report that for better efficiency the optimal concentration should be around 1,000,000 platelets/µL, i.e. about 5× above the baseline concentration.7,8
The sources of PRP can be autologous, homologous or heterologous, the former having the advantage of reducing the transmission of infectious diseases and immunological rejection reactions.5,8 Among cases where the use of autologous PRP is limited are those of patients with wounds associated with chronic diseases such as cardiovascular conditions, diabetes mellitus and hypertension, because they have poor health, and in the long term develop a framework of thrombocytopenia, preventing successive blood collections. 9 There are also situations of extensive wounds caused by burns. 10 In these cases the possibility of using other sources of PRP would be interesting. However, a possible disadvantage of using homologous or heterologous sources could be the inability of the platelets to adequately secrete all the growth factors in addition to containing the antigenicity factors.5,8 In recent studies, satisfactory results have been achieved for the treatment of lower limb wounds in diabetic patients treated with PRP gel obtained from compatible homologous platelets, and corneal ulcer healing in rabbits using a combination of PRP and heterologous blood serum.9,11
Thus, this study aimed to compare the morphometric feasibilities of using autologous, heterologous and homologous PRP in experimentally-induced wounds in rabbits.
Materials and methods
Animals
The animal ethics committee (protocol number 1639) of the University of Oeste Paulista, Presidente Prudente, Brazil approved this work, in accordance with Institutional and National guidelines. Twenty-four healthy adult white rabbits (
Induction and treatment of wounds
The rabbits were divided into the following groups: autologous GA (
Initially, skin antisepsis was performed using 70% alcohol, followed by demarcation of the lesion site and subcutaneous application of 1.0 mL of a local anaesthetic (lidocaine hydrochloride 2% with epinephrine) at the lesion site.
An 8 mm punch was used to perform the surgical wound in the dorsal region of both the left (A) and right (B) sides. 13
The skin was removed with the aid of anatomical forceps, preserving the musculature. The wound on the left, considered as the control (side A) was treated with sodium chloride 0.9%® and the wound on the right (side B) was treated with PRP in gel form. A dressing was then placed over both wounds (A and B) with sterile rayon and an adhesive plaster (Band Aid®). After the surgical procedure, the animals received tramadol hydrochloride (0.5 mg/kg) intramuscularly, administered twice a day (every 12 hours) 12 for three consecutive days in order to minimize the initial discomfort.
The first dressing change took place three days after wound induction. From this point on, the dressings were changed every four days, according to the protocol established by Vendramin et al., 14 with modifications, until the end of the 16th day of the experiment. On the 17th day, a wound biopsy was performed (using an 8 mm punch and anatomical forceps) for histopathological evaluation of the wounds and to observe the early phase of scar remodelling.
Collection and processing of the autologous, homologous and heterologous PRP
For the autologous PRP each rabbit’s own blood was used, and for the homologous PRP one healthy rabbit was used for each rabbit with a wound.
For the heterologous PRP the dog was manually restrained and blood was collected from the external jugular vein. In the autologous and homologous groups, the blood was collected from the auricular vein, after anaesthesia, adopting the same protocol used for the induction of the wound. Following anaesthesia, 4 mL of blood was collected from the central ear artery with a 25 G surgical scalp vein needle. The material was transferred to a vial containing the anticoagulant sodium citrate, where one aliquot was removed for automated platelet counting (automatic counter Sysmex pocH Diff 100iV; Roche Diagnóstica Brasil Ltda, São Paulo, São Paulo, Brazil) and then centrifuged, first at 200
At this stage two distinct levels were formed: an upper level with a slightly yellowish coloration, denominated platelet-poor plasma (PPP), and a lower level with reddish coloration. The PPP was withdrawn and a new platelet count was conducted on the remainder, with the objective of determining whether there was a concentration six times greater than the initial. 15 The platelets were counted in both automated and manual (hemocytometer; Boeco, Hamburg, Germany) systems. 16
As an adequate platelet concentration was detected, the liquid PRP was transformed into a gel through the addition of calcium gluconate 10%, in the proportion of 4:1. 14 The final volume of PRP in gel form was approximately 0.5 mL.
Morphometric evaluations
The wound edges, in millimeters, were measured (width and height) with the aid of digital callipers (DC-60 Western®; São Paulo, São Paulo, Brazil). The measurements obtained were used to calculate the wound area using the formula: A = π × R × r, where A is the area, R is the largest radius and r is the smallest radius.17,18
The percentage of contraction of the wound was calculated using a mathematical model where the percentage of contraction (Pc) equalled the final area (Af) minus the initial area (Ai) times 100 ( × 100) divided by the initial area (Ai), in other words: Pc = (Af – Ai) × 100/Ai. 19 In each case the wound was photographed with a digital camera (Nikon®; Nikon Corp, Tokyo, Japan).
Histopathological evaluation
Prior to the removal of the fragments (17th day), the same anaesthetic protocol was performed as at the induction of the wounds. The degeneration, necrosis and regeneration of the epidermis were evaluated, along with the oedema, haemorrhage, degree of neovascularization and fibrosis type of inflammatory infiltrate in the dermis. For all the analysed parameters a scoring system was applied where: (0) corresponded to absence, (1) to slight alteration, (2) to moderate alteration and (3) to marked alteration. The skin samples were fixed in a solution of 10% formalin buffered at pH 7.0 for 24 to 48 h, and then washed in running water for one hour. Following this, the fragments were transferred to a solution of 70% alcohol. Next they were processed in accordance with the routine histological technique for optical microscopy and with inclusion in paraffin. Using a rotary microtome, 5 µm thick sections were obtained for all samples and stained with haematoxylin–eosin (H/E) and Masson’s trichrome for identification of collagen fibres.20,21 All evaluations were performed by a single observer in a blinded manner.
Statistical analysis
To validate the assumptions of data normality and homogeneity of variances the Shapiro–Wilk and Levene tests, respectively, were performed. To determine whether the histopathological scores differed between groups on side A and side B, we used the non-parametric Kruskal–Wallis contrasted with the method of Student–Newman–Keuls tests. To determine whether these scores differed between sides A and B, within each group, we used the Wilcoxon test.
To verify whether the averages of the percentage of wound contraction of side A in relation to side B differed between groups within each moment, we used one-way analysis of variance (one-way ANOVA).
To verify whether the final percentages (M17-M0) of wound contraction differed between sides within each group, we used the paired
Simple linear regression was used to model the percentage of wound contraction (dependent variable) versus the time in days (independent variable) for each of the groups and for both sides, A and B. To determine the regression coefficients and different intercepts between the lines adjusted for each group, we used the unpaired
Results and discussion
In our study, no clinical sign was observed over time. The use of tramadol hydrochloride postoperatively might have contributed to the welfare of the animals. 22 Furthermore some authors, working with PRP in wound healing, have reported a possible analgesic effect of the use of concentrated platelets in that they contain endocannabinoids, which, under inflammatory conditions, significantly increase their production to confer analgesia. 23
As shown in Figure 1, the appearance of the wounds (A and B) in all three types of treatment remained pinkish in color throughout the experiment, without the macroscopic characteristics of inflammation.
Photograph demonstrating the process of skin contraction of wounds in rabbits treated with different sources of platelet-rich plasma (PRP). Macroscopic characteristics after 17 days of treatment. A, B and C correspond to the control side (not treated) of the autologous, heterologous and homologous PRP groups, respectively. D, E and F correspond to the treated side in the respective PRP groups.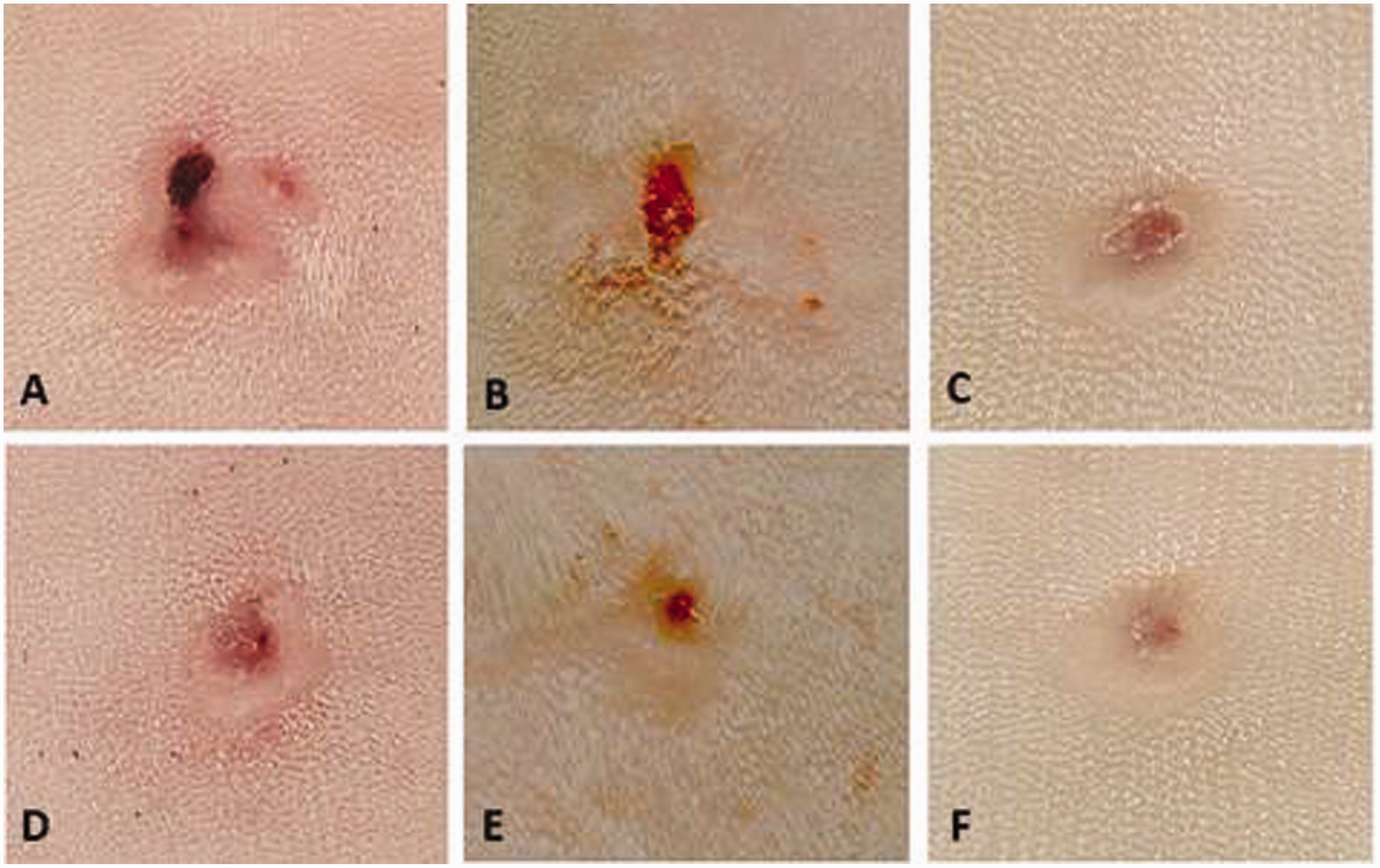
With respect to the histopathological evaluation, it was found that all animals treated with the three types of PRP showed complete re-epithelialization. The presence of a fibrin-leukocyte crust and angiogenesis was observed in wounds A and B of all three groups (
The inflammatory infiltrate seen in the GA and GHE groups was of the lymphocytic type, with a discrete presence of macrophages, greater than or equal to 50% in both the A and B wounds. There was already a discrete presence of neutrophils in the GHE group. Despite this finding, the use of heterologous PRP did not result in the presence of infection or an allergic reaction, corroborating other studies that have utilized heterologous sources of PRP.11,25
Collagen fibres were present in all the groups (Figure 2), with the highest quantities in the rabbits treated in the GA and GHO groups ( Light microscopy showing the skin of rabbits treated with different sources of platelet-rich plasma (PRP). A, B and C correspond to control side (not treated) of the autologous, heterologous and homologous groups, respectively. D, E and F demonstrate the treated side of the respective PRP groups. A and C: small quantity of collagen fibres. B and E: moderate quantity of collagen fibres. D and F: large quantity of collagen fibres. Masson’s trichrome, magnification (100 × ).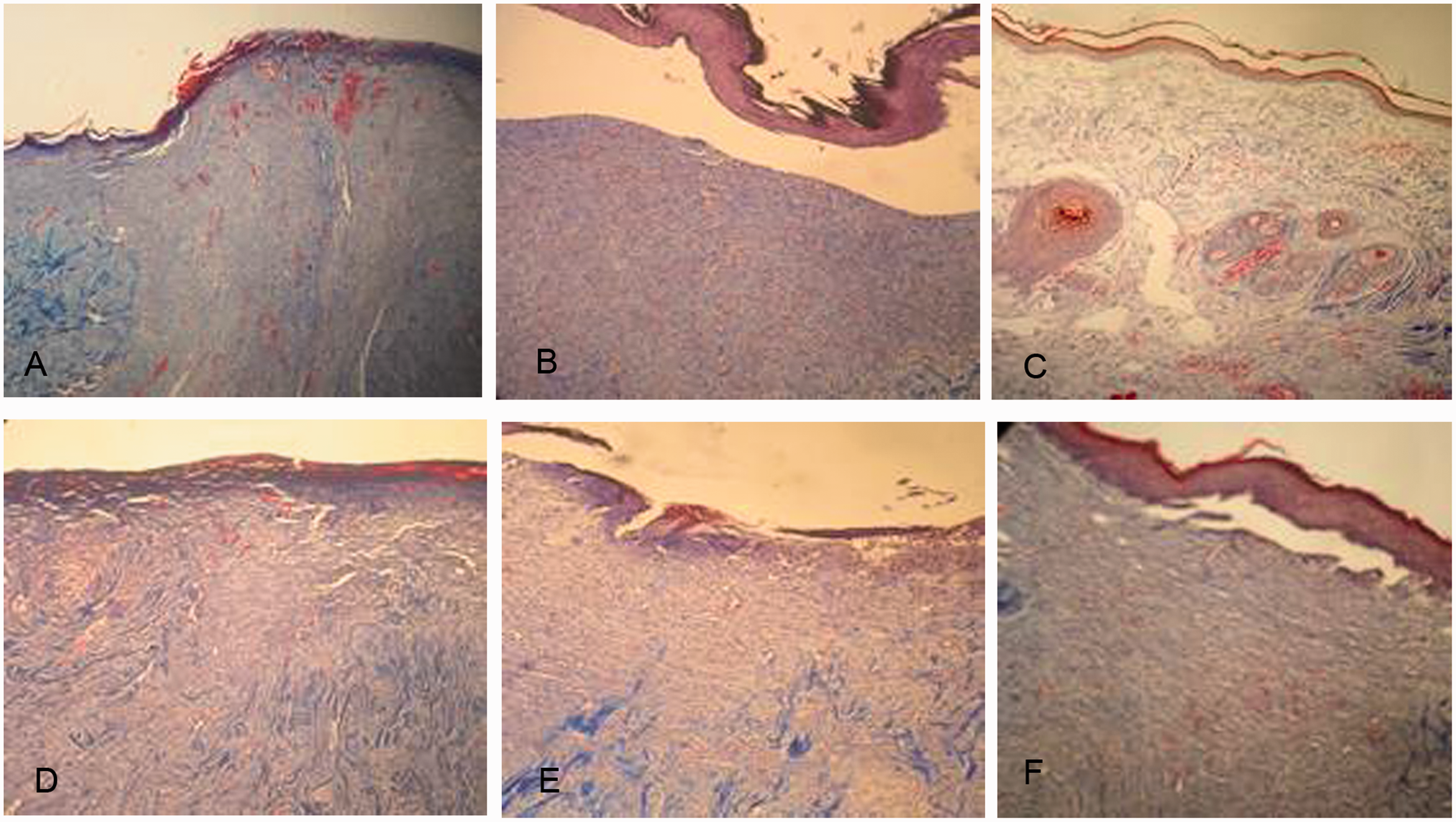
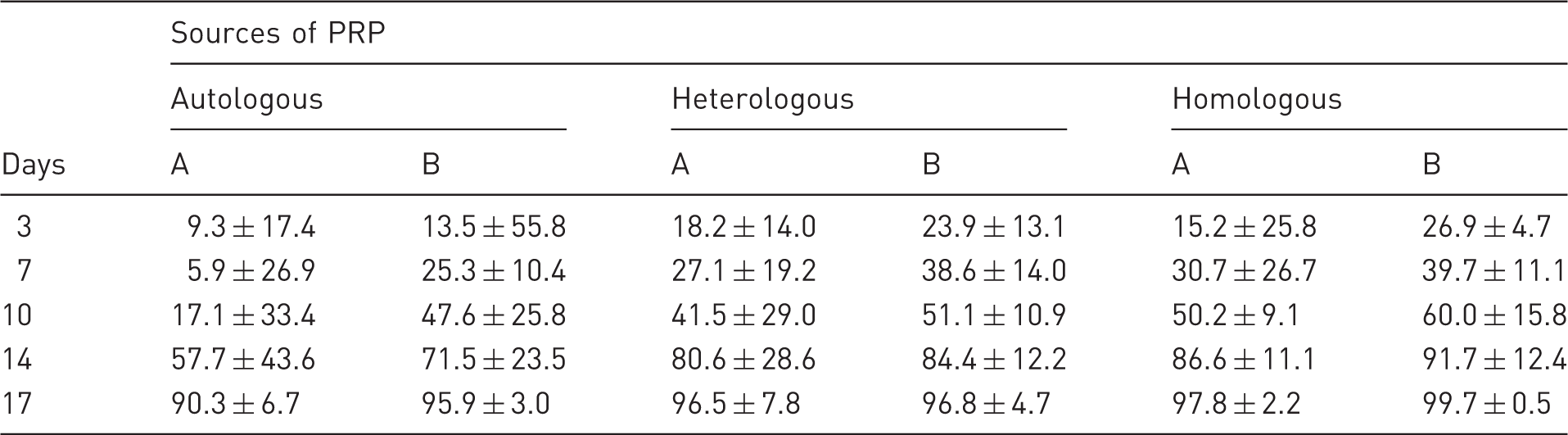
Percentage (average ± standard deviation) of skin contraction of wounds (A: control side; B: treated side) of rabbits treated with autologous, heterologous and homologous platelet-rich plasma (PRP) on different days.
Footnotes
Conclusion
It was concluded that PRP improves and accelerates the healing process, demonstrating its therapeutic potential for cutaneous lesions, and its possible use in cases where obtaining autologous blood is not feasible. Despite the heterologous group not presenting a statistically significant difference, it is suggested that this therapy be used only after giving priority to the homologous source.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
