Abstract
Although environmental enrichment is known to improve laboratory rodent wellbeing and enhance scientific data collection, relatively little is known with regards to the type of enrichment that might be useful for zebrafish (Danio rerio). Therefore, this study explored if zebrafish displayed preferences for a range of enrichments, including substrates, artificial plants, combinations thereof and airstones. Tanks divided into two compartments containing different enrichment cues were used to determine the preferences of zebrafish housed in pairs and groups of eight. When comparing time spent in enriched versus barren compartments, dominant individuals in a pair displayed a preference for substrate and behaviourally excluded the subordinate (p < 0.05). In groups there was a preference for all substrate (p < 0.01) and plant (p < 0.05) enrichments over barren conditions. The strongest preference was for gravel substrate and images of gravel attached to the bottom of the tank. When preferences were compared for different enrichments, gravel (both sexes, p < 0.01) again emerged as the cue attracting the most significant preferences, with any combination featuring gravel substrate preferred over any combination featuring sand (p < 0.05). The study has demonstrated that zebrafish reared in barren conditions preferred structural enrichment over standard conditions; however, when fish were held in pairs this was influenced by dominance status and in groups this was influenced by gender.
Fish are now the second most popular experimental model in the UK, with more than 560,000 procedures carried out in 2011. 1 This represents an increase of 15% compared with the previous year. Zebrafish (Danio rerio) have emerged as a popular model of early embryonic development and mutant phenotypes, and are used in large numbers for developmental and genetic studies. Anecdotal evidence suggests most zebrafish are housed in barren tanks with no added complexity such as refuge, plants or substrate. 2 However, the European convention no. 123 for the protection of vertebrates used in research (2005) has proposed that enrichment should be applied to all captive animals in order to improve their welfare.
Enrichment relates to a broad spectrum of husbandry improvements and includes structural enrichment as well as social context. Structural enrichment has been categorized into naturalistic enrichment, aiming to recreate the wild habitat in a captive environment, and behavioural engineering, prompting motivated behaviour by offering rewards, usually food. 3
Laboratory animal facilities provide standardized accommodation, designed to promote the animals' physical health but not necessarily prioritizing the performance of their natural behaviours. In rodents, natural behaviours are still observed even after generations of captive breeding. 4 For example, mice prefer spaces with nest-building materials to those with a completed nest box. 5 The provision of a nest box for gerbils has been found to reduce stereotypical digging. 6 Enrichment has also been endorsed for laboratory rabbits, where animals kept in an enriched cage system displayed less stress-related and stereotypical behaviour. 7
Until recently, published studies on the effects of structural enrichment in fish tanks have focused on anxiety or fearfulness assessment (boldness, neophobia) and cognition, 8 using wild-caught sticklebacks, as well as physiological parameters indicative of biological functioning such as egg clutch size (Berriman, 2011, unpublished data). Housing zebrafish in tanks containing clear plastic pillars or with a background attached to the outside of the tank resulted in a larger clutch size, which may suggest improved welfare, whereas other forms of enrichment, including plastic plants, have had no obvious effect (Berriman 2011, unpublished data). Studies have investigated the behavioural response of zebrafish to increased structural complexity. Zebrafish spent twice as long in a compartment equipped with artificial plants and clay pots compared with a barren compartment. At the same time, no significant differences were observed in terms of aggressive and sociopositive behaviour. 2 Some findings have suggested the addition of sanitizable artificial plants as standard practice in zebrafish facilities. 9 A study on sanitizable enrichment objects designed for use in toxicology facilities found that zebrafish housed with glass rods sustained aggression levels longer than those from barren tanks. Rather than imposing enrichment it is vital that we provide these fish with choices to understand their subjective preferences. The present study aimed to create a preference order from binary choice tests by providing zebrafish with a range of items routinely used as enrichment in ornamental and laboratory aquaria. Where the other studies prescribe one particular enrichment design, one naturalistic, 2 one according to toxicological requirements, 10 this investigation utilizes a variety of different enrichment types in order establish a hierarchy of preferences. Furthermore, the wild zebrafish habitat has been extensively characterized, describing a range of plant types and substrates. 11 This may help design tank environments accordingly, extrapolating from natural habitat preferences. 12
With our simple choice test we hypothesize that fish will prefer enrichment items over barren conditions and that gender and/or social relationships may impact upon this preference. Females and males differ in their motivation and resulting behaviour; males defend a spawning area and females aggressively target other females in midwater. 13 Therefore, we expect males to prefer substrates to other types of enrichment more than females. If the provision of cover or complexity improves welfare it would be expected that fish will prefer the provision of plastic plants. Dominant individuals tend to constrain the behaviour of subordinates, thus we expect that subordinates may be excluded from preferred resources. Zebrafish in groups exhibit much less aggression, and as such their preferences may differ from fish held in male–female pairs. By applying the enrichment preference assay to two distinct social contexts this study also aims to evaluate how group sizes can affect the fishes' capacity to exhibit preferences.
Materials and methods
Animals and housing
General
In order to reduce numbers used, the experiment incorporated repeated assays on a small number of zebrafish groups (six pairs and five groups of eight) that allowed statistical analyses to be performed. All zebrafish, Danio rerio (N = 52, mean weight 0.58 ± 0.021 g, 9 months at start of experiment; AB strain), were bred and reared at the University of Liverpool zebrafish facility. These fish had been held in barren 10-litre plastic tanks (see below), in a semi-closed recirculating system (replacing approximately 20% of system water per day, average 2.5 water changes/h; water quality monitored daily, parameters available on request) with UV filtration at 28 ± 1℃; 12:12 h light:dark regime in mixed-sex groups averaging 10 individuals. Before and during the experiment animals were fed once a day with Tetra® Tropical Flakes aquarium fish food ad libitum, supplementing twice a week with enriched 48 h-old brine shrimp (Artemia sp.). During the experiment, all feeds were administered halfway between the two observations.
Pair study
Six males (0.38 ± 0.03 g wet weight, 2.88 ± 0.06 cm standard length) and six females (0.69 ± 0.07 g wet weight, 3.27 ± 0.08 cm standard length) were paired by randomly taking 12 fish from different stock tanks. The animals were weighed (to 0.01 g) and measured (to 0.1 cm) at the beginning of the experiment; this was followed by 7 days of acclimation in barren experimental tanks (described below). After this period all fish were transferred into the preference experimental tanks and allowed 48 h to acclimate, with behavioural observations conducted on the following 5 days.
Group study
Forty (20 males, 0.47 ± 0.02 g wet weight, 3.00 ± 0.04 cm standard length; 20 females, 0.69 ± 0.03 g wet weight, 3.17 ± 0.04 cm standard length) D. rerio were taken from 40 different stock (group) tanks and arranged into five groups of four males and four females at random, then placed into experimental tanks with two barren compartments. This was to prevent previous social relationships confounding the results. After 7 days of acclimation the fish were transferred into new tanks designed as described below, randomly assigning four fish to either side of the division. This was followed by a further 48 h acclimation period before observations commenced.
This research was approved by the Ethics Committees at the University of Liverpool and the University of Nottingham and conducted humanely under a UK Home Office Project Licence (PPL 40/3534). No fish sustained injuries or exhibited signs of stress during the behavioural assays.
Tank design
Tanks
Ten-litre volume PVC tanks (26 × 22 × 14.5 cm; Aquatic Habitats, Apopka, Florida, USA) were divided lengthwise using an opaque grey plastic partition, creating two equal 5-litre compartments (Figure 1). Each partition had a 5 × 5 cm opening starting 15 mm from the bottom to allow transfer of the fish between the compartments. To prevent visual disturbance with adjacent tanks, the sides of the tanks were covered with opaque polythene. During the experiment equal amounts of feed were simultaneously introduced into both tank compartments.
Design of the experimental tanks with examples of cues presented in the preference tests.
Enrichments
The competing cues presented to zebrafish to determine their preferences for one of the two cues. The resources were presented on opposing sides and the fish could choose which side to enter.

aGroup study only
Data collection
Pair study
Observations were conducted from a vantage point 1.8 m (6 ft) away from the subjects, hidden behind shelving. This distance still allowed the unaided eye to recognize sexual traits (males and females were differentiated by sexual dimorphism, with females being larger, more rounded and more silvery while males appear more streamlined and golden 16 ). Each pair was exposed to all nine preference tests. Order of presentation of the tank designs was randomized to limit any sequential bias. So with each tank design assigned a letter between A and B, the sequence of designs was AEFGBIDCH for one pair and DIHCGAFBE for another, with similar random sequences for the remaining zebrafish pairs.
For each test an observation period of two 10 minute (10am ± 2 h and 5pm ± 2 h) observation sessions were conducted each day for each tank, randomizing observation order for each observation session.
Data were collected on duration (s) of time spent in the left or right chamber for each individual. The animals were categorized into dominant and subordinate for each observation session. Individuals chasing their partner at least once during the observed period with little or no reciprocation were categorized as dominant, in which case the other animal was classed as subordinate. If no interactions occurred, no classification was made for the observation session. At the end of every week for each tank the animal classified as ‘dominant’ in most observation sessions was determined. As dominance was recalculated every week, in some cases dominant and subordinate roles reversed from one week to another (N = 3) and this was included in the analysis.
Group study
As with the pair study, order of presentation of the tank designs was randomized to limit any sequential bias. All data were collected through direct observation, taking instantaneous samples at 15 second intervals over a 5 minute period, thus generating 21 sampling points. This was carried out once in the morning and once in the afternoon at similar times as above. In this experiment only the numbers of males and females in the least populated chamber were counted then subtracted from ‘4’ to determine the number of fish in the other chamber. An inter-observer reliability test was conducted for sex and occupancy to ensure consistency and precision, yielding a correlation coefficient of R = 0.96 (df = 41, p < 0.001) demonstrating the reliability of the protocol.
Statistical analysis
Pair study
The effect of different order of presentation of the enrichment cues on occupancy time was tested (Kruskal–Wallis test), and found to be non-significant across all categories: p = 0.57 for males, p = 0.78 for females, p = 0.69 for dominant and p = 0.88 for subordinate fish.
For six of the nine preference tests, there was sufficient dominance behaviour to allow a comparison between dominant and subordinate individuals. The Kolmogorov–Smirnov-test (applied separately for each preference test) established that the data did not adhere to a normal distribution. Therefore non-parametric statistical methods were employed for the further analysis.
Data were further processed by analysing the effects of time of day (morning samples versus afternoon samples) and effect of absolute time spent in the tank for each tank design (day 1 to day 5) using the Kruskal–Wallis test for both. As the tests showed that there was neither a significant effect of time spent in the experimental setup (p-values ranging from 0.34 to 0.99) nor of the time of day at which the observations were made (p = 0.31), the data were converted into daily then weekly means for the rest of the analyses.
To explore differences between different compartments, between males and females as well as dominant and subordinate individuals in occupancy time, mean values were calculated for each tank design and week. The resulting values were analysed with the Wilcoxon Signed Rank test.
The results were also presented in a preference order, according to percentage occupancy derived from the mean occupancy time for the six tanks used for each preference test.
Group study
For each tank design tested, all 21 occupancy counts for each observation period were totalled for each tank. From these values weekly cumulative occupancy counts were calculated for each individual tank, for each preference test. As in Kistler et al. (2011),
2
data were analysed by first converting occupancy counts into ratios, then using the ratios to calculate Jabob's preference index
17
:
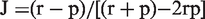
R is the ratio of the number of fish in the enriched compartment to the number of fish in the structured compartment plus the number of fish in the empty compartment, and p is the available proportion of each compartment of the experimental space in the aquarium, respectively, in this case p = 0.5. The index ranges between +1 for maximum preference, and −1 for maximum avoidance. To examine preference for a given compartment for the whole observation period (1 week with 8–10 sampling points) the index was calculated per aquarium. Data were first analysed in SPSS 21 for Windows with two-way Analysis of Variance (ANOVA), using the factors <enrichment type> and <sex>, also testing the effect of <order of presentation> of the enrichment cues as covariate. To test for non-random use of structures (significant difference from zero) a one-sample t-test was conducted (with n−1 degrees of freedom, n is the number of aquaria in the analysis). We did not use a correction factor (such as the Bonferroni method) to account for the increased probability of type I error for multiple t-tests, because with this the interpretation of a preference test depends on the number of other tests performed, with the likelihood of type II errors also increased so that important differences may be deemed non-significant. 18
Results
Preferences of paired zebrafish
Occupancy
The dominant individuals within the pairs (5 females, 1 male) exhibited a preference for the sand compartment (most preferred cue 76%, W = −2.20, p = 0.028, Table 2; Figure 2(a)), whereas subordinate individuals (5 males, 1 female) stayed mainly in the barren side (71.1%, W = −1.99, p = 0.046). Dominant zebrafish (4 females, 2 males) also displayed a preference for gravel (76% occupancy, W = −1.99, p = 0.046), whereas subordinate individuals appeared to be excluded and restricted to the barren area (71% occupancy), although this was not statistically significant (W = −1.57, p = 0.116, Figure 2(b)). For both substrates there was a difference in time spent in the enriched compartment between dominant and subordinate individuals as subordinates were constrained by the dominant (W = −2.20, p = 0.028 for sand and W = −1.99, p = 0.046 for gravel; Figure 2). No preference was displayed for a submerged plant versus a barren area for either dominance category (dominant fish, W = −0.94, p = 0.345, N = 6; subordinate fish, W = −0.31, p = 0.753).
Mean percentage of time spent by paired zebrafish in (a) a tank compartment with sand versus a barren compartment; and (b) gravel versus barren (N = 6). Preference order based upon percentage occupancy in the enriched compartment versus a barren compartment in group-housed (N = 5, 4 males and 4 females in each tank) and pairs of zebrafish (N = 6). In pairs dominance status is also shown. *significant at 5% level **significant at 1% level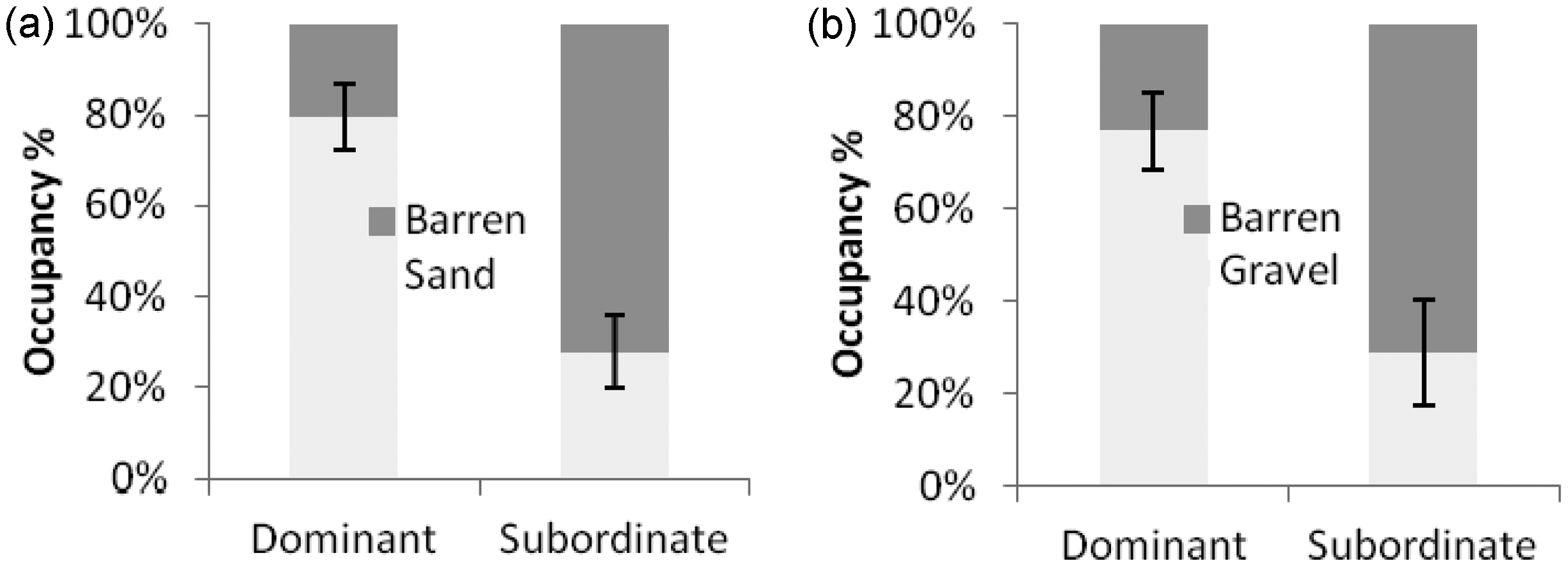
The tests involving comparisons between substrates, plants, plant/substrate combinations and airstones versus barren showed no effect of sex or dominance status.
Preferences of grouped zebrafish
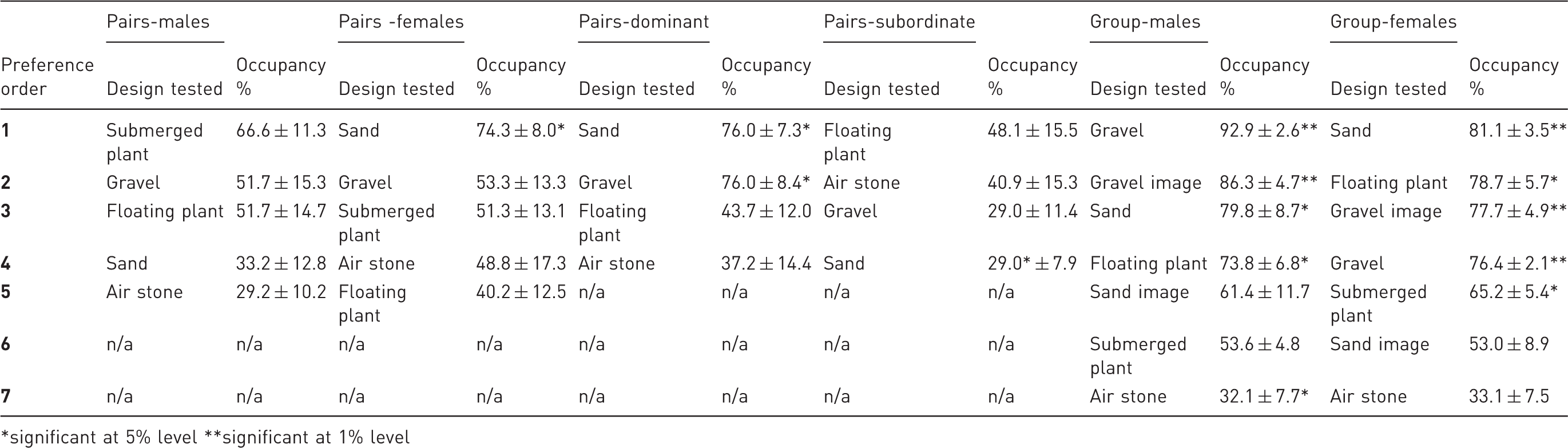
Overall, occupancy for grouped fish was affected by enrichment type (F = 12.54, p < 0.001) but not by sex (F = 1.02, p = 0.32) or order of presentation (F = 0.02, p = 0.88).
Occupancy rates in substrate-enriched compartments were significantly higher than in barren compartments (Table 2; Figure 3(a, b)): For males (gravel, one-sample t-test: t = 16.47, p < 0.001) and females (sand, t = 7.29, p = 0.002) the substrate compartments featured highest in the order of preferences. The preference for substrate over barren also extended to images of gravel placed underneath the tank compartment (t = 7.72 for males, p = 0.002 and t = 5.64 for females, p = 0.005, Figure 3(j)). Fish did not exhibit a similar preference for images of sand substrate compared with a barren area (t = 0.89 for males, p = 0.424; and t = 0.23 for females, p = 0.83, Figure 3(h)). Both sexes significantly preferred gravel over sand substrate (t = 11.53 for males, p < 0.001; t = 7.92 for females, p = 0.001, Figure 3(c)).
Mean percentage of time spent by zebrafish kept in groups of eight in (a) gravel vs. barren; (b) sand vs. barren; (c) sand vs. gravel; (d) floating plant vs. barren; (e) submerged plant vs. barren; (f) floating plant vs. submerged plant; (g) airstone vs. barren; (h) sand picture vs. barren; (j) gravel image vs. barren; (k) first choice substrate (gravel) and plant (floating plant) vs. second choice substrate (sand) and plant (submerged plant); (l) first choice substrate and second choice plant vs. second choice substrate and first choice plant (N = 5).
Preference order based upon percentage occupancy in a compartment containing one or a combination of enrichment cues versus another in group-housed (N = 5) and pairs of zebrafish (N = 6).
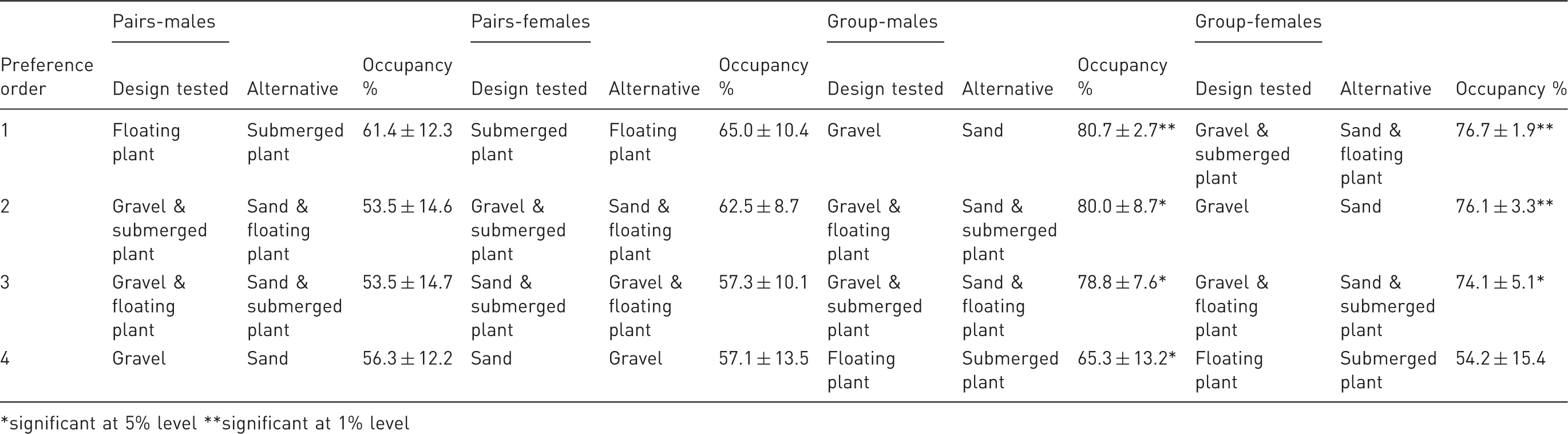
*significant at 5% level **significant at 1% level
Both male and female zebrafish preferred combinations featuring the first choice substrate (gravel) over those including the second choice substrate (Table 3, Figure 3(k, l)). The occupancy for males was similar for both tests for gravel and floating plant (t = 3.36, p = 0.028) and for gravel and submerged plant (t = 3.81, p = 0.019) as it was for females (t = 14.235, p < 0.001 for gravel and submerged plant; t = 4.67, p = 0.01 for gravel and floating plant). Finally, male (t = −2.96, p = 0.04) and female (t = −2.22, p = 0.09) zebrafish spent more time in the barren compartment when airstones were installed on the other side (Table 2, Figure 3(g)).
Discussion
The present study supplied a range of enrichment cues that were preferred by zebrafish over standard barren holding conditions except for airstones. When fish were held in pairs, dominance status influenced preferences with subordinates generally found more in the area less preferred by the dominant. Group-housed zebrafish displayed a strong affinity for gravel substrate, whether as individual cue or combined with either plant type. There were differences linked to gender, with males preferring simulated overhanging vegetation to rooted artificial plants. These findings demonstrate the importance of social context and gender in zebrafish behaviour and underline the utility of providing structural enrichment as part of their husbandry regime.
Paired dominant zebrafish tended to prefer the half with substrate as opposed to the barren half of their tank. Effects of gender were confounded by dominance relationships, with the dominant obviously excluding the subordinate from its preferred compartment (pers. obs.). This is exemplified by the preference for gravel or sand by dominant animals and the concomitant exclusion of the subordinate individuals. In zebrafish, dominance is linked to size, with larger animals more likely to dominate smaller ones. 19 Female zebrafish are usually larger than males, 16 which was also true for the individuals used in the pair assay (W = −3.96, p = 0.003, df = 10). As a result, the majority of animals identified as dominant in the pair study (five for sand, four for gravel) were also female, which may explain why both female and dominant individuals spent significantly more time in the sand compartment. The relative absence of subordinates from the sand compartment suggests that they were prevented from spending more time there by the dominant individuals, who defended their preferred area. This is supported by the fact that while subordinate individuals entered these compartments as much as dominant ones they spent significantly less time there, confirming our hypothesis that the higher aggression levels in smaller groups may deter subordinate individuals from choosing their preferred physical environment, in order to avoid conflict or due to social interactions with the dominant. Structural enrichment has been shown to modulate dominance behaviour in several other fish species, with individuals more likely to defend their territory when plants and pots were added. 20
All of the substrate and plant enrichment cues presented to the zebrafish housed in the group tanks were preferred by either one or both sexes when a barren compartment was offered as an alternative. The cues were chosen to resemble substrate and foliage from a natural environment, yet appealed to zebrafish which were laboratory bred and reared in barren conditions. This is consistent with another preference assay showing that captive-bred zebrafish prefer enriched environments, 2 suggesting that zebrafish may have some behavioural needs that are not met by barren conditions. Diverse animal species carry a range of innate needs and responses which are undiminished after generations of captive breeding and which elicit pro-enrichment preference behaviour. 21 This is exemplified in studies on laying hens, 22 mice,23,24 rats 25 and blue foxes in fur farms. 26
There was a pronounced preference for gravel, alone or combined with other cues, over other substrate and combinations. This may not be entirely expected, as the natural environment of zebrafish features a range of substrates, including silt (which equates to very fine sand) – the most common substrate in the wild habitat survey – sand and boulders. 11 The appeal may lie in camouflage from predators or conspecifics, which may explain why images of substrate alone were sufficient to elicit preference behaviour. This was especially true for gravel images, which attracted occupancy rates almost as high as the actual substrate. Preference by both sexes for gravel images over the adjacent barren compartment was far more pronounced than that for images of sand, where only a weak preference by male zebrafish was found. Teleosts generally have well-developed vision and are able to recognize a range of properties of visible objects, including size, texture, pattern and brightness as well as colour contrast. 27 In this case, in the absence of texture, the interrupted pattern of the images seems to have sufficed in simulating actual substrate. At the same time, the relatively uniform pattern of sand and its thus reduced utility for camouflage may explain why this substrate and its images were less desirable. One could argue that substrate acts as lid over the clear tank bottom and that this alone may have been desirable for the fish. However, as there was no preference when sand pictures were offered, a ‘lid effect’ seems very unlikely.
The affinity to artificial plants, especially to the simulated overhanging vegetation, could be attributed to the diet choices displayed by zebrafish in their natural habitat, with terrestrial insects, particularly ants, constituting a substantial proportion of the stomach contents of wild zebrafish. 14 Arguably these insects can be associated with terrestrial plants breaking through the water surface from above.
The preferences shown in this study are consistent with research 2 investigating preference and behavioural response of zebrafish to increased structural complexity, where zebrafish spent twice as long in a compartment equipped with artificial plants and clay pots compared with a barren compartment. In the present study, which investigated several different enrichment cues, occupancy rates for the compartments with added structural complexity proved even higher, especially those for substrate. Similar results have been published for other fish species: brown trout (Salmo trutta) also preferred gravel when given the choice between that and a uniform bright plastic sheet. 28 Three-spined stickleback (Gasterosteus aculeatus) preferred a ‘complex’ substrate (red and brown gravel ranging 5–20 mm, unevenly distributed) over a ‘simple’ (sand) substrate. 29 Red snapper (Lutjanus campechanus) also preferred coarse substrate (shell pieces) over a sand substrate. 30 This suggests that the provision of substrate may be beneficial to teleosts, as areas of high structural complexity at the microhabitat level (such as heterogenous substrate) are linked to increases in macroinvertebrate numbers and diversity. 29 This may promote increased foraging and is likely to relieve boredom in a barren environment. These results are reflected in an investigation of the neural consequences of enrichment in a zebrafish tank showing that cell proliferation in the forebrain of Danio rerio kept in tanks with gravel and artificial plants was significantly higher than in fish from barren tanks, perhaps reflecting improved brain development. 31
Substrate has been shown to produce substantial welfare benefits for many other species, in both zoo 32 and research animals. 33 For example, it would be hard to envisage rodent cages without substrate such as bedding and nesting material, which constitutes an essential hygiene and behavioural implement. Mice and rats have clearly shown that they prefer certain substrates over others. 34 With zebrafish facilities commonly avoiding the use of substrates for reasons of hygiene, less problematic replacements such as substrate images could help in providing a better captive environment.
While fish spent more time in most structural enrichment designs compared with the adjacent barren compartments, airstones were not preferred. This may be attributed to neophobia. Proximity to the air stones may also require an increased energetic effort. Over time one could expect the zebrafish to be less deterred by the novel stimuli if neophobia or aversion was the correct explanation for this avoidance. This study merely reflects on the role of airstones in a preference assay. The authors are not intending to cast doubt over the importance of using these devices as air supplies in aquatic facilities.
When housed in pairs, only the provision of substrate elicited preference behaviour while in the group setting, various enrichment elements were preferred to a barren area. This suggests that the physical needs of zebrafish are modulated by their social situation. The effects of small group sizes on zebrafish dominance relationships bring about a cascade of behavioural changes, as already discussed. 12 Group-housed fish showed relatively less dominance behaviour and showed a distinct range of preferences. For example, compartments with artificial plants were preferred by the grouped fish but not by the pairs, as the resulting cover could be vital for small shoals (which a group of eight may arguably constitute) who, with their high visibility and relatively low vigilance, are particularly exposed to predator attacks. 35 In contrast gravel and, to a lesser degree, sand substrate have a more universal appeal as they provide camouflage while also constituting feeding and spawning habitat. Substrate and substrate images could also make the tanks on that side appear deeper to the zebrafish, as demonstrated in another recent study. 36
In our study sexual differences in enrichment preferences were mainly associated with plant type. Sexual dimorphism in habitat choices, linked to vegetation, has been also been reported for the pipefish (Stigmatopora spp.), where males and females preferred different types of seagrass. 37 In that investigation vegetation was primarily linked to camouflage and feeding, and different habitat preferences attributed to higher energetic effort by the females requiring more food intake.
This study shows that zebrafish make choices with regard to their physical environment, preferring structural complexity over barren standard conditions. The results indicate that in a group context zebrafish show clear preferences for substrates and plants over barren conditions. In contrast, when zebrafish are held in pairs, habitat choices are confounded by dominance relationships. With clear preference patterns emerging for specific enrichment elements, the group preferences provide a clearer perspective regarding enrichment choices; this offers insight into how refinements in zebrafish housing may be achieved, not least with the substrate images representing a simple and hygienic pathway for providing environmental enrichment.
Further research could address the question how a range of welfare parameters is influenced by long-term exposure to enrichment when compared with a barren environment, as it is hard to imagine that attitudes on how zebrafish should be housed will be shifted on preference data alone. A recent study investigated behavioural and endocrine responses to enrichment designs consistent with toxicological requirements (vertical glass rods), finding no significant improvement in welfare. 10 A similarly designed study, but with tank designs determined by preference testing, might produce different results.
Footnotes
Funding
Paul Schroeder is funded by the University of Nottingham School of Veterinary Sciences, and the Wellcome Trust. Lynne Sneddon and Iain Young are grateful for a NC3Rs research grant.
Acknowledgements
We are grateful to Gregor Govan at the Institute of Integrative Biology at Liverpool for assistance with constructing the tanks, fish husbandry and advice; Jack Thomson for performing behavioural observations for inter-observer reliability tests; and to Abigail Sunderland, Angela Sims and Sam Barlow for assistance with fish husbandry. We also thank Sergei Maslo for advice on statistical analysis.
