Abstract
Lactating mice in laboratory conditions are thought to be under considerable stress. Dams may manifest this stress through a decrease in milk yield and/or increase in infanticide. The purpose of this study was to examine the effect of access to an environmental enrichment device called the RatLoft on milk yield, circulating serotonin, and pup mortality using both wild-type mice and mice genetically deficient in tryptophan hydroxylase 1 (TPH1−/−; the rate-limiting enzyme in the non-neuronal serotonin synthesis pathway). Presence or absence of the RatLoft did not affect milk yield or circulating serotonin concentrations overall, but serotonin concentrations decreased throughout the 21-day lactation period. Serotonin concentrations were increased in TPH1-deficient mice with access to the RatLoft compared with those without the RatLoft on day 21. Pup mortality tended to decrease for dams with access to the RatLoft as compared to no RatLoft. Within the TPH1−/− groups, dams with access to the RatLoft tended to kill less pups per dam than dams without the RatLoft. These results demonstrate that access to the RatLoft during lactation decreases pup infanticide by dams. This environmental enrichment may be particularly beneficial to TPH1−/− dams that, in addition to decreased infanticide, had increased serotonin concentrations, possibly indicating enhanced well-being. Use of the RatLoft could prove beneficial to researchers working with lactating mouse models to increase the number of pups weaned and positively impact the welfare of the dam.
Standards of care for animal welfare are based on the assumption that laboratory animals experience emotions such as pain or sadness and therefore require guidelines to protect their well-being. 1 Rodent dams undergoing studies post-partum are under particular demand to care for their pups and, like women, may be at risk for developing mood disorders related to stress and depression.2–4 Depression-like behaviors that can manifest from the laboratory environment, such as not feeding or killing pups, adversely affect both the dam and the researcher.
Intimately related to both depression and lactation is serotonin (5-hydroxytryptamine [5-HT]), which regulates mood at a neuronal level and the mammary gland during lactation. 5 5-HT is synthesized from the amino acid L-tryptophan in a two-step process carried out by the rate-limiting enzyme tryptophan hydroxylase-1 (TPH1) in non-neuronal tissues and TPH2 in neuronal tissues.6–8 The inhibition of 5-HT synthesis induces depression-like symptoms, as does the absence of tryptophan, in laboratory animals. 9 In laboratories, stress is thought to be provoked by factors such as lighting, caging, and the separation of a dam from her pups, and may have adverse effects on neuroendocrine systems that affect welfare.3,10
Environmental enrichment (EE) has been shown to decrease depression-like behaviors in rodents.
11
One such form of EE is the RatLoft, in this case the contoured tube loft, which is a hollow metal tube that hangs over the side of the cage, offering a lactating dam respite from the demands of her pups (Figure 1; Research Animal Welfare Equipment, LLC, Madison, WI, USA). Our objective was to determine whether access to the RatLoft affected maternal welfare in laboratory conditions. We hypothesized that the use of the RatLoft during lactation would increase circulating 5-HT levels and decrease depression-like behaviors, resulting in less infanticide and a greater number of pups that can be weaned and used for further studies.
Contoured tube RatLoft in a solid-bottomed shoebox cage. The RatLoft is a hollow metal tube that hangs over the side of the cage, offering a lactating dam respite from the demands of its pups, with access to food and water. It was used as a form of environmental enrichment in this study.
Materials and methods
Animals
Eight-week-old C57B6/J female mice were obtained from the breeding colony of Dr Laura Hernandez (University of Wisconsin–Madison IACUC approval number: A01473). Of the dams used in the study, 11 were wild-type C57B6/J mice and 13 had a C57B6/J strain background, but with an additional mutation in the TPH1 gene (Tph1tm1Bdr; tryptophan hydroxylase 1; targeted mutation 1, Michael Bader; MGI: 2450301). These TPH1-deficient mice were a gift from Dr Nelson Horseman at the University of Cincinnati and were originally described by Walther et al. in 2003. 7 This mutant line has been utilized in studies targeted at the role of 5-HT in mammary gland function.12,13 The TPH1-deficient mice express normal amounts of 5-HT in neuronal serotonergic systems, but lack 5-HT in the periphery except for the duodenum, where 5-HT levels are about 4% of wild type, likely due to serotonergic neurons in the small intestine. 14 All mice were individually housed in standard shoebox cages (29 cm × 18 cm × 12 cm) with autoclaved Aspen kiln-dried hardwood bedding shavings, (American Wood Fibers, Columbia, MD, USA), changed weekly, in the Animal Sciences vivarium at the University of Wisconsin–Madison. Mice were given ad libitum access to autoclaved, acidified water and pelleted, autoclaved, 2019 Teklad Global 19% Protein Extruded Rodent Diet with 9% fat content (Harlan Laboratories, Madison, WI, USA). Mice were maintained at a room temperature of 25℃, constant humidity, with a 12 h light–dark cycle with the light cycle beginning at 06:00 h. All experiments were performed under protocols approved by the College of Agricultural and Life Sciences Animal Care and Use Committee at the University of Wisconsin–Madison.
Pregnant mice deficient for TPH1−/− (n = 13) and wild-type mice (WT, n = 11) were randomly assigned to the loft (L; n = 7 for TPH1−/− and n = 6 for WT, respectively) and to the no loft (NL; n = 6 for TPH1−/− and n = 5 for WT, respectively) treatment groups. The RatLoft (Research Animal Welfare Equipment, LLC) is a stainless steel tube that hangs from the side of the cage and provides access to food (Figure 1). It is elevated off the cage floor enough so that the pups are not large enough to access the RatLoft until late lactation. RatLofts were put in cages at the time of mating and remained in the cage through day (d) 21 of lactation. Males were removed from the cages at the time of parturition and litter sizes were not standardized.
Sample collection
Approximately 100 µL of blood was collected into 500 µL Capiject micro-collection tubes (Terumo, Somerset, NJ, USA) from the dam’s maxillary vein on d1, d9, and d21 of lactation. Blood was centrifuged at 3000 ×
Sample analysis
5-HT assay
Serum 5-HT concentrations were measured by enzyme-linked immunosorbent assay (ELISA) using a Serotonin EIA kit (Enzo Life Sciences, Farmingdale, NY, USA) according to the manufacturer’s instructions. The inter-assay coefficient of variation (CV) for the WT mice was 16.4% and the inter-assay CV for the TPH1-ablated mice was 15.3%.
Statistical analyses
All data were analyzed using Prism GraphPad software (version 6.00 for Macintosh; GraphPad Software Inc, San Diego, CA, USA) using the mouse as the experimental unit. Two mice were not included in the statistical analysis due to aberrant physiological and maternal behaviors throughout lactation. One additional mouse was not included in the study because it failed to become pregnant. This left a final total of 24 mice in the treatment groups: WT (n = 6 for L and n = 5 for NL) and TPH1−/− (n = 7 for L and n = 6 for NL). Since litter sizes were not standardized at the time of parturition, MY and pup weight were standardized on an individual pup basis by dividing the overall litter weight and MY by the number of pups the dam had on any given day. Because some dams induced mortality, litter size was often variable by day, which was accounted for in all calculations by dividing the appropriate parameters, such as MY and pup weight, by the number of pups on any given day of lactation. All analyses were performed using a two-way analysis of variance (ANOVA) or the Mann–Whitney unpaired t-test for non-normally distributed data. Differences between means were considered significant with a P ≤ 0.05 and tending towards significant with a P ≤ 0.10. All values are reported as a least squared mean plus or minus the standard error of the mean (SEM).
Results
Mean and standard error as well as statistical significance (set at P < 0.05) of pup mortality, serotonin serum concentration, body weight per pup, and milk yield per pup within WT and TPH1−/− groups for L versus NL dams.
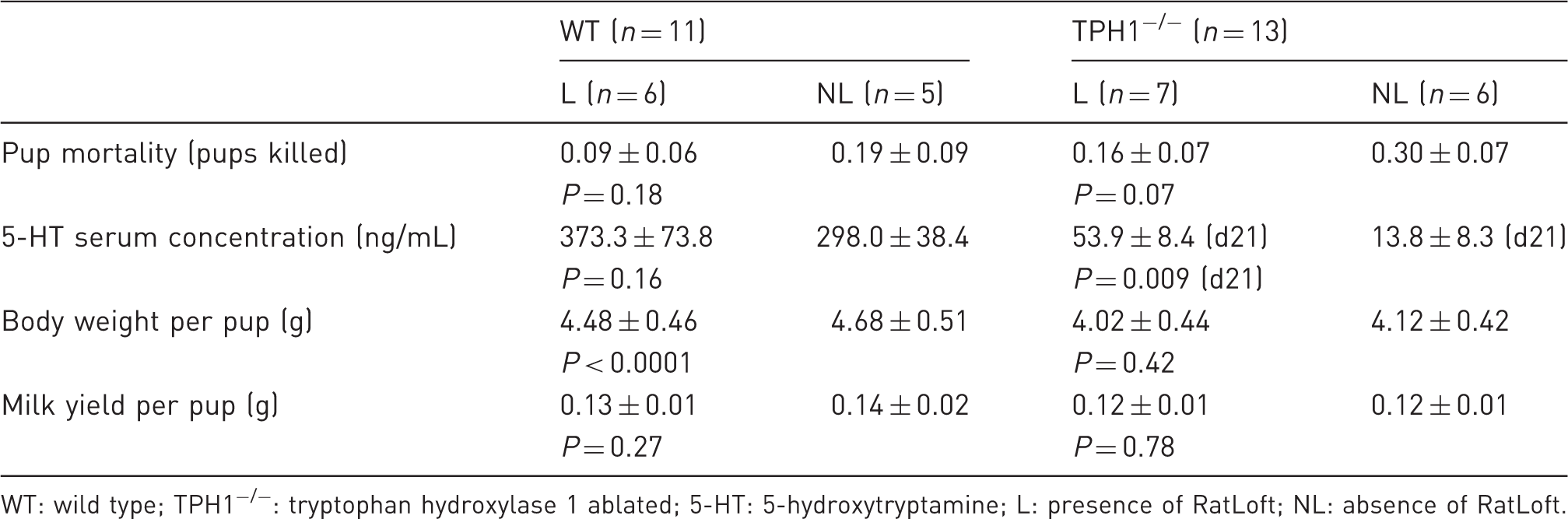
WT: wild type; TPH1−/−: tryptophan hydroxylase 1 ablated; 5-HT: 5-hydroxytryptamine; L: presence of RatLoft; NL: absence of RatLoft.
Mean and standard error as well as statistical significance (set at P < 0.05) of pup mortality, serotonin serum concentration, body weight per pup, and milk yield per pup between WT versus TPH1−/− dams and L versus NL dams.
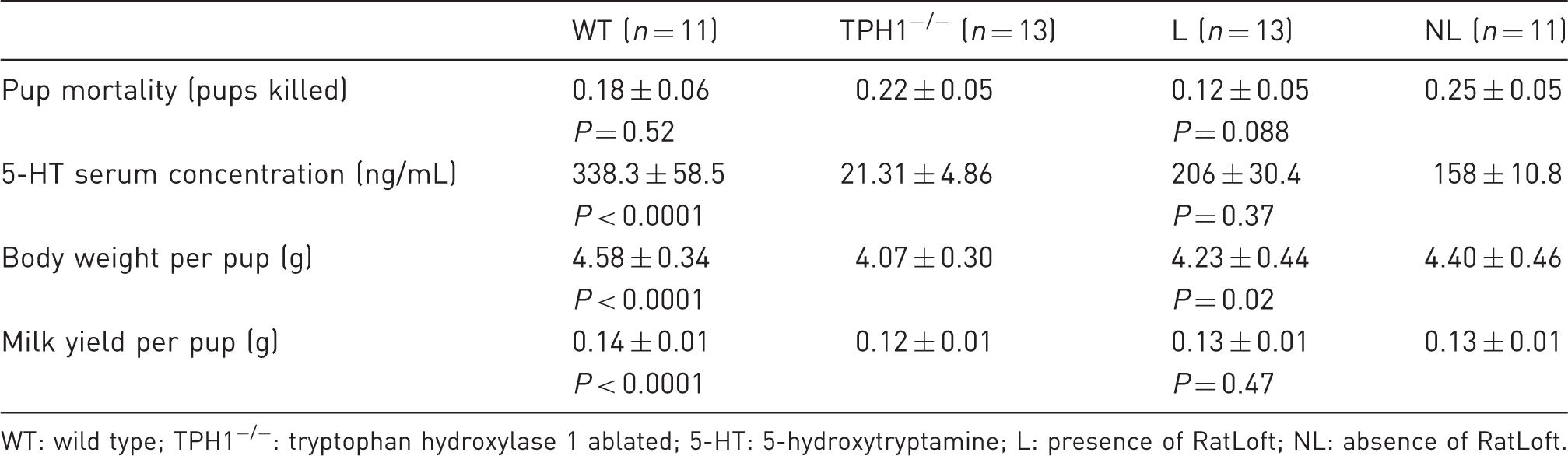
WT: wild type; TPH1−/−: tryptophan hydroxylase 1 ablated; 5-HT: 5-hydroxytryptamine; L: presence of RatLoft; NL: absence of RatLoft.
Mortality
Within the TPH1−/− group, dams with access to the RatLoft (L) tended to kill less pups per dam than those without access to the RatLoft (NL) (P = 0.07; 0.16 ± 0.07 and 0.30 ± 0.07 pups killed per dam for L and NL, respectively; Table 1). The presence or absence of the loft did not affect mortality rate of mice alone within the WT group (P = 0.18; 0.09 ± 0.06 and 0.19 ± 0.09 pups killed per dam for L and NL, respectively; Table 1). Overall, however, all dams with access to the loft tended to kill fewer pups than all dams without access to the loft (P = 0.088; 0.12 ± 0.05 and 0.25 ± 0.05 pups killed per dam for L and NL, respectively) (Figure 2; Table 2). Disregarding presence or absence of the loft, there were no differences in mortality rates of TPH1−/− dams compared with WT dams (P = 0.52; 0.18 ± 0.06 and 0.22 ± 0.05 pups killed per dam for WT and TPH1−/−, respectively; Table 2).
Pups killed per dam in WT (n = 11 dams) versus TPH1−/− (n = 13) dams with the presence or absence of the RatLoft. Dams with the TPH1 ablation were unable to produce non-neuronal serotonin and were randomly assigned a cage with a loft (n = 7) or no loft (n = 6). WT mice were also randomly assigned a loft (n = 6) or no loft (n = 5). Pup mortality was then counted throughout the experiment. Within the TPH1−/− group, L mice tended to kill less pups per dam than NL mice (P = 0.07) (Table 1). Overall, L mice tended to kill less pups per dam than NL mice (P = 0.088) (Table 2). WT: wild type; TPH1−/−: tryptophan hydroxylase 1 ablated; L: presence of RatLoft; NL: absence of RatLoft.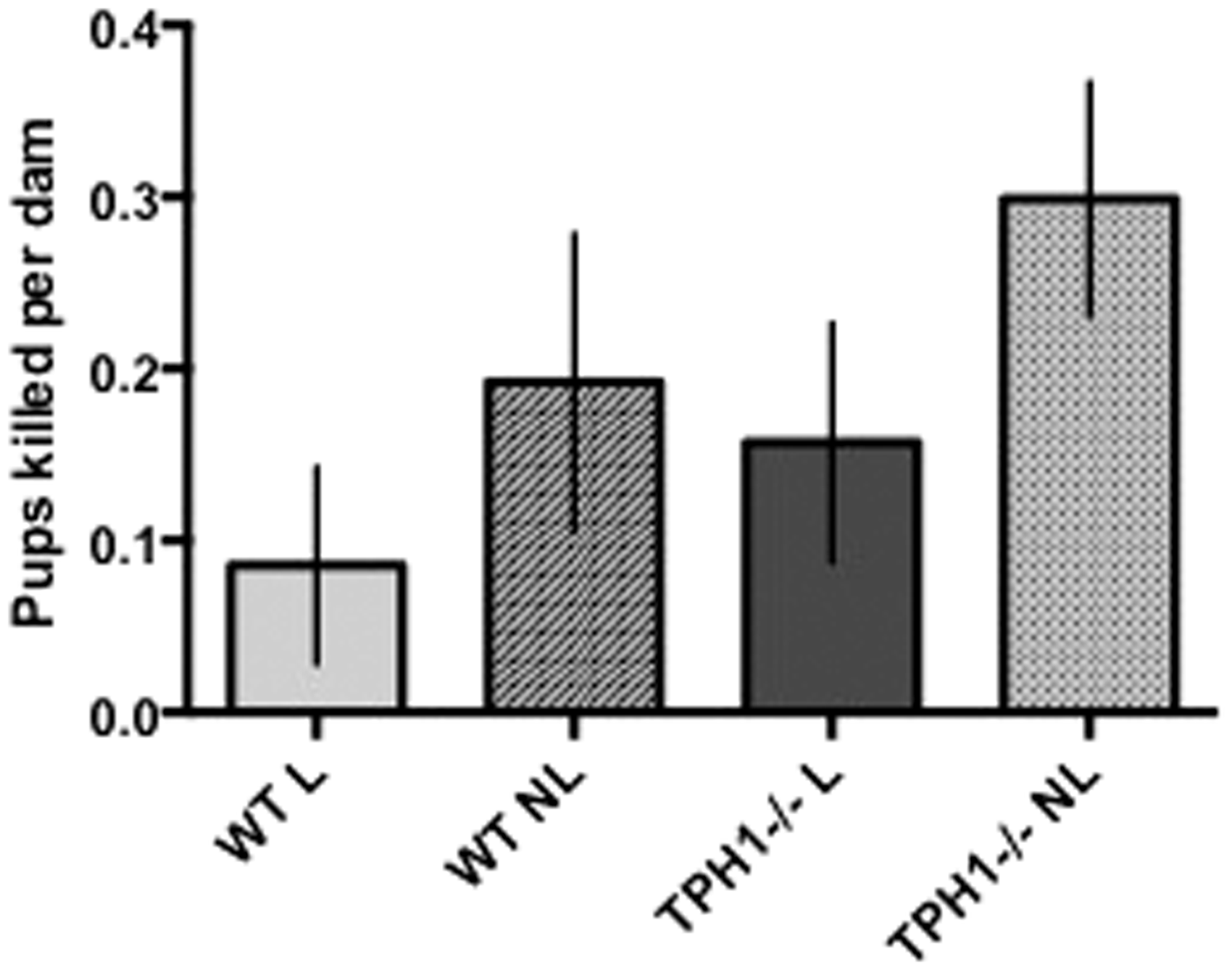
5-HT serum concentration
The presence or absence of the loft overall did not affect serum 5-HT concentrations (P = 0.37; 206 ± 30.4 and 158 ± 10.8 ng/mL for L and NL, respectively; Table 2) and there was no interaction between 5-HT concentration in L versus NL and time (P = 0.82). WT mice had more circulating 5-HT than TPH1−/− mice (P < 0.0001; 338.3 ± 58.5 and 21.31 ± 4.86 ng/mL for WT and TPH1−/−, respectively) (Table 2; Figure 3). Within the TPH1−/− group, dams with access to the loft had significantly higher 5-HT serum concentrations than NL mice on d21 (P = 0.009; 53.9 ± 8.4 and 13.8 ± 8.3 ng/mL for L and NL, respectively) (Table 1; Figure 3). Within the WT group, the presence or absence of the loft did not affect 5-HT serum concentrations (P = 0.16; 373.3 ± 73.8 and 298.0 ± 38.4 ng/mL for L and NL, respectively) and there was no interaction between presence or absence of the loft and time (P = 0.65) (Table 1). There was a time effect, with overall 5-HT concentrations decreasing in the serum throughout lactation with the exception of d21 in TPH1−/− mice (P = 0.036) and there was an interaction between the genotype of the dam and the day of lactation with respect to 5-HT concentration (P = 0.014) (Figure 3).
Serum serotonin (5-HT) concentrations in WT (n = 11 dams) versus TPH1−/− (n = 13) dams with the presence or absence of the RatLoft. Dams with the TPH1 ablation were unable to produce non-neuronal serotonin and were randomly assigned a cage with a loft (n = 7) or no loft (n = 6). WT mice were also randomly assigned a loft (n = 6) or no loft (n = 5). Serum was collected from the maxillary vein on days 1, 9, and 21 of lactation and 5-HT concentrations were determined with an ELISA. WT dams had higher 5-HT concentrations than TPH1−/− dams (P < 0.0001) (Table 2). Within the TPH1−/− group, dams with access to the loft had significantly higher 5-HT serum concentrations than NL mice on d21 (P = 0.009) (Table 1). 5-HT concentrations decreased overall throughout lactation with the exception of d21 in TPH1−/− mice (P = 0.036) and there was an interaction between genotype (WT and TPH1−/−) and time (P = 0.014). WT: wild type; TPH1−/−: tryptophan hydroxylase 1 ablated; 5-HT: 5-hydroxytryptamine; ELISA: enzyme-linked immunosorbent assay; L: presence of RatLoft; NL: absence of RatLoft.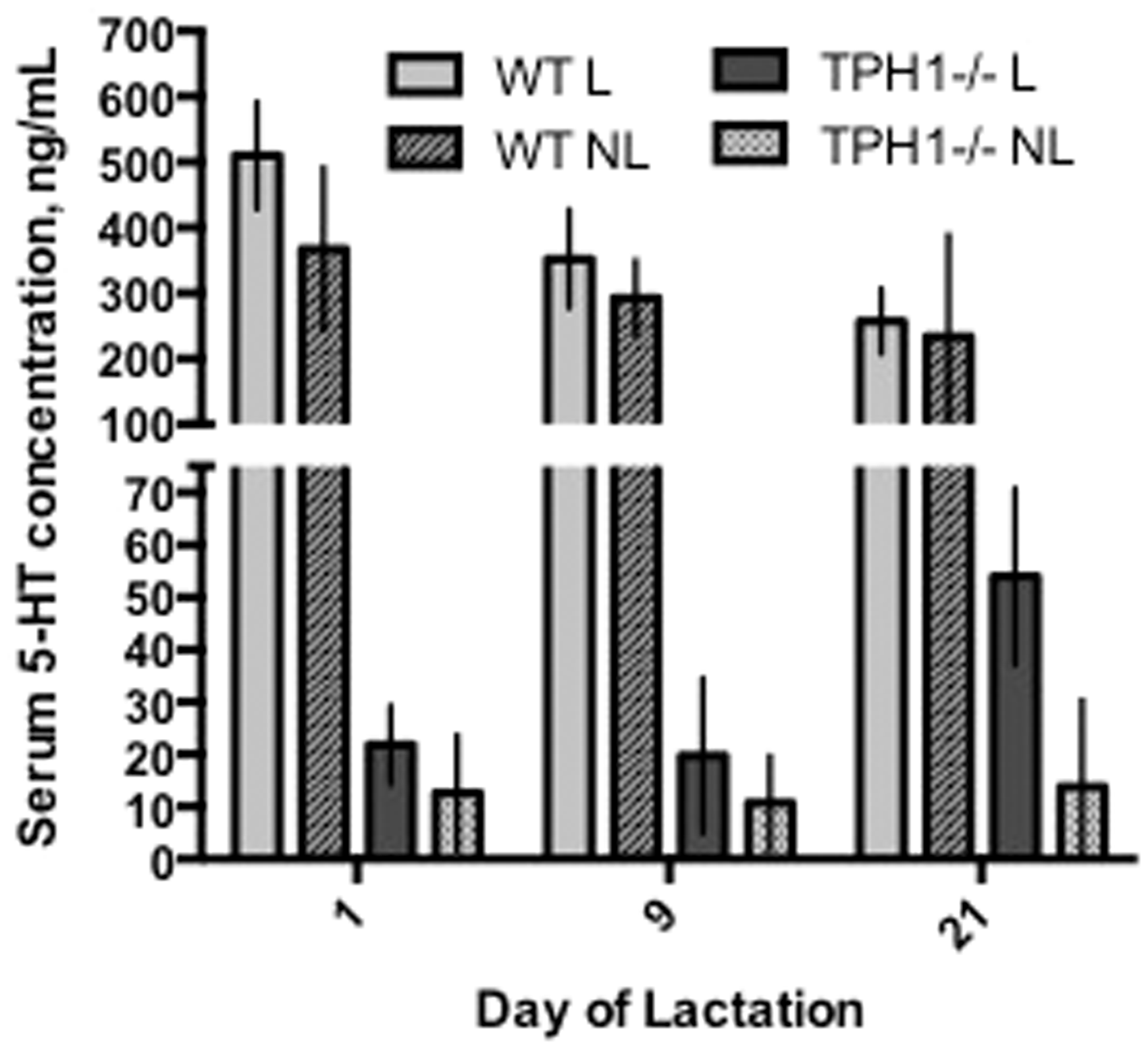
Animal body weight
The BW per pup of WT dams was greater than the BW per pup of TPH1−/− dams throughout lactation (P < 0.0001; 4.58 ± 0.34 and 4.07 ± 0.30 g for WT and TPH1−/−, respectively) (Table 2; Figure 4a). Overall, pups whose mothers had access to the loft weighed less than pups whose dams did not have access to the loft (P = 0.02; 4.23 ± 0.44 and 4.40 ± 0.46 g for L and NL, respectively) (Table 2; Figure 4b). Following a similar trend, WT dams with access to the loft had smaller pups than WT dams that did not have access to the loft (P < 0.0001; 4.48 ± 0.46 and 4.68 ± 0.51 g for L and NL, respectively) (Table 1; Figure 4c). Within the TPH1−/− group, however, BW per pup was not affected by the presence or absence of the loft (P = 0.42; 4.02 ± 0.44 and 4.12 ± 0.42 g for L and NL, respectively) (Table 1). BW per pup increased throughout lactation for all mice (P < 0.0001). There was no interaction in any of the groups between BW per pup and day of lactation. Average dam weight was not affected by the genotype or by the absence or presence of the loft on any given day (P = 0.14), although there was an overall time effect with dams gaining weight throughout lactation (P < 0.0001).
BW per pup of: (a) WT dams’ pups (n = 11 dams) versus TPH1−/− dams’ pups (n = 13 dams); (b) all dams’ pups with access to the RatLoft (n = 13 dams) versus all dams’ pups without access to the RatLoft (n = 11 dams); (c) WT dams’ pups with access to the RatLoft (n = 6 dams) versus WT dams’ pups without access to the RatLoft (n = 5 dams). Dams with the TPH1 ablation were unable to produce non-neuronal serotonin and were randomly assigned a cage with a loft (n = 7) or no loft (n = 6). WT mice were also randomly assigned a loft (n = 6) or no loft (n = 5). BWs were then collected daily throughout the entire 21-day lactation period and standardized to per-pup by dividing the total BW of the litter by the number of pups in each dam’s litter. Throughout lactation, pups of WT dams weighed more than pups of TPH1−/− dams (P < 0.0001); pups of L mice weighed less than pups of NL mice overall (P = 0.02); and pups of WT L dams weighed less than pups of WT NL dams (P < 0.0001) (Tables 1 and 2). BW: body weight; WT: wild type; TPH1−/−: tryptophan hydroxylase 1 ablated; L: presence of RatLoft; NL: absence of RatLoft.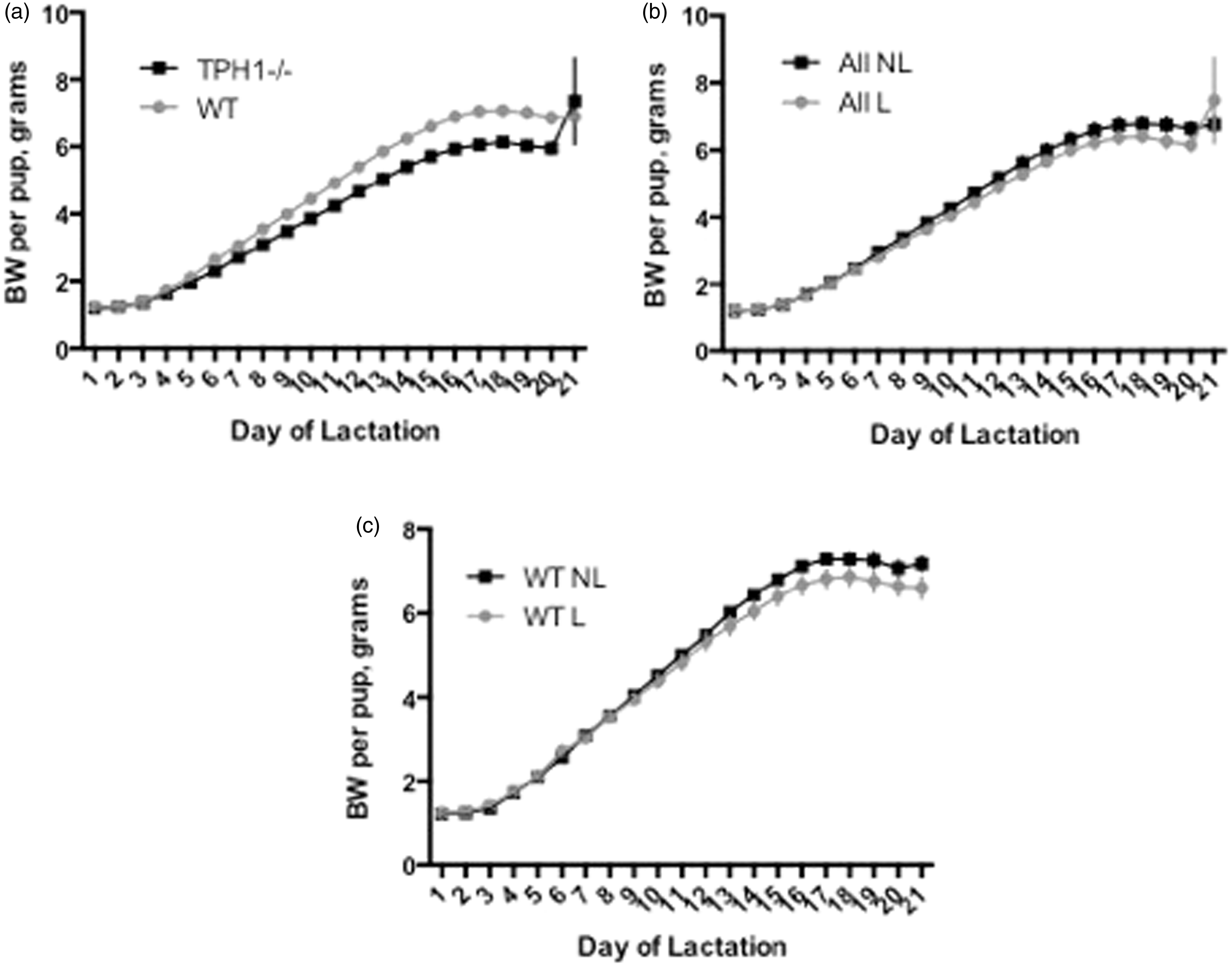
Milk yield
The MY per pup was not affected by the presence or absence of the loft (P = 0.47; 0.13 ± 0.01 and 0.13 ± 0.01 g/pup for L and NL, respectively) (Table 2). Overall, TPH1−/− dams produced significantly less milk than WT dams throughout the lactation period (P < 0.0001) (Figure 5). Additionally, the average daily MY per pup was greater for WT dams than for TPH1−/− dams (0.14 ± 0.01 and 0.12 ± 0.01 g/pup for WT and TPH1−/−, respectively) (Table 2). Within the WT or TPH1−/− groups respectively, the presence or absence of the loft did not affect MY (P = 0.27 for WT dams; 0.13 ± 0.01 and 0.14 ± 0.02 g/pup for L and NL, respectively; P = 0.78 for TPH1−/− dams; 0.12 ± 0.01 and 0.12 ± 0.01 for L and NL, respectively) (Table 1). There was no interaction in any of the groups between MY per pup and day of lactation. There was a time effect for all mice, with MY peaking at d13 in WT dams and d14 in TPH1−/− dams (P < 0.0001) (0.22 ± 0.02 and 0.18 ± 0.02 g on d13 in WT dams and d14 in TPH1−/− dams, respectively) (Figure 5).
Milk yield per pup of WT (n = 11 dams) versus TPH1−/− (n = 13) dams. Dams with the TPH1 ablation were unable to produce non-neuronal serotonin. Milk yields were collected using the weigh–suckle–weigh method daily throughout 21 days of lactation and was standardized to per-pup by dividing the total milk yield by the number of pups in each dam’s litter. WT dams had overall higher milk yields per pup than TPH1−/− dams (P < 0.0001) (Table 2). WT: wild type; TPH1−/−: tryptophan hydroxylase 1 ablated.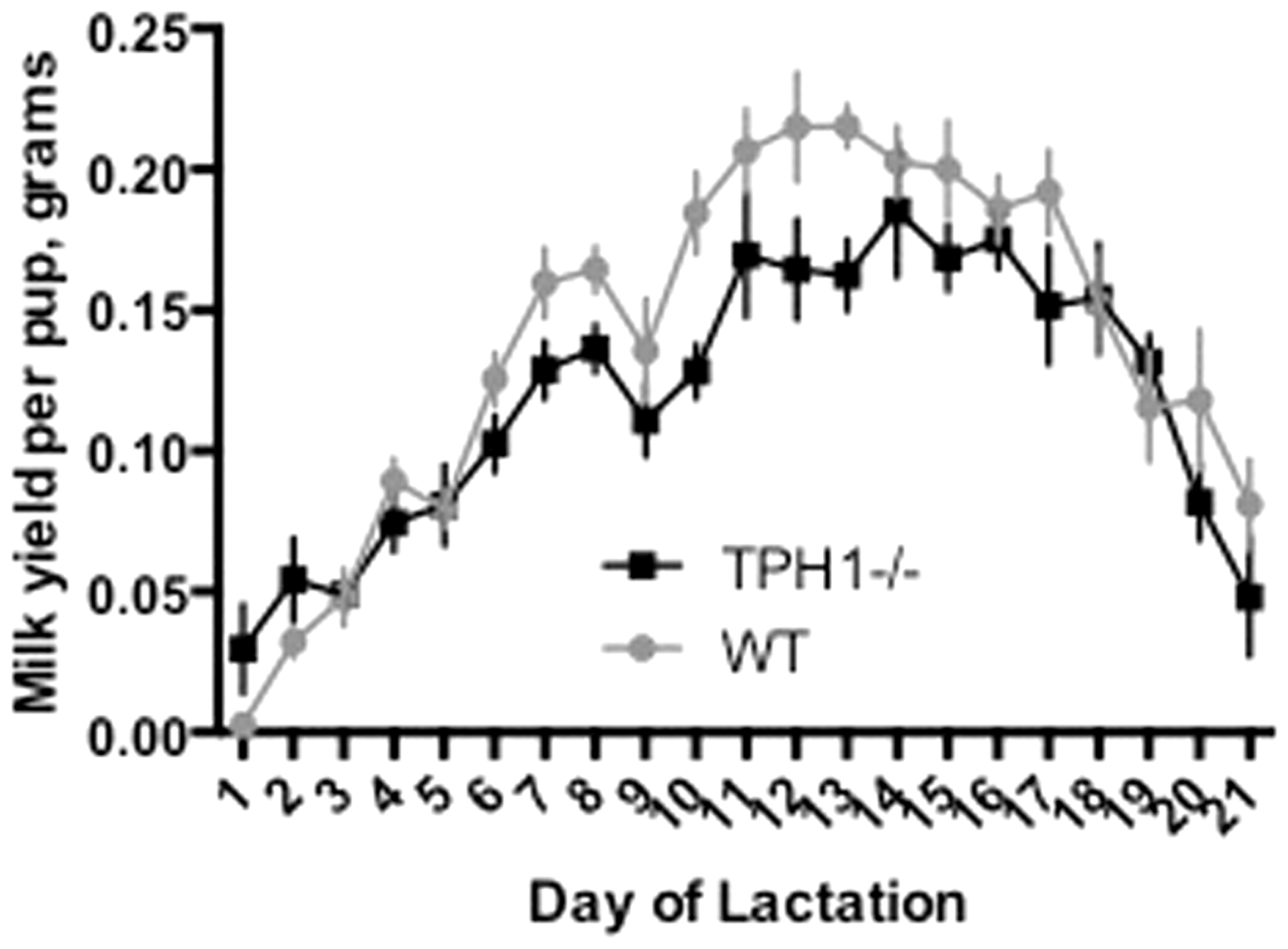
Discussion
The welfare of laboratory animals is a fundamental component of any animal care and use program. Selection of an EE device depends on the animal, ability to autoclave the device, and durability of the device, with the ultimate end goal of reducing the animal’s stress. 15 Stress can be provoked in a laboratory setting by factors such as caging, transport, noise, lighting, routine cleaning, and social disruption and the physiological and behavioral adaptations to these stressors can have negative effects on the animal and hence the experiment. 3 Specific aspects of this study, and lactation studies in general, such as use of the weigh–suckle–weigh method, which involves separation of the dam from its pups, may be particularly stressful. EE devices are thought to improve the quality of animals’ lives by providing objects that allow structure and control, thereby engaging them with their otherwise monotonous environment.3,16,17 The current study utilized a contoured tube loft, which is a hollow metal tube that hangs over the side of the cage. Food is accessible from the loft and it is only large enough to fit one adult-sized mouse, precluding pups even when they are large enough to climb inside the loft at about d16 of lactation.
The loft is thought to be beneficial to nursing rodent dams, providing respite from the demands of lactation in an otherwise relatively small cage. Early work by Bateman (1957) showed that nursing dams spend approximately 70% of their day on the nest suckling pups, and that lactating and non-lactating dams spend comparable time eating. 18 Therefore, although lactation is physiologically demanding in general, it is thought to be more so when in a confined cage environment that may provoke stress. Additional stress is provoked through handling by researchers. For example, anxiety is invoked in the dam when pups and the dam are isolated in lactation studies using the weigh–suckle–weigh method, one of the primary means of determining MY.19,20 In addition to the ethical concerns of provoking this separation anxiety, researchers must be cognizant of the potential effects on their data. The weigh–suckle–weigh method is the definitive means of determining MY in dams, but as a result of this and similar procedures, dams may undertake a variety of harmful behaviors, including killing several or all of their pups by not feeding them (indicated by low MYs) or by not keeping them warm, thus provoking hypothermia.21,22
Some behaviors that result in infanticide may be adaptive. Dams have been shown to reduce their litter size with food restriction, for example, in a presumed response to the inability of the mother to produce sufficient milk. 23 Importantly, there is debate as to whether dams are actually practicing infanticide on entire litters, as one study has found no evidence of infanticide in dams that lost their entire litter, or whether some pups are simply stillborn and are only later cannibalized by the mother. 21 In the present study, however, a reduced number of pups from the first day of lactation to the next negates this possibility, as pups were handled daily beginning on the day of birth and any dead pups were removed from the cage immediately. In cases like these, where the mother is likely to be responding to environmental stressors, EE has been shown to diminish negative behaviors that may lead to infanticide. 11 In fact, Whitaker et al. demonstrated that, when compared with pups from non-enriched cages, B57BL/6Tac pups from enriched cages weighed more and had an increased survival to weaning age, paralleling the results presented in this study. 24
Various factors other than EE have been shown to improve pup survival in laboratory settings. Strain and parity may have particularly important roles in determining pup survival, as several studies have found that C57BL/6 mice have high mortality rates when compared with both DBA/2J and BALB/c strains, despite C57BL/6 mice showing higher engagement in maternal behaviors.22,25 Indeed, even Jackson Laboratory has reported that C57BL/6J females frequently lose their first litter, even though mice with this genetic background are commonly used in breeding colonies. 26 Although parity was not specifically controlled for in this study, the large mortality rates previously reported in C57BL/6 strains (as high as 32% of complete litter loss and 36% of individual pup loss) have demonstrated the acute need to develop methods for supporting the well-being of lactating dams used in research.22,25
In addition to EE, serotonin (5-HT) concentrations may contribute to the well-being of laboratory animals by regulating mood and acting as homeostatic regulators of the mammary gland, vitally influencing lactating mammals.5,8 Serotonin is synthesized in a two-step process from the amino acid L-tryptophan, using the rate-limiting enzyme TPH1 in non-neuronal tissues and TPH2 exclusively in neuronal tissues. 12 The genetically-modified mouse model used in this study had a non-functional TPH1 gene (TPH1−/−) and, therefore, would theoretically only produce neuronal 5-HT. Neuronal 5-HT is produced both in the central nervous system and from enteric neural cells in the gut.14,27 While it is unlikely that 5-HT can cross the blood–brain barrier, its precursor (5-hydroxytryptophan or 5-HTP) can cross and enter the circulation. Our TPH1-ablated mice, therefore, could have minimal levels of 5-HT in their peripheral circulation either from neuronal enteric cells or as a result of neuronal 5-HTP crossing the blood–brain barrier and completing the 5-HT synthesis pathway in various tissues, including the mammary gland.
The inhibition of neuronal 5-HT synthesis induces depression-like behaviors, as does the absence of tryptophan, in laboratory animals. 9 Based on the neurochemistry of 5-HT, selective serotonin reuptake inhibitors (SSRIs) have proven to be particularly effective in the treatment of depression and post-partum depression (PPD), demonstrating the association between 5-HT and mood in both humans and laboratory animals.28–30 Brenes et al. have recently found that EE may augment neuronal 5-HT production in laboratory animals, further reducing depression-like behaviors in dams. 19 Our laboratory has observed that TPH1-deficient mice have significantly lower pup weights and MYs than WT mice. For these reasons, we hypothesized that circulating 5-HT levels would be elevated in TPH1-deficient mice with access to the loft relative to WT mice. Additionally, we expected depression-like behaviors such as infanticide to be reduced in mice with access to the RatLoft.
Access to the RatLoft increased circulating 5-HT levels in mice deficient for TPH1. Since TPH1 is the rate-limiting enzyme for non-neuronal 5-HT, we are confident that increases in circulating 5-HT levels had to be produced via neuronal pathways. Given the correlation between increased 5-HT and enhanced mood and behavior, the presence of elevated 5-HT levels in TPH1−/− dams given access to the loft suggests that these dams had improved moods. Dams that had access to the loft killed fewer of their pups, which improves maternal welfare and results in increased productivity in breeding colonies. Pup mortality is a direct barrier to building up breeding colonies, especially in genetically-modified models where maternal behavior may be unintentionally affected. 21 This is an especially important point in the present study, as the TPH1−/− dams may be responding more acutely to modulations in mood stimulated by EE given the known roles of 5-HT in mediating maternal behavior. Given that the study employed a relatively small sample size, and the parameters, such as MY using the weigh–suckle–weigh method, were stressful in and of themselves, it is possible that this particular genetic background responded in a more robust way to the EE than dams of different strains might have experienced. Importantly, given the overall decreased pup mortality demonstrated in the present study with the use of EE, the RatLoft can and should be considered a useful tool in assuring that breeding colonies will be more efficiently and effectively maintained.
Studies have shown that stressors during pregnancy produce depression-like behaviors. 31 Several of our dams are concurrently pregnant and lactating, typical of breeding colonies, and therefore the results of depression-related parameters such as 5-HT may have been affected. The BW of pups may also be confounded by time spent away from the dam and by litter sizes. While attempts were made to standardize per pup, milk production is typically proportional to litter size and therefore, smaller litters would result in decreased MYs. Time away from the dam when pups were in the same cage was not specifically measured, as this would require 24 h monitoring, but growth data may also be confounded by this factor. Additionally, at about d15 of lactation, pups have their eyes open and have the ability to wander freely around the cage. Our results demonstrated that around this day, WT pups whose mothers had access to the loft weighed less than their NL counterparts (Figure 5). This is possibly because the pups were climbing up into the RatLoft and self-weaning on the dam’s food, resulting in lower BWs than they would have if they were suckling alone. This hypothesis is supported by work demonstrating that pups begin to nibble solid food around d15 and energy expenditures are lessened around this time on the lactating dam. 32 Further research would be required to determine the direct effects of the RatLoft on energy-related expenditures such as growth and MY.
Further research such as the effect of the RatLoft on fertility, litter size, weaning weights, and parity should be considered. Additionally, given the aforementioned impact of genetic strain on pup mortality, it would be interesting to employ this form of EE in rodent models other than C57BL/6 mice.22,25 This study shows that use of the RatLoft reduces pup mortality in lactating dams, providing benefits both in terms of their welfare as laboratory animals and to the laboratory researchers. The RatLoft should therefore be considered in studies involving mouse colonies based on the demonstrated improvement in both colony yield and dam welfare.
Footnotes
Acknowledgements
The authors would like to thank the RARC Animal Care Committee for daily care and maintenance of all mice used in the study. We also appreciate Dr Nelson Horseman for the gift of his mice to begin our colony and Peter Crump for his help with the statistical analysis. Additionally, we would like to acknowledge the Cargill–Benevenga Research Stipend for the financial support of the authors for this project.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
