Abstract
Expansion of physiological knowledge increasingly requires examination of processes in the normal, conscious state. The current study describes a novel approach combining surgical implantation of radio-telemeters with vascular access ports (VAPs) to allow repeated hemodynamic and pharmacological measures in conscious rats. Dual implantation was conducted on 16-week-old male lean and obese Zucker rats. Continued viability one month after surgery was observed in 67% of lean and 44% of obese animals, giving an overall 54% completion rate. Over the five-week measurement period, reliable and reproducible basal mean arterial pressure and heart rate measures were observed. VAP patency and receptor-independent vascular reactivity were confirmed by consistent hemodynamic responses to sodium nitroprusside (6.25 µg/kg). Acutely, minimal hemodynamic responses to repeated bolus administration of 0.2 mL saline indicated no significant effect of increased blood volume or administration stress, making repeated acute measures viable. Similarly, repeated administration of the β-adrenoceptor agonist dobutamine (30 µg/kg) at 10 min intervals resulted in reproducible hemodynamic changes in both lean and obese animals. Therefore, our study demonstrates that this new approach is viable for the acute and chronic assessment of hemodynamic and pharmacological responses in both lean and obese conscious rats. This technique reduces the demand for animal numbers and allows hemodynamic measures with minimal disruption to animals’ welfare, while providing reliable and reproducible results over several weeks. In conclusion, dual implantation of a radio-telemeter and VAP introduces a valuable technique for undertaking comprehensive studies involving repeated pharmacological tests in conscious animals to address important physiological questions.
Scientific research is increasingly searching for new approaches to examine biological systems under normal, conscious physiological conditions. This shift towards conscious measures is driven by the need for data that are highly physiologically and clinically relevant, which creates a demand for developing new techniques of experimentation in conscious animals.
Hemodynamic measures are particularly prone to confounding factors such as anesthetics and experimental stress. Conscious hemodynamic data have for some time been obtained indirectly via the tail-cuff technique. 1 This method is non-invasive and relatively inexpensive making it particularly useful for multiple measures in individual animals. However, tail-cuff measures are significantly affected by the need for physical restraint of the animal, the stress of which leads to higher blood pressure and heart rate (HR) recordings in restrained versus unrestrained rats.2,3 In addition, restraint stress has disparate hemodynamic effects on hypertensive versus control rats and therefore may also affect experimental conclusions in disease models.4,5 Alternatively, hemodynamic measures have been obtained invasively through externally tethered arterial cannulae, and although the animals are conscious and can move within their cage; this has also been shown to markedly augment HR and blood pressure.6,7
Telemetry has been used for hemodynamic measures in conscious rats for over two decades.7,8 Radio-telemeters consist of a pressure sensing catheter and implantable transmitter device, making them particularly advantageous for obtaining hemodynamic measures in conscious, unrestrained rats. 1 Telemeters have previously been utilized for chronic monitoring of hemodynamic changes such as circadian rhythms or long-term pharmacological effects.9,10 Chronic pharmacological administration has generally been achieved via mini osmotic pumps.9,11 However, accurate repeated assessments of the acute effects of pharmacological agents require ongoing intravenous access.
Acute pharmacological testing of hemodynamic responses in tandem with radio-telemetry has previously been achieved through oral administration,7,12 repeated cannulation,9,11 intraperitoneal injection13,14 or commonly via indwelling intravenous catheters.15–17 While the latter provides the vascular access required for accurate assessment of acute pharmacological responses, open catheters increase the risk of infection and thus are generally only used for short-term investigations requiring patency for only a few days.15,18–20 This style of catheter also requires the animal to be tethered for the length of the experimental period,15,20,21 maintaining a degree of restraint. Although mild, as stated above, this increases stress on the animal, and affects quality of life and introduces a confounding stress factor to the hemodynamic measures.6,7
To overcome the restraint issues, fully implantable vascular access ports (VAPs) allow experiments to be undertaken over weeks or months, while not inhibiting the animals’ normal behavior.22,23 To date, VAPs have primarily been used for blood sampling and pharmacological testing in larger animal species.23–25 However, VAPs have also been used to allow repeated conscious pharmacological evaluations in rats.22,26,27 Collection of blood samples via a VAP has been shown to have limited side-effects and exert minimal stress.22,28
Various disease models have been shown to have different responses to surgery and various stressors compared with control animals. Obese patients and animals exhibit prolonged post-surgical recovery and a higher incidence of infection.16,29 Additionally, as with the hypertensive models mentioned above, obesity is associated with augmented hemodynamic responses to multiple stressors.30–32 Therefore, it is important to validate both the instrumentation and experimental procedures of new techniques in target disease states.
Thus, in the present study we aimed to assess a novel approach combining radio-telemeters and VAPs to allow repeated hemodynamic and pharmacological measures in conscious rats. While disease models can introduce unique difficulties, we also aimed to assess whether or not our new approach is viable in both lean and obese animals. Our results showed that the dual implantation of a radio-telemeter and VAP introduces a valuable technique, which is able to reduce the demand for animal numbers and allows repeated pharmacological and hemodynamic measures to be undertaken with minimal disruption to the animal, while providing reliable and replicable results over several weeks. This emerging technique will therefore allow measures to be obtained in conscious animals to address important scientific questions, which will greatly enhance investigation of biological processes under repeated physiological and pathophysiological conditions.
Materials and methods
Animals
All procedures were approved by the University of Otago Animal Ethics Committee and were conducted in accordance with the New Zealand Animal Welfare Act (1999). Zucker obese rats have a homozygous missense mutation in the leptin receptor gene (fa/fa) leading to impaired satiety signaling and hyperphagia.33–35 Male lean and obese Zucker rats (Crl:ZUC–Leprfa, n = 28; Charles River Laboratories, Wilmington, MA, USA) were housed at 20 ± 1℃ under a 12 h light–dark cycle and provided with chow (Rat and Mouse Cubes; Specialty Feeds, Glen Forrest, WA, Australia) and water ad libitum. Animals were gentled for approximately 10 min daily for one week prior to surgery to minimize the discomfort associated with the experimental manipulations and reduce data variability.
At the completion of the experimental procedures, or at any point during the study they met the defined humane endpoints (including weight loss greater than 10% in 24 h or 20% of the total; lack of hindlimb function; malaise), the animals were euthanized with sodium pentobarbitone (80 mg/kg intravenously; Provet Pty Ltd, Auckland, NZ).
Surgical procedures
Dual implantation of a radio-telemeter and VAP was performed on 16-week-old animals (Figure 1). Anesthesia was induced with 5% isoflurane (Attane; Minrad Inc, Bethlehem, PA, USA) and maintained at 2–2.5% isoflurane in 1 L/min oxygen to ensure complete loss of pedal withdrawal response. Body temperature was maintained using a heating blanket. To minimize corneal desiccation, the eyes were lubricated (Tricin; Jurox Pty Ltd, Rutherford, NSW, Australia). The animals were provided with preoperative analgesia and antibiotic coverage subcutaneously, using the non-steroidal anti-inflammatory drug carprofen (5 mg/kg Carprieve; Norbrook, Newry, Northern Ireland, UK) and combined trimethoprim and sulphamethazine (30 mg/kg Amphoprim; Virbac, Carros, France), respectively. Surgical sites were prepared by the removal of fur with clippers.
Surgical schematic of dual implantation. The body of the radio-telemetric device is positioned in the abdomen, with the pressure sensor tip in the aorta between the renal arteries and the iliac bifurcation. The vascular access port (VAP) cannula is inserted into the femoral vein, connected to the VAP reservoir positioned subcutaneously between the scapulae.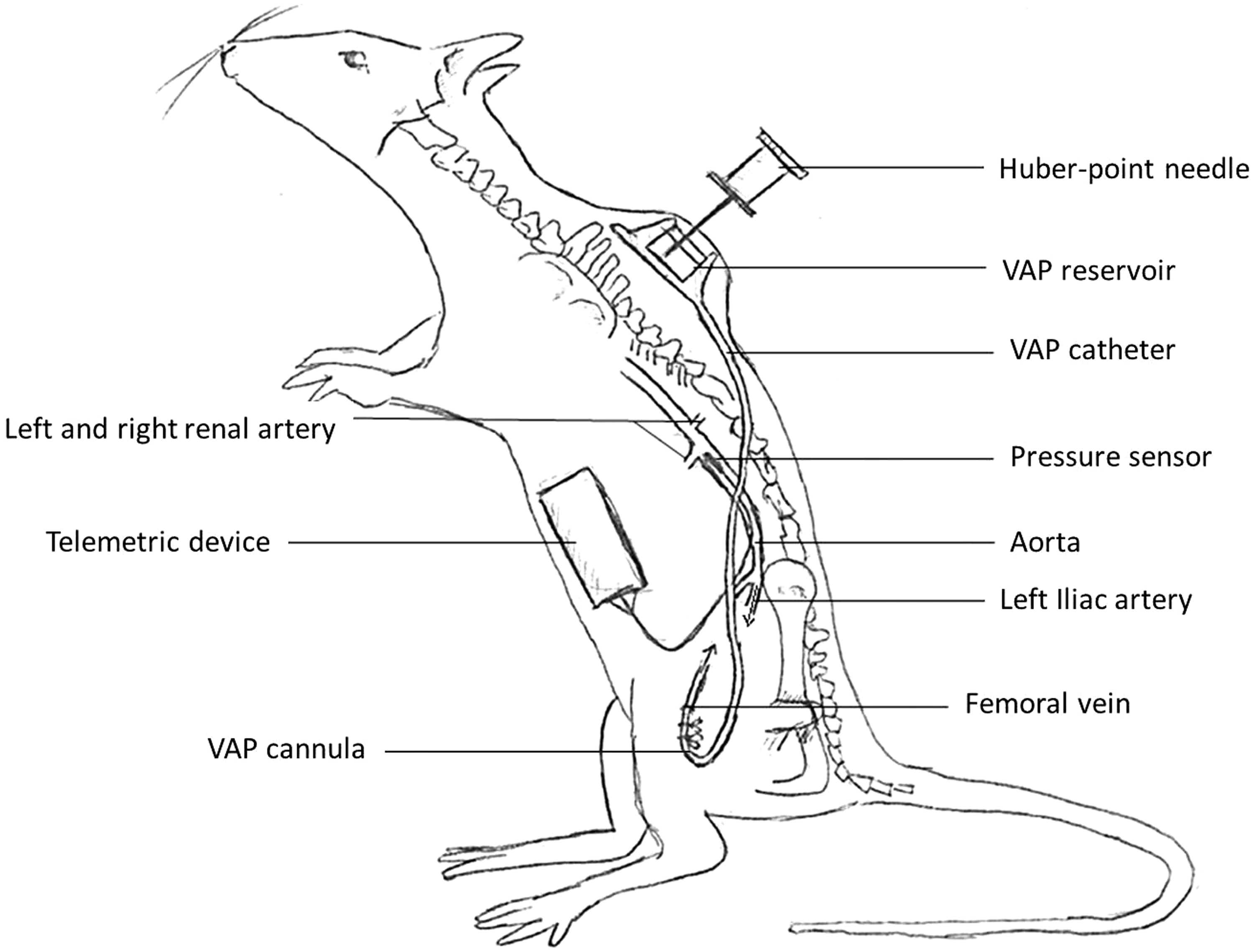
The aseptic condition of the equipment was established by autoclaving or the use of disinfectant (0.5% chlorhexidine in 70% ethanol; Jaychem Industries, Auckland, NZ), as appropriate, and aseptic conditions were maintained at all times. To allow manipulation of the animal and access to multiple surgical sites, sterile elasticated tubular bandage of appropriate size for the animal (7.5 cm for lean and 10 cm for obese; London Healthcare Ltd, Saddleworth, UK), was used as an alternative surgical drape. The bandage was carefully placed, by sliding the animal into the elastic tube, ensuring the outer remained aseptic, and the draped animal was then placed onto the sterile field. The surgical sites were exposed by cutting through the bandage, and the skin was thoroughly disinfected with chlorhexidine.
The VAPs were provided sterile by Access Technologies (ROP-3H, hydromer-coated polyurethane, 3Fr; Skokie, IL, USA). Round-tipped cannulae were used to minimize endothelial damage and the risk of thrombosis. Cannulation was conducted via the femoral vein, rather than the commonly used jugular vein.18,19,36,37 Placement in the femoral vein is preferable for extended cannula patency, and to avoid potential complications associated with catheter placement in the atrium or augmented cerebral blood flow following jugular vein ligation.21,22,28 The VAP was primed with heparin sodium (100 IU/mL; Hospira Australia, Mulgrave, Australia). Subcutaneous placement of the VAP was achieved by the use of a trocar, which was inserted from the proximal end of an incision in the inguinal triangle, around the side of the animal to a dorsal skin pocket and exited between the scapulae via a back incision. The VAP reservoir was positioned within the subcutaneous pocket offset from the incision site and secured to the underlying musculature. Blunt dissection was used to isolate the femoral vein, which was permanently occluded with a 3/0 silk ligature immediately proximal to the intersection with the superficial epigastric vein. The VAP cannula was introduced into the femoral vein and advanced beyond the abdominal muscle into the iliac vein. The VAP catheter was further secured to the underlying musculature. Skin incisions were closed with 3/0 silk ligatures, using subcuticular sutures at the inguinal site to reduce the incidence of self-mutilation. VAP functionality was confirmed by blood withdrawal, or alternatively by a positive pharmacological hemodynamic response.
A radio-telemeter with pressure sensitive tip (TRM53P; Telemetry Research, Millar Instruments, Houston, TX, USA) was implanted into the abdominal aorta.8,38,39 Telemeters were disinfected in 2% glutaraldehyde overnight. The abdominal aorta was exposed via a ventral midline incision. The aorta was occluded using vessel clamps (Aesculap, Tuttlingen, Germany), one immediately above the iliac bifurcation and another approximately 2 cm proximal, between the renal and iliolumbar arteries. The pressure tip of the telemeter was inserted 1.5–2 cm into the aorta, retrograde from the distal to proximal clamp positions, and the insertion point was sealed with tissue glue (Histoacryl; B Braun, Melsungen, Germany). The vessel clamps were carefully removed, ensuring the aorta was completely sealed, and hindlimb reperfusion was supported with topical lignocaine (2% Lopaine; Ethical Agents, Auckland, NZ) to reduce vascular spasm. Total occlusion time of the aorta was on average approximately 15 min (see Results). The abdomen was flushed with 5 mL of warmed sterile saline to keep tissues moist and avoid tissue adhesion, and the body of the telemeter was secured to the abdominal muscle during wound closure with 3/0 silk ligatures. Warmed sterile saline (10 mL) was provided subcutaneously to replace fluids and support recovery.
Post-operative monitoring
Animals were closely monitored for 4 h post-surgically, with heating blanket and oxygen support. Post-operative animals were monitored twice daily for three days and then daily until completion of the seven-day recovery period. Monitoring included assessment of body weight, food and water intake by daily weight, fecal production, wound condition, and animal appearance and behavior. The rats received daily analgesia until post-operative day 2 and antibiotic support twice-daily for two days, as detailed above. The animals were supplemented with subcutaneous saline where water intake was insufficient or dehydration became apparent via insufficient skin turgor. The VAP was flushed on day 3, and at minimum twice-weekly with 0.5 mL heparin sodium (100 IU/mL) to ensure it remained patent.
Initial work identified that obese animals in particular were prone to develop post-surgical damp and infected abdominal wounds, because of their lack of mobility and pressure necrosis of the ventral skin surface, which was exacerbated by embedded bedding material. Thus, for 2–3 days following surgery, all the animals were housed on paper, before returning them to normal corncob bedding (The Andersons, Maumee, OH, USA). All post-operative animals were individually housed to protect the wound sites from damage. To minimize isolation stress, animals were housed within close proximity of each other and provided environmental enrichment primarily in the form of wooden gnaw blocks and paper wool.
Vascular access port operation
At all times, the VAPs were accessed under strict aseptic conditions using Huber point needles (PG24-625; Access Technologies) to avoid coring, in accordance with the manufacturer’s instructions. A short-acting local analgesic (5% lignocaine/prilocaine; AstraZeneca, North Ryde, NSW, Australia) was applied topically 10 min before needle placement. Prior to needle insertion, the skin over the port was disinfected with chlorhexidine, or iodine solution (Betadine; Sanofi-Aventis, Virginia, Australia) to reduce irritation if the skin was broken. An equilibration period of 15 min was allowed following the brief restraint and needle insertion, before experimental hemodynamic measures were conducted. Solutions were administered using 5 mL Luer Lock syringes (Terumo, Tokyo, Japan) to limit pressure development. Upon completion of hemodynamic measures, access via the VAP was concluded by injection of 0.4 mL heparin sodium (100 IU/mL) to prevent coagulation, and the needle was removed from the port using positive pressure.
Experimental procedures
Experiments were performed twice-weekly to reduce stress to the animals, as well as to ensure complete drug clearance and avoid potential desensitization of receptors to experimental drugs, such as catecholamines. Blood pressure data were derived from the telemeter according to the manufacturer’s instructions, and acquired using LabChart 7 software (ADInstruments, Dunedin, NZ). Basal hemodynamics were assessed before needle insertion while the animals were at rest. The nitric oxide donor sodium nitroprusside (SNP; 6.25 µg/kg) was administered at the beginning of each experimental session to confirm VAP patency, as well as to provide a measure of receptor-independent vascular reactivity over the course of the five-week study. Repeated boluses of either saline or the β-adrenergic agonist dobutamine (30 µg/kg) were administered at 5 or 10 min intervals to assess volume effects and acute reproducibility, respectively. All boluses were 0.2 mL, with drug doses calculated to account for 0.1 mL dead space in the VAP, so that each subsequent dose comprised half the injection and half the previous dose. Solutions were eliminated from the VAP by consecutive flushes with 0.4 mL and 0.2 mL of saline. All chemicals were from Sigma-Aldrich (St Louis, MO, USA) and diluted in saline (0.9% NaCl; Baxter, Toongabbie, Australia) unless otherwise stated.
Data and statistical analysis
Heart rate and mean arterial pressure (MAP) were derived from the blood pressure data using the LabChart blood pressure module (ADInstruments), and averaged over 10 consecutive cycle bins. Student’s t-test, Fisher’s exact test or two-way repeated measures analysis of variance (ANOVA) were performed using SigmaPlot 12.0 (Systat Software Inc, Chicago, IL, USA). Differences between groups were assessed using the Student-Newman-Keuls post-hoc test, with significance assumed at the level of P < 0.05. Data are expressed as mean ± standard error.
Results
Surgical outcomes and post-operative complications
Surgical outcomes.
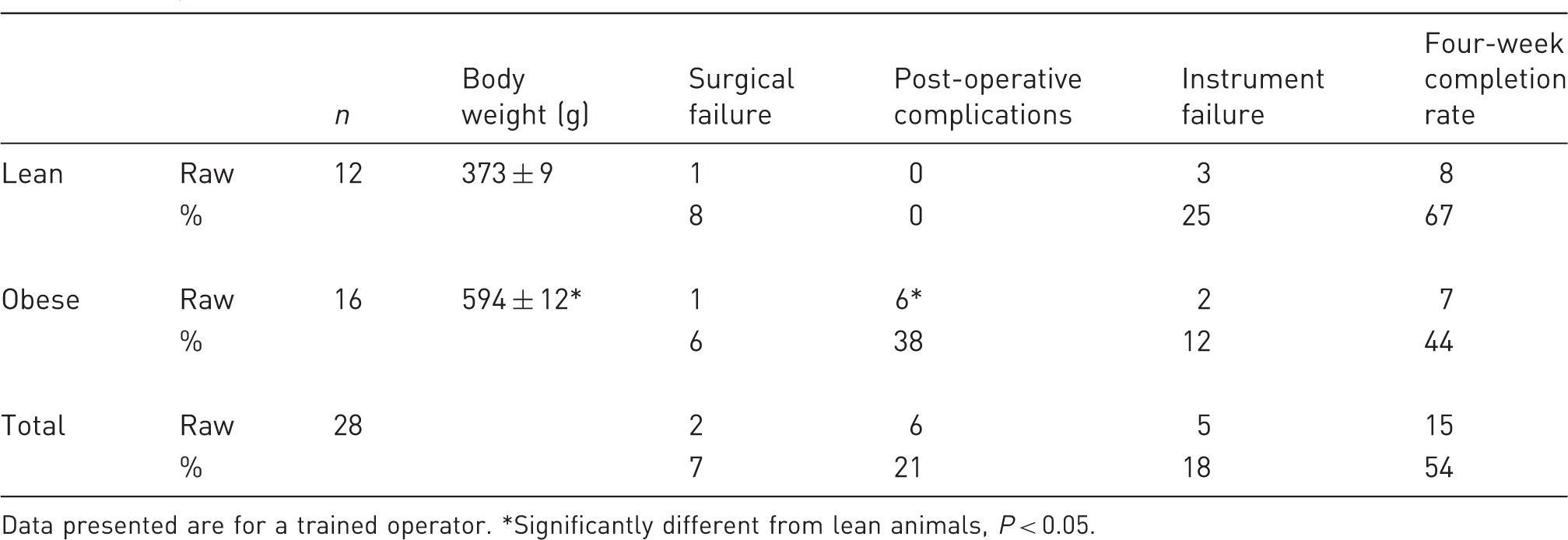
Data presented are for a trained operator. *Significantly different from lean animals, P < 0.05.
Obese animals exhibited an average body weight more than 60% greater than their lean littermates (Table 1). This contributed to post-operative complications including infection due to reduced movement, hindlimb paralysis, gastrointestinal disturbance and sudden cardiac failure. Such complications were likely related to increases in anesthetic requirement, surgical manipulation, average length of aortic occlusion (12 ± 1 min in lean versus 17 ± 2 min in obese animals, P = 0.125; occlusion >20 min greatly increased the chance of surgical complications) and underlying pathological factors in the obese animals.
Post-operative recovery
The post-operative state of the animals was assessed using the institutional Animal Welfare Office’s standard Bright, Alert, Responsive (BAR) score. This scale assesses the animals’ demeanor on a scale of 0–3, with 0 being normal behavior and 3 indicating a lack of response to the researcher. Following dual implantation surgery, none of the animals were designated a score of 3. All but one of the animals showed signs of modified behavior 4 h after completion of the surgery, with lean animals displaying poorer scores (1.7 ± 0.1 versus obese animals 1.1 ± 0.2). However, all the lean animals were rated 1 or less by post-operative day 1 (mean 0.8 ± 0.1), while the obese animals took until post-operative day 3 to return to this level (mean 0.6 ± 0.2). One week after surgery, almost all of the animals had resumed normal behavior (lean 0.2 ± 0.1 and obese 0.3 ± 0.2). In addition, three obese animals with post-operative complications requiring euthanasia were consistently scored at 2, confirming the BAR score is effective for assessing the state of post-operative animals.
Implantation increased the weight of each animal by 14 ± 1 g (not significant [NS] as determined by two-way repeated measures ANOVA) due to the added mass of the telemeter and VAP. Lean rats exhibited a significant reduction in body weight in the days following surgery (Figure 2a), primarily attributable to a reduced food intake (Figure 2b). However, food intake in lean animals returned to a plateau by post-operative day 10 and all animals were restored to pre-surgical weight by day 20.
Surgical recovery. Change in body weight (a), food intake (b) and water intake (c) following implantation surgery (day 0) in lean and obese Zucker rats (n = 3–12, decreasing over time). *Significantly different, lean versus obese;§Significantly different from day 1 within lean;#Significantly different from day 1 within obese;##Significantly different from day 3 within obese; P < 0.05.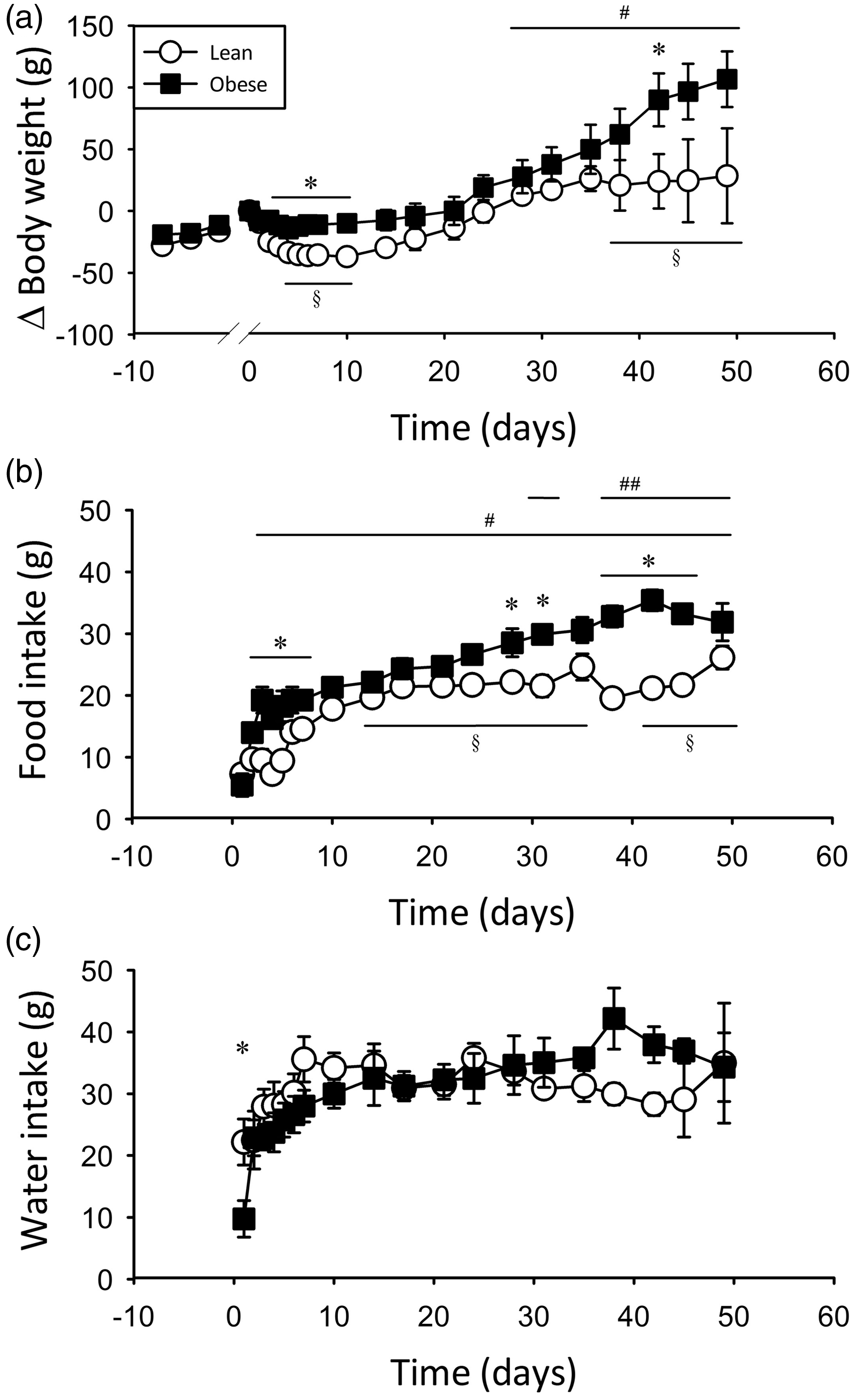
Obese animals displayed reduced food intake on days 1 and 2, but with significant increases each day, and reached a plateau by day 3 following surgery (Figure 2b). This rapid return to plateau feeding levels is likely to be responsible for the absence of significant weight loss in obese animals following surgery (Figure 2a). This unusual post-operative food intake in obese animals can be attributed to their excessive appetite due to leptin receptor deficiency, which induces an ‘insatiable hunger’ state. A significant increase in body weight, accompanied by a further increase in food intake above the plateau, was apparent in obese animals in the second month after recovery. Plateau food intake was comparable with previous reports for both lean and obese rats. 40
Water intake was generally consistent, both between lean and obese rats and across the study (Figure 2c). The only exception was that obese animals in the 24 h following surgery showed a significantly reduced fluid intake compared with all other time points. This may be related to reduced mobility, but would be offset by post-operative fluid administration and did not result in visible dehydration.
Overall these results indicate that animals recover well from the surgery, and return to normal behavior within one week of surgery.
Chronic consistency
Baseline hemodynamic measures in conscious rats were consistent across the course of the study (Figures 3a and b), even with individual data points representing only a ‘snapshot’ of 10 cardiac cycles. Lean rats displayed normal MAP with an overall average of 106 ± 5 mmHg, while measures in obese rats were significantly higher with an overall average of 129 ± 5 mmHg, indicating hypertension in these animals (P < 0.05). HR fluctuated around 400 bpm, creating small but significant differences between lean and obese animals. This small variability in baseline HR is unlikely to have meaningful physiological impact, and is overcome when data can be averaged across days to compare groups; and pharmacological responses are consistent irrespective of baseline, as shown below. Within the groups, however, there was no significant difference between any of the time points, indicating reliability in the conscious hemodynamic measures.
Chronic hemodynamic consistency. Timeline of basal mean arterial pressure (a) and heart rate (b) in conscious rats over a five-week measurement period. Sodium nitroprusside (6.25 µg/kg) mean arterial pressure (c) and heart rate (d) response consistency demonstrating ongoing catheter patency (n = 3–7, decreasing over time). *Significantly different, lean versus obese; P < 0.05.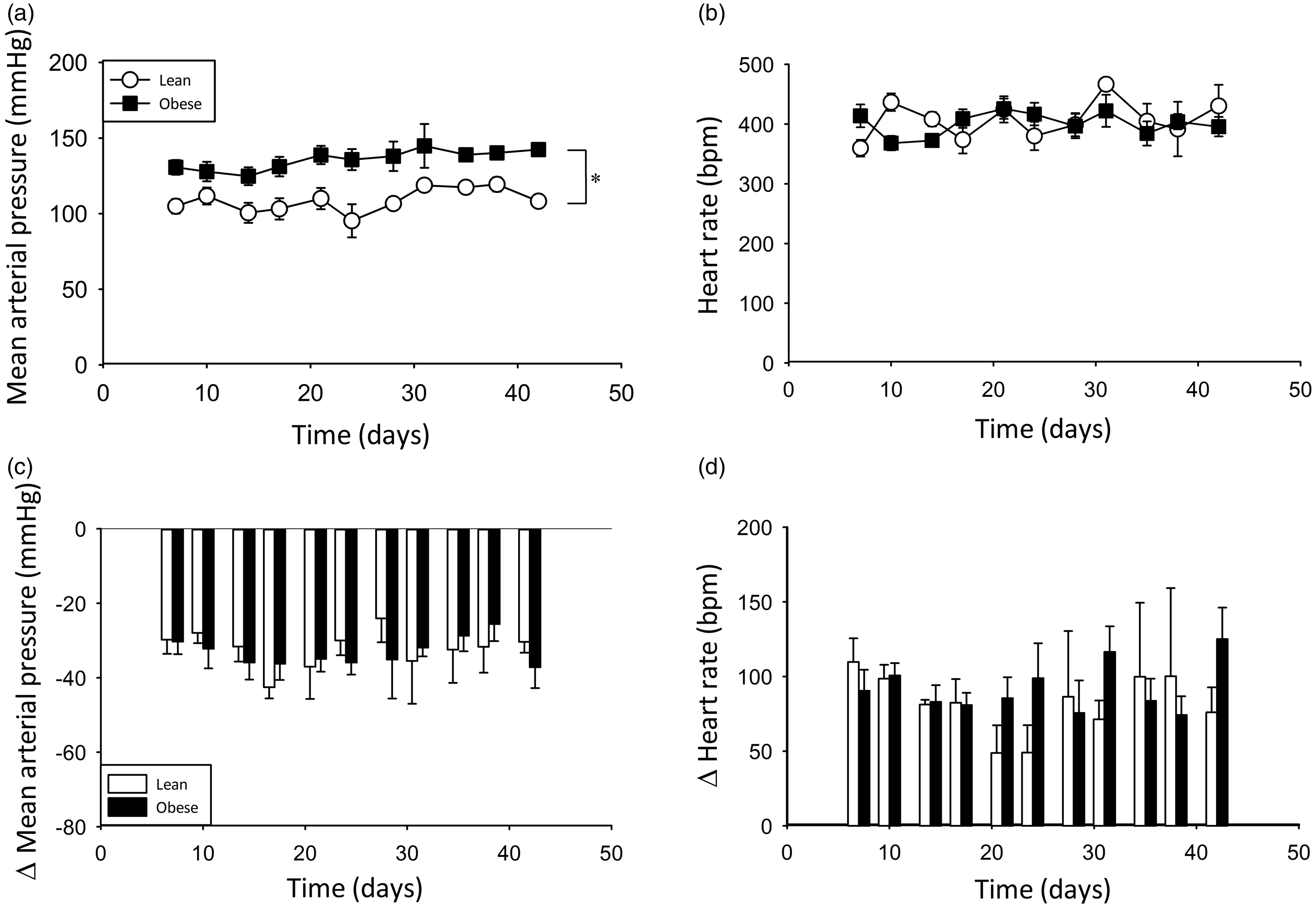
The patency of the VAP was tested using SNP, a nitric oxide donor which causes broad vasodilation and consequently a drop in MAP. Moreover, repeated pharmacological administration of SNP allows assessment of chronic changes in receptor-independent vascular reactivity to determine the chronic consistency in the effectiveness of the VAP for pharmacological and hemodynamic responses. To this end, conscious animals were administered SNP (6.25 µg/kg) via the VAP at twice-weekly intervals for five weeks. SNP caused the expected drop in MAP (Figure 3c) and consequently a baroreflex-mediated increase in HR (Figure 3d). While specific responses fluctuated minimally, there was no significant difference in SNP response between groups or at any time point. Therefore, the patency of the VAP and the vascular reactivity remained constant and repeated pharmacological testing was seen to produce consistent hemodynamic responses for up to 50 days.
Acute response consistency
Repeated boluses of saline were administered at 5 min intervals to assess baseline hemodynamic responses to experimental manipulation and volume effects (Figure 4a). There was no significant difference in peak response between any of the time points, indicating that responses to injection itself are consistent across repeated manipulations (Figures 4b and c). There was also no change in baseline hemodynamic measures following repeated saline boluses (Figure 4a), suggesting a potential increase in blood volume had no effect. Therefore, the limited hemodynamic response to injection itself indicates that animals experience minimal stress during administration via the VAP. Importantly, obese animals also experienced minimal stress-induced hemodynamic changes using this method, avoiding the disparate stress responses other techniques elicit in hypertensive animals.4,5,30,32. These data suggest that this technique allows pharmacological testing of hemodynamic effects with minimal confounding responses.
Acute response consistency. Hemodynamic responses following repeated bolus administration of saline; representative trace of blood pressure and calculated heart rate (a), average change in mean arterial pressure (b) and heart rate (c; n = 4). Hemodynamic responses to repeated bolus administration of 30 µg/kg dobutamine; representative trace of blood pressure and calculated heart rate (d), average change in mean arterial pressure (e) and heart rate (f; n = 2).▾ Indicates bolus administration.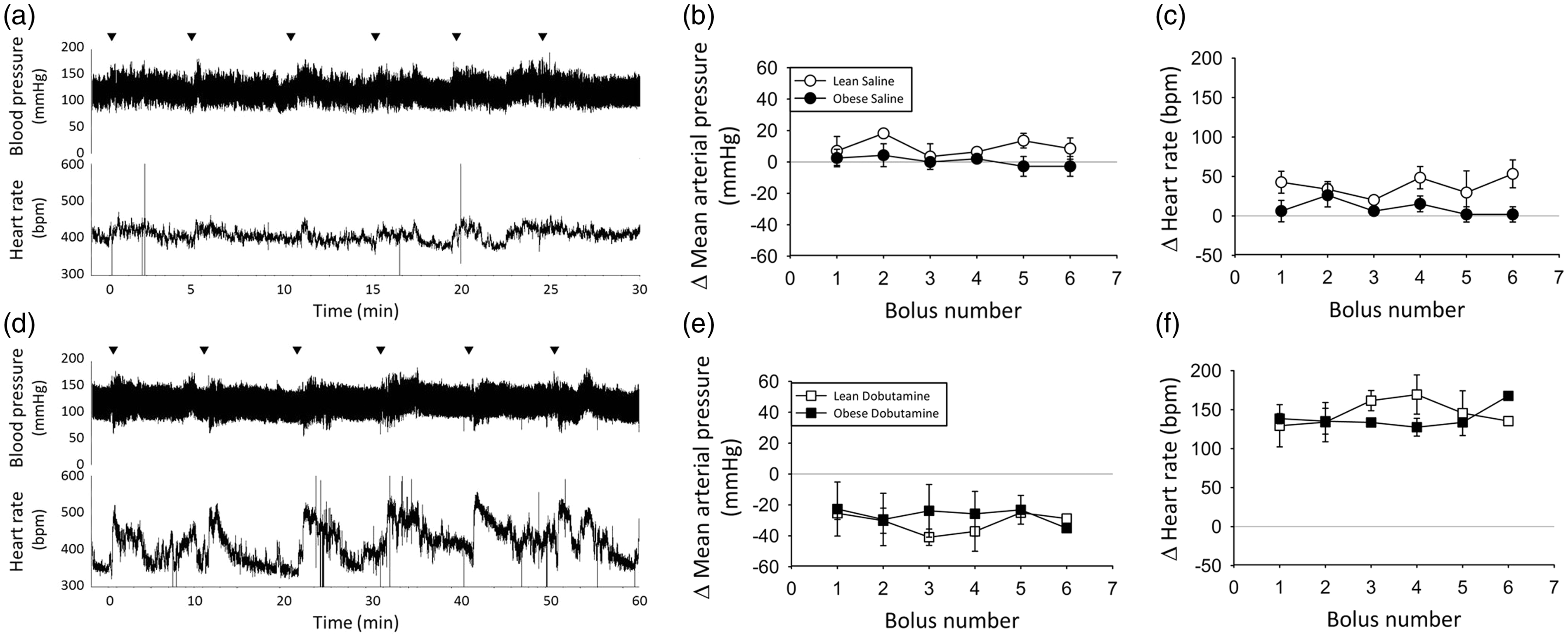
To assess consistency in acute pharmacological responses, the experiment was repeated using multiple bolus administration of the β-adrenoceptor agonist dobutamine (30 µg/kg; Figure 4d). Stimulation of β-adrenoceptors on the heart enhanced cardiac output, in part via increased HR, while β2-adrenoceptors in the vasculature elicited vasodilation. Dobutamine reliably caused the expected HR increase and a brief MAP decrease, which were easily distinguishable from saline effects. Variability in these peak responses was minimal across repeated bolus injections at 10 min intervals in both lean and obese rats (Figures 4e and f). Therefore, repeated pharmacological administration via a VAP elicited reliable and replicable changes in both HR and MAP in conscious animals.
Discussion
The present study describes a novel technique combining radio-telemetric hemodynamic measures with fully implantable VAPs. The achievement of a 67% success rate in normal animals indicates that this is a viable technique for physiological studies. Animals recovered well from the dual implantation surgery, in agreement with literature reports of normalized behavior within three days of surgery. 16 Basal and pharmacological hemodynamic measures were consistent when assessed chronically over five weeks or during repeated acute administration. Reaction to the injection alone was limited, indicating that the procedure induces minimal stress, allowing accurate physiological data to be obtained from both normal and obese conscious animals. Therefore, our novel combination provides an improved platform to undertake comprehensive studies involving cardiovascular physiological measures and repeated pharmacological tests in conscious animals, while providing both acute and chronic reliability.
Combining radio-telemetry with VAPs provides fully implantable instrumentation to allow physiological measures to be undertaken in unrestrained, conscious rats over extended periods. As the instrumentation is fully contained within the animal, this technique achieves extended catheter patency, 22 with the observed 79% patency after four weeks higher than reports for externalized catheters. 41 By allowing numerous repeated tests, both acute and chronic, to be undertaken in an individual animal, data output is maximized. The ability to produce extensive data from a single animal greatly reduces the number of animals required. Additionally, repeated measures designs have greater statistical power, thereby further reducing demand for animals and improving scientific interpretation. In addition, as the animals are completely unrestrained, this method avoids interfering with normal behavior and exerts minimal physical and mental stress on the animal. To support the animals through surgical recovery, individual housing is required, which may contribute to some isolation stress. However, individual housing does have the advantage of allowing experiments to be performed in the animal’s home cage. This reduces the acute experimental stress experienced by the animal, which not only improves animal welfare but also has scientific benefits through improved data quality. Importantly, removal of the confounding stress effects associated with restraint greatly improves interpretation of hemodynamic and pharmacological responses. Improved methods of conducting acute pharmacological examinations under conscious conditions open up new avenues of scientific investigation and enhance clinical relevance.
In this study, obese rats presented unique surgical difficulties in respect to maintenance of anesthesia and restricted accessibility of the surgical sites; creating a greater demand for surgical expertise. In addition, obese animals experienced distinct post-operative complications including infection due to sedentary behavior and cardiac complications due to underlying pathological factors. Interestingly, increased perioperative complications are also observed in the obese cohort of human patients undergoing surgery.42,43 In our study, many obese-specific difficulties were overcome with minimal adaptations to the procedure, making dual implantation studies in obese animals nevertheless feasible. As in the lean animals, the obese rats demonstrated consistent hemodynamic and pharmacological measures. In the absence of restraint stress, hemodynamic responses in obese and lean animals were comparable, avoiding confounding factors of disease models reported with other techniques.4,5,30
Exceptionally long-term applications of this technique were not investigated here. We achieved an overall completion rate of 54%, with greater success in lean animals, at termination of our primary study one month following surgery. As our primary experimental measurements concluded after four weeks, only half of the animals were monitored beyond this point, so the actual achievable VAP lifespan was not determined. However, a one-month time frame is likely to be adequate in the majority of physiological studies, and this greatly improves upon the lifespan of days generally achieved with open, tethered catheters. 22
Furthermore, the frequency of our measures (experiments were performed twice-weekly) was restricted by the potential for catecholamine-induced receptor desensitization. The absence of this restriction in the majority of investigations using alternative pharmacological agents should significantly increase the opportunity to undertake additional measures within this period.
In conclusion, the dual implantation of radio-telemeters and VAPs introduces a valuable technique for addressing physiological questions. The improved capacity for repeated measures within an animal reduces the animal numbers required and opens up possibilities for addressing new research questions. The absence of physical restraint avoids the issue of disparate stress responses in many disease models, as exemplified in the obese rat. Therefore, this approach provides both acute and chronic reliability for comprehensive hemodynamic investigations involving repeated pharmacological tests in conscious animals.
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
Department of Physiology, Otago School of Medical Sciences, University of Otago, New Zealand.
