Abstract
Prolonged ischemia of skeletal muscle tissue, followed by reperfusion, leads to ischemia/reperfusion injury (IRI), which is a feared local and systemic inflammatory reaction. With respect to the 3Rs, we wanted to determine which parameters for assessment of IRI require a reperfusion time of 24 h and for which 2 h of reperfusion are sufficient. Rats were subjected to 3 h of hind limb ischemia and 2 h or 24 h of reperfusion. Human plasma derived C1 inhibitor was used as a drug to prevent reperfusion injury. For 2 h of reperfusion the rats stayed under anesthesia throughout (severity grade 1), whereas for 24 h they were awake under analgesia during reperfusion (grade 2). The femoral artery was clamped and a tourniquet was placed, under maintenance of venous return. C1 esterase inhibitor was systemically administered 5 min before the induction of ischemia. No differences in local muscle edema formation and depositions of immunoglobulin G and immunoglobulin M were observed between 2 h and 24 h (P > 0.05), whereas lung edema was only observed after 24 h. Muscle viability was significantly lower after 24 h vs 2 h reperfusion (P < 0.05). Increased plasma creatine kinase (CK)-MM and platelet-derived growth factor (PDGF)-bb could be detected after 2 h, but not after 24 h of reperfusion. By contrast, depositions of C3b/c and fibrin in muscle were only detected after 24 h (P < 0.001). In conclusion, for a first screening of drug candidates to reduce IRI, 2 h reperfusions are sufficient, and these reduce the severity of the animal experiment. Twenty-four-hour reperfusions are only needed for in-depth analysis of the mechanisms of IRI, including lung damage.
Lower extremity ischemia may occur due to embolic/thrombotic or traumatic vascular occlusion, or alternatively due to tourniquet application during surgical intervention. Reperfusion of the ischemic extremity is crucial for tissue survival. However, as a result of reperfusion, complex inflammatory cascades are activated which can aggravate ischemic tissue damage and lead to local as well as systemic inflammatory reaction. In this phenomenon, called ischemia/reperfusion injury (IRI), key roles are attributed to the complement, the coagulation and the kinin systems.1–3 Locally, lower extremity IRI is manifested in muscle edema formation, rigidity, loss of muscle viability, apoptosis and necrosis. In addition, local injury is often accompanied by distant organ damage, affecting organs like the lung, heart, kidney as well as the liver and may result in multiple organ dysfunction syndrome.4,5 In particular, acute lung injury is a consequence of lower extremity IRI and is characterized by increased microvascular permeability and pulmonary edema. 6
Lower extremity IRI has been extensively studied in different species such as pigs, dogs and rabbits.7–9 Although 75% of all experimental animals are rodents, comparisons are often impossible. A review of the literature reveals a variety of methods with respect to the induction of ischemia, which is done by placement of an elastic rubber band or a tourniquet on the proximal part of the limb, and/or clamping the femoral or iliac artery, or even the infrarenal aorta.10–12 However, not only does the technique of ischemia induction vary, but the time periods of ischemia and reperfusion also show strong heterogeneity. In acute models rats are subjected to periods of ischemia ranging from 2 h to 4 h, whereas reperfusion times range from 1 h to 24 h.11,13,14
The aim of the present study was to establish an acute hind limb IRI model with shortened reperfusion times in consideration of the 3 R (replacement, reduction, refinement) principles, 15 which would minimize the potential pain and suffering of animals, and still represent a good model for analyzing the effect of drug candidates on IRI. To demonstrate the efficacy of the present rat model we used C1 esterase inhibitor (C1 INH) as a model drug candidate, which is a crucial regulator of plasmatic cascades activated by inflammation. We hypothesized that for a first screening of drug candidates, 2 h of reperfusion would be sufficient, whereas for detailed analysis of mechanisms as well as for distant lung injury 24 h reperfusions are required. Furthermore, we wanted to compare the effect of exogenous human plasma-derived C1 INH on tourniquet-induced limb IRI after 2 h and 24 h of reperfusion in a rat model.
Materials and methods
Animals
All the animal experiments were conducted in compliance with the Guide for the Care and Use of Laboratory Animals and Swiss national guidelines and were approved by the animal ethics committee (permission number 71/10) of the Cantonal Veterinary Service (Canton of Bern, Switzerland). 16 Male Wistar rats (wild type, bred at the central animal facility, University of Bern, Switzerland, body weight 250–350 g) were kept together in threes in a clear 1500 cm2 Euro-standard type IV S cage (Tecniplast, Buguggiate, Italy) with overall dimensions of 48 × 37.5 × 21 cm in order to enable social interactions and welfare. However, after surgical interventions the rats were kept singly for 24 h of reperfusion to prevent mutual injury. The rats were housed under standard conditions with food (maintenance diet; Provimi Kliba, Kaiseraugst, Switzerland) as well as water ad libitum and poplar wood as bedding material (Lignocel; Provimi Kliba). For cage enrichment the rats were provided with paper tissue, pieces of wood and tubes. Cages were individually ventilated at 20 ± 2℃ and 45–65% relative humidity with a circadian rhythm of 12/12 h. During the light cycle animals were exposed to an intensity of 200 lux.
Reagents
C1 INH (Berinert®) was provided by CSL Behring (CSL Behring GmbH, Marburg, Germany).
Experimental groups
The rats were divided into five groups. The experimental groups of 2 h reperfusion (n = 7) and 24 h reperfusion (n = 6) received 50 IU/kg (50 IU/mL) of exogenous human C1 INH. The control groups, which were subjected to either 2 h (n = 7) or 24 h (n = 7) of reperfusion received no C1 INH. In addition, tissue and plasma samples of normal, healthy rats (n = 4) were included in the study for comparison. The timelines of the experimental procedures are given in Figure 1a.
Experimental design and surgical procedure. (a) Male Wistar rats were subjected to 3 h of ischemia, followed by either 2 h or 24 h of reperfusion. During 2 h of reperfusion rats were kept under anesthesia, whereas for 24 h of reperfusion rats were awake. (b) For induction of ischemia, the femoral artery was occluded by using an atraumatic microclamp. Additionally, a tourniquet was placed underneath the femoral vessels under maintenance of venous return (for details see Materials and methods section). C1 INH: C1 esterase inhibitor.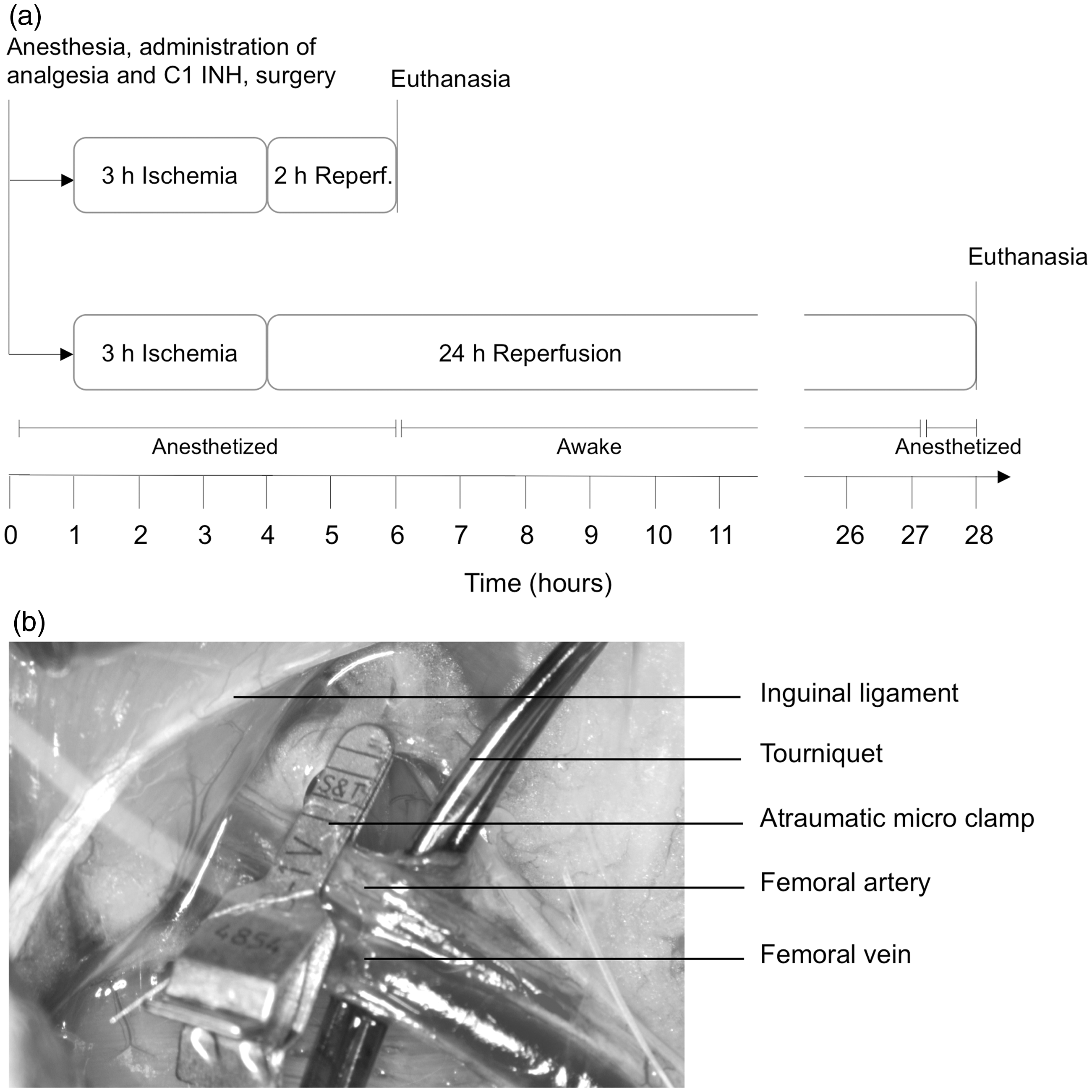
Anesthesia and analgesia
Induction of anesthesia was performed with 2.5% isoflurane in oxygen in an anesthetic induction chamber and later maintained by inhalation of 1.5% isoflurane via a nose mask. Arterial oxygen saturation, heart rate, breath rate, rectal temperature, pulse distention, and breath distention were continuously monitored using the mouse ox plus system (Starr Life Sciences, Oakmont, PA, USA). Thirty minutes prior to surgical intervention, 0.05 mg/kg of buprenorphine (Temgesic, 1 mg/mL; Reckitt Benckiser (Switzerland) AG, Zurich, Switzerland) was injected subcutaneously into the anesthetized rats to provide analgesia. The total duration of anesthesia was approximately 6 h, after which the rats of the 24 h reperfusion groups were allowed to wake up, whereas the rats which underwent only 2 h of reperfusion remained under anesthesia until euthanasia by exsanguination (Figure 1a). When the animals of the 24 h reperfusion groups were completely awake, buprenorphine injection was repeated to provide adequate analgesia. The condition of the rats was continuously monitored for the first 5 h of reperfusion and then after 19 h and 24 h. The administration of analgesia was strictly controlled to avoid pica behavior. After completion of 24 h of reperfusion, the rats were anesthetized again as described above and sacrificed by exsanguination during organ removal.
Surgery
The rats had an acclimatization period of at least six days prior to surgical intervention. Their fur was completely removed from both hind limbs with an electric shaver for an assessment of limb perfusion by Laser Doppler (Moor LDI, Auxminster, UK). To maintain a body temperature of 37℃ the rats were kept on a heating pad (T/pump professional system; Gaymar Industries, Inc, Orchard Park, NY, USA). For the induction of unilateral hind limb ischemia, approximately 30 min after induction of anesthesia, the femoral artery and vein were exposed via a groin incision, and a tourniquet (standardized weight of 450 g) was placed underneath the femoral vessels to block collateral circulation. 11 The femoral artery was occluded for 3 h with two microvascular clamps (B1-V; S&T, Neuhausen, Switzerland) (Figure 1b). The rat hind limbs were not exsanguinated, but a comparable state was achieved by allowing venous return during the entire period of ischemia in order to prevent venous congestion and additional injury through microcirculatory impairment, which would not represent the clinical situation. After 3 h of ischemia the limb was reperfused for either 2 h or 24 h. At the end of the experiments, tissue samples of both the ischemic and contralateral gastrocnemius muscles as well as the lungs were taken for subsequent analyses.
Assessment of edema formation
Edema formation was assessed by calculating the wet weight to dry weight ratio (wet/dry ratio). Two samples of the gastrocnemius muscle from both legs as well as the left lobe of the lung were taken and immediately weighed to obtain the wet weight. Muscle and lung samples were then dried for 24 h at 80℃ after which a constant dry weight was achieved. Subsequently, the wet/dry ratio was calculated.
Analysis of muscle viability
IRI severely affects muscle viability, which may ultimately result in muscle necrosis. For analysis of muscle viability an MTT (3 -(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide; Sigma, St Louis, MO, USA) assay was performed. The muscle samples from the gastrocnemius muscle were taken, washed in phosphate-buffered saline (PBS), blotted dry and incubated in 0.1 mg MTT/mL PBS in a total volume of 3 mL at 37℃, rotating in the dark for 2 h. Thereafter, the muscle samples were blotted dry and incubated in 100% isopropanol at 37℃, rotating in the dark overnight to elute the formazan crystals from the tissue for measurement of the optical density (OD). Two hundred microliters of the thus obtained supernatant was measured using a microplate (Nunc, 96 well, maxisorp, transparent; Nunc A/S, Roskilde, Denmark) with a microplate reader at 560 nm (Ref. 690 nm, Infinite M1000 spectrophotometer; Tecan, Männedorf, Switzerland). After drying the muscle samples at 80℃ for 24 h the OD per milligram dry weight was calculated and compared with values of the contralateral legs.
Histological assessment of damage
Hemorrhage and total myocyte damage as well as infiltration of neutrophil granulocytes were assessed in tissue samples from the gastrocnemius muscle. The tissue samples were fixed in 4% formaldehyde in PBS for 24–72 h, embedded in paraffin, cut into 3 µm thick sections and stained with hematoxylin and eosin (H&E).
Analysis of creatine kinase-MM level
For analysis of the creatine kinase-MM (CK-MM) level, a solid phase enzyme-linked immunosorbent assay (ELISA; Life Diagnostics, West Chester, PA, USA) was performed. According to the manufacturer’s instructions, standards and EDTA-plasma samples, taken after 2 h or 24 h reperfusion, were incubated in microtiter wells for 45 min at room temperature. Subsequently, the wells were washed, horseradish peroxidase conjugated detection antibody was added and incubated for 45 min. After washing, a TMB reagent was added, incubated for 20 min and stopped by adding stop solution. Finally, the OD was spectrophotometrically measured at 450 nm. GraphPad Prism 5 software (GraphPad Software Inc, San Diego, CA, USA) was used for analysis.
Analysis of platelet-derived growth factor-bb levels
An in-house developed immunoassay consisting of carboxylated non-magnetic beads (for platelet-derived growth factor [PDGF]-bb, region: 38, Cat. No: 171506038; Bio-Rad, Hercules, CA, USA) conjugated with specific capture (rabbit anti-PDGF-bb, 500-47; PeproTech, Hamburg, Germany) and detection antibodies (biotinylated rabbit anti-PDGF-bb, 500-P47Bt; PeproTech) was used to detect PDGF-bb. Briefly, EDTA-plasma was diluted 1:3 in a cytokine assay buffer (Bio-Rad) and incubated with antibody-coupled magnetic beads. A washing step was followed by incubation with biotinylated detection antibody. After streptavidin–phycoerythrin (Qiagen, Hilden, Germany) incubation the PDGF-bb concentration was measured. Recombinant protein (rat PDGF-bb, 520-BB; R&D Systems, Minneapolis, MN, USA) was used to establish a standard curve. PDGF-bb concentrations were calculated using the Bio-Plex Manager 6.0 software (Bio-Rad).
Immunofluorescence analyses of tissue samples
Immunofluorescence staining using specific antibodies was used to quantify the depositions of immunoglobulin M (IgM) (3020-08; Southern Biotech, Birmingham, AL, USA) and immunoglobulin G (IgG) (3030-08; Southern Biotech), C1q (A0136; Dako, Baar, Switzerland), C4b/c (LSB 4228; LifeSpan BioSciences Inc, Seattle, WA, USA), C3b/c (A0062; Dako), factor B (341272; Calbiochem, Darmstadt, Germany) and fibrin (F0111; Dako). The tissue samples from the gastrocnemius muscle of both legs were taken and washed in PBS, blotted dry and embedded in OCT matrix (Tissue tek; Sakura, Torrance, CA, USA) on dry ice. The samples were immediately stored at –20℃ until cryosections were cut. The sections were fixed in acetone and rehydrated in tris-buffered saline (TBS). Primary antibodies were incubated overnight at 4℃ and secondary antibodies were incubated for one hour at room temperature. Subsequently, the slides were mounted and covered. Pictures were taken with a fluorescent microscope (Leica DMI 4000B; Leica Microsystems Schweiz AG, Heerbrugg, Switzerland) and analyzed using Image J (National Institutes of Health, Bethesda, MD, USA) and GraphPad Prism 5 software (GraphPad Software Inc).
Statistical analysis
Data are expressed as means ± standard deviation (SD). One-way analysis of variance (ANOVA) followed by Bonferroni’s post hoc test for significance was achieved by using the GraphPad Prism 5 software. A 95% confidence interval was established and *P < 0.05; **P < 0.01; ***P < 0.001; ****P < 0.0001 were considered to be statistically significant. Determination of n numbers per group was performed without formal power analysis, based on preliminary experiments with C1 INH in our laboratory.
Results
Edema formation in muscle and lung as well as muscle viability
Edema formation in the muscle and lung tissue was assessed from the wet/dry ratio. Non-treated animals showed clear edema formation in the reperfused gastrocnemius muscle tissue after 2 h (wet/dry ratio = 5.4 ± 0.4, ratio for normal control animals = 4.3 ± 0.05, P < 0.0001) as well as after 24 h of reperfusion (5.6 ± 0.7), whereas C1 INH treated rats presented significantly less edema after 2 h (4.6 ± 0.2, P < 0.001 vs non-treated) and 24 h (4.7 ± 0.2, P < 0.0001) of reperfusion (Figures 2a–e). As compared with normal rats (4.6 ± 0.1) no lung edema was detected after 2 h of reperfusion for non-treated (4.7 ± 0.2) as well as for C1 INH treated rats (4.7 ± 0.1). However, after 24 h of reperfusion significant edema formation was found in non-treated rats (5.1 ± 0.2), which was significantly reduced (P < 0.001) in C1 INH treated rats (4.7 ± 0.1, Figure 2f). Muscle viability was analyzed using the MTT assay. Results are given as a viability index in % of the contralateral control leg, which was set to 100%. As compared with normal rats (96.5 ± 7.0%) muscle viability was not significantly decreased after 2 h (80.5 ± 21.1% for non-treated and 88.1 ± 7.3% for C1 INH treated rats). By contrast, after 24 h of reperfusion, C1 INH treatment (96.2 ± 12.9%) significantly improved viability compared with non-treated rats (63.3 ± 9.5%, Figure 2g). Analysis of correlation between the muscle viability index in % and wet/dry ratio revealed a significant (P < 0.001) negative correlation (Pearson coefficient = –0.53, Figure 2h).
Effect of C1 esterase inhibitor (C1 INH) on edema formation in the muscle and lung as well as muscle viability after 2 h and 24 h of reperfusion. (a) Muscle edema formation was analyzed in contralateral (con) and reperfused (rep) legs. Edema is indicated by an increase in wet to dry ratio. (b–e) Representative images of baseline and edema formation after 2 h and 24 h of reperfusion. (b) Baseline. (c) Non-treated (n-t) rat after 2 h of reperfusion. (d) Non-treated (n-t) rat after 24 h of reperfusion. (e) C1 INH treated rat after 24 h of reperfusion. (f) Lung edema formation was analyzed after 2 h and 24 h by using wet/dry ratio. (g) Assessment of viability of gastrocnemius muscle after 2 h and 24 h by MTT assay. (h) Analysis of correlation between muscle viability and muscle edema formation. One-way ANOVA followed by Bonferroni’s post hoc test for significance was used. Error bars indicate mean ± SD. *P < 0.05; **P < 0.01; ***P < 0.001; ****P < 0.0001.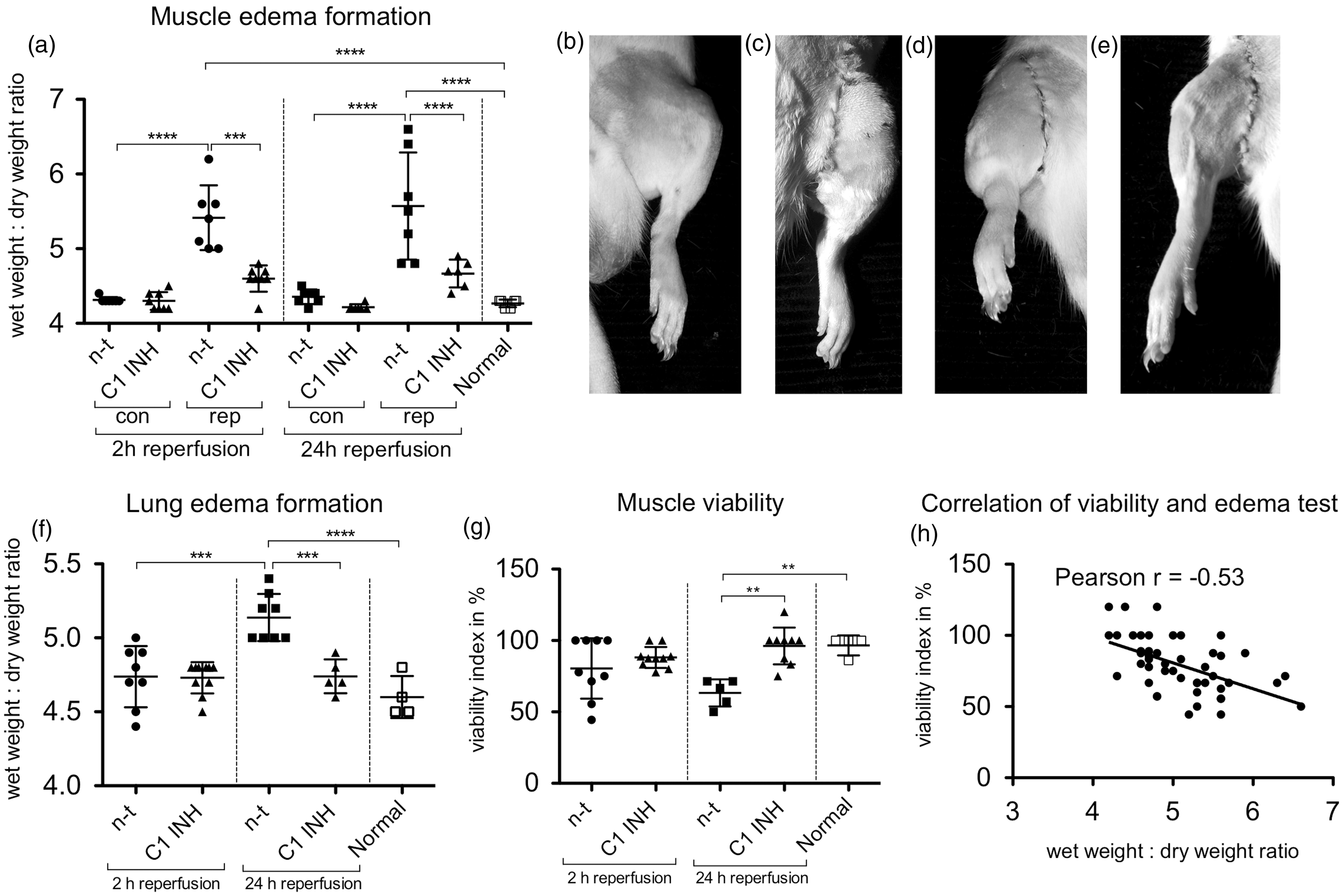
Histopathology and measurement of CK-MM and PDGF-bb plasma levels
The H&E stained muscle sections were assessed for overall muscle damage. The C1 INH treated rats showed only minimal damage after 2 h as well as 24 h of reperfusion, whereas non-treated rats presented strong edema formation, myocyte destruction and infiltration of neutrophil granulocytes (Figures 3a–d). ELISA and singleplex bead-based assays were performed for analysis of CK-MM and PDGF-bb plasma levels, respectively. After 2 h of reperfusion C1 INH treated animals (1122 ± 468 ng/mL) showed a significantly decreased level (P < 0.01) of CK-MM as compared with non-treated rats (1989 ± 485 ng/mL), whereas PDGF-bb levels were not significantly decreased by C1 INH treatment (2468 ± 390 pg/mL) in comparison to the non-treated rats (2219 ± 566 pg/mL). However, after 24 h of reperfusion CK-MM as well as PDGF-bb levels were significantly decreased and returned to baseline levels (Figures 3e and f).
Histopathological assessment of muscle damage as well as plasma levels of creatine kinase (CK)-MM and platelet-derived growth factor (PDGF)-bb. (a–d) Representative images of gastrocnemius muscles from non-treated (n-t) as well as C1 esterase inhibitor (C1 INH) treated rats, which were subjected to either 2 h or 24 h of reperfusion. (e, f) Analysis of CK-MM and PDGF-bb plasma levels after 2 h and 24 h of reperfusion. One-way ANOVA followed by Bonferroni’s post hoc test for significance was used. Error bars indicate mean ± SD. *P < 0.05; **P < 0.01; ***P < 0.001; ****P < 0.0001.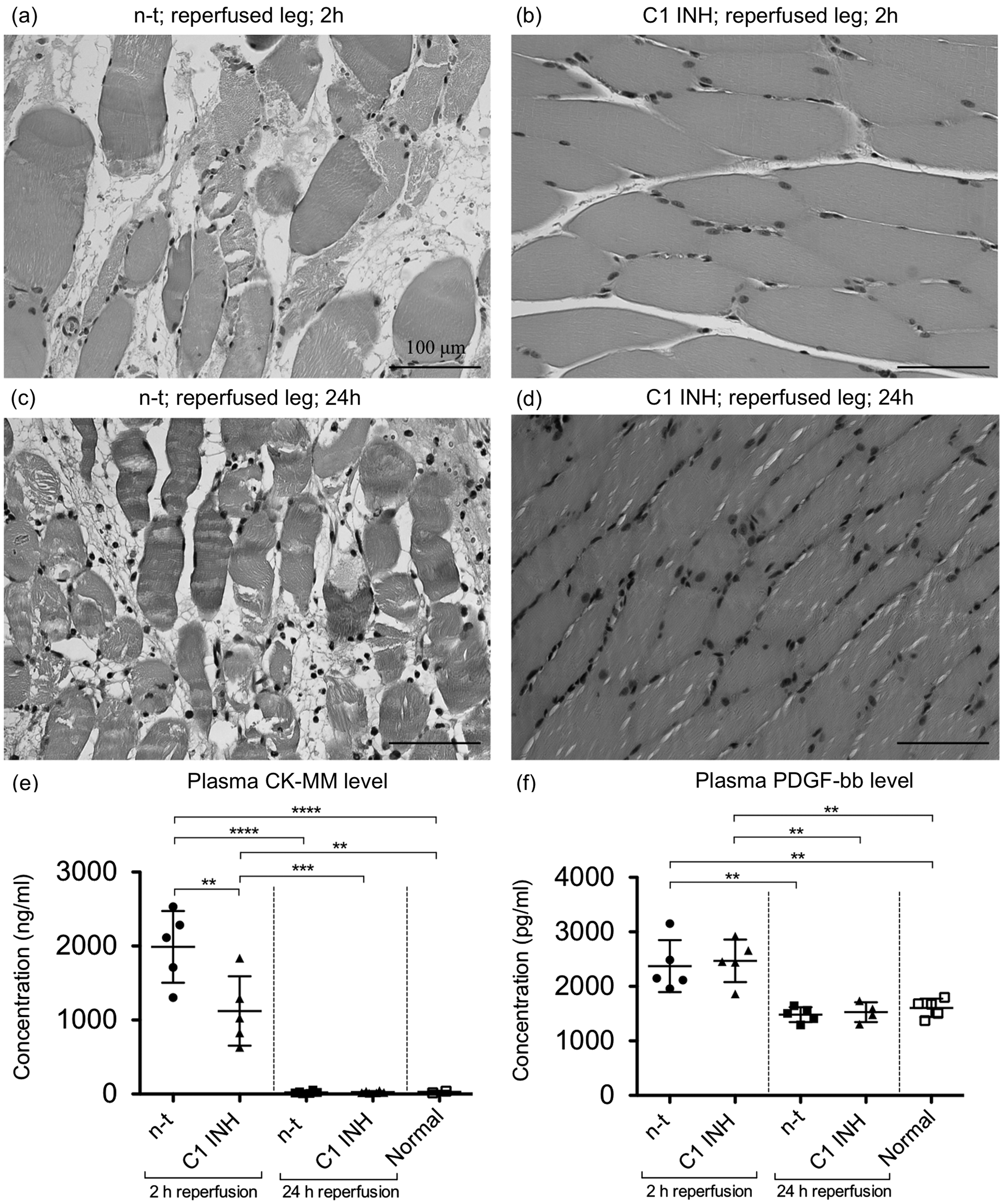
Antibody deposition in muscle tissue
Depositions of IgG and IgM in the gastrocnemius muscle tissue were assessed by immunofluorescence staining. As compared with normal rats only minimal antibody deposition was detected in the contralateral legs after 2 h and 24 h of reperfusion, respectively, whereas in the reperfused leg IgG deposition was increased at both time points (P < 0.0001). Treatment with C1 INH led to a significant decrease of IgG deposition after 2 h (P < 0.0001) as well as after 24 h (P < 0.05) of reperfusion as compared with non-treated rats (Figure 4a). Analogous results were found for IgM deposition, for which C1 INH treated rats also showed significantly less than non-treated rats after 2 h (P < 0.05) and 24 h (P < 0.0001) of reperfusion (Figure 4f).
Effect of C1 esterase inhibitor (C1 INH) on immunoglobulin G (IgG) and immunoglobulin M (IgM) depositions in gastrocnemius muscle after 2 h and 24 h of reperfusion. (a, f) Quantitative analysis of immunofluorescence stainings. (b–e) and (g–j) These show representative images of IgG and IgM depositions, respectively. One-way ANOVA followed by Bonferroni’s post hoc test for significance was used. Error bars indicate mean ± SD. *P < 0.05; **P < 0.01; ***P < 0.001; ****P < 0.0001. con: contralateral leg, rep: reperfused leg, n-t: non-treated.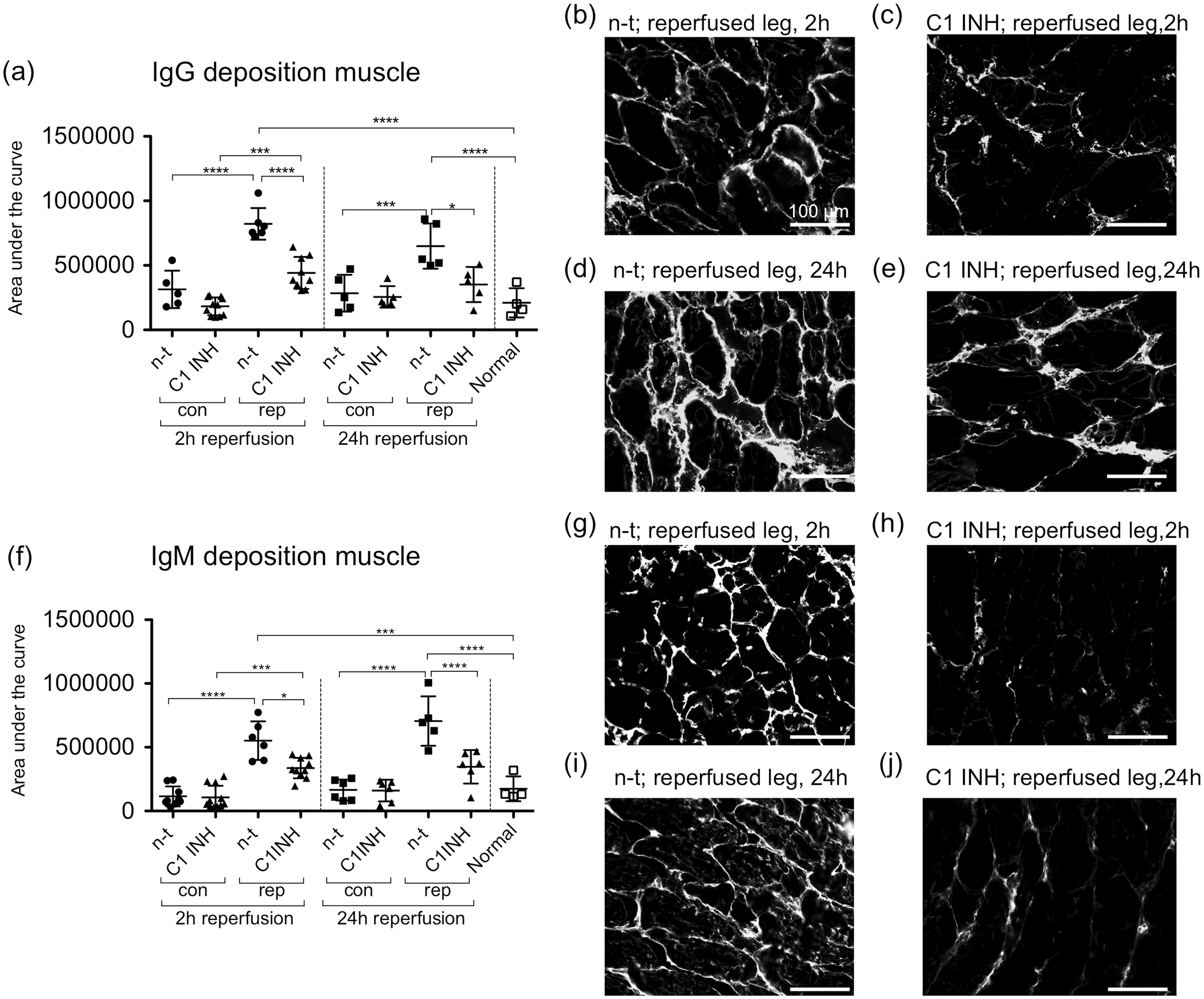
Analysis of C3b/c, factor B and fibrin depositions in muscle tissue
Analysis of C3b/c deposition revealed no significant differences between all groups after 2 h reperfusion. However, after 24 h of reperfusion non-treated rats presented C3b/c deposition in the contralateral as well as the reperfused legs, which was significantly reduced (P < 0.001) in the C1 INH treated rats (Figure 5a). In contrast to C3b/c deposition, factor B deposition was found in the contralateral and reperfused legs after 2 h and 24 h of reperfusion and was significantly reduced in the C1 INH treated rats (P < 0.01). However, after 24 h of reperfusion C1 INH could not significantly reduce factor B deposition in the reperfused legs (Figure 5f). Furthermore, we analyzed the effect of C1 INH on fibrin deposition after 2 h and 24 h of reperfusion and found no fibrin after 2 h of reperfusion as compared with the normal rats. After 24 h of reperfusion non-treated rats demonstrated fibrin deposition in the contralateral and reperfused legs, which was significantly reduced (P < 0.05) in the C1 INH treated rats (Figure 5k).
Assessment of C3b/c, factor B and fibrin depositions after 2 h and 24 h of reperfusion. (a, f, k) Quantitative analysis of immunofluorescence stainings. (b–e), (g–j) and (l–o) These show representative images of C3b/c, factor B and fibrin depositions, respectively. ‡All non-treated groups are significantly different from normal rats. One-way ANOVA followed by Bonferroni’s post hoc test for significance was used. Error bars indicate mean ± SD. *P < 0.05; **P < 0.01; ***P < 0.001; ****P < 0.0001. C1 INH: C1 esterase inhibitor, con: contralateral leg, rep: reperfused leg, n-t: non-treated.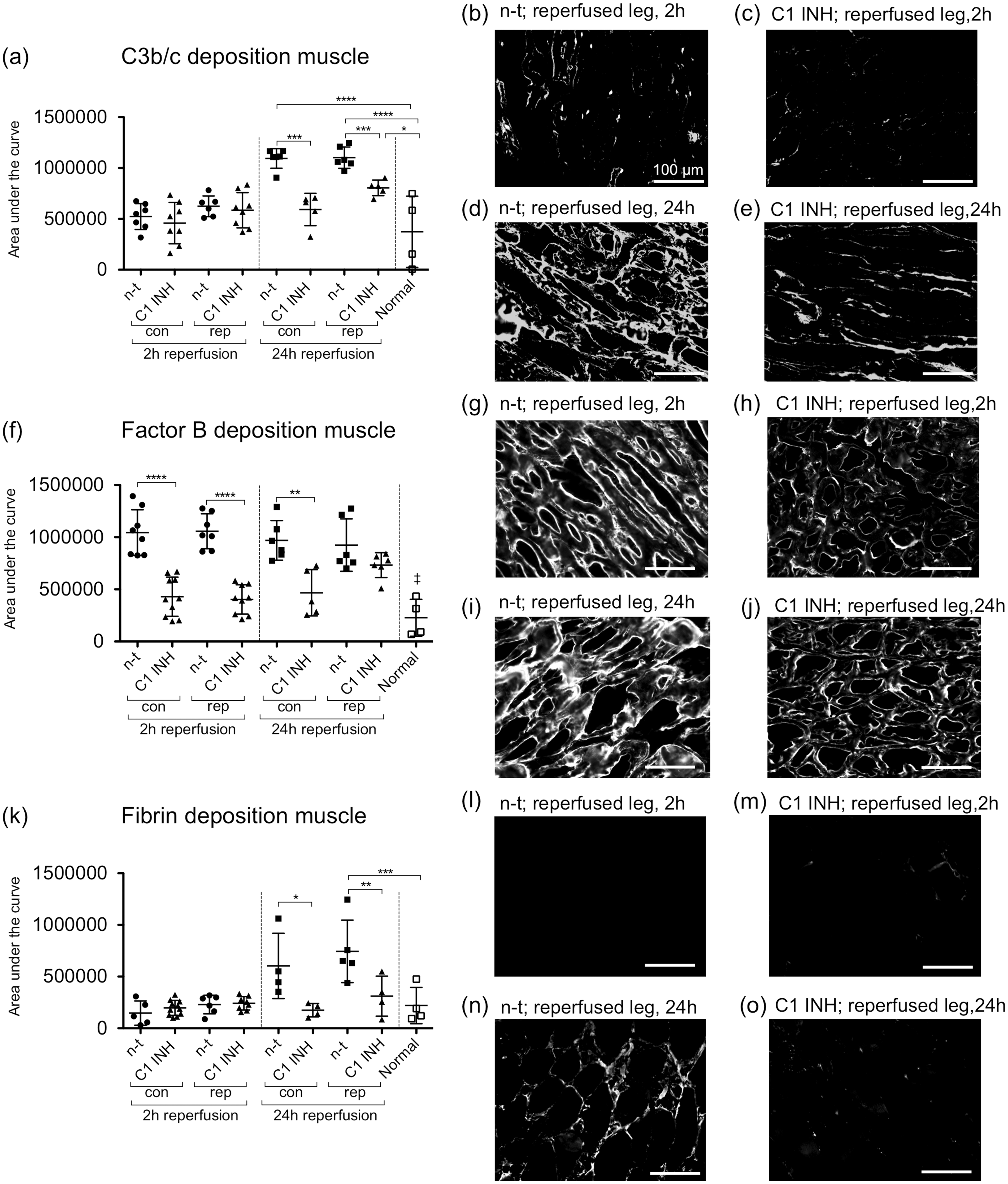
Discussion
In order to refine rat IRI models and to enable comparability between different studies we wanted to determine the parameters for which it is necessary to subject rats to 24 h of reperfusion and for which 2 h of reperfusion are sufficient to reduce potential pain and distress. To assess differences between early (2 h) and late (24 h) reperfusions in a model of acute limb IRI, we selected eight parameters, including muscle edema formation, muscle viability, distant lung edema formation, plasma CK-MM level, plasma PDGF-bb level, antibody, complement components C3b/c and factor B as well as fibrin depositions, and analyzed the effect of the drug candidate C1 INH. The muscle edema test was used for a first screening of the efficacy of C1 INH to reduce the accumulation of body fluids in muscle tissue. Edema formation was detected after 2 h as well as after 24 h of reperfusion and was significantly reduced by C1 INH treatment. C1 INH is a potent regulator of the kinin system, which is a key mediator in edema response. 17 C1 INH interacts with kallikrein and FXIIa and may therefore prevent edema formation.18,19 Another key parameter in the present study was the assessment of distant lung injury, by analyzing lung edema formation. Lung edema formation caused by limb IRI is a severe clinical problem, which should also be considered in animal experiments. 20 Investigation of lung injury by analysis of lung edema revealed that after 2 h of reperfusion no detectable lung edema was present, whereas after 24 h of reperfusion significant edema formation occurred, which was reduced by C1 INH treatment. MTT assay was used for the assessment of muscle viability. Analysis of the effect of C1 INH treatment on muscle viability showed significant improvement after 24 h of reperfusion as compared with non-treated groups, whereas after 2 h of reperfusion viability was not severely affected. Furthermore, we measured CK-MM in plasma samples, as it plays a significant role in energy homeostasis of cells with high energy requirements, such as myocytes, and its level is routinely used as an indicator of skeletal muscle injury. 21 Under normal conditions CK-MM is located in the cytosol and mitochondria of tissues and leaks into the blood due to muscle cell rupture, cell damage and disease. 22 High CK-MM levels in plasma were detected after 2 h of reperfusion and could be significantly reduced by C1 INH treatment. However, after 24 h of reperfusion only very low levels of CK-MM were measured, as CK-MM is a marker of early detection of muscle injury and has a serum half-life of only about one hour in rats. 23 Additionally, we looked at the plasma level of PDGF-bb, which is known to have angiogenic effects, regulates the tonus of blood vessels and is involved in wound healing.24,25 At low levels PDGF-bb is expressed in the endothelium, platelets and macrophages, which increases following mechanical injury and atherosclerosis. 26 As compared with normal values, we found elevated plasma levels after 2 h, but not after 24 h of reperfusion. Similar results were shown in a study of tourniquet-induced IRI in hand surgery, where PDGF-bb was significantly increased compared with normal values as soon as after 10 min of reperfusion. 27 Our data showed that C1 INH treatment did not reduce plasma PDGF-bb levels after 2 h of reperfusion. However, we cannot conclude to which source the increased plasma PDGF-bb level can be attributed.
Furthermore, we analyzed the deposition of antibodies, complement fragments, as well as fibrin, which provide reliable and quantifiable data which reveal the underlying mechanisms of actions of drug candidates in IRI. Analysis of antibody as well as complement deposition was selected, as the hypoxic state of ischemia leads to an alteration of proteins, such as non-muscle myosin heavy chain type II or annexin IV, which function as neoepitopes for natural antibodies.28,29
We found depositions of IgM and IgG in the reperfused leg after 2 h and 24 h of reperfusion, which were significantly reduced by C1 INH treatment. In the contralateral legs only minimal antibody deposition was detected, suggesting that neoepitopes are only locally exposed and that IRI did not cause systemic formation of neoepitopes.
Furthermore, we investigated the key component of the complement system C3b/c. After 24 h of reperfusion C3b/c was significantly deposited in the contralateral and reperfused legs of non-treated rats and could be markedly reduced by C1 INH treatment, whereas after 2 h of reperfusion only low amounts of C3b/c deposits were detected. Analysis of the complement component factor B revealed high depositions after 2 h and 24 h of reperfusion in the contralateral and reperfused muscles, which were significantly reduced by C1 INH treatment. After 24 h of reperfusion no significant reduction of factor B by C1 INH was detected in the reperfused leg. It is known that C1 INH down-regulates the function of the alternative pathway, including C3b/c as well as factor B, which fits in with our data. 30
Reperfusion following ischemia mediates the activation of the coagulation system. We analyzed the role of the coagulation system by measuring fibrin deposition in the gastrocnemius muscle. After 2 h of reperfusion fibrin deposition in the reperfused leg was not detected, but it was after 24 h of reperfusion. C1 INH treatment significantly reduced fibrin deposition compared with the non-treated rats, which is in line with earlier findings, where it was shown that C1 INH plays a crucial role in the coagulation system, as it is the main inhibitor of factor XIa and also modulates thrombin activity.31,32
Usefulness of measurement of parameters after 2 h or 24 h reperfusion.

Beneficial effects on acute lung injury were shown through anti-platelet and even anti-apoptotic mechanisms of isoflurane.33,34 However, any bias due to reduced edema formation as well as attenuated reperfusion injury attributed to isoflurane anesthesia can be excluded in our study, since non-treated as well as C1 INH treated rats received identical anesthetics. No control group without buprenorphine analgesia was included, as analgesic treatment is mandatory for reducing pain and post-surgical stress response during the 24 h reperfusion period. The effect of drug candidates on antioxidant enzymes, including superoxide dismutase, catalase, glutathione, malondialdehyde and also protein oxidation levels are frequently measured in reperfusion injury models. Here we did not analyze these factors because this study focused on the role of the plasma cascade systems in IRI. 35
In contrast to Thaveau et al., who reported that the contralateral leg may be used as a control in the experimental setting of unilateral hind limb ischemia, we also found C3b/c, factor B and fibrin depositions in the contralateral muscle. 36 Whether or not the contralateral leg may be used as a control in unilateral hind limb ischemia should therefore be carefully considered based on the parameters to be assessed.
In summary, this study demonstrates that an acute model with 2 h of reperfusion is sufficient to assess basic parameters of IRI in the rat hind limb setting and may therefore be suitable for a first screening of drug candidates. However, for in-depth analysis of mechanisms of actions, muscle viability and distant organ damage, rats need to be subjected to 24 h of reperfusion.
Footnotes
Acknowledgments
The authors wish to thank Dr Rolf Spirig, Dr Sylvia Miescher and Dr Martin Spycher (CSL Behring AG, Bern, Switzerland) as well as Dr Marc W Nolte (CSL Behring GmbH, Marburg, Germany) for providing C1 INH (Berinert) and helpful discussions. Further, we wish to thank Dr Yara Banz and Dr Boris Leuenberger and team at the Institute of Pathology for the preparation of histology slides. This study was performed with the support of the Microscopy Imaging Center (MIC), University of Bern.
