Abstract
Inhalation anesthesia with isoflurane is a well-established and safe method used in small laboratory animals. In most cases oxygen is used as a carrier gas for isoflurane, but room air or mixtures of oxygen with air or nitrous oxide are also being used. Anesthesia is therefore administered using different fractions of inspired oxygen (FiO2), and this may have consequences for the outcome of experiments. The aim of the present study was to investigate the influence of FiO2 on rat hind limb ischemia/reperfusion injury and to refine the used inhalation anesthesia. Male Wistar rats were subjected to 3.5 h of ischemia and 2 h of reperfusion, and divided into three groups according to FiO2 in the O2/air/isoflurane anesthesia gas mixture: 40%, 60%, and 100% O2. Normal, healthy rats were used as controls. Muscle edema and creatine kinase MM, a marker for myocyte necrosis, were significantly increased with 40% FiO2 as compared with 100% FiO2 (P < 0.05). Partial pressure of oxygen, oxygen saturation, and oxyhemoglobin were significantly higher in the 100% O2 group as compared with 40% O2. No significant differences were detected for other parameters, such as the oxidative stress markers malondialdehyde and superoxide dismutase. We conclude that a refined inhalation anesthesia setting using 40% FiO2, reflecting more or less the clinical situation, leads to a more severe and more physiologically relevant reperfusion injury than higher FiO2. Oxidative stress did not correlate with FiO2 and seemed to have no influence on reperfusion injury.
Inhalation anesthesia systems using isoflurane are often used in laboratory animal studies because they allow for a safe and easily adjustable anesthesia. In many studies,1–3 the carrier gas for isoflurane is oxygen. However, room air or a mixture of oxygen and air or nitrous oxide have also been used,4–6 resulting in differences in the fraction of inspired oxygen (FiO2) in experimental rodent anesthesia protocols. FiO2 is the fraction or percentage of oxygen in the anesthesia gas mixture inhaled by the patient or laboratory animal. Room air is composed of 21% oxygen and 79% nitrogen, which is equivalent to an FiO2 of 21%, while pure oxygen has an FiO2 of 100%. We investigated whether different FiO2 in the anesthesia gas mixture might influence the outcome of our rat hind limb ischemia/reperfusion injury (IRI) model.
Reperfusion injury of the lower extremity is a common clinical consequence of surgical interventions performed under tourniquet application as well as the treatment of peripheral vascular diseases such as thrombotic occlusion and embolism. Basically, IRI is caused by prolonged lack or massive restriction of blood supply to a tissue or organ, followed by re-establishment of the circulation of oxygenated blood. Paradoxically, reperfusion can aggravate ischemic tissue damage, causing an inflammatory reaction which may lead to both local injury and systemic complications as seen in clinical studies,7,8 and experimentally in rat hind limb IRI models.9,10
During IRI, a complex cascade of inflammatory reactions occurs, which have been reviewed in detail previously.11,12 During the ischemic stage, nutrient depletion and hypoxia may lead to neoepitopes being expressed particularly in the affected endothelium, which may be recognized by preformed antibodies circulating in the blood. This triggers the classical and lectin pathways of complement activation, as shown in mouse models of intestinal IRI.13,14 Deposition of complement proteins on the endothelial surface was shown to be involved in the vascular leakage in human intestinal IRI 15 and in a mouse mesenteric IRI model. 16 Complement activation also leads to anaphylatoxin C5a formation, which is a potent chemoattractant for proinflammatory cells. It also initiates transforming the endothelium from an anticoagulant and anti-inflammatory to a procoagulant and proinflammatory state. Models of rat liver, renal and intestinal IRI showed that to be the case.17–19
It has also been proposed that reintroducing oxygen during the reperfusion phase may be a triggering factor for an overproduction of reactive oxygen species (ROS), which are generated by activated endothelial cells.20,21 Overproduced ROS thus causes oxidative stress. For instance, in rat liver and cerebral IRI studies,22,23 oxidative stress has been demonstrated to occur and plays an important role in the pathophysiology of reperfusion injury, and experimental treatments based on ROS-scavenging have shown beneficial effects.24–26 However, in a recent clinical study on kidney and heart reperfusion injury, biomarkers of oxidative damage were not released upon reperfusion, so the causal involvement of ROS and oxidative stress in IRI has been questioned. 27 Additionally, tissue oxygen tension (tpO2) decreased during ischemia and recovered to baseline levels during reperfusion in a skeletal muscle IRI model. 28 However, so far no studies have investigated whether the oxidative stress, blood oxygenation and tpO2 may be linked to FiO2 in inhalation anesthesia. Nor is it clear if they affect experimental reperfusion injury.
To address the above issues, and to refine FiO2 in small animal inhalation anesthesia, a rat hind limb IRI model was used, in which FiO2 was varied from 40% to 100%. The effect of FiO2 on reperfusion injury was assessed by measuring edema formation, muscle viability, creatine kinase MM (CK-MM) in plasma, deposition of complement and fibrin in tissue.
Materials and methods
Ethics statement
Animal housing and experimental protocols were approved by the Cantonal Veterinary Office, Bern, Switzerland, under license No. BE 05/12, and were in accordance with Swiss Animal Protection Law and conform to Directive 2010/63/EU of the European Parliament and of the Council on the Protection of Animals used for Scientific Purposes, as well as to the National Institutes of Health (NIH) Guide for the Care and Use of Laboratory Animals.
Animals
Adult male Wistar rats aged 7–9 weeks, weighing between 250 and 350 g, were purchased from Janvier Labs (Le Genest, France) and used in all the experiments. The rats were kept in groups of three animals in clear 1500 cm2 Euro-standard type IV S cages (Tecniplast, Buguggiate, Italy) with overall dimensions of 48 × 37.5 × 21 cm (length × width × height). Unrestricted feeding was with standard pelleted maintenance diet (Provimi Kliba, Kaiseraugst, Switzerland) and water ad libitum. Poplar wood chips were used as bedding material (Lignocel, Provimi Kliba). Paper tissue, a piece of poplar wood with a size of 20 × 3.5 × 3.5 cm (length × width × height) and a black polyvinyl chloride (PVC) tube of 16 cm length and 7.3 cm diameter were provided as environmental enrichment. Cages were individually ventilated at 20 ± 2 ℃ and 45–65% relative humidity with a circadian rhythm of 12/12 h. During the light cycle the animals were exposed to a light intensity of 200 lux.
Experimental design
Animals were subjected to 3.5 h of ischemia and 2 h of reperfusion, and divided into three groups according to FiO2 in the anesthesia gas mixture (O2/air/isoflurane): group 1 (n = 7) 40% O2, group 2 (n = 6) 60% O2, and group 3 (n = 8) 100% O2. Four normal, healthy rats were used as controls for comparison. No substances were administrated to the animals. The numbers of animals per group were calculated based on the paper ‘Guidelines for the design and statistical analysis of experiments using laboratory animals’ 29 and the ARRIVE guidelines. 30 Data on edema formation (wet/dry ratio) of previous experiments 31 have revealed a difference in the means between experimental and control groups of 1.1 and a standard deviation of 0.5. Sample size calculations based on these data have resulted in at least six animals per group. Due to technical problems with blood gas analysis for some animals, the n numbers had to be increased to n = 7 for the 40% and n = 8 for the 100% FiO2 groups. For normal rats (control group), we observed a very small standard derivation (<1%) in our earlier study. 31 We therefore decided to limit this group to n = 4 in order to keep the number of animals to a minimum.
Anesthesia and monitoring
Anesthesia was induced by 3% isoflurane in the oxygen/air mixture according to the experimental group in a clear, polystyrene induction chamber. Anesthesia throughout the whole IRI procedure was then maintained with 2% isoflurane in the same oxygen/air mixture in a standard rat nose mask (Provet, Lyssach, Switzerland). The animals were placed on a heating pad and rectal temperature kept at 37 ± 1 ℃. Basic parameters (heart rate, breath rate, pulse distension, breath distension and oxygen saturation [sO2]) were continuously monitored and recorded every 5 min using a MouseOx plus system (Starr Life Sciences Corp, Oakmont, PA, USA) and MouseOx plus software version 1.4.9.
Surgical procedure
All animals were placed supine, and the fur on their neck and on both hind limbs was shaved. The left carotid artery was cannulated with a polyethylene tubing (product code: 800/100/200; Portex, Smiths Medical, Labhardt AG, Basel, Switzerland) for blood sampling. The cannula was flushed with approximately 1 mL of heparin solution (100 IE/mL in saline; Laboratory Dr G Bichsel AG, Interlaken, Switzerland) prior to insertion in the carotid artery and filled with heparin solution after each blood sampling to prevent clogging. Care was taken to prevent heparinization of the animals.
Baseline measurements for blood gas and blood chemistry analyses and tpO2 in the gastrocnemius muscle (details below) were performed before surgery. The used rat hind limb IRI model was performed as described earlier.32,33 In brief, two microvascular clamps (B1-V; S&T, Neuhausen, Switzerland) were used to block arterial blood supply proximal to the deep femoral artery branch while a tourniquet, connected to a weight of 450 g, was placed underneath the femoral artery, vein and the sciatic nerve around the thigh as high as possible. The rats underwent 3.5 h of ischemia followed by reperfusion for 2 h and stayed under isoflurane anesthesia during the whole time. At the end, the rats were sacrificed by exsanguination and tissue samples from the soleus and gastrocnemius muscles as well as the lung were taken for further analysis.
Measurement of muscle and lung edema
To assess edema formation in the muscle and lung, two pieces of tissue from the gastrocnemius muscle in the contralateral and reperfused legs, as well as the lung, were taken, washed in phosphate-buffered saline (PBS), blotted dry and weighed immediately to get the wet weight. The pieces were then dried at 80 ℃ overnight until constant dry weights were obtained. The ratio of wet:dry weight was regarded as an indicator for edema formation.
Muscle viability
Muscle viability was determined using MTT (3-(4,5-dimethyl-2-thiazolyl)-2,5-diphenyl-2H-tetrazolium br-omide, M2128; Sigma-Aldrich, St Louis, MO, USA). The principle is that mitochondrial dehydrogenases of living cells reduce the MTT tetrazolium compound to its purple formazan product. Briefly, muscle tissue was incubated with 0.1 mg MTT/mL PBS at 37 ℃ overnight, then transferred to 100% isopropanol at 37 ℃ for 6 h. At the end, optical density (OD) of the supernatant was read at 570 nm (reference wavelength 690 nm, Infinite M1000 spectrophotometer; Tecan, Männedorf, Switzerland). OD/mg dry weight was used to quantitate muscle viability.
CK-MM in plasma
To assess myocyte damage, CK-MM was measured in EDTA plasma using an enzyme-linked immunosorbent assay (ELISA) kit (2112-2; Life Diagnostics Inc, West Chester, PA, USA). The assay was performed according to the manufacturer’s protocol and the samples were analyzed in duplicate at baseline and at the end of reperfusion. Concentrations were determined by comparing with standards provided with the kit.
Histopathological examination
To assess histopathological changes, one piece of gastrocnemius muscle from both the contralateral and reperfused legs, as well a piece from the lung, were washed in PBS, blotted dry and fixed in 4% formalin until paraffin embedding and staining with hematoxylin and eosin (H&E). Hemorrhage, neutrophil infiltration, myocyte damage, and tissue edema were examined by a professional pathologist who was blinded to this study.
Immunofluorescence
To detect deposition of antibodies, complement components and fibrin, as well as heparan sulfate expression, immunofluorescence staining was performed. Briefly, the gastrocnemius muscle from both the contralateral and reperfused legs was taken and stored at –25 ℃ for further use. Sections with 5 µm thickness were fixed in acetone and rehydrated in tris-buffered saline (TBS). Primary antibodies were added and incubated at 4 ℃ overnight, followed by incubation of secondary antibodies and 4′,6-diamidino-2-phenylindole (DAPI; Boehringer, Roche Diagnostics, Indianapolis, IN, USA) for staining of the nuclei at room temperature for 1 h. Finally, the slides were mounted and covered. The following primary antibodies were used: goat anti-rat IgM (3020-08; Southern Biotech, Birmingham, AL, USA), goat anti-rat IgG (3030-08; Southern Biotech), rabbit anti-human C3b/c (A0062; Dako, Baar, Switzerland), goat anti-human C6 (A307; Quidel, San Diego, CA, USA), rabbit anti-human fibrinogen (F0111; Dako), and mouse anti-heparan sulfate (370255; Amsbio, Abingdon, UK). Secondary antibodies and conjugates were donkey anti-goat Alexa488 (C 2306; Sigma-Aldrich), goat anti-mouse FITC (115-097-020; Jackson ImmunoResearch Laboratories, West Grove, PA, USA), and streptavidin-Cy3 (S6402; Sigma-Aldrich). Images were taken with a fluorescent microscope (Leica DMI 4000B; Leica Microsystems Schweiz, Heerbrugg, Switzerland) and analyzed by ImageJ (NIH, Bethesda, MD, USA) and GraphPad Prism 6 software (GraphPad Software Inc, San Diego, CA, USA).
Blood gas and blood chemistry analyses
Samples of 150 µL heparinized arterial blood were taken from the carotid catheter at baseline, end of ischemia, 1 h of reperfusion and 2 h of reperfusion. Partial pressures of oxygen and carbon dioxide (pO2, pCO2), sO2, pH, base excess (BE), the concentration of total hemoglobin (tHb), oxyhemoglobin (O2Hb), and deoxyhemoglobin (HHb), the hematocrit (Hct), the concentrations of the electrolytes Na+, K+, Ca2+, HCO3− and Cl−, as well as concentrations of glucose (Glu), lactate (Lac), and osmotic concentration (Osm) were measured on a Cobas b 123 POC blood gas analyzer (Roche, Basel, Switzerland).
Tissue oxygen tension
To monitor oxygen delivery and transport from vessels to tissue during IRI, tpO2 was measured by inserting an oxygen probe (CC8, Licox; Integra Neurosciences, Plainsboro, NJ, USA) and a temperature probe (CC1.SB, Licox; Integra Neurosciences) into the gastrocnemius muscle through a 16 gauge introducer catheter (393229, BD Venflon Pro Safety; BD Europe, Allschwil, Switzerland). The probes were connected to a digital Licox monitor (Licox CMP; Integra Neurosciences). Time points at which tpO2 was recorded were baseline, 1 h of ischemia, 2 h of ischemia, 3 h of ischemia, 3.5 h of ischemia, 1 h of reperfusion, and 2 h of reperfusion.
Oxidative stress
The oxidative stress markers, malondialdehyde (MDA) formed from lipid peroxidation, and superoxide dismutase (SOD), were determined in plasma by colorimetric and fluorometric methods, respectively, using a lipid peroxidation (118970; Abcam, Cambridge, UK) and an SOD assay kit (19160; Sigma-Aldrich). The assays were performed according to the manufacturers’ protocols. Concentration of MDA was determined by interpolation from a standard curve, while SOD activity was calculated using the equation provided by the manufacturer.
Statistical analysis
All data are presented as a mean ± standard deviation (SD). Statistical analyses were done by one-way analysis of variance (ANOVA), followed by two post hoc tests with GraphPad Prism 6 software: Bonferroni’s (to compare a set of selected means) and Dunnett’s (to compare means with the control group). To compare different inter-group time points (such as breath rate and heart rate, blood gas analysis, tpO2 and oxidative stress), the areas under the curve (AUC) were calculated for each experiment, and the groups were compared by one-way ANOVA with Bonferroni’s post hoc test. P values <0.05 were considered statistically significant.
Results
Edema formation
In all groups, ischemia and reperfusion led to significantly higher wet/dry ratios – and thus edema formation – in the reperfused limbs as compared with the contralateral ones as well as with the normal, healthy controls. A tendency for increased edema formation after 3.5 h of ischemia and 2 h of reperfusion was observed with decreasing FiO2, with a significant difference between the 40% and 100% O2 groups. The observed wet/dry ratios were 6.0 ± 0.62 in the 40% O2 group versus 5.4 ± 0.36 in the 100% O2 group, P = 0.049 (Figure 1a). No edema was formed in the contralateral limbs of all three groups when compared with the hind limbs of the normal, healthy rats.
Edema formation in the muscle (a) and lung (b), muscle viability (c), and creatine kinase MM (CK-MM) in plasma (d). Error bars indicate mean ± SD. *P < 0.05; **P < 0.01; ***P < 0.001; ****P < 0.0001. C: contralateral limb; R: reperfused limb.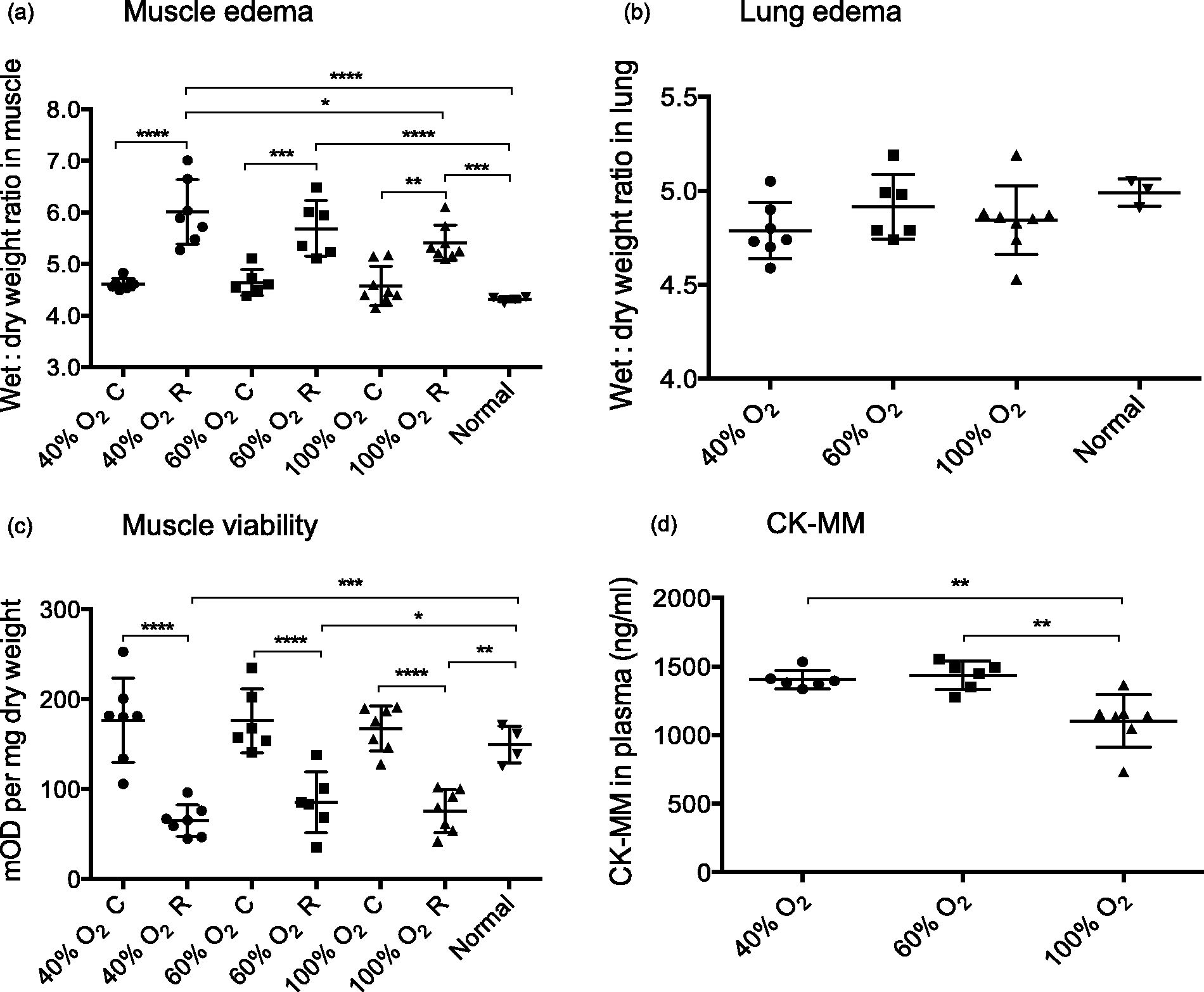
To assess distant organ damage, wet/dry ratios of the lungs were also measured, but no edema formation was found in the lungs in this model with 2 h of reperfusion (Figure 1b).
Muscle viability
Muscle viability was reduced in the reperfused limbs of all three experimental groups as compared with the normal controls as shown in Figure 1c. However, no significant differences were found between experimental groups. No reduction of muscle viability was found in the contralateral limbs as compared with the normal controls.
CK-MM in plasma
CK-MM levels in plasma at the end of reperfusion – indicating muscle cell necrosis – were 1405 ± 68.2 ng/mL in the 40%, 1436 ± 103.0 ng/mL in the 60%, and 1103 ± 190.8 ng/mL in the 100% O2 groups. The values in the 40% and 60% O2 groups were significantly elevated as compared with the 100% O2 group (P = 0.0031 and P = 0.0013, respectively; Figure 1d). Baseline values were all below the detection limit of the ELISA kit, i.e. less than 90 ng/mL.
Histopathological findings
Histopathological evaluation by H&E staining revealed intermediate damage to the gastrocnemius muscle tissue after 2 h of reperfusion. Myocyte destruction and muscular edema were observed in each experimental group to a similar extent, while only low levels of neutrophil infiltration were seen (Figures 2a–d). No hemorrhage was observed in all three experimental groups. Moreover, no histopathological changes were detectable in the lungs after 2 h of reperfusion (data not shown).
Representative images of hematoxylin–eosin stained muscle tissue in the 40% O2 (a), 60% O2 (b), 100% O2 (d) groups and normal controls (d), as well as deposition of IgG – quantitative analysis (e) and representative images (f–i), and IgM – quantitative analysis (j) and representative images (k–n). Error bars indicate mean ± SD. **P < 0.01; ***P < 0.001; ****P < 0.0001. C: contralateral limb; R: reperfused limb. Scale bar represents 100 µm.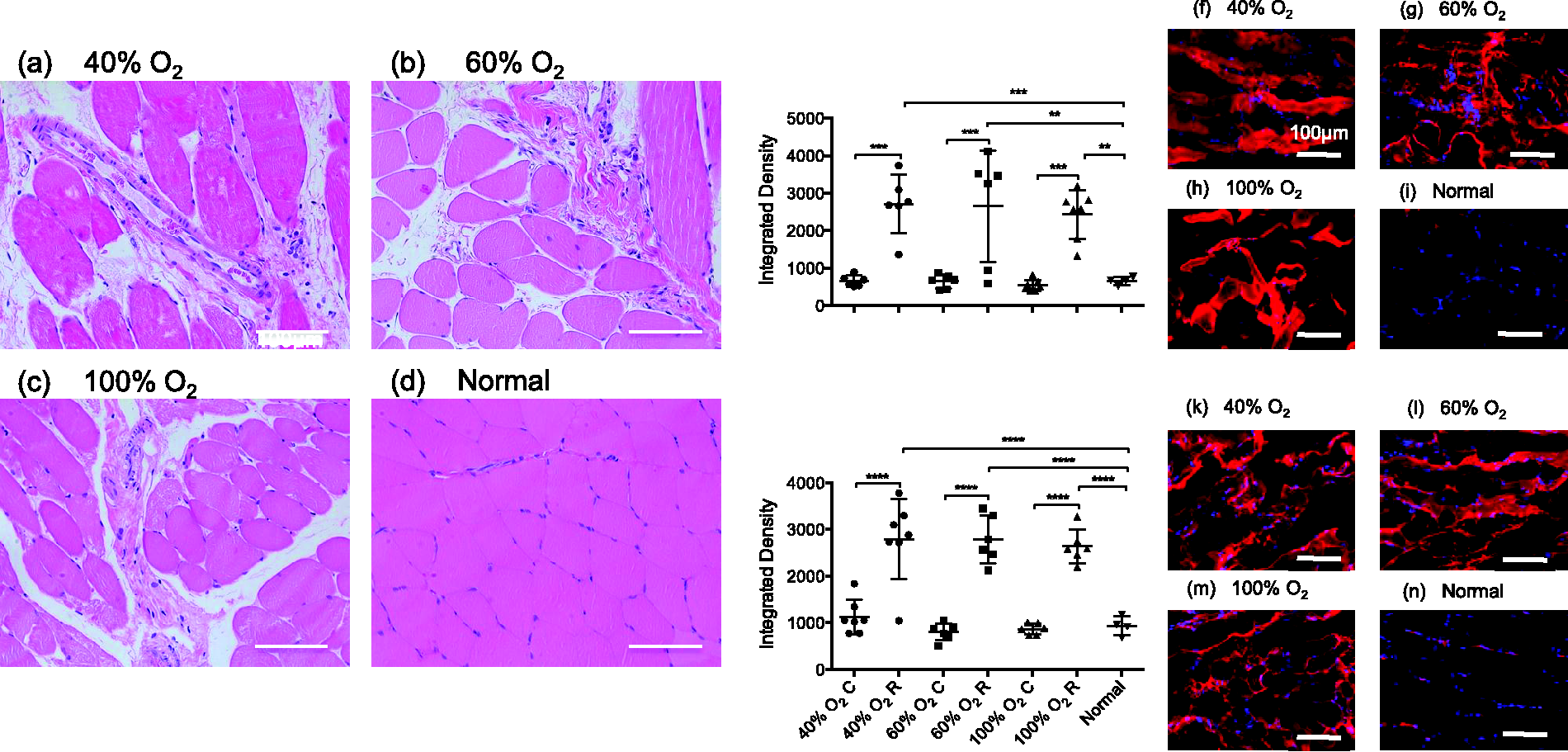
Deposition of antibodies and complement
Depositions of IgG and IgM, and the complement proteins C3b/c and C6 were detected by immunofluorescence. Fluorescence intensities were quantified as integrated density by Image J and compared between groups. Both IgG and IgM deposits were significantly increased in the reperfused limbs of all experimental groups as compared with the normal controls (P < 0.0001; Figures 2e–n). For complement, an FiO2 of 40% and 60% led to significantly higher deposition of C3b/c and C6, as compared with the normal control group (P < 0.0001), whereas deposition of complement was not significantly elevated in the rats with an FiO2 of 100% (Figures 3a–j). However, no significant differences were found in the tissue of reperfused limbs between the 40%, 60% and 100% FiO2 groups for deposition of antibodies and complement. Similarly, no significant differences were found for these markers between the contralateral limbs and the normal controls.
Deposition of C3b/c (a–e), C6 (f–j), fibrin (k–o), and heparan sulfate expression (p–t). Error bars indicate mean ± SD. *P < 0.05; **P < 0.01; ***P < 0.001. C: contralateral limb; R: reperfused limb. Scale bar represents 100 µm.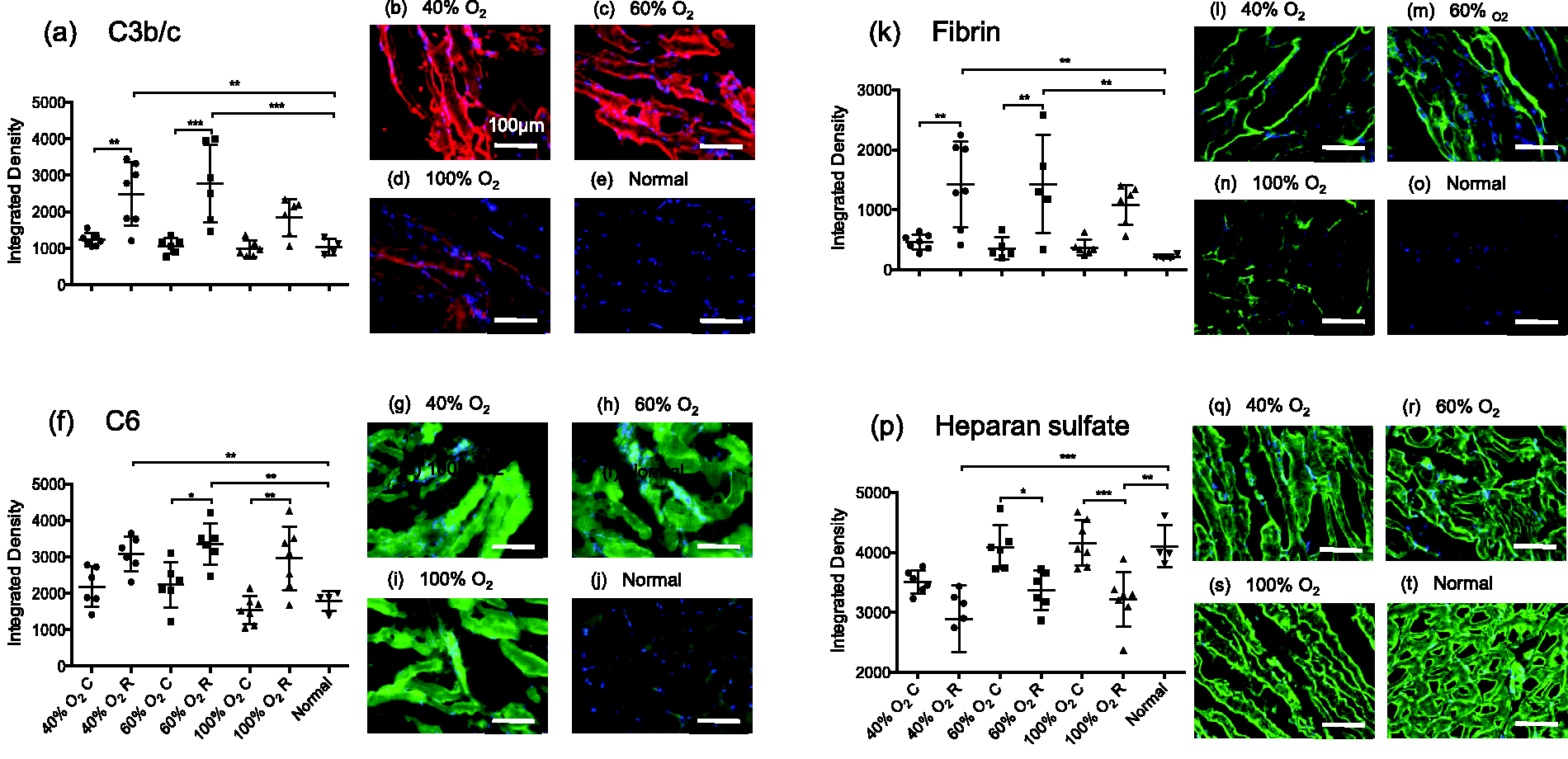
Deposition of fibrin and expression of heparan sulfate
Deposition of fibrin was measured as a marker of the activation of the coagulation system. A significantly higher level of fibrin deposition was observed in the tissue of the reperfused limbs in the 40% and 60% FiO2 groups as compared with the normal controls (P = 0.0016 and P = 00036, respectively; Figures 3k–o), but this was not the case for the 100% FiO2 group. Expression of heparan sulfate in tissue is known to be reduced as a consequence of inflammation.34,35 Both the 40% and 100% O2 inhalations led to significantly lower levels of expression of heparan sulfate in the tissue of the reperfused legs as compared with the normal controls (P = 0.0004 and P = 0.0112 respectively; Figures 3p–t). However, no significant differences were detected for the above markers in the reperfused tissue between the experimental groups, as well as between the contralateral limbs and the normal controls.
Breath rate and heart rate
All animals showed an increase in breath rates at the beginning of ischemia. By assessing AUC over the whole duration of the experiments, a significantly higher breath rate was observed in the 40% FiO2 group as compared with both the 60% and 100% FiO2 groups (P = 0.0157 and P = 0.0461, respectively; Figure 4). For heart rates, no significant intra-group changes over time or inter-group differences (as assessed by AUC) were observed.
Breath rate and heart rate. Values of breath rate (a) and heart rate (b) over time are shown on the left panels for each group. The corresponding areas under the curve are presented on the right panels. Error bars indicate mean ± SD. *P < 0.05.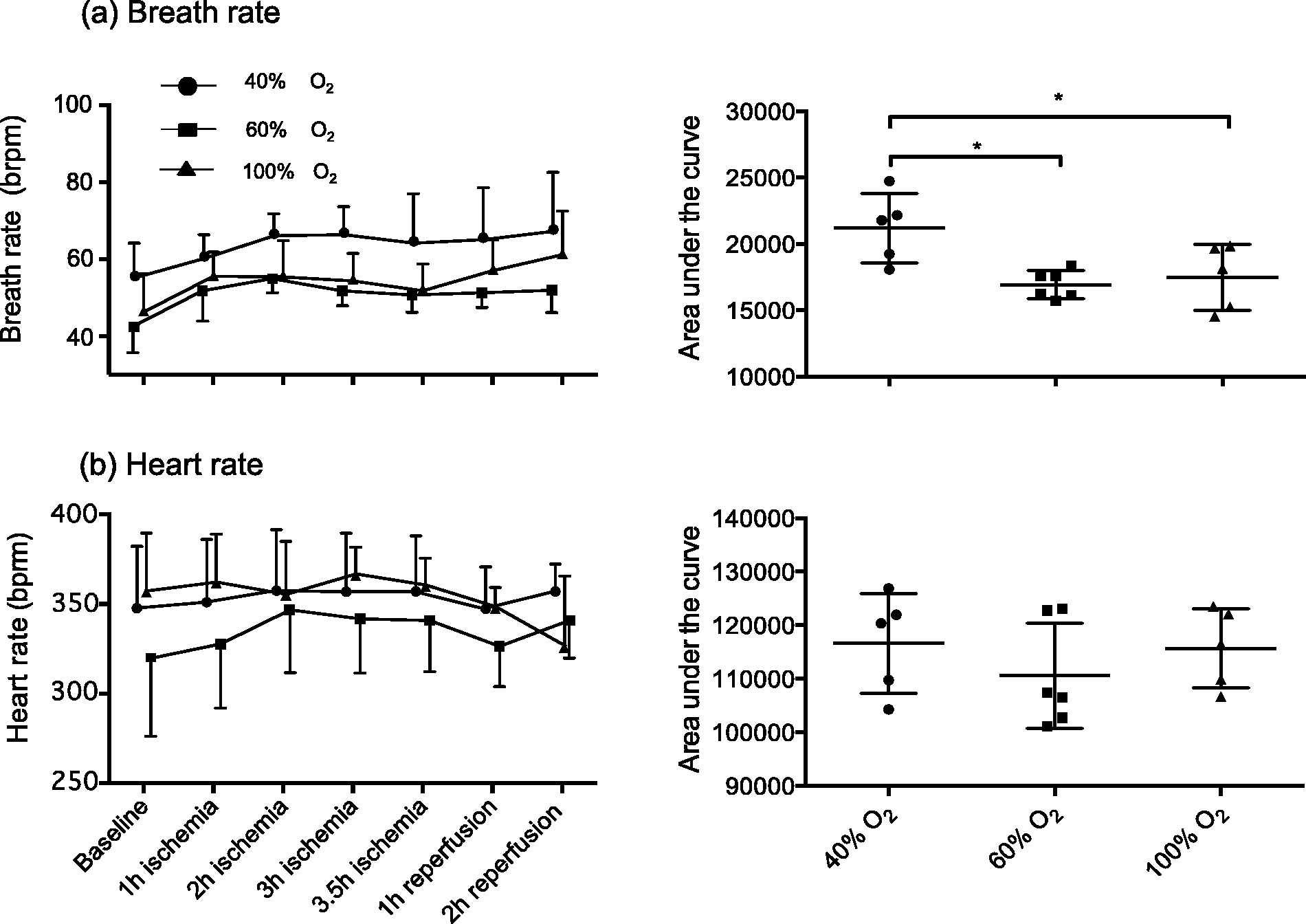
Blood gas and blood chemistry analyses
Arterial blood gas and blood chemistry analyses were performed to assess the effect of FiO2 on blood oxygenation and related parameters. For inter-group comparison, AUC data were calculated for all parameters over time. As shown in Figures 5a–d, an FiO2 of 100% led to significantly higher levels of pO2, sO2 and O2Hb over time as compared with 40% (P < 0.01), while HHb was reduced significantly (P < 0.01) in the 100% as compared with the 40% group. No significant inter-group differences were found for the rest of the parameters over time by comparing AUC data. However, we found increased concentrations of K+ in each group, while concentrations of Ca2+ and Glu dropped over time (supplementary Table 1, see http://lan.sagepub.com).
Blood gas and blood chemistry analyses. Values of pO2 (a), sO2 (b), O2Hb (c), and HHb (d) over time are shown on the left panels for each group, while the corresponding areas under the curve are presented on the right panels. Error bars indicate mean ± SD. **P < 0.01; ****P < 0.0001. pO2: partial pressure of oxygen, sO2: oxygen saturation, O2Hb: oxyhemoglobin, HHB: deoxyhemoglobin.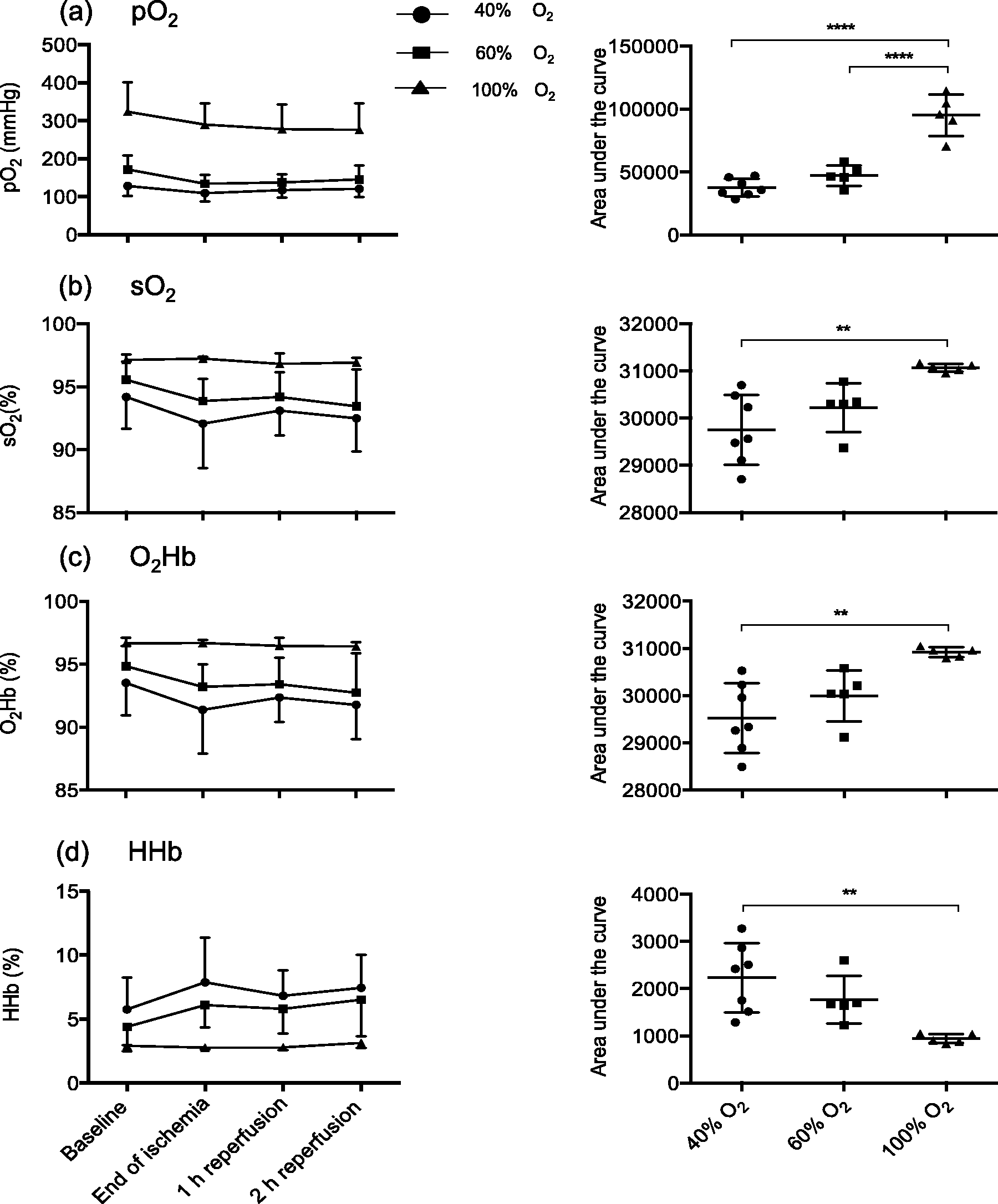
Tissue oxygen tension
Tissue oxygen tension was measured every hour to assess oxygen delivery from the blood to the tissue under different inhaled oxygen concentrations. At baseline tpO2 was between 20 and 39 mmHg, it then dropped to below 10 mmHg during ischemia and rose again during reperfusion. However, significant differences between groups for tpO2 were not observed over time (Figure 6).
Tissue oxygen tension (tpO2). Data of tpO2 over time are shown on the left panel, while the corresponding areas under the curve are displayed on the right panel. Error bars indicate mean ± SD.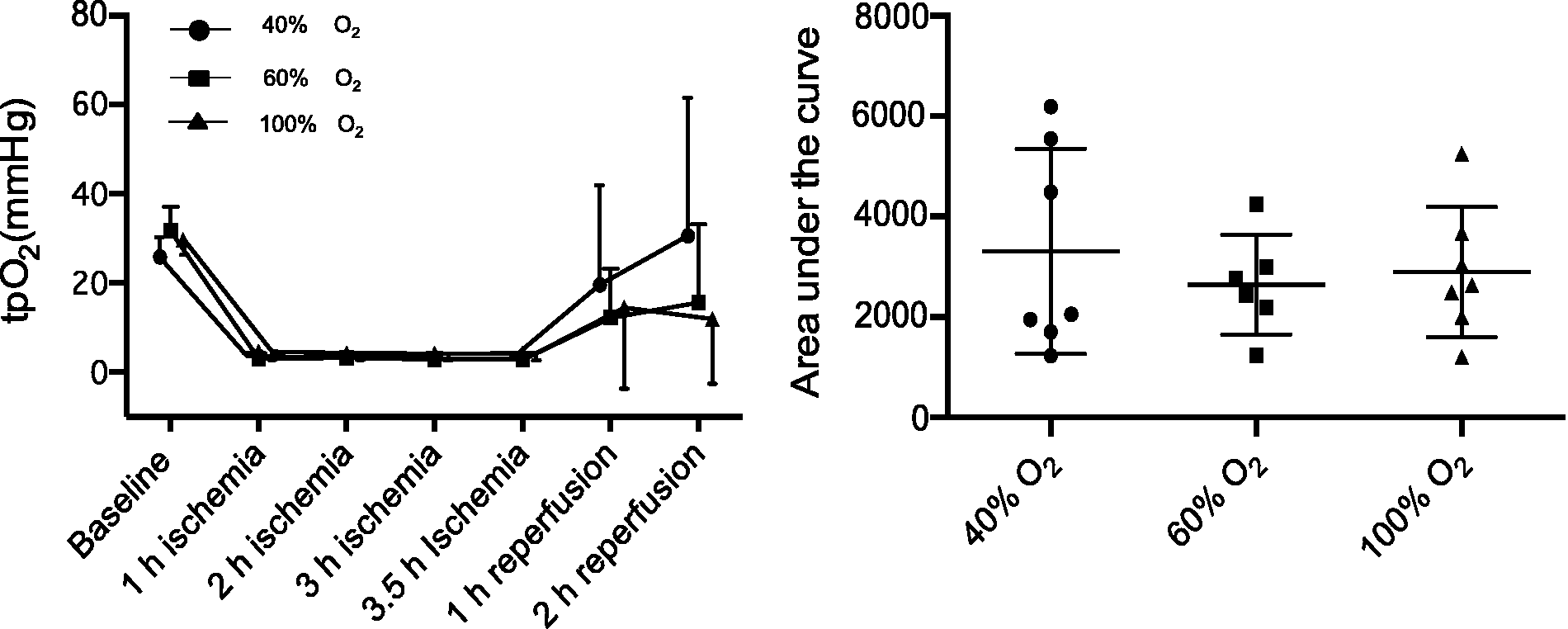
Oxidative stress
Analysis of oxidative stress markers was performed to investigate the influence of different inspired oxygen concentrations on MDA levels and SOD activities in citrate plasma. No statistically significant inter-group differences were found for concentrations of MDA or SOD activities by comparing AUC data (Figure 7a, right panel, ANOVA with Bonferroni’s post hoc test). Also intra-group analyses of MDA values over time did not reveal significant changes (Figure 7a, left panel, ANOVA with Dunnett’s post hoc test versus baseline). However, the SOD activities showed a trend for reduction over time in all experimental groups, with the value at 2 h of reperfusion being significantly lower than baseline in the 40% O2 group (P = 0.026, Figure 7b).
MDA concentration and SOD activity in plasma. Concentrations of MDA (a) and levels of SOD activity (b) are displayed on the left panels, while the corresponding areas under the curve are shown on the right panels. Error bars indicate mean ± SD. *P < 0.05. MDA: malondialdehyde, SOD: superoxide dismutase.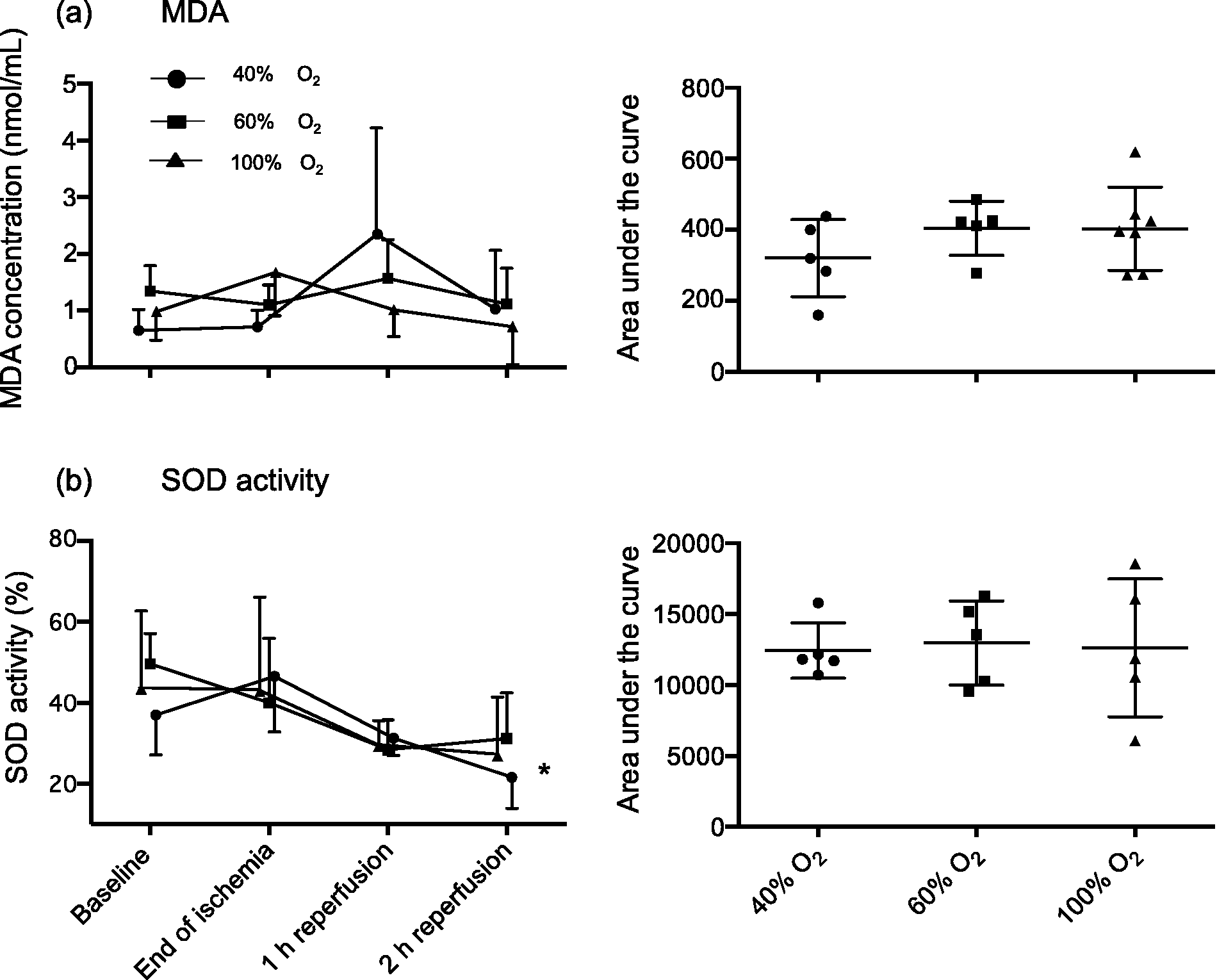
Discussion
In this study a rat hind limb IRI model was used in which FiO2 was varied from 40% to 100% in order to assess the effect of inhaled oxygen in isoflurane anesthesia on reperfusion injury. A more severe muscle edema formation and a significantly higher level of CK-MM in plasma were detected in the 40% FiO2 as compared with the 100% FiO2 group. Clinically, edema formation and plasma levels of CK-MM, reflecting myocyte necrosis, are the most important factors to determine the extent of reperfusion injury. These tissue injury data are supported by the finding of C3b/c and fibrin deposition, indicating activation of the complement and coagulation systems, respectively. In the 40% and 60% FiO2 groups significantly higher C3b/c and fibrin depositions were found in the reperfused limbs as compared with the contralateral ones and the normal controls, whereas this was not the case in the 100% FiO2 group. As expected, pO2, sO2, O2Hb were significantly higher in the 100% FiO2 group than in the group with 40% FiO2. In addition, breath rate was significantly elevated in the 40% FiO2 group as compared with the other two groups. For the rest of the parameters, no significant inter-group differences were detected.
Muscle edema was assessed as a basic parameter to evaluate the severity of reperfusion injury. Edema is caused by increased vascular permeability due to substantial inflammation.33,36 Vascular leakage has been linked to endothelial cell activation, which can be caused by the deposition of complement 37 or by the presence of cytokines and chemokines.38,39 In our study, muscle edema was more pronounced in the rats which underwent hind limb ischemia and reperfusion while breathing 40% O2 as compared with the rats breathing 100% O2. This is in line with a study of rat intestinal IRI 40 and a canine model of myocardial infarction and reperfusion, 41 both of which have shown that an FiO2 of 100% attenuated reperfusion injury. Also CK-MM, the predominant CK isoenzyme in skeletal muscle and an indicator for muscle injury, was less present in the plasma of rats breathing 100% O2 as compared with 40% and 60% O2, providing further evidence for reduced muscle damage in the 100% O2 group.
Higher FiO2 did not lead to higher tpO2 in the muscle tissue at the end of reperfusion. This finding seems to be in contrast to an earlier report, which showed that tpO2 was significantly elevated over normoxic conditions (21% O2) with FiO2 between 30% and 100% in normovolemic rats. 42 Normoxic conditions were not used in this study because in our (unpublished) experience too many animals died when using room air/isoflurane anesthesia for hind limb IRI experiments, presumably due to the long duration (>6 h) of anesthesia. It is also possible that in our study the tpO2 of 40%, 60% and 100% FiO2 was higher than at normoxic conditions. Furthermore, breath rate was significantly elevated in the 40% FiO2 group as compared with both the 60% and 100% O2 groups. This can be explained by the reduced acidosis at the end of reperfusion (higher pH, supplementary Table 1, see http://lan.sagepub.com) in the 40% FiO2 group as compared with the other two groups. So these animals breathed at an increase rate to fully exhale their acidosis. Lower oxygen and increased metabolic acids are the trigger for an increased breath rate.
We did not find any significantly elevated concentrations of MDA in plasma, nor did we find significantly reduced SOD activities in the experimental groups, with the exception of the 2 h reperfusion value in the 40% FiO2 group. These results are consistent with a recently published clinical study, 27 as well as earlier data from rat IRI studies,40,43 which question the role of oxidative stress in IRI. Yet our data are different from those in previous skeletal muscle IRI studies in rabbits.44,45 The controversial results might be due to differences in animal species and/or experimental techniques.
Taken together, different FiO2 in the O2/air/isoflurane mixture indeed affected the outcome of the rat hind limb IRI model in this study. An FiO2 of 40% led to more severe reperfusion injury than inhalation of higher oxygen concentrations. The FiO2 of 40% represents more or less the clinical conditions for standard anesthesia during surgical interventions and it is more physiological than the often used 100% FiO2 setting in rodent models. The latter in fact seems to provide a certain protection from reperfusion injury. Perhaps the high pO2 value (200–400 mmHg) in the 100% O2 group causes a decreased vasodilator response during hind limb reperfusion,46,47 and this leads to slower reperfusion, which has been shown to reduce IRI. 33
Conclusions
Our data suggest that anesthesia with an FiO2 of 40%, reflecting more or less the situation in a clinical setting, leads to more severe limb IRI than the use of higher FiO2. Using an FiO2 of 40% for rat anesthesia, in particular for IRI experiments, may therefore represent refinement of the animal experimentation model with more clinical relevance. In addition, we found no link between FiO2 and the occurrence of oxidative stress and no indication for an important role of oxidative stress in our IRI model.
Footnotes
Acknowledgements
We wish to thank Anjan Bongoni (Department of Clinical Research, University of Bern) for support and helpful discussions, as well as Dr Yara Banz and her team at the Institute of Pathology for preparation of histological slides and examination of histopathological changes. This study was performed with the support of the Microscopy Imaging Center (MIC), University of Bern.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Swiss National Science Foundation, grants no. 32003B_135272 and 320030_156193 were awarded to RR. The Chinese Scholarship Council in Switzerland awarded grant support to SZ.
