Abstract
Compared with single nodular liver cancer, the prominent biological characteristics of multinodular liver cancer include rapid progression and short survival. Here, we developed a multinodular liver cancer model in mice and assessed the biological characteristics of the resulting neoplasms. H22 hepatoma cells at a dose of 2 × 105/mouse, suspended in 1.6 mL, 0.8 mL, or 200 µL saline were injected via the tail vein of BALB/c mice at a velocity of 200 µL per second. The mice were sacrificed at different time points after injection. And at the time of death the liver, lungs, spleen, kidneys and heart were removed for morphological study. The biological characteristics of the tumor nodules were evaluated by immunohistochemistry. In the mice treated with a large volume injection of H22 cells, by day 7, there was a 100% occurrence of multinodular tumors in the livers, determined by histology. At the time of death, there were 100%, 100%, 37.5% and 37.5% occurrences of tumors in the lungs, kidneys, spleen and heart, respectively. The neoplastic cells in the liver nodules showed pleomorphism, and exhibited high expression of proliferating cell nuclear antigen (PCNA), c-myc, vascular endothelial growth factor (VEGF) and matrix metalloproteinase 2 (MMP-2). In mice treated with a small or medium volume injection, no tumor cells were identified in the livers, spleen, kidneys or heart at any of the examined time points. By day 7 and at the time of death, there was a 100% occurrence of tumor in the lungs. A multinodular liver cancer model in mice was achieved using a large volume injection of H22 cells.
In the advanced stage of either primary or metastatic liver cancer, patients frequently present with expansive multinodular lesions that can involve the liver or other organs. Compared with single nodular liver cancer, the prominent biological characteristics of multinodular liver cancer include rapid progression, a strong tendency for invasion of adjacent tissue, short survival, and recurrence after treatment. 1 To date, there is no effective therapy for this group of diseases.
Animal models can be helpful to our understanding of the mechanisms underlying the pathogenesis of liver cancer and may provide information with regard to new therapeutic strategies and drug screening. To date, attempts to model multinodular liver cancer in mice have utilized three experimental approaches: (i) chemically-induced tumors,2,3 (ii) intraportal 4 or intrasplenic 5 injection of tumor cells and (iii) transgenic models.2,3,6 Chemically-induced models are time-consuming. 2 Intraportal or intrasplenic injection of tumor cells requires a surgical procedure and the possibility of peritoneal seeding.4,5 Development of transgenic mouse models is time-consuming and expensive.2,6,7 The ideal model of liver cancer should faithfully reproduce the key biological characteristics, be reliable, reproducible, affordable and easy to manipulate.2,7,8 None of the currently available mouse models meets all the criteria of an ideal animal model, which include biological, genetic, etiological and therapeutic criteria.9,10
In 1999, Liu et al. reported that intravenous injection of a large volume of solution at a high velocity may transfer naked DNA to the liver, lungs, kidneys, spleen, and other viscera in mice. 11 Since then, this simple technique, termed, ‘hydrodynamic injection’ has been widely applied to deliver DNA, siRNA, PCR-amplified products and antisense oligonucleotides to the liver, lungs, kidneys, spleen, etc.12,13 Recently, hydrodynamic injection (large volume injection) has been successfully employed to deliver tumor cells to the liver, lungs14,15 and kidneys 15 in tumor models designed to generate models of multiple organ metastases in mice. Nevertheless, the dynamic characteristics and biological behaviors of these metastatic tumors in the liver need to be further investigated.
In the present study, therefore, murine H22 hepatoma cells were delivered into mice by rapid intravenous injection of a large volume solution. The dynamic morphological characteristics of the resulting tumor formation were examined by light and electron microscopy. The biological characteristics of these tumors were evaluated by immunohistochemistry using antibodies that reflect cell proliferation, angiogenesis and tumor invasion.
Materials and methods
Animals and cell line
Female BALB/c mice, aged 6 to 8 weeks (16–18 g), were purchased from the Laboratory Animal Research Center of Jilin University (Changchun, China). The murine ascitic H22 hepatoma cell line was obtained from the China Center for Type Culture Collection (Wuhan, China). After the recovery of frozen cells, H22 cells were suspended in saline and then cultured in the peritoneal cavity of the mice. All the animal experimental procedures were carried out in accordance with institutional guidelines of the Jilin University Animal Care and Use Committee.
Intravenous injection of H22 cells and animal treatment
H22 cells were collected from ascites after centrifugation. The cells at a dose of 2 × 105/mouse, diluted in 0.2 mL (small volume injection), 0.8 mL (medium volume injection), or 1.6 mL (large volume injection) of saline, were injected via the tail vein of the mice at a velocity of 200 µL per second using a syringe with a 23-gauge needle. Five of the mice were sacrificed 15 min, 30 min, 1 h, 8 h, 24 h, 3 days and 7 days after injection in the small and large volume injection groups. Three of the mice were sacrificed 8 h, 24 h, 3 days and 7 days after injection in the medium volume injection group. The liver, spleen, lungs, kidneys and heart were removed for tumor examination and morphological analysis. For each of the different volumes, one group of animals was allowed to survive until death in order to investigate late stage tumor growth and survival.
Tumor examinations
The organs were weighed and examined visually to identify the presence of macroscopically visible lesions. Subsequently, they were fixed in 10% neutral-buffered formalin and paraffinized, and 4 µm sections were stained with hematoxylin and eosin (HE) to examine the neoplastic lesions and their numbers under the microscope. The areas of tumor nodules in histological sections were measured by Image-Pro Plus 5.0 (Rockville, MD, USA).
Immunohistochemistry
Immunohistochemical staining of proliferating cell nuclear antigen (PCNA), c-myc, vascular endothelial growth factor (VEGF) and matrix metalloproteinase-2 (MMP-2) was performed on paraffinized sections of the livers according to the instructions of the UltraSensitiveTM S-P kit (Maixin, Fuzhou, China). The primary antibody (1 : 500, PCNA, Santa Cruz, CA, USA; VEGF, MMP-2 and c-myc, 1 : 100, Biosynthesis Biotechnology Co Ltd, Beijing, China) was applied overnight at 4℃. Phosphate-buffered solution (PBS) instead of the primary antibody was the negative control. The reaction was detected by incubating using a DAB kit (Maixin). Sections were counterstained with hematoxylin. Positive staining was indicated by brownish-yellow granules in the nuclei for PCNA, in the cytoplasm for VEGF, MMP-2 and c-myc. For analysis, three to five fields in each section were chosen. Positive cells were counted successively under the light microscope to calculate the percentage of positive cells.
Electron microscopy
Blocks of liver tissue, measuring 1 mm3 were fixed in 2.5% glutaraldehyde in 0.2 mol/L phosphate buffer (pH = 7.4), post-fixed in 1% osmium tetroxide and embedded in epoxy resin blocks. Ultra-thin sections were stained with uranyl acetate and lead citrate. The samples were subjected to electron microscopic examination using a JEM-1200EX transmission electron microscope (JEOL Ltd, Tokyo, Japan).
Statistical analysis
Data are presented as the mean ± SD. Differences between groups were analyzed by a two-tailed unpaired t-test (GraphPad InStat, San Diego, CA, USA). A P value < 0.05 was considered to be statistically significant.
Results
Survival of mice and liver weights after injection of tumor cells
In the group treated by large volume injection, the mice became transiently immobile immediately after injection and recovered several seconds to minutes later. The mice gained one-tenth of their body weight (18.3 g) which remained elevated for 30 min and then slowly returned to the baseline (16.7 g) by 2.5 to 3 h. The mice began to die from day 12, and by day 17 all were dead. The median survival time was 14 days. The liver weights were markedly higher than those of the control (untreated) mice from 15 min to 1 h after injection (Figure 1a), because of the accumulations of solution injected. Eight hours later, the liver weights declined and became similar to those of the controls. Then the livers gradually increased in weight from 24 h and were significantly higher than those of the controls when the mice died (Figure 1a).
Hepatic weights and macroscopic changes after injection of tumor cells. H22 hepatoma cells were delivered into the mice intravenously by large, medium or small volume injection. The livers were removed, weighed, fixed in 10% neutral-buffered formalin and photographed. Untreated mice were employed as controls. (a) Liver weight. Compared with control, *P < 0.05, **P < 0.01, ***P < 0.001. Compared with 7 days, #P < 0.05. (b) Representative photographs of livers studied at various times after large volume injection. (c) Representative photographs of livers studied at various times after medium volume injection.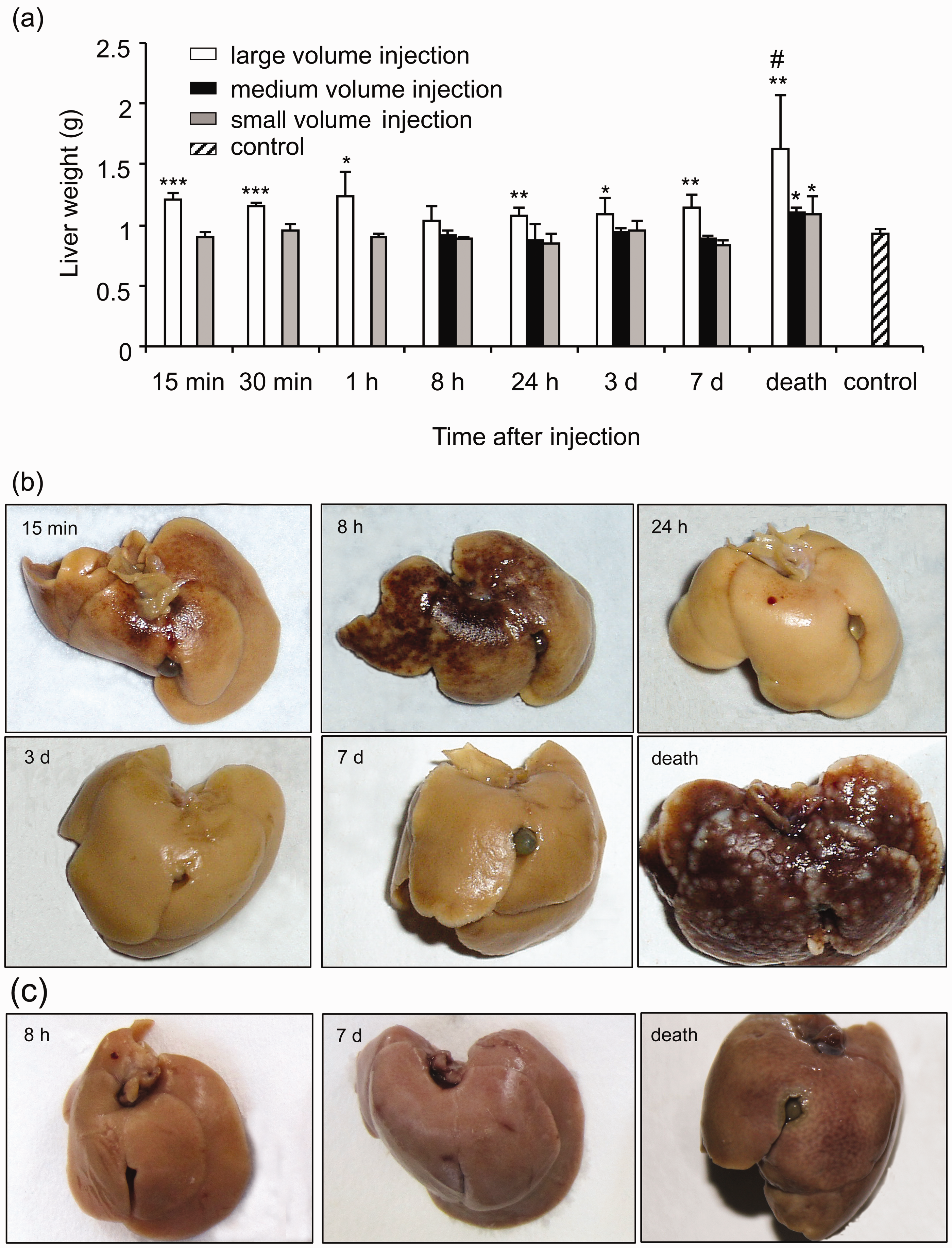
In the group treated by medium volume injection, the mice showed normal behavior after injection. The mice gained 0.8 g in body weight (median weight = 17.8 g), which returned to the baseline (17.1 g) within 2 h. Three mice died on days 15, 16 and 21 respectively. No significant increases in liver weight occurred from 8 h and 7 days post-injection compared with the controls, but the liver weights increased by the time of death (Figure 1a).
By contrast, after small volume injection, the mice treated exhibited neither changes in behavior nor increases in body weight. The mice began to die from day 15, and by day 25 all were dead. The median survival time was 19 days. No significant increases in liver weight occurred from 15 min to 7 days post-injection compared with the controls, but the liver weights increased by the time of death (Figure 1a).
Macroscopic features of the livers
As shown in Figure 1b, the mice treated with large volume injection had red hemorrhagic spots on the surfaces of their livers from 15 min to 8 h post-injection. The sizes of the hemorrhagic spots decreased by 24 h and became minimal by day 3 (when there were no noticeable liver changes) and regressed by day 7. The surfaces of the livers became irregular by day 7. When the mice died, numerous small grey–white nodules were distributed on their liver surfaces.
By contrast, in the mice treated with small or medium volume injection (Figure 1c), there were neither hemorrhages nor nodules on the surfaces of the livers at all the examined time points.
Light microscopic changes in the livers
In the mice treated with large volume injection, from 15 min to 1 hour after injection, the hepatocytes around the central veins were swollen, their cytoplasms had become vacuolar, and the cells were stained less with eosin (Figure 2a). By contrast, the hepatocytes around the portal triads were much less swollen, although some of these cells contained large vacuoles (Figure 2b). Small hemorrhagic foci were observed in the areas between the swollen hepatocytes and the normal-looking hepatocytes (Figures 2a and 2b). A few recognizable tumor cells demonstrated pleomorphism and contained weakly basophilic cytoplasm with prominent eosinophilic nucleoli. These appeared in small portal veins, within the hepatic sinusoids (Figure 2c), and inside or between damaged hepatocytes together with red blood cells (Figure 2d).
Pathological features of the livers after rapid intravenous injection of H22 cells in a large volume. H22 hepatoma cells were delivered to the mice by a large volume injection. The mice were sacrificed at different time points. The paraffinized sections of the livers were stained with hematoxylin and eosin (HE) (a–j) and using an immunohistochemical kit (k, l). (a–d) 15 min to 1 h. (e) 24 h. (f, g) 3 days. (h) 7 days. (i, j) Death at 15 days. (k) Death. (l) 7 days. PCNA: proliferating cell nuclear antigen, VEGF: vascular endothelial growth factor, MMP-2: matrix metalloproteinase-2.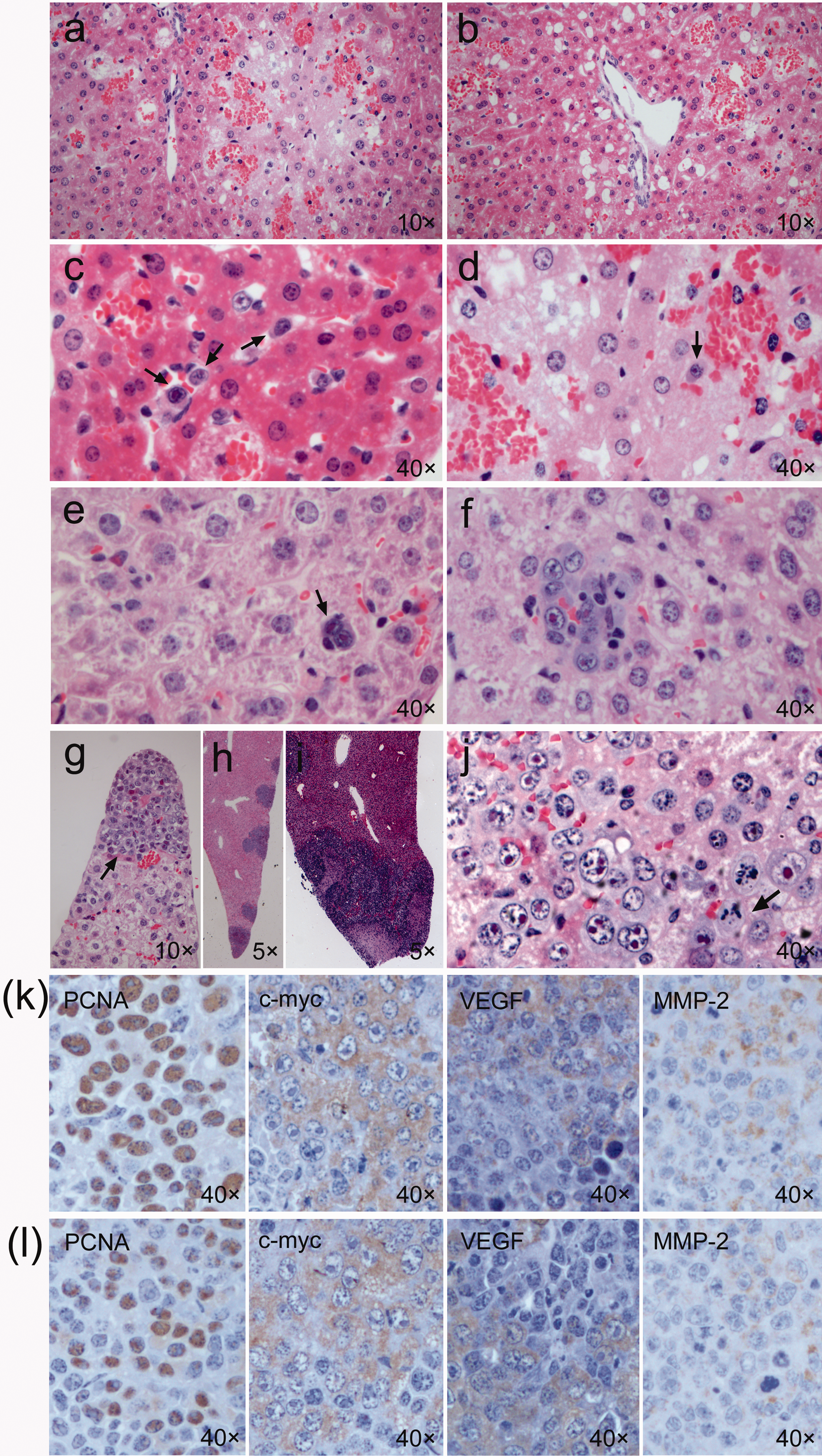
Tumor nodules in the livers.

The H22 hepatoma cells in a large volume were intravenously injected into the mice. The total number of tumor nodules in each mouse was calculated from the microscopic examination of seven sections in a total of seven lobes from each liver. The area of each nodule was measured by software Image-Pro Plus 5.0. The data are expressed as the mean ± SD. aP < 0.05 vs 3 days; bP < 0.05 vs 7 days.
In mice where a small or medium volume of tumor cells had been injected, neither swollen and necrotic hepatocytes nor hemorrhagic foci were seen in the livers at all time points. Until the mice died, the liver tissues were congested but no tumor cells or nodules were found there (data not shown).
Biological characteristics of tumor cells in the livers after large volume injection
Expression of PCNA, c-myc, VEGF and MMP-2 (%) in hepatic tumor cells located in tumor nodules.

The data are expressed as the mean ± SD. aP < 0.05 vs 7 days. PCNA: proliferating cell nuclear antigen, VEGF: vascular endothelial growth factor, MMP-2: matrix metalloproteinase-2.
Electron microscopic findings of the livers
In the mice treated with large volume injection, ultrastructual examination of the livers at periods ranging from 15 min to 1 h after injection, demonstrated injury to the endothelial cells and hepatocytes. Red blood cells had entered the hepatocytes (Figure 3a). The endothelial gaps enlarged; vesicles of varying size were observed near the sinusoidal side of the plasma membranes of the hepatocytes (Figure 3b). Tumor cells were found in the cytoplasms of the swollen hepatocytes whose rough endoplasmic reticulum had apparently expanded. Some mitochondria appeared to be swollen and others condensed (Figure 3c). In each case the tumor cells were mostly located between the hepatocytes and were characterized by a large nucleus, irregular nuclear membrane, much euchromatin, a large nucleolus and scanty cytoplasm, fewer organelles, more glycogen, few mitochondria and rough endoplasmic reticulum (Figure 3d) than hepatocytes. By 8 h, the vesicles had disappeared and organelles of the hepatocytes appeared normal. Neutrophils and Kuppfer cells were present in the hepatic sinusoids. By 24 h and 3 days, sinusoidal endothelial cells had an apparently normal appearance (Figure 3e). Increased numbers of lysosomes were seen in the cytoplasm of the Kuppfer cells. By 7 days, the injured hepatocytes and endothelial cells had a normal appearance (data not shown); the tumor cells grew in groups, and compressed the adjacent liver cells (Figure 3f).
Electron microscopic features of the liver after rapid injection of a large volume tumor cell solution. (a) Injury of cytoplasm of endothelial cells and hepatocytes. (b) Enlargement of sinusoidal endothelial gap, neutrophils in sinusoid and vesicles in the cytoplasm of hepatocytes. (c) Tumor cell inside a hepatocyte. (d) Tumor cells between the hepatocytes. (e) Intact hepatocytes and sinusoidal endothelial cells by day 3. (f) Tumor cells in a group on day 7. RBC: red blood cell.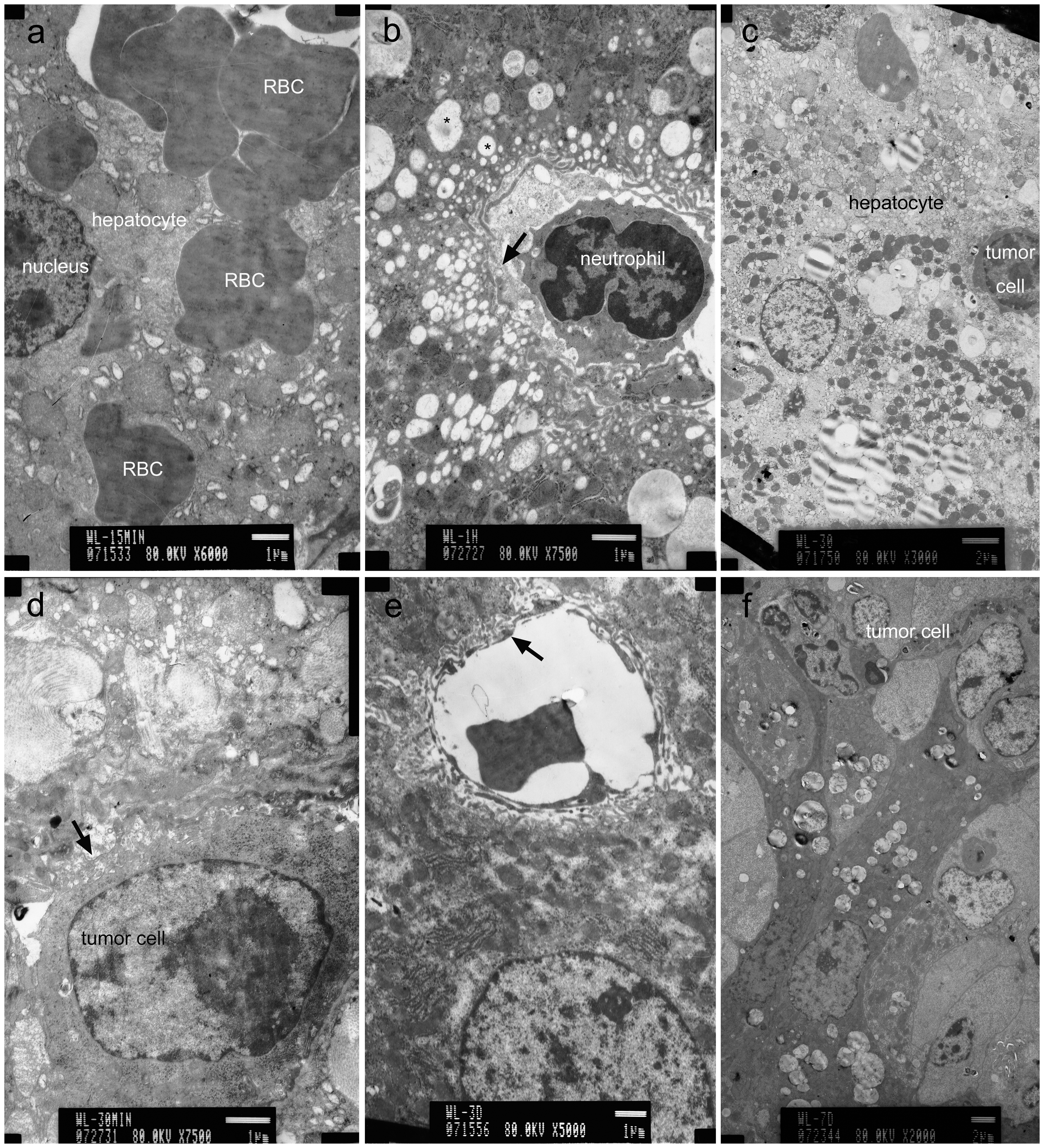
By contrast, in the mice treated with small volume injection, there were no significant changes from 15 min to 7 days compared with the untreated mice. No tumor cells were identified. Ultrastructual examination was not performed in the medium volume injection group.
Morphology of the lungs after injection of tumor cells
In the mice treated with large volume injection, observed macroscopically, the lung surfaces were smooth from 15 min to 7 days, but extensive fine surface granularity was revealed after the mice had died (Figure 4a). Microscopically, the alveolar walls were observed as thickened and hyperemic. From 15 min to 1 h, small groups of 3–6 tumor cells were seen in the alveolar walls (Figure 4b). The tumor cells gradually decreased in number by 8 h, and were rarely found by 24 h and day 3. However, by day 7, 100% of mice (5/5) had clusters of tumor cells in the lung tissues (Figure 4c). After the mice died, 100% (8/8) demonstrated tumor cells mainly growing along the alveolar walls (Figure 4d). Less commonly, the tumors formed solitary nodules. Extensive hemorrhagic areas were present in the lung tissue.
Tumor formation in the lung, kidney, spleen and heart. (a–d) Pulmonary changes after large volume injection. (a) Representative photograph of the lung of a dead mouse on day 13. (b) Histological section of lungs at 15 min to 1 h. (c) 7 days. (d) Death at 15 days. (e–h) Pulmonary changes after medium volume injection. (e) Representative photograph of the lung of a dead mouse on day 16. (f) 8 h. (g) 7 days. (h) Death at 16 days. (i–l) Pulmonary changes after small volume injection. (i) Representative photograph of the lung of a dead mouse on day 18. (j) 15 min to 24 h. (k) 7 days. (l) Death at 15 days. (m–p) Tumor formation in the kidney, spleen and heart after a large volume injection. (m) Tumor cells in a glomerulus. (n) Renal interstitium. (o) Splenic red pulp. (p) Myocardium.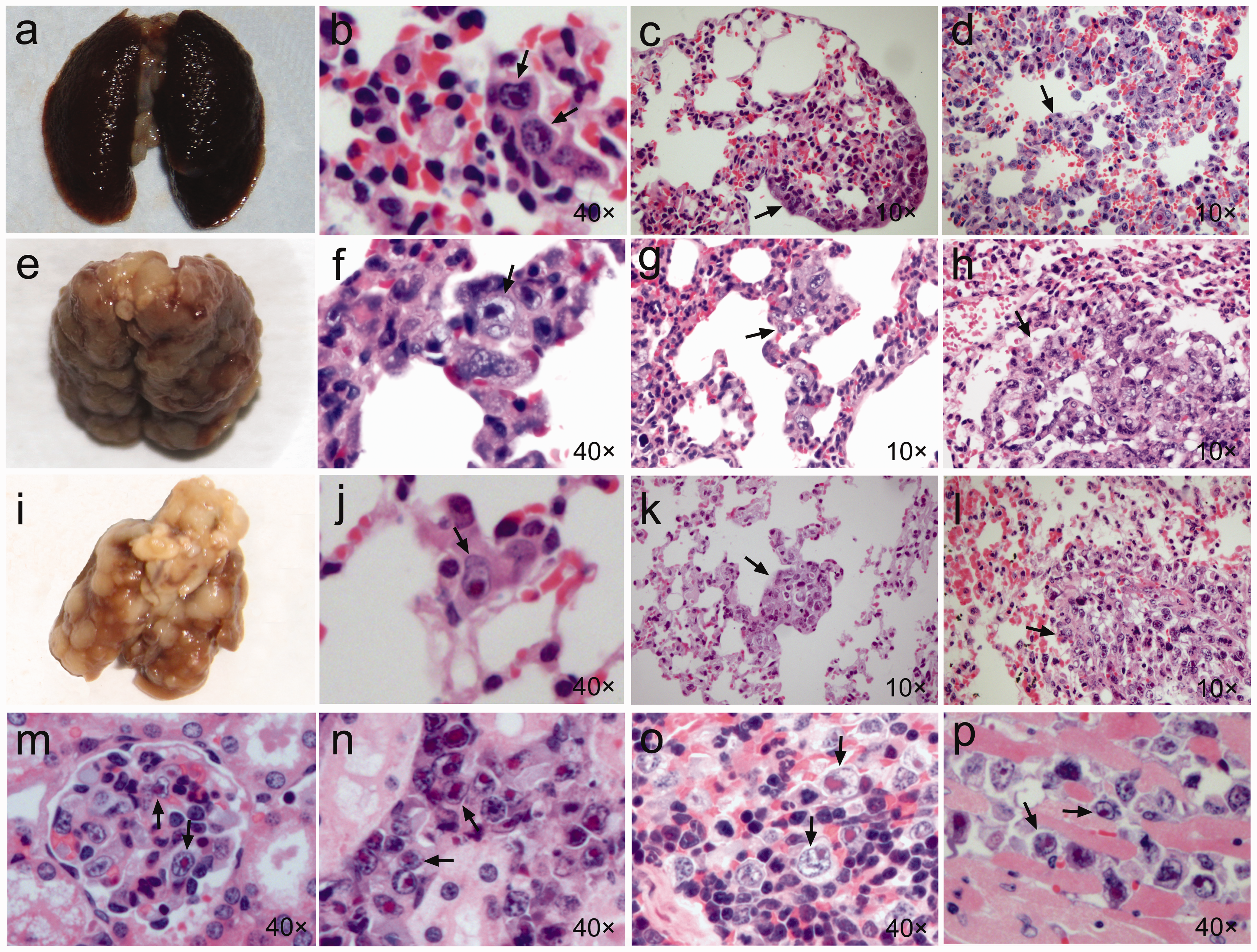
In the mice treated with small or medium volume injection, the surface of the lungs appeared when observed macroscopically to be smooth until day 3 after injection. However, by day 7, the lungs had coarse surfaces. When the mice died, grey–white nodules of varying sizes were distributed on the surface of the lungs (Figures 4e and 4i). Microscopically, the pulmonary septa did not appear widened. From 15 min to 24 h, single recognizable tumor cells were distributed on the walls of the alveoli (Figures 4f and 4j). By day 3, 40% of the mice (2/5) in the small volume injection group, and 33% of the mice (1/3) in the medium volume group demonstrated tumor cell nodules in the lung parenchyma. By day 7, 100% of the mice (5/5 in the small volume injection group and 3/3 in the medium volume injection group, respectively) exhibited tumor nodules in the lung and the nodules had increased in size in comparison to their size on day 3 (Figures 4g and 4k). By the time of death, 100% of the mice (12/12) in the small volume injection group, and 100% (3/3) in the medium volume injection group had tumor cells growing in multiple nodular areas throughout their lungs (Figures 4h and 4l). Although the nodules were non-encapsulated, they were sharply demarcated from the adjacent normal tissue. Even when individual nodules coalesced to form larger lesions, the margins of the nodules were still clearly defined.
The effects of tumor cells injection on the kidney, spleen and heart
Macroscopically, no apparent nodules were seen on the external surface of the kidney, spleen or heart in the mice, whether treated with large, medium or small volume tumor cell injection.
Microscopically, in the kidneys of the mice treated with large volume injection, the tumor cells were not recognizable microscopically in either the cortex or the medulla from 15 min to day 3. However, by day 7, they were found in the interstitial tissue of the cortex of these mice. At death, the tumor cell groups were located in the cortex, medulla and hilum, mainly in the glomeruli (Figure 4m) and the interstitium (Figure 4n). In the spleen, no tumor cells were recognized from 15 min to 7 days after cell injection. At death, the tumor cells were located diffusely throughout the splenic red pulp (Figure 4o). In the heart, tumor cell growth was found in the myocardium at death (Figure 4p). By contrast, in the mice given small or medium volume injection, the kidneys and spleen were congested, and no tumor cells were observed in the renal parenchyma, spleen or myocardium at these time points.
Discussion
Laboratory mice are one of the best models for studying cancer. This is due to their small size, low cost, and genetic consistency when compared with the use of larger animals for similar experiments.16,17 In the present study, a large volume injection was applied to mice to generate multinodular liver cancers with the additional involvement of multiple organs. Rapid injection of a large volume solution (about 8%–10% of body weight) induces immediate congestion in the right-sided heart, and leads to an elevation of intravascular pressure. 18 As a result, the hydrostatic pressure developed in the inferior vena cava drives the injected tumor cell solution into tissues such as the liver, kidneys and heart that are directly linked to the inferior vena cava.
H22 can be grown in vitro in a cell culture system, but this is time-consuming. H22 is a murine ascites hepatoma cell line which allows the passage of the cells into the peritoneal cavity. Culturing tumor cells in the mouse peritoneal cavity may avoid microbial contamination and facilitates the collection of large numbers of tumor cells which are then adapted to growth in vivo.
In our study the mice survived well following the rapid injection of a large volume solution of tumor cells. Despite the fluid overload which induces a temporary dysfunction of the cardiac system and structural changes of the liver, animals tolerated the procedure well.18–20 The rapid injection of a large volume solution led to an increase in liver weight. Increasing hydrostatic pressure enlarged the gap between sinusoidal endothelia, damaged the cytoplasm of endothelial cells and hepatocytes and induced the formation of vesicles and large vacuoles in hepatocytes. We postulate that these changes facilitated the entry of tumor cells by blood circulation into or between the hepatocytes. Histological and electron microscopic examination showed that the mice recovered within one week from the above injuries to the liver caused by increasing hydrostatic pressure. In order to lessen the hepatic injury induced by a large volume of injection fluid (1.6 mL), the effects of a rapid injection of medium volume (0.8 mL) was investigated. Although no morphological evidence of hepatic injury was found, this medium volume injection only produced pulmonary nodules, but did not successfully generate hepatic nodules in mice.
In the early stage (15 min to 24 h) after a large volume injection, the H22 tumor cells were not easily recognized in the liver. This may have been due to tumor cell injury from hemodynamic shear forces 21 or due to the host immune system 22 which caused some cell loss in blood circulation. A major cause may have been dormancy of extravasated cells. 23 However, by 3 days, tiny tumor cell foci were microscopically present in many lobes of the liver, presumably because the surviving tumor cells continued to grow after extravasation. 23 By day 7, the tumor cells escaped dormancy and grew as tumor foci in 100% of the mice. Under microscopy, the majority of nodules were observed to be located mainly next to the capsule of the liver. The proportion of lobes bearing tumor nodules as well as the number and size of tumor nodules increased with time.
Carcinogenesis is a multistage process involving proliferation, angiogenesis, invasion and metastasis. PCNA is a nuclear protein and c-myc is a proto-oncogene which play a key role in cell proliferation and correlate with the degree of malignancy, metastasis and survival of hepatocellular carcinoma (HCC).24,25 VEGF and MMP-2 perform a critical role in angiogenesis, invasion and metastasis of HCC.26,27 Our immunohistochemical staining showed a high expression of PCNA, c-myc, VEGF and MMP-2 in the hepatic tumor nodules of the late stage tumors at 7 days and at death. These resemble the biological characteristics of liver cancer in humans.24–27 However, objective quantification such as real time PCR or western blot analysis is needed to support this observation.
In this study, the rate of extrahepatic tumor occurrence was 100% in the lungs and kidneys, and 37.5% in the spleen and heart, respectively. In a clinical setting, the most common site of distant metastasis is the lung. 28 Metastasis of liver cancer to the kidney, 29 spleen28,30 and heart 31 have been documented but are unusual.
Rapid injection of a large volume solution delivered the tumor cells to the heart, liver, lungs, kidneys and spleen. In the lung, the tumor cells grew mainly along the alveolar wall and rarely formed nodules. By comparison, in mice with small volume injection, all injected cells enter the inferior vena cava and are then transferred into the right-side of the heart and then to the lungs to form multiple nodules there, rather than in other organs. We postulate this is why the survival of the mice with large volume injection was shorter than the mice with small or medium volume injection. However, the reason why the tumor cells grew along the alveolar walls rather than formed nodules in the lungs of the mice treated with large volume injection should be investigated further.
In summary, an advanced stage of liver cancer with multiple nodules was achieved in mice by the rapid injection of a large volume of H22 hepatic tumor cell solution. Kang et al. 14 and Li et al. 15 have also reported that multinodular tumor formation in the liver can be produced by a rapid injection of murine B16, 4T1 or Renca cells in a large volume. The involvement of multiple organs seems to be independent of the tumor cell type. However, there is no evidence that human liver cancer cell lines yield similar or comparable results to the H22 cell line. The advantages of this technique include its simplicity, eliminating the need for a surgical procedure, the rapid formation of tumors, high tumor occurrence and low cost. Avoiding the potential exposure of operators to carcinogenic agents is also beneficial. These findings provide an alternative for mouse models of advanced stage liver cancer.
Since metastasis is a process involving a complex interaction between tumor cells and the host, animal models are still necessary for examining this phenomenon and are unlikely to be entirely replaced by in vitro studies. However, our experiments have shown that a rapid injection with a large volume of tumor cells is more efficient as a method of inducing multinodular hepatic tumors than the injection of small or medium volumes. This fulfils the goal of reducing the number of animals needed in experiments to generate such neoplasms. Refinement of the hydrodynamic model is an objective in our next experiments.
Footnotes
Acknowledgements
This work was supported in part by grants from the Program for New Century Excellent Talents in University of Ministry of Education of China (NCET-04-0303), and the Natural Science Foundation of Jilin Province, China (201115112). The authors thank Dr William Orr, Department of Pathology, University of Manitoba, Canada for his help in the preparation of this manuscript.
