Abstract
Mice with genetic alterations are used in heart research for the extrapolation of human diseases. Echocardiography is an essential tool for evaluating cardiac and hemodynamic functions in small animals. The purpose of this study was to compare the effect of different anesthetic regimens and the conscious state on the evaluation of cardiac function by echocardiography. Mice were examined in the conscious state after three days of training, and then for a 7 min period after a single intraperitoneal injection of ketamine at 100 mg/kg, etomidate at 10, 20 or 30 mg/kg, or after inhalation of isoflurane at 1.5% with or without a short period of induction with isoflurane 3%. Intra- and inter-observer variabilities were assessed. The operator’s comfort was also assessed. Heart rate, left ventricular end diastolic diameter, fraction shortening and cardiac output were measured using echocardiography. Ketamine at 5 and 7 min after induction and isoflurane at 3, 5 and 7 min after induction provided good anesthetic conditions and a quick awakening time, and did not influence cardiac performance, whereas the conscious state was associated with a non-physiological sympathetic activation and other anesthetic drugs induced a significant decrease in heart rate. Etomidate 10 mg/kg and 20 mg/kg were not enough to provide adequate anesthesia. Etomidate 30 mg/kg induced a good anesthetic condition but influenced cardiac performance and had a long awakening time. Our results indicate that ketamine and isoflurane with a short induction period are better anesthetic drugs than isoflurane without induction or etomidate for evaluating cardiac function in healthy mice.
Mice with genetic alterations are used in heart research for the extrapolation of human diseases.1–3 Echocardiography is an essential tool for evaluating cardiac and hemodynamic functions in small animals (measurement of remodeling, hypertrophy, systolic and diastolic dysfunctions). Parameters such as fractional shortening (FS) and heart rate (HR) are used in assessing cardiac function; and wall thickness and left ventricular (LV) diameters are used for assessing LV remodeling. Echocardiography is a non-invasive method, which can be realized (and repeated) in conscious mice or under general anesthesia.4–7 Ideally, studies of organ physiology should be performed on conscious animals to avoid confounding effects associated with anesthesia. Indeed, anesthesia causes a cardiac depression: alteration of systolic function, HR and cardiac output (CO). Under anesthesia, negative inotropic and chronotropic effects are observed. 8 However, the stress related to handling mice during conscious echocardiography may also alter their cardiac function.9,10 In fact, restraint of conscious small animals enhances sympathetic activity.9–11 Autonomic nervous system plays a key role in the observed differences in cardiac function between anesthetized and conscious mice. 12 The ideal drug must be safe and simple to use, and must induce reproducible sedation and immobility without changes in cardiac function and HR. The combination of ketamine and xylazine is a frequent choice in the anesthesia management of mice, but various anesthetic agents have been compared in different studies (sometimes versus the conscious state) to determine the optimal anesthetic regimen: ketamine and xylazine, pentobarbital, tribromoethanol (Avertin) and isoflurane,5,12–18 and the best time to realize echocardiography after drug administration. Etomidate is a short-acting hypnotic and is associated with hemodynamic stability and minimal respiratory depression. This is the reason why etomidate is used for induction in patients with cardiovascular disease. 19 Etomidate has not been assessed for the evaluation of cardiac function by echocardiography in mice.
The aim of this study was to assess the better anesthetic regimen to perform echocardiography in mice, integrating for the first time the cardiac effects of etomidate.
Materials and methods
Animals
Mice were housed in a pathogen-free facility and handled in accordance with the procedures outlined in Council Directive 86/609/EEC. The investigation conforms with the Guide for the Care and Use of Laboratory Animals published by the US National Institutes of Health (NIH publication No. 85–23, revised 1996). Experimental procedures were conducted according to the official edict presented by the French Ministry of Agriculture (Paris, France) and the recommendations of the Helsinki Declaration. These experiments were conducted in an authorized laboratory and under the supervision of an authorized researcher. Male BALB/c mice (Janvier, Le Genest St Isle, France) at 10–12 weeks of age and weighing 22.7 to 25.0 g were used. After arrival, the animals were allowed to recover for one week in groups of six animals in plexiglas cages (Tecniplast, Lyon, France). R04-10 water and food were provided ad libitum (SAFE Diet, Augy, France). The animals were housed in standard conditions. During the whole experiment they were housed under a controlled 12 h light–dark cycle and temperature (21℃) regime. A total of eight mice were used for testing each anesthetic.
Animal training
For conscious echocardiography, mice were trained daily for three days before echocardiography. Training included holding the mice for 5 min in the position required for echocardiography and touching the chest with a probe and a gel heated at 34℃.
Anesthesia and sedation
The depth of anesthesia was determined by assessing the withdrawal reflex at the left paw. Awakening time was measured from the beginning of the anesthetic regimen administration to the fully awake state (anesthetic duration). The mouse was considered to be ‘fully awake’ when it was able to walk normally.
Echocardiography and cardiac parameters
Echocardiography was performed using a Vivid 7 ultrasound machine and a 13 MHz linear transducer (General Electric Medical System, Horten, Norway). After a short-axis 2-D image of the left ventricle was obtained at the level of the papillary muscles, 2-D guided M-mode images were acquired at a sweep speed of 100 mm/s and stored digitally. The following parameters were measured digitally from M-mode tracings using the leading-edge technique:
17
left ventricular diameter at the end diastole (LVDd) and systole (LVDs), anterior and posterior wall thickness at diastole and systole, and HR. All LV dimensions were presented as the average of five consecutive selected beats. HR was determined from the cardiac cycles recorded on the M-mode tracing, using at least three consecutive beats. The other parameters were calculated from M-mode-derived LV dimensions:
Fractional shortening (FS): (LVDd − LVDs)/LVDd × 100 (%) End diastolic volume (EDV) and end systolic volume (ESV) were calculated by the cube method: EDV = (LVDd)3 and ESV = (LVDs)3 Ejection fraction (EF): EF = (EDV − ESV)/EDV × 100 (%) Stroke volume (SV): SV = EDV − ESV Cardiac output (CO): CO = SV × HR
Anesthetic regimen
Mice were kept warm using a heating lamp during examination. As it was a short-time procedure, a feedback system for controlling body temperature was not used. During echocardiography in the conscious state (Aw), mice were picked up by the nape of the neck and restrained in the prone position. Then, the following agents were assessed: etomidate, ketamine and isoflurane. Mice were anesthetized with a single intraperitoneal injection of 10 mg/kg etomidate (Eto 10); 20 mg/kg etomidate (Eto 20); 30 mg/kg etomidate (Eto 30); 100 mg/kg ketamine (K 100); or with inhalation of isoflurane at 1.5% (Iso 1.5); and isoflurane at 1.5% after induction with isoflurane at 3% for 2 min (Iso ind). The intraperitoneal route was chosen because it is the easiest way to administer the anesthetic regimen for a short-time repeated procedure. Echocardiography measurements were recorded at 3, 5 and 7 min after the drug administration. The awakening time was measured in each group. At the end of the procedure, the comfort of the operator in performing the echocardiography was assessed on a scale ranging from 0 (very uncomfortable) to 5 (very comfortable).
Statistical analysis
Inter-observer variability was determined as the difference between the measurements (HR, FS and LVDd) of the two observers (OL and LL) divided by the mean between the measurements of these two observers: (OL value − LL value)/([OL value + LL value]/2). Intra-observer variability was determined as the difference between the measurements of a single observer (OL) divided by the mean between the measurements of this single observer: (OL value 1 − OL value 2)/([OL value 1 + OL value 2]/2). The results were not normally distributed and were thus analyzed non-parametrically. To assess whether the measurements changed over time, Friedman’s test was used. When Friedman’s test was significant (P < 0.05), pairwise comparisons were made using Wilcoxon’s signed rank test. Time point comparisons between groups were made using a non-parametric Kruskal–Wallis first. When the Kruskal–Wallis test was significant (P < 0.05), then pairwise comparisons were made using the Mann–Whitney U-test.
Data are presented as mean ± SD. P < 0.05 was considered statistically significant.
Results
Anesthetic conditions/data quality
Eto 10 was insufficient to adequately anesthetize the animals (three mice were still awake at 3 min and all at 5 min), so these measurements were not taken. K 100 at 5 and 7 min, Eto 30 at 3 and 5 min and Iso ind at 3, 5 and 7 min provided acceptable anesthetic conditions in which echocardiography can be performed, as shown by the intra- and inter-observer variabilities (Figure 1). Awakening time was different between groups. Ketamine and isoflurane (with or without induction) were associated with the quickest awakening time when compared with Eto 20 and Eto 30 (Iso 1.5 = 7.5 ± 0.5 min, Iso ind = 7.6 ± 0.5 min, Eto 20 = 40 ± 9 min, Eto 30 = 49 ± 13 min, K 100 = 10 ± 2 min; P < 0.01). Even if they were trained and the images were of good quality, awaken mice were agitated causing discomfort for the operator. Comfort in performing the echocardiography was better in fully anesthetized mice (K 100, Eto 30, Iso 1.5 and Iso ind; comfort rate = 4.4 ± 0.5) when compared with awaken or lightly anesthetized mice (Aw, Eto 20; comfort rate = 1.1 ± 0.2).
Intra- and inter-observer variabilities according to the anesthetic regimens. Grey bars represent intra-observer variability; Black bars represent inter-observer variability.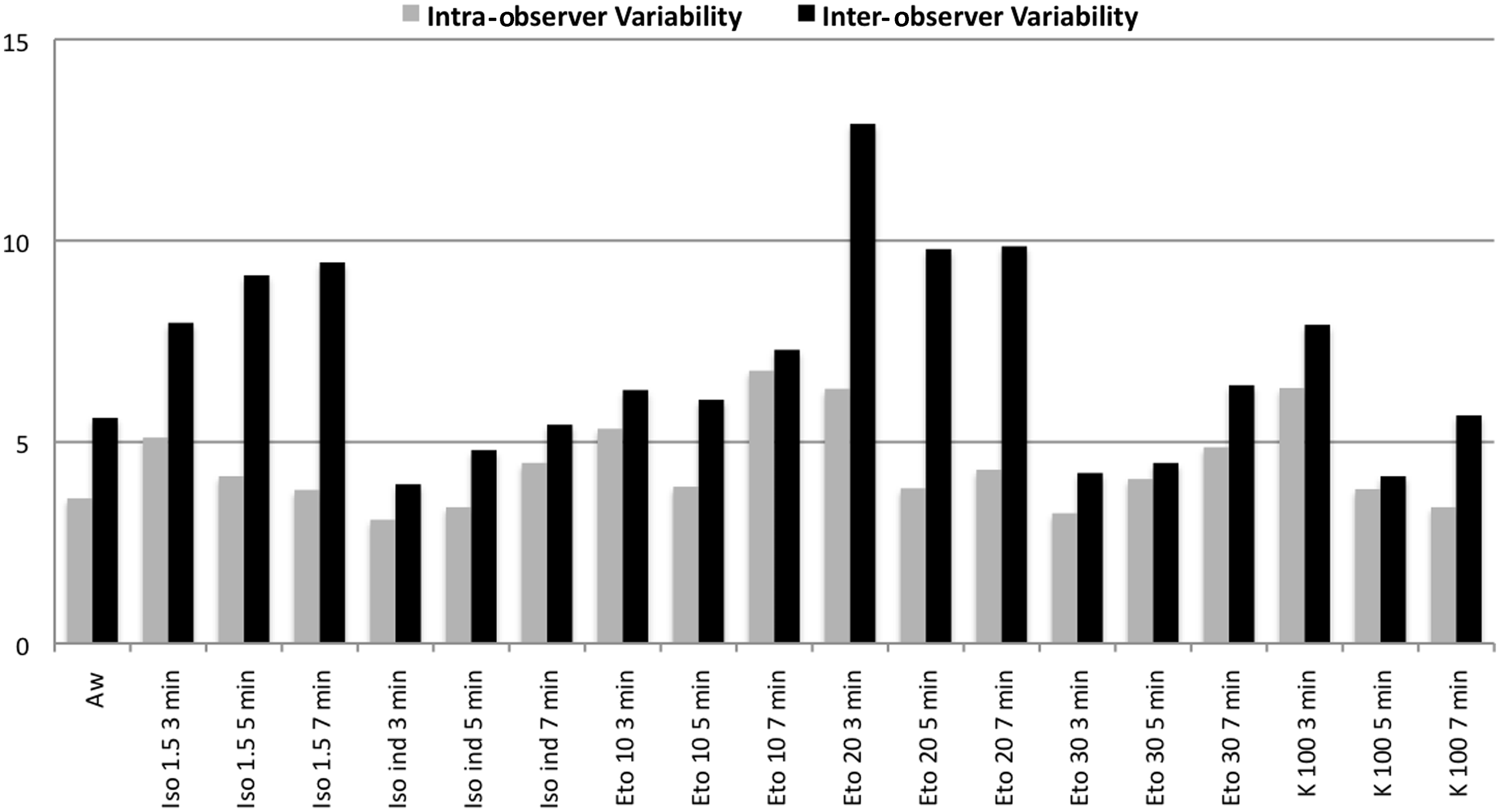
Assessment of echocardiography parameter
Echocardiographic measurements of heart rate in mice anesthetized with different anesthetic regimens.
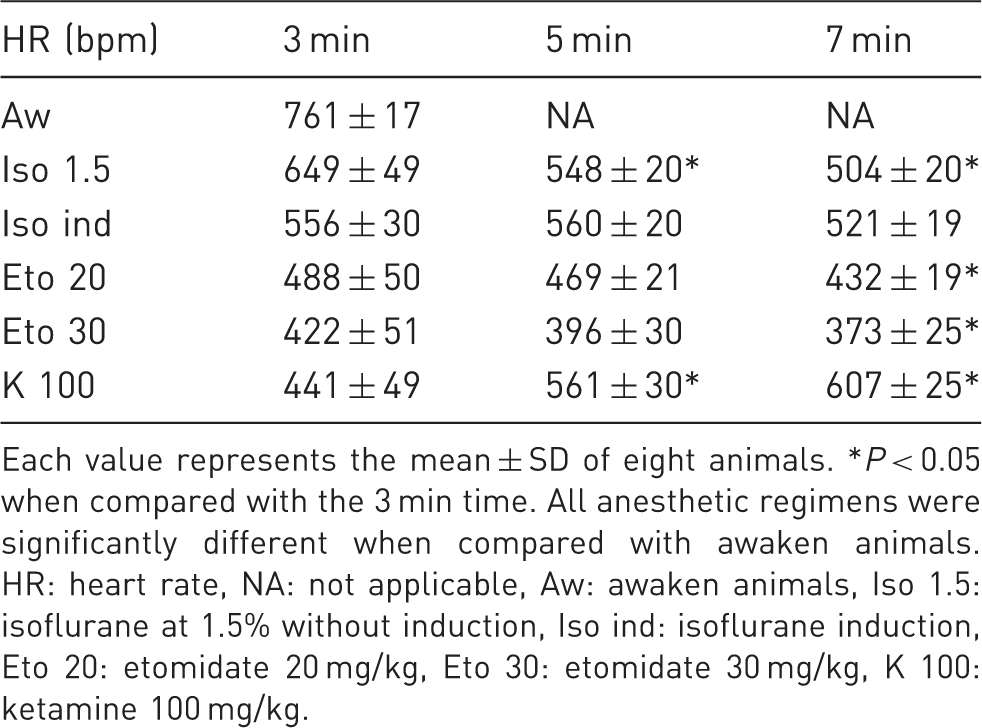
Each value represents the mean ± SD of eight animals. *P < 0.05 when compared with the 3 min time. All anesthetic regimens were significantly different when compared with awaken animals. HR: heart rate, NA: not applicable, Aw: awaken animals, Iso 1.5: isoflurane at 1.5% without induction, Iso ind: isoflurane induction, Eto 20: etomidate 20 mg/kg, Eto 30: etomidate 30 mg/kg, K 100: ketamine 100 mg/kg.
Echocardiographic measurements of fractional shortening in mice anesthetized with different anesthetic regimens.
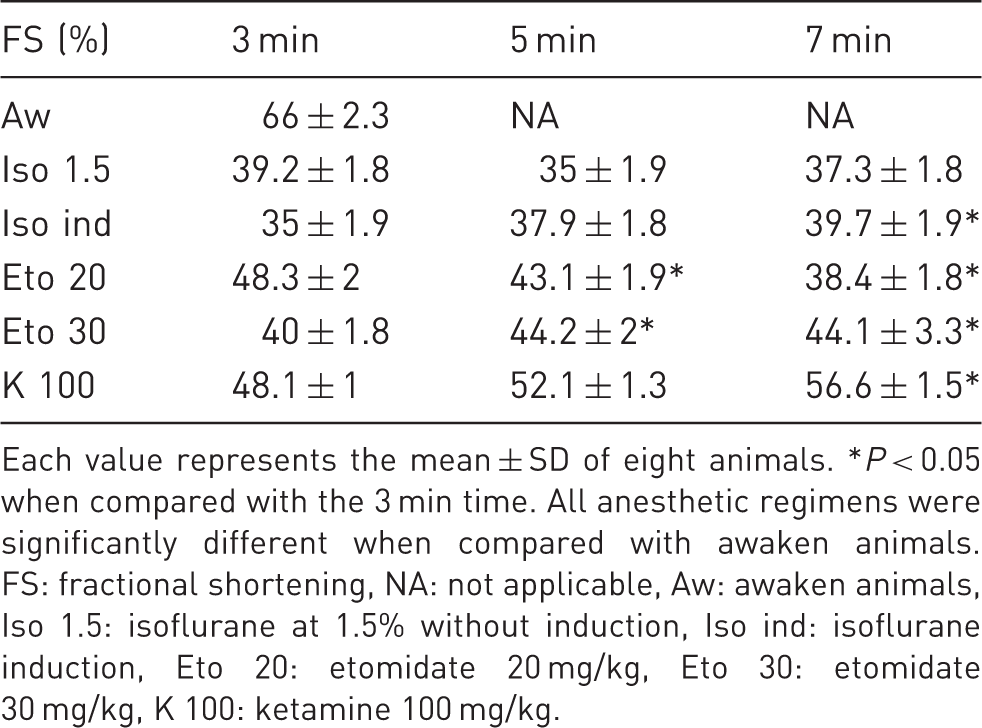
Each value represents the mean ± SD of eight animals. *P < 0.05 when compared with the 3 min time. All anesthetic regimens were significantly different when compared with awaken animals. FS: fractional shortening, NA: not applicable, Aw: awaken animals, Iso 1.5: isoflurane at 1.5% without induction, Iso ind: isoflurane induction, Eto 20: etomidate 20 mg/kg, Eto 30: etomidate 30 mg/kg, K 100: ketamine 100 mg/kg.
Echocardiographic measurements of left ventricular end diastolic diameter in mice anesthetized with different anesthetic regimens.

Each value represents the mean ± SD of eight animals. All anesthetic regimens were significantly different when compared with awaken animals. No difference during the time was found for each anesthetic regimen. LVDd: left ventricular diameter at the end of diastole, NA: not applicable, Aw: awaken animals, Iso 1.5: isoflurane at 1.5% without induction, Iso ind: isoflurane induction, Eto 20: etomidate 20 mg/kg, Eto 30: etomidate 30 mg/kg, K 100: ketamine 100 mg/kg.
Echocardiographic measurements of cardiac output in mice anesthetized with different anesthetic regimens.
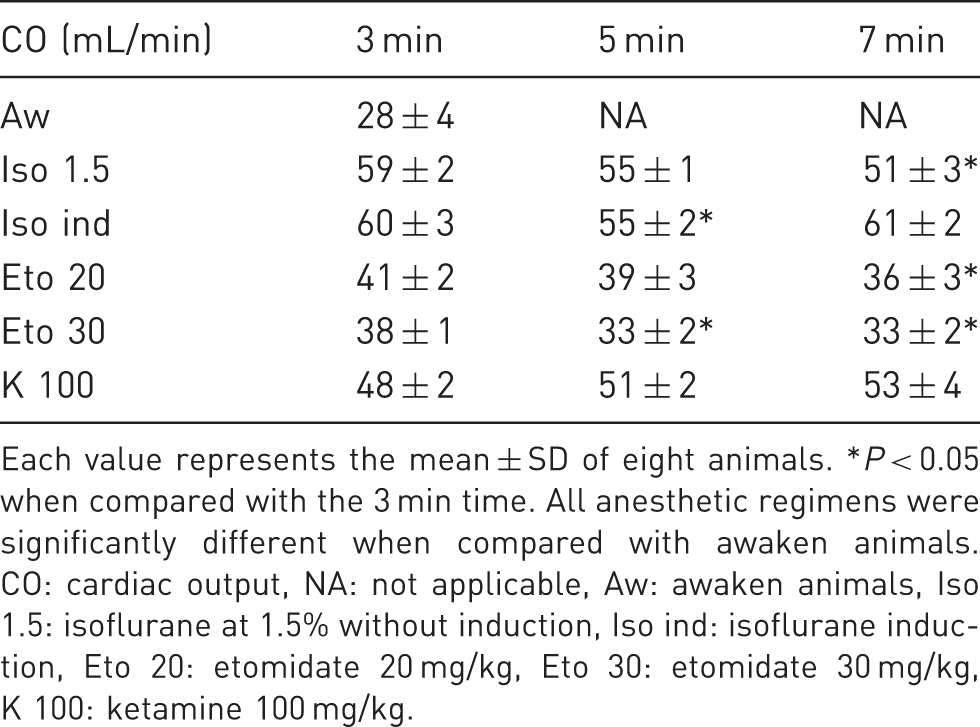
Each value represents the mean ± SD of eight animals. *P < 0.05 when compared with the 3 min time. All anesthetic regimens were significantly different when compared with awaken animals. CO: cardiac output, NA: not applicable, Aw: awaken animals, Iso 1.5: isoflurane at 1.5% without induction, Iso ind: isoflurane induction, Eto 20: etomidate 20 mg/kg, Eto 30: etomidate 30 mg/kg, K 100: ketamine 100 mg/kg.
HR and cardiac function
HR is correlated with FS (R2 = 0.064, P < 0.01) and with LVDd (R2 = 0.074, P < 0.01) whatever the anesthetic regimen chosen, meaning that drugs which modify HR also modify LV dimensions and remodeling indexes. Thus, the anesthetic regimen is important when echocardiography has to be performed.
Discussion
In the present study, we evaluated the cardiac function of mice by echocardiography under different conditions: anesthesia or the conscious state. K 100 and Iso ind provide the better conditions for performing echocardiography in mice. A combination of ketamine and xylazine is usually chosen for this kind of study.20–23 Nevertheless, these drugs are associated with a decrease in HR and FS.5,8,12–16,18,24,25 Ketamine induces relaxation, sedation and analgesia. Its safety range is relatively large and it has few side-effects. 26 Xylazine is a potent alpha-2 agonist agent and provides a deeper and prolonged sedation. Xu et al. found that xylazine is the cause of cardiac depression, leading to a lower and unstable HR. In their study, the optimal dosage for studying echocardiography in mice was ketamine alone at 100 mg/kg. 13 Our study confirms that K 100 does not affect cardiac performance. Ketamine procures the highest HR and FS levels in comparison with isoflurane and etomidate. The use of etomidate is associated with a significant decrease in HR (<450 bpm). 27 In wild-type mice, an intravenous injection of etomidate showed a transient and dose-dependent hypertensive response accompanied by a mild bradycardia. 28 In their study, Erhardt et al. found that etomidate (+carfentanyl) produced few hemodynamic variations; but they did not keep these drugs as first-line anesthesia because muscle spasms were observed. 29 Inhaled volatile anesthetics have already been tested. Isoflurane induces a steady-state level of anesthesia with a minimal cardiac depression.5,14,30,31 In the study by Tan et al., HR and FS% are decreased less by isoflurane than by other drugs; but a significant decrease is noted in HR and FS, compared with measurements in conscious mice. 12 However, as in our study, echocardiography in conscious mice is a very stressful event. The animals are maintained in a holder during the whole procedure. As the animals are handled often and repeatedly, performing this procedure in conscious mice is not ideal for animal welfare. Moreover, as stated in our study the tachycardia induced by stress could modify the results. Halothane provides a more stable HR and FS than a ketamine–xylazine combination, and is associated with more reproducible measurements. 8 In our study, CO was maintained in isoflurane groups by increasing the LVDd, even if HR was lower than in the ketamine group. Many anesthetic drugs lead to cardiodepressive effects. Tribromoethanol is associated with a significant decrease in HR and FS.12,14,17 Hart et al. found that the decrease in HR is less important in comparison with ketamine–xylazine. 18 Kawahara et al. showed that intraperitoneal injection of 30 mg/kg pentobarbital provides a stable HR and FS, 15 however, they did not assess the awakening time, which is a problem with the use of pentobarbital, leading to hypothermia. Moreover, larger doses (40 or 50 mg/kg) are associated with an unstable cardiac function.12,15,16,25
HR is known to be very fast in mice, compared with humans, and is crucial in the evaluation of cardiac function in mice. For assessing cardiac function, physiological levels of HR must be maintained. HR is correlated positively with FS and negatively with LVDd. The relationship between HR and FS is expected because of the force-frequency effect. 32 Rottman et al. described a logarithmic relationship between HR and FS, independent of the presence or choice of anesthesia. 33 Diastolic filling depends on the diastolic duration. When HR is lowered, LVDd increases; nevertheless, even if the LV preload increases, it is insufficient for the maintenance of CO. HR is the major pathway for controlling CO. Normal values have been evaluated in C57BL/6 mice by Rottman et al. 33 They found HR = 683 (SD 63). In other studies, normal HR is noted at 550–620 bpm.34,35 This is also what we have found with Iso 1.5, Iso ind and K 100 (Table 1).
The anesthetic state enables easier animal handling and repeated examinations in a relative short-time period. But anesthesia is associated with a complete sympathetic withdrawal; sympathetic suppression and parasympathetic activation result in cardiac dysfunction. Moreover, there are variabilities in the time to onset, maintenance and recovery from anesthesia between the different drugs. The risk is in the misinterpretation of the obtained data. To avoid the effects of anesthesia, some studies have used echocardiography in conscious animals. In conscious mice, repeated measurements are possible with high reproducibility using this method. Nevertheless, the stress provoked by animal handling induces an activation of sympathetic nervous activity.9,10,12 Exogenous increasing of the sympathetic tone leads to higher HR and FS. 35 Animal training can also prevent severe bradycardia (due to vagal stimulation).12,16,17 In our study, even if mice were trained, the conscious state was associated with non-physiological results: increases in HR and FS, and decreases in LVDd and CO. Nevertheless, it is necessary to limit variations in sympathetic and parasympathetic tones and the anesthesia state allows a better placement of the transducer. Pre-existing sympathetic dysfunction probably limits the effects of anesthetics (sympathetic blockade) or restraint (sympathetic activation). Whether or not to evaluate under anesthesia remains a matter of debate. Anesthesia may mask LV dysfunction, and cardiomyopathy may be better evaluated by echocardiography in conscious mice.
The choice of drug remains crucial. The ideal anesthesia should be easy to administer, onset and recovery should be rapid, it should be safe and should provide reproducible measurements near to the physiological cardiac state. The optimal dose regimen must be sufficient to induce a depth of anesthesia adequate for a proper restraint of the animal, without any cardiovascular effects. Moreover, measurements must be done within a time window. It is important to acquire images at a fixed and defined time to minimize the variations seen with drugs (time between onset of anesthesia and measurements). In fact, the critical variables for echocardiography in mice are: anesthetic drug, timing of echocardiography measurements and genetic background (pre-existing cardiomyopathy). K 100 and Iso ind offer good anesthetic conditions and a quick awakening time, and do not affect cardiac performance in healthy mice. Intravenous administration could be an alternative approach, but it remains more complex. The interest of our results is that it provides two routes of anesthesia: intraperitoneal and inhaled.
The limitations of this study are firstly that it included only healthy mice. The results could have been quite different in pathological mice, in genetically-modified mice or in mice with extreme weight distribution. Secondly, gender or age could also have influenced the results. Stypmann et al. found no gender or age-related changes in cardiac function (minimal increase of HR in younger mice), but this study was made using high doses of xylazine. 24
Conclusion
For evaluating cardiac function in healthy mice K 100 and Iso ind are better anesthetic drugs than isoflurane without induction or etomidate. They are associated with good anesthetic conditions and quick awakening times, and induce no cardiac depression. Examinations in conscious animals produce a sympathetic activation leading to misinterpretation in data. Other studies should be done to evaluate the effects of ketamine and isoflurane in pathological animals.
Footnotes
Acknowledgement
The authors thank the Animal Facility of Rangueil and Cédric Baudelin (Anexplo, UMS US006/Inserm, Toulouse, France) for there technical support.
Declaration of conflicting interests
Support for this study was provided solely from institutional and department sources.
