Abstract
The manner in which an animal's environment is furnished may have significant implications for animal welfare as well as research outcomes. We evaluated four different housing conditions to determine the effects of what has been considered standard rodent enrichment and the exercise opportunities those environments allow on disease progression in the amyotrophic lateral sclerosis mouse model. Forty-eight copper/zinc superoxide dismutase mice (strain: B6SJL-TgN [SOD1-G931]1Gur) (SOD1) and 48 control (C) (strain: B6SJL-TgN[SOD1]2Gur) male mice were randomly assigned to four different conditions where 12 SOD1 and 12 C animals were allotted to each condition (n = 96). Conditions tested the effects of standard housing, a forced exercise regime, access to a mouse house and opportunity for ad libitum exercise on a running wheel. In addition to the daily all-occurrence behavioural sampling, mice were weighed and tested twice per week on gait and Rotor-Rod™ performance until the mice reached the age of 150 days (C) or met the criteria for our humane endpoint (SOD1). The SOD1 mice exposed to the forced exercise regime and wheel access did better in average lifespan and Rotor-Rod™ performance, than SOD1 mice exposed to the standard cage and mouse house conditions. In SOD1 mice, stride length remained longest throughout the progression of the disease in mice exposed to the forced exercise regime compared with other SOD1 conditions. Within the control group, mice in the standard cage and forced exercise regime conditions performed significantly less than the mice with the mouse house and wheels on the Rotor-Rod™. Alpha motor neuron counts were highest in mice with wheels and in mice exposed to forced exercise regime in both mouse strains. All SOD1 mice had significantly lower alpha neuron counts than controls (P < 0.05). These data show that different enrichment strategies affect behaviour and disease progression in a transgenic mouse model, and may have implications for the effects of these strategies on experimental outcomes.
As advancements in animal housing continue to mature in biomedical research, the use of environmental enrichment for rodents has become more popular. What constitutes as environmental enrichment is subject to more recent research efforts, which analyse varieties of enrichment that actually produce a measurable difference in parameters typically considered to represent animal wellbeing. Although the term ‘wellbeing’ has not been universally defined, many agree that it can be properly maintained not only by eliminating pain, distress and behavioural abnormalities, but also by allowing an animal to perform species-specific behaviours. 1–4 Environmental enrichment has been defined by Reinhardt and Reinhardt 5 as, ‘the provision of stimuli which promote the expression of species-appropriate behavioural and mental activities in an under-stimulating artificial environment’. Deprivation of such possibilities may result in psychological distress and induce abnormal behaviours or other pathologies. 6–10 Several studies have described animals in under-stimulating conditions that thwart highly motivated behaviours as inappropriate research models. 11–16 Despite this consensus, many researchers are often cautious about participating in enrichment plans because of the importance of standardization and reproducibility in laboratory science. However, reproducibility between laboratories conducting the same experiment has been questioned due to an increased variability between animals from different laboratories 15,17,18 and systematic variation of environmental factors has been suggested as a more robust approach 18 than standardization. Moreover, when signs indicate that the standard housing conditions negatively affect the wellbeing of animals, the question again arises whether the animal models being used are appropriate for scientific experiments and whether the reliability of results and conclusions remain valid. 3 To ensure that the goals of sound science and responsible animal care are met, it is important to examine the effects of extraneous variables, such as enrichment on research endpoints and evaluate how it may change results and potentially improve the validity of data.
For example, Hockley et al. 19 examined the effects of enrichment on disease progression in the R6/2 Huntington's mouse model. They found that even limited enrichment (addition of 120 mm × 30 mm cardboard tube and food placed on the floor) slowed decline in Rotor-Rod™ performance, despite disease progression. However, further enrichment (additional space and bedding, running wheel and extra toys) was required to induce marked improvement in behavioural tests for normal littermates. Lazarov et al. 20 found that levels of cerebral antibody peptides and amyloid deposition, factors associated with the clinical features of Alzheimer's disease, were reduced in the hippocampus and cortex of enriched vs. standard housed transgenic mice. Environmental enrichment consisting of increased cage space with toys, platforms, tunnels and a running wheel reversed the long-term deficits of lead-exposed rats during learning. This model offers new insights in ameliorating the effects of lead on cognition in children. 21 Such results suggest that increasing environmental stimulation could have a potent effect on disease progression in humans and creates changes in the performance of research models utilized for human disease.
Our objective in this study was to look at the effect of specific kinds of housing and exercise opportunities on disease progression in the amyotrophic lateral sclerosis (ALS or ‘Lou Gherig's disease’) mouse model. This can be done by providing what is historically considered rodent enrichment, such as mouse houses, wheels, nestlets, etc., to standard caging as a means to provide different modes of activity in the form of species-specific behaviour. The definition of enrichment is often debated and is thus beyond the scope of this paper; therefore, any physical environmental addition will be further addressed as ‘furnished’. We prefer to use the term ‘furnished’ until there has been clear scientific evidence that the wellbeing of the animals has been improved. The primary goal is to enrich the animals' lives in measurable ways rather than enriching the environment.
The ALS mouse model was chosen for evaluation in furnished conditions and for assessment of exercise parameters for ALS. ALS is the most common neuromuscular disorder in adult humans and is characterized by progressive muscle wasting and weakness 22 as well as degeneration of cortical and spinal motor neurons followed by paralysis and eventual death. 23 Familial ALS has been attributed to mutations in the SOD1 gene in 20% of the cases. 24,25 Transgenic mice that express mutant SOD1 (mSOD1) develop an adult-onset paralytic condition that reproduces the clinical and pathological hallmarks of ALS. 23,26 In these animals the first clinical signs of disease consist of minor limb shaking and/or tremor at 91 ± 14 days of age. Shortening of stride appears at 125 ± 11 days and death occurs at 136 ± 7 days. Histological analysis reveals loss of motor neurons starting at day 90. 27
The role of physical activity for patients with ALS is controversial. 28 It is frequently recommended that patients avoid physical activity in order to preserve their muscle strength and minimize overwork muscle damage. 29 Recent insights from human clinical studies, however, suggest that a moderate amount of exercise can improve functional rating and decrease spasticity for ALS patients. 29 This research is limited because of the high dropout rate in these types of studies. Human subjects cannot be used to assess the appropriate level of exercise to maximize function without the possibility of accelerating the progression of the disease. Current conservative approaches limit the amount and intensity of exercise physical therapists and/or physicians recommend to patients with ALS. Our particular design allowed us to monitor disease progression through motor performance when animals were provided different levels of exercise via furnished environments. It also allowed us to examine the ways in which control and ALS mice chose to utilize the furnishings provided to them. Therefore, results from this model could provide a preliminary guide to assist in the establishment of appropriate exercise parameters for humans with ALS, as well as ascertain the impact of particular types of furnishings on individuals of this transgenic strain of mice.
Materials and methods
Animals
Forty-eight copper/zinc superoxide dismutase mice (strain: B6SJL-TgN[SOD1-G93A] (SOD1), Stock No. 002726, JAX®) and 48 control (strain: B6SJL-TgN[SOD1]2Gur) (C) male mice aged six to eight weeks were obtained from Jackson Laboratory (Bar Habor, ME, USA). Mice were randomly allotted to four different housing conditions with 12 SOD1 and 12 C animals per condition (n = 96). Mice were allowed to acclimatize to the assigned conditions for one week before data collection. The ALS transgene is expressed in the hemizygous state requiring the use of all male mice. Initially housed in pairs, mice were separated and housed individually two to three days following arrival owing to intense male aggression observed pre-study. Data collection commenced one to two weeks postarrival. Health scores were given for each SOD1 mouse twice a week by an observer without prior knowledge of mouse experimental group assignment. Scores were based on a 4-point scale, where 1 was equivalent to a healthy normal mouse and 4 was equivalent to a mouse in the last stages of ALS. Scores were defined by activity, body posture, balance, smoothness of coat, interest in and ability to eat and drink, and hydration utilizing the skin turgor test (Table 1). The study was reviewed and approved by the Institutional Animal Care and Use Committee prior to obtaining the animals.
Chart used by blind observer to document disease progression
Animals were euthanized at stage 4, unless 15% of body weight was lost prior to this stage
Environment
All animals were maintained in the same conventional room at a temperature of 21 ± 1ºC and relative humidity of 47–52%. In order to observe active behaviour during daylight hours, the room was programmed for a 12/12 h reversed light/dark cycle using red light (light intensity of 75 ± 45 lux during the dark cycle). Behavioural observations were made during the dark cycle within the hours of 10:00–11:00 and 15:00–16:00. Four different conditions were tested and are shown in Figures 1a–d. The first condition was the standard mouse cage (29.2 × 19.1 × 12.7 cm) (Figure 1a). The second condition required removal of each mouse from a standard cage to participate in a daily forced exercise regime for 30 min, six days of the week (Figure 1b). These mice were placed in a motorized exercise wheel on the Forced Running/Walking System® (Lafayette Instrument; Lafayette, IN, USA) at 20 s per revolution during the 30 min. Once completed, the mouse was returned to its cage. The third condition consisted of a standard cage with a red Techniplast mouse house™ with dimensions of 11.7 × 11.7 × 6.5 cm (Figure 1c). The fourth condition provided ad libitum access to a single mouse exercise wheel in a cage with dimensions (30.5 × 19.1 × 19.1 cm) (Figure 1d). Sanichip™ (PJ Murphy Forest Products Corp, Montville, NJ, USA) bedding was provided in all cages. Cages and bedding were changed once a week. All conditions were provided with nesting material (cotton fiber Nestlets®, 5 cm × 5 cm, Ancare, Bellmore, NY, USA) every week. Tap water in drinking bottles and pelleted food containing 20.0% protein, 9.0% fat, 4.0% fibre, 6.5% ash and 2.5% added mineral (Lab Diet®, PMI, Richmond, IN, USA) were provided ad libitum.

Four housing conditions evaluated: (a) standard caging; (b) forced exercise was provided for 30 min six days a week in wheels rotating at 20 s per revolution, mice are returned to standard housing when complete; (c) mouse house; (d) ad libitum access to a wheel (running wheels in a cage size of 30.5 × 19.1 × 19.1 cm)
Behaviour
Observations
Mice were observed six days per week. Daily all-occurrence behavioural sampling was utilized to estimate time budgets. This method consisted of four cycles of 60 s intervals per cage, repeated twice for a total of 8 min of observation per cage 6 days/week. Observations were completed in two sets of 4 min separated by 30 min each. Since the first set of four needed to be taken before providing forced exercise, and the second set of four completed after the 30 min forced exercise, this time span was applied between the two sets of observations for all conditions. All occurrences of rest, groom, ingestion and locomotor activity (cage exploration and wheel use) were documented for each mouse during this time. Cages were observed four at a time; however, data were recorded for individual mice. Changes in behavioural states were calculated by summing the tallies of each behavioral state and subtracting one. This is equivalent to the number of times the animal changed its behavioural state during the sampling period. Mouse house entry was documented for mice with a mouse house. The use of exercise wheels by mice was recorded continuously by the 86060 Activity Wheel Counter® by Lafayette Instrument. The Counter in each cage used an optical sensor to monitor revolutions of the mouse activity wheel. Data collected from each wheel were automatically downloaded and stored in a computer via Starter Interface (Lafayette Instrument) using Activity Wheel Monitor software® (Lafayette Instrument). Data for mice voluntarily running on wheels were recorded in 1 h intervals. Measures recorded included revolutions per interval (rpi), revolutions per minute (rpm), total revolutions and total metres run.
Tests
All mice were weighed and tested twice per week on gait stride length and Rotor-Rod™ performance (Lafayette Instruments, rotating axle 3.6 cm diameter; rpm 16) until mice reached the age of 150 days (C) or met the criteria for our humane endpoint (SOD1) described in the section below. The two behavioural tests were selected as a parallel to the patient ALS Functional Rating Scale (ALSFRS) used in human patient assessment. The ALSFRS assesses upper and lower extremity functions as well as bulbar functions such as chewing, swallowing, speaking and breathing. Lower extremity function was assessed using a footprint analysis first described by Mullenix et al. 30 This test has been shown to be sensitive to subtle changes in gait following central and peripheral nervous system deficits. 30 Briefly, the animal's hind paws are gently painted with non-toxic ink and the animal is placed in a narrow corridor lined with paper. If the animal hesitates, stops or turns around, the process is repeated until there are at least three good strides with each lower extremity. Footprints from middle of recorded walk are then measured for stride length (left footprint to next left footprint) and width.
Use of upper and lower extremities was tested on a rotating rod. The period for which a mouse could remain walking on a Rotor-Rod Treadmill™ without releasing was recorded to determine locomotor coordination and limb function. Each mouse received three trials with the best result recorded, unless the maximum of 120 s was reached.
Endpoints, histology and stereology
Mice were euthanized if they had lost 15% of their baseline body weight or exhibited an inability to reach for food or water. Control mice were euthanized at 150 days of age. Animals were sacrificed by overdose of sodium pentobarbital (200 mg/kg), followed by cervical dislocation. The brain and spinal cord were removed and submerged in 10% formalin. The lumbar and cervical portions of the spinal cord were then isolated. Beginning at L2–L3, the spinal cord was quickly frozen and exhaustively sectioned on a sliding microtome at a nominal thickness of 14 μm. Every 15th section was collected, Nissl-stained, dehydrated and mounted onto glass slides.
The dorsal horn motor neurons were counted using the optical fractionator stereological procedure to quantify neuronal loss. Details of the method are available in protocol format. 31 Briefly, the method combines systematic random sampling of known fractions of the total structure (the fractionator) with direct counts of motor neurons generated using the optical dissector counting probe. 32–34 The fractionator sampling design estimates the total number of neurons by multiplying the number counted in dissector frames by an inverse of the product of all fractions of the sampling hierarchy. This sampling scheme consisted of the spinal cord fraction (SCF) and the section sample fraction (SSF). SCF represented the original fraction collected during sectioning (1/15th). SSF represented the fraction of collected sections used for counting. The SSF for each animal was adjusted so that six sections were examined from each animal using systematic random sampling rules. In this case, SSF was determined by counting the total number of sections from a set of sections of an animal and dividing by six (e.g. if there were 85 sections, then every 14th section would be counted). The entire area of each dorsal horn (left or right side randomly selected for each animal) was tiled with dissector frames. Preliminary investigation indicated that there was no difference between right and left sides (data not shown).
Estimation of neurons in the sections was performed using a BX-40 (Olympus, Melville, NY, USA) compound trinocular microscope equipped with a high precision motorized stage with motorized z-focus (Ludl Electronic Products, Hawthorne, NY, USA), and a MicroFire (Optronics, Goieta, CA, USA) digital video camera. The video image and control of the microscope stage and z-focus, and count sampling functions are integrated and controlled by StereoInvestigator (version 5.0, MicroBrightField, Colchester, VT, USA) software running on a PC style computer.
Neurons were counted according to the counting rules for the optical dissector. 33,35,36 The individual performing the counts was blind with regards to treatment. Briefly, the dorsal horn of either the right or left side (randomly selected) was outlined using the stereology software tools, and a dissector frame overlay grid was randomly positioned over the dorsal horn. Sequential dissectors were placed so as to completely tile the dorsal horn, effectively counting all neurons of interest in each outlined section. The nucleolus of the neuron's nucleus was used as the counting unit, and supporting histology featured were used to classify the cell as a neuron. These features included size, shape, presence of Nissl and location. A neuron was counted if the nucleolus was in focus within the section without touching the apparent bottom surface of the section (forbidden plane), and without touching the forbidden lines of the dissector counting frame. Large neurons, believed to represent both alpha and gamma neurons, were combined for the purpose of counting motor neurons. Alpha motor neurons were considered to be the larger neurons, whereas gamma motor neurons were the smaller neurons of the neuron classification.
Statistics
Traditionally, eight subjects are considered the minimum number of animals for statistical significance in transgenic studies. To allow for some loss during transport and that typical to the disease, 12 animals were obtained for each condition in this study (four different housing conditions for each strain, n = 96). Behaviour and behavioural test data were analysed using PROC MIXED in SAS version 8.0 using Mixed Models in a repeated measures design. All data were adjusted for normality by carrying out log transformations. Correlations between behavioural tests and neuron counts were tested using Pearson product-moment method of PROC CORR (SAS version 8.0). The stereological-derived neuron counts were analysed by independent t-tests performed on appropriate pairs of treatment groups using SPSS Base 10 (SPSS Inc, Chicago, IL, USA) statistical software. Statements of statistical significance are based upon P < 0.05.
Results
Behaviour
Although individual housing eliminated a more desirable social housing element, it did help avoid injuries and deaths related to fighting as well as deducted group housing factors on behavioural data, such as wheel and house use. Average lifespan for all mouse groups is demonstrated in Figure 2. SOD1 mice housed in the mouse house condition averaged significantly lower in lifespan than controls did (P < 0.05). Although, SOD1 mice in the forced exercise regime and mice with wheels averaged higher in lifespan than mice in standard caging or mice with houses, no significant differences were observed across conditions within treatments.
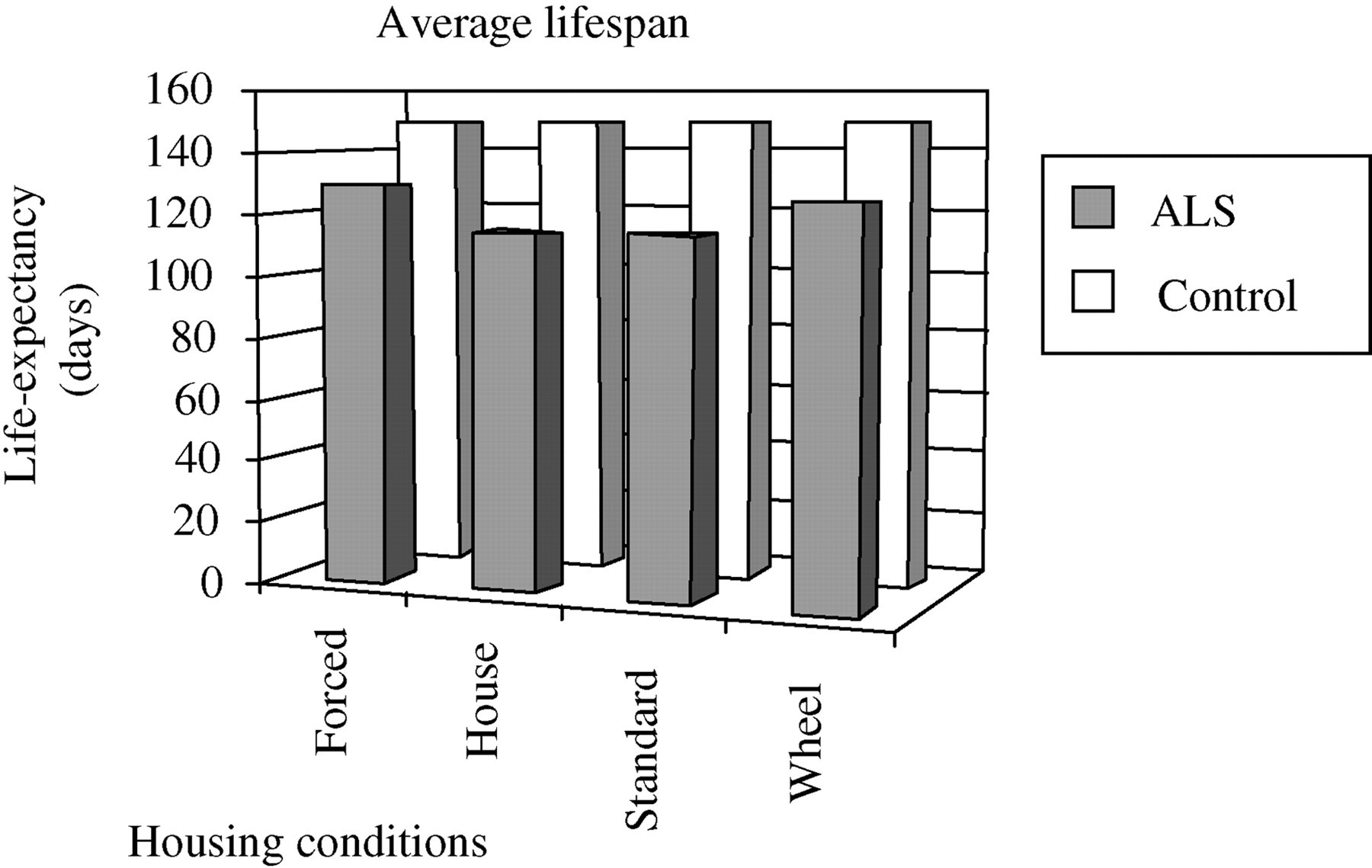
Average lifespan for all mice according to each housing condition. Data reflected as mean and (SE). Statistically significant (df = 95, P < 0.05) difference between mouse strains for mice in the mouse house condition
Changes in behavioural states (Figure 3), derived from the number of occurrences of behaviour changes recorded per sample, were not significantly lower for SOD1 compared with control mice (P > 0.05). Among SOD1 mice, changes in behavioural state were less frequent in standard housed mice (P < 0.001) compared with the three other conditions. Different furnished conditions did not result in statistically significant changes in behavioural states among control animals.
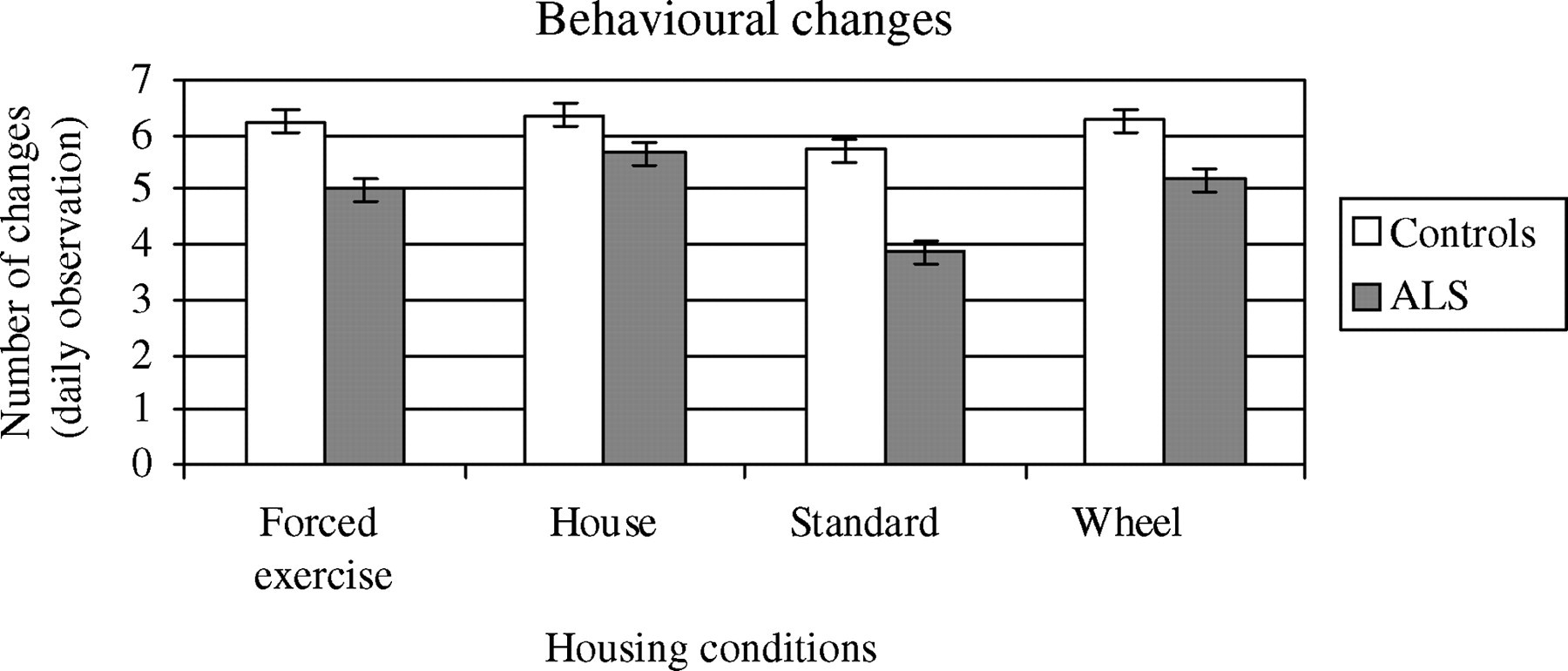
Changes in behavioural states, derived from the number of occurrences of behaviour changes recorded per sample. Statistically significant (df = 55, P < 0.001) difference between SOD1 mice in standard caging compared with three other conditions. Data reflected as mean and (SE)
Among controls, mice with houses rested significantly more often than all other conditions (P < 0.05, Table 2). Resting did not necessarily always occur in the house however. We expected most mice to utilize the mouse house for sleeping and nesting, but found mice to be quite variable in their use of the houses across and within the SOD1 and control groups. Also, control mice in the forced exercise regime and standard caging rested significantly more often than control mice with access to a wheel (P < 0.05; Table 2). Among SOD1 mice, significant differences across housing conditions for frequency of rest did not exist.
Frequency of behavioural change and treatments in control and SOD1 mice according to housing condition
Data reflected in frequency, using chi-square analysis. Rotor-Rod™ data reflected in mean and (SE) *Statistically significant (df = 95, P < 0.005) difference between each measurement indicated: control forced exercise* and control standard* vs. control house* and control wheel*
Wheels were utilized vigorously by all mice regardless of strain in the ad libitum wheel condition. All wheel use measures showed significantly lower performance by SOD1 mice compared with controls as expected (P < 0.0001; Table 3), primarily in the 41–80-day age group (P < 0.005, Table 4). Significantly fewer metres were observed in the SOD1 mice age group 81–120 than controls (P < 0.0001, Table 4). In addition, SOD1 mice increase their rpi and rpms in the latter stage of life (opposite to the controls' response), while metres decrease as represented in Table 4. Mice with access to wheels in the SOD1 strain entered their wheel to run/walk less often than did control mice with wheels (P < 0.0001; Table 3). They also rested significantly more often than control mice with wheels (P < 0.0001, Table 2).
Mean and (SE) of control and SOD1 mice using wheels voluntarily
*Statistically significant (df = 23, P < 0.0001) difference between mouse strains for each measurement indicated. rpi: revolutions per interval; rpm: revolutions per minute
Mean and (SE) of control and SOD1 mice using wheels voluntarily according to age group (age 1–40 not collected)
Statistically significant (*df = 291, P < 0.005; **df = 674, P < 0.0001) difference between each measurement indicated rpi: revolutions per interval; rpm: revolutions per minute
Tests
Within the controls, mice in the forced exercise regime and mice in standard caging performed significantly lower on the Rotor-Rod™ than mice with houses and mice with wheels (P < 0.005, Table 2). Within the SOD1 group, mice with wheel access performed higher on the Rotor-Rod™ and weighed more than forced exercise mice.
Control mice with houses and wheels weighed significantly less (Figure 4) than control mice in standard caging and forced exercise (P < 0.0001). In SOD1 mice, weights of mice with houses were significantly less than mice with standard caging (P < 0.0001).
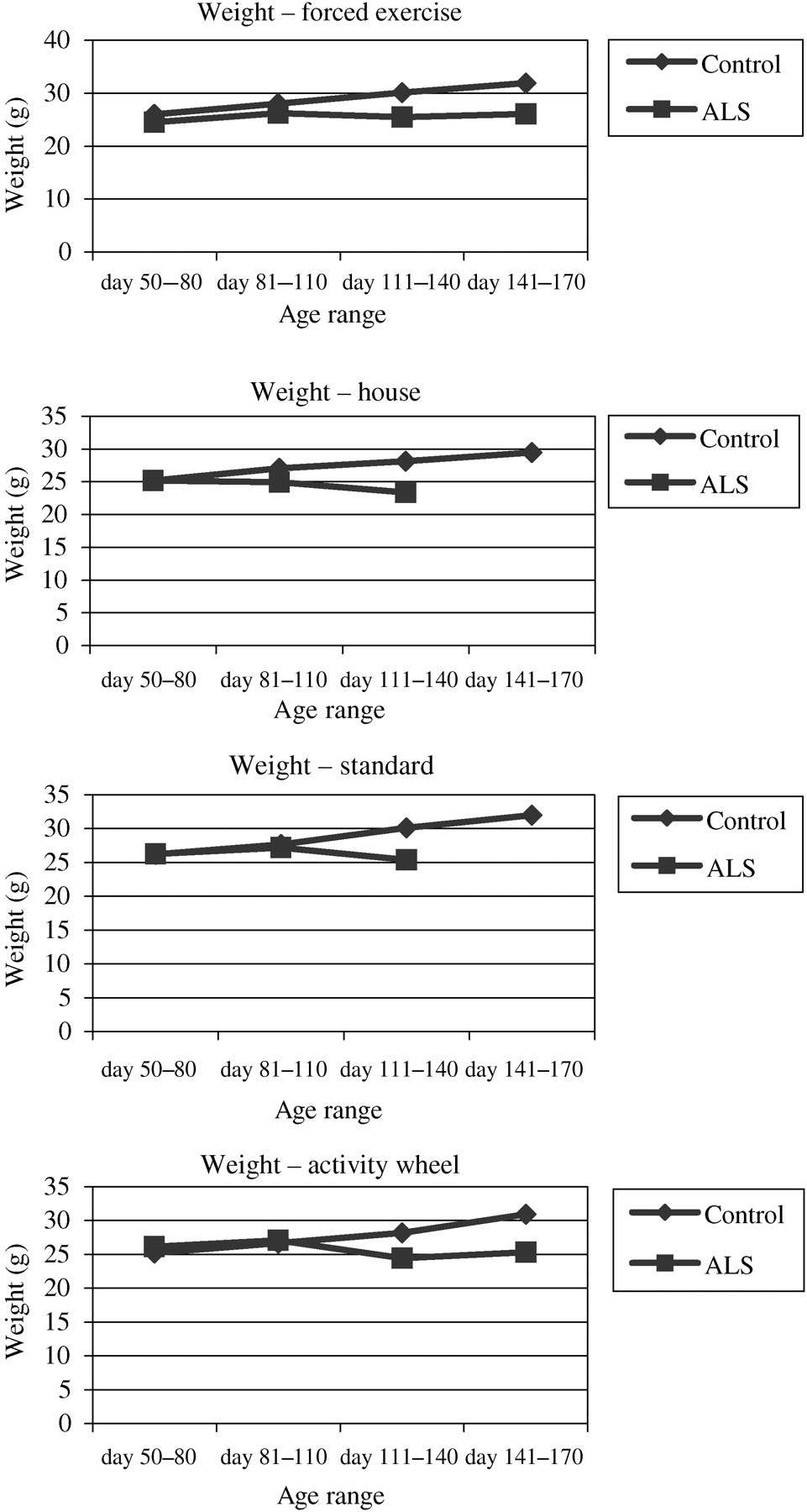
Average weights for all mice according to each housing condition. Data reflected as mean and (SE). *Statistically significant (df = 7, P < 0.0001) difference between mouse strains for each measurement indicated: SOD1 house vs. SOD1 standard*, control forced exercise and control standard vs. control house* and control wheel*
Gait stride length means (Table 5) for SOD1 mice were lower for each housing condition than controls in the last two age groups (days 81–160). Also, in the final age group (days 121–160), gait stride lengths for SOD1 mice were significantly lower than controls in each housing condition (P < 0.0001, Figure 5). Stride length was also longest among control mice in the forced exercise regime but shortest in control mice standard caging (P < 0.05) in the final age group. Starting at day 81 for SOD1 mice, stride length was longest among mice in the forced exercise regime and shortest among mice with wheels (P < 0.05).
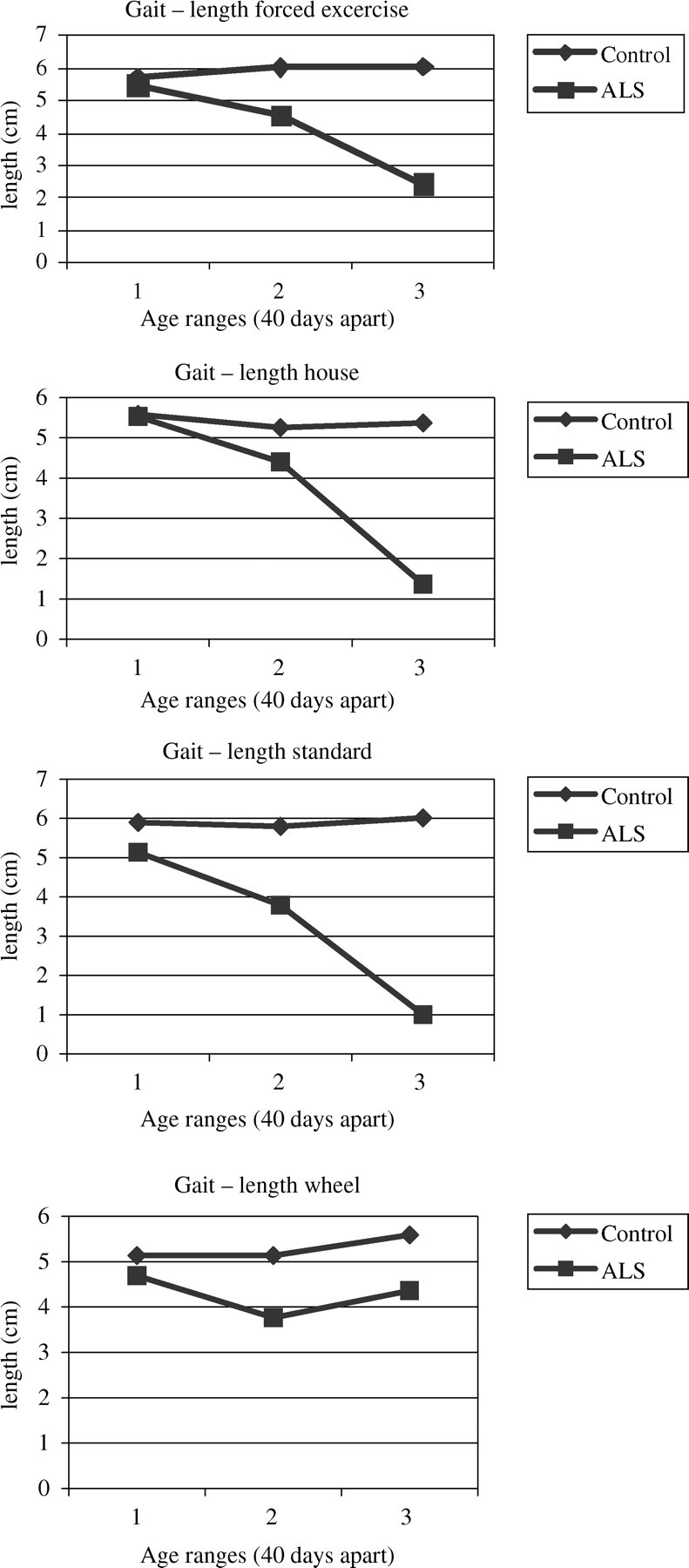
Gait stride lengths for final age group (days 121–160) according to each housing condition. Data reflected as mean and (SE). Statistically significant (P < 0.0001) difference between mouse strains for each housing system
Gait stride lengths for all age groups (ages 1–40 not collected) according to each housing condition
Data reflected in mean and (SE)
**Statistically significant (P < 0.0001) difference between mouse strains for each measurement indicated
Histology and stereology
The mean numbers reported for control mice in three conditions are roughly similar in magnitude and are thus not significantly different. For example, the number of alpha neurons was found to be 15,270, 13,567 and 14,330, respectively, for forced exercise, mice with houses and mice with wheels. Unfortunately, standard housing counts were lost in processing. Alpha motor neurons among SOD1 mice equalled 6293, 5028 and 8306 for forced exercise mice, mice with houses and mice with wheels, respectively. There was a significant difference among alpha neurons for SOD1 mice with houses and SOD1 mice with wheels (P < 0.05, Table 6). All SOD1 mice had significantly lower alpha neuron counts than controls (P < 0.05). Although not significant, SOD1 forced exercise mice had lower (P = 0.10) alpha neuron counts than SOD1 mice with wheels.
Mean and (SE) of motor neuron counts for control and SOD1 mice
*Statistically significant (df = 9, P < 0.05) difference between mice strains for alpha motor neurons
Discussion
To assess the effects of enrichment or ‘furnishings’ on disease progression in ALS mice, we contrasted the standard housing condition with three different levels of cage furnishings/exercise opportunities, a daily forced exercise regime, provision of a mouse house only, and opportunity to exercise voluntarily on a wheel in a cage with more cage space. Mice of the SOD1 strain exposed to the forced exercise, as well as those assigned to the voluntary running wheel condition, demonstrated higher averages in lifespan and Rotor-Rod™ performance over mice in standard and mouse house conditions.
Similar studies in neurodegenerative disease found that participation in any physical activity would decrease the risk of aggravating the symptoms of individuals diagnosed with Alzheimer's, or Huntington's disease. 19,20 Drory et al. 29 also found that a regular moderate physical exercise programme has a short-lived positive effect on disability in ALS patients and should be recommended. In our study, changes in behavioural state among SOD1 mice were significantly less frequent in standard housed mice compared with the other three conditions, indicating a decreased activity level in the standard caging. SOD1 mice with access to a mouse house exhibited the highest frequency of behavioural change, demonstrating high variability of house utilization by mice. We observed mice using the house as a place to groom only, or strictly as a platform to reach the top of the cage bars. Although 90% of mice created a nesting area with the nestlet, nearly half of the total mice with houses built nests outside the house throughout the duration of the study. This shows the variability of enrichment utilization and implies that nesting material is valuable for non-breeding males despite provision of shelter. This observation may suggest that nest building has an important thermoregulation function for adult mice rather than exclusively for pups in a breeding situation. Such information is valuable in terms of meeting the behavioural and physiological needs of mice in biomedical research.
Our findings also suggest that mice provided with an opportunity to exercise with wheels will voluntarily choose to do so, while less interactive furnishings, such as the mouse house produced more variable behavioural responses from mice. Even mice with some debilitating conditions, such as SOD1 mice, exhibited a strong motivation to use an exercise wheel. Sherwin's 37 review of wheel-running research suggests that although there is no consensus on why it occurs, a possible motivating factor for wheel-running is that it is self-reinforcing. In our study, the SOD1 mice continued to utilize the wheel, albeit at a slower pace and for shorter distances, indicating that wheel running may have been regarded by the mice as an important activity despite disease progression. Attempts to run short spurts at higher speeds were made as their physical handicap grew, suggesting the motivation to run remains if not intensifies during disease progression. This condition was thus categorized as the highest level of exercise for this study.
Fischer and Peduzzi 38 found rats with chronic spinal cord injuries housed in furnished environments that scored significantly better on combined behavioural score testing and BBB locomotor test 39 than rats with the same injury housed in standard laboratory cages. Our study similarly found that SOD1 mice exposed to the forced exercise regime and mice with wheels did better in average life-span and Rotor-Rod™ performance than SOD1 mice in standard cages and those with houses. Stride length remained longest throughout disease progression in SOD1 mice in the forced exercise regime compared with the other SOD1 conditions. Similar to other studies looking at neurodegenerative diseases, 19 we have found Rotor-Rod™ performance and gait stride to be reliable measures of disease progression in ALS mice.
Alpha motor neuron counts were highest in mice with wheels and mice exposed to the forced exercise regime in both mice strains. All SOD1 mice had significantly lower alpha motor neuron counts than controls (P < 0.05). The stereology methods used in the present report, based upon the optical fractionator method, have been successfully used to characterize total motor neuron number for specific spinal cord segments in the SOD1 mouse model, 40,41 as well as in humans. 42 Combining the optical fractionator stereology method with the SOD1 model for ALS suggests that this model would be of value in testing various therapeutic measures including exercise regimes, new drug candidates or combinations of each.
Mice in furnished and super-furnished cages have been shown in another study to gain more weight than mice in non-furnished cages, however, the level of exercise provided by different enrichment will likely have an impact on weight gain. The same study also showed the strain of the mouse, and not the enrichment condition, had the greatest impact on both mean values and variation. 43 Barnard et al. 44 also found that mice provisioned with ‘furnished’ or ‘enriched’ cages had a pattern of increases in body weight. This trend was also stated by Chamove, 7 where male mice in such environments had an increase in weight. Olson and Dahlborn 45 did an extensive literature review of the impact of enrichment and its impact on weight gain.
Within our study, SOD1 mice in standard caging weighed the most, and SOD1 mice with a mouse house weighed the least. SOD1 mice exposed to the forced exercise regime and mice with wheels only weighed slightly less than mice in standard caging; however, SOD1 mice with wheels showed the highest frequency of ingesting behaviour and SOD1 mice exposed to forced exercise were observed ingesting the least in the behavioural data. These findings may indicate that when considering the frequency of ingesting behaviours observed in both SOD1 mice exposed to forced exercise and SOD1 mice with wheels, both groups maintained good weights. However, with such variability among reports, effects of cage furnishings should continue to be tested on specific models and strains frequently used in research.
These data reflect that different enrichment strategies affect behaviour and disease progression in a transgenic mouse model, and may have implications for the effects of these strategies on experimental outcomes. Also, results from this model could provide a preliminary guide to assist in the establishment of appropriate exercise parameters for humans with ALS.
Footnotes
ACKNOWLEDGEMENTS
Partial funding of this project was provided by the Center for Alternative Animal Testing at John Hopkins Bloomberg School of Public Health. We would like to acknowledge Larisa Kuchta, Dena Van Lierop, Stephanie Weiler and Marsha Melnick for their work on this study. We would also like to thank Linda Prentice for providing her expertise in sectioning and staining slides as well as Dr Larry Carbone, Anne Neil and Dr Lisa Heath for their assistance in isolating spinal cords.
