Abstract
Mouse parvoviruses are among the most prevalent infectious pathogens in contemporary mouse colonies. To improve the efficiency of routine screening for mouse parvovirus infections, a multiplex polymerase chain reaction (PCR) assay targeting the VP gene was developed. The assay detected minute virus of mice (MVM), mouse parvovirus (MPV) and a mouse housekeeping gene (α-actin) and was able to specifically detect MVM and MPV at levels as low as 50 copies. Co-infection with the two viruses with up to 200-fold differences in viral concentrations can easily be detected. The multiplex PCR assay developed here could be a useful tool for monitoring mouse health and the viral contamination of biological materials.
Rodent parvovirus is one of the most common diseases in experimental rodents. Two parvoviruses have been identified in mice, minute virus of mice (MVM) and mouse parvovirus (MPV).1,2 These viruses have high affinities for dividing cells, such as enteroepithelial cells, lymphatic cells, haematopoietic cells and neoplastic cells.3–5 MVM and MPV usually induce asymptomatic infections in mice. In addition, both viruses have been found to contaminate cultured cells.2,3,6 Infection of either in vivo or in vitro systems could alter experimental data collected regarding the immune system, neoplasm and transplantation.5,7,8
Because MVM and MPV infections are often subclinical with no specific histopathology lesions observed, antibody detection is routinely used as the diagnostic assay for rodent parvovirus infections. 9 While serological assays may be able to survey a large number of serum samples with high sensitivity, such an assay could not be applied to screen immunodeficient mice, cell lines and biological materials. Polymerase chain reaction (PCR) has the advantages of high sensitivity and high specificity, which enables it to detect infections in experimental mice and biological materials and even environmental contamination.
While single PCR assays specific for either MVM or MPV have long been established,10,11 no assay has been reported that can detect both viruses simultaneously. This study established a multiplex PCR assay to detect MVM and MPV with a housekeeping gene as an internal control. This multiplex PCR assay remains sensitive with a 200-fold difference in the viral copy number.
Materials and Methods
Animals
The study protocol was performed under the terms of the Animal Protection Act in Taiwan, was approved by the National Taiwan University's Institutional Animal Care and Use Committee, and was conducted in compliance with the Guide for the Care and Use of Laboratory Animals in Taiwan. A total of 174 mice were obtained from seven different laboratory mouse colonies in Taiwan. They were in six different genetic backgrounds, including 63 ICR (CD-1) mice, one ICR genetically-engineered mouse (ICR GEM), 15 FVB/NJNarl, one C57BL/6JNarl, 50 C57BL/6J genetically-engineered mice (C57BL/6J GEM), 10 BALB/cByJNarl, five BALB/cAnN.Cg-Foxnlnu/CrlNarl, 24 CB17/lcr-Prkdcscid/CrlNarl and five NOD.CB17-Prkdcscid/JNarl (Table 1). These animals varied in age, including 115 young adults (6 weeks ≤ age ≤ 8 weeks), 35 middle-aged mice (8 weeks < age ≤ 16 weeks) and 24 adult mice older than 16 weeks (Table 1). These colonies and animals were selected because they were representatives of animal research colonies in Taiwan and/or were readily available through research collaboration or routinely health monitoring services. Tissues and serum samples were collected after CO2 euthanasia and stored at –80°C until use.
Mouse strains/stocks, age group and the results of the MVM/MPV/actin multiplex PCR
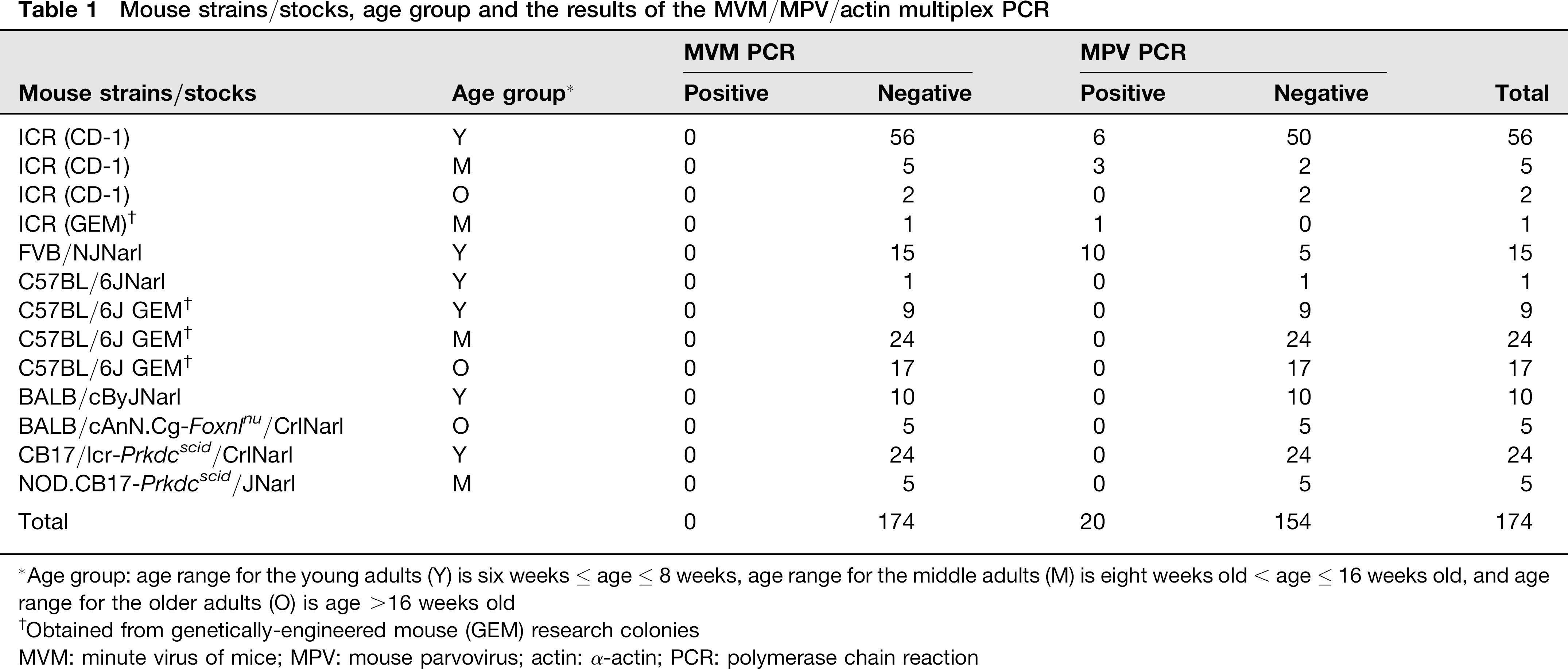
Age group: age range for the young adults (Y) is six weeks ≤ age ≤ 8 weeks, age range for the middle adults (M) is eight weeks old < age > 16 weeks old, and age range for the older adults (O) is age >16 weeks old
Obtained from genetically-engineered mouse (GEM) research colonies
MVM: minute virus of mice; MPV: mouse parvovirus; actin: α-actin; PCR: polymerase chain reaction
Viral, tissue and cultured cell DNA
Liver tissues of MPV-1f-infected mice were gifts from Dr Ching-Fong Liao of Academia Sinica, and rat parvovirus (RPV–NTU)-infected tissues were collected from naturally infected rats in Taiwan. MVM, Kilham rat virus (KRV), Toolan's H-1 virus (H-1), and rat minute virus (RMV) were gifts from Dr Lela Riley of the Research Animal Diagnostic Laboratory of the University of Missouri, Columbia, USA. MPV and RPV–NTU DNA were extracted from tissues with the QIAamp® DNA Mini Kit, and viral DNAs of MVM, KRV, H-1, and RMV were extracted with the QIAamp® MiniElute® Virus Spin Kit (Qiagen, Hilden, Germany).
MVM and MPV plasmids, containing the MVM viral protein (VP) gene and the MPV VP gene, were used to transform Escherichia coli TOP10 competent cells, and the transformed cells were screened for the presence of MVM and MPV DNA. Plasmid DNA preparations of MVM VP and MPV VP were prepared, and their concentrations were quantified by our laboratory. The plasmid copy number was calculated using the molecular weight of the plasmid and Avogadro's constant. Concentrated plasmid DNA stocks were stored at –20°C.
Spleens of 174 mice and 16 different cultured cell samples were collected for surveillance of the MVM and MPV infections. DNA was extracted from spleens and cell lines with the QIAamp® DNA Mini Kit and stored at –80°C. Tissue DNA extracts of mice obtained from a colony documented to be free of MVM and MPV infections based on previous serology and PCR monitoring results were used as negative controls in the PCR assay. The DNA concentration and the purity of the tissue DNA extracts were determined using a spectrophotometer.
Primers
Primers for the MVM/MPV/actin multiplex PCR assay were selected based on alignments performed with the Lasergene® analysis program (DNASTAR, Inc, Madison, WI, USA). The MVM primers 3163F and 3627R (Table 2), located in the VP region, were designed to be highly specific for MVM versus all other rodent parvoviruses. The MPV primers 4107F and 4388R (Table 2), located in the VP region, were designed from MPV sequences that were distinct from those of other rodent parvoviruses. The housekeeping gene primers Ac1 and Ac2, located in the α-actin gene region, were designed by Müller et al. 12 Oligonucleotide primers were synthesized at Invitrogen (Carlsbad, CA, USA).
Oligonucleotide primers used for the MVM/MPV/actin multiplex PCR assay

MVM: minute virus of mice; MPV: mouse parvovirus
PCR
Each PCR mixture contained various amounts of DNA template or 5 μL of tissue DNA extract, 0.6 μmol/L each of MPV primers (4107F and 4388R), 0.8 μmol/L each of MVM primers (3163F and 3627R), 0.05 μmol/L each of Ac1 and Ac2, 0.2 mmol/L each of dNTP (OneStar, Taipei County, Taiwan, ROC), 1X GoTag® Flexi buffer (Promega, Madison, WI, USA), 1.75 mmol/L MgCl2 (Promega) and 1.25 U of GoTag Flexi DNA polymerase (Promega). The following thermocycling profile was used for the MVM/MPV/actin multiplex PCR: 95°C for 60 s (1 cycle); 95°C for 30 s, 56°C for 30 s, 72°C for 30 s (35 cycles), and then 4°C thereafter. PCR products (10 μL) were electrophoretically separated in 2% agarose gels, stained with ethidium bromide and visualized by ultraviolet light. To assess the specificity of the MVM/MPV/actin multiplex PCR assay, extracted MVM DNA (102 copies), MPV DNA (102 copies) and 105 DNA copies of KRV, H-1, RPV and RMV used as templates were amplified under the conditions described above. To mimic diagnostic samples, 1.25 μg of DNA extracted from the spleens of parvovirus-free mice were added into various parvovirus tubes to assess the tissue background in the multiplex PCR described above. Tissue DNA extracts from mice free of MPV and MVM and a no-template control were included as negative controls. To determine the sensitivity of the PCR assay, various amounts of MVM and MPV DNA (103, 102, 50, 10 and 5 copies) were used as templates. An MPV/MVM-free tube was included as a negative control in every reaction. To evaluate the efficiency of detecting co-infection with MVM and MPV, serial dilutions (104, 103, 102, 50 or 10 copies) of one virus were co-amplified with a fixed copy number (104 copies) of the other viral DNA samples.
Surveillance of MVM and MPV infections in laboratory mice and the contamination of cultured cells in Taiwan
To understand the MVM and MPV status of laboratory mice in Taiwan, samples from 174 mice from seven different laboratory facilities were screened using the MVM/MPV/actin multiplex PCR assay. In addition, to detect MVM and MPV contamination of cultured cells in laboratories, 16 different cultured cell specimens (Table 3) were collected and screened using the multiplex PCR assay.
Screening results for contamination with MVM and MPV in 16 cell cultures
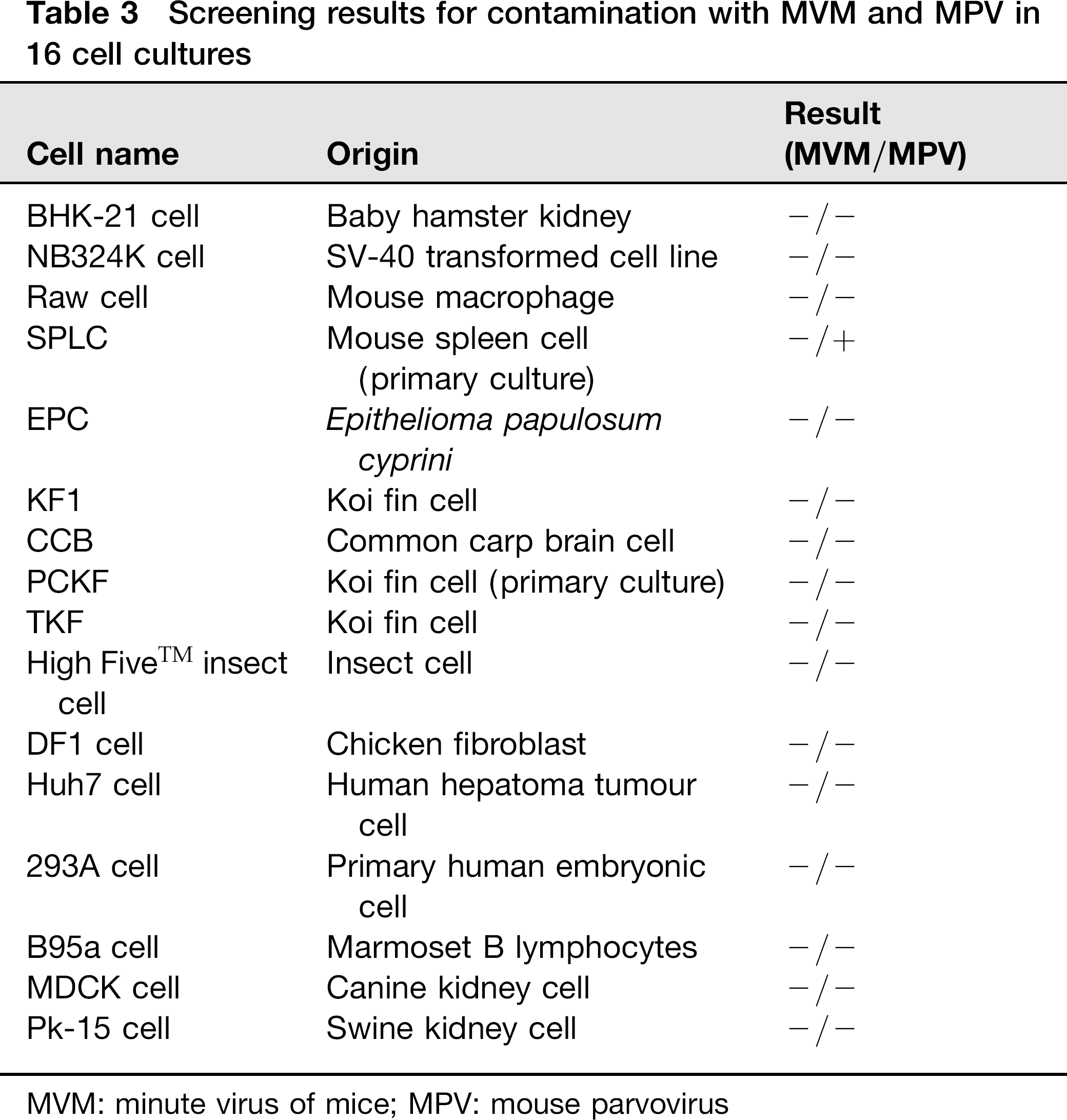
MVM: minute virus of mice; MPV: mouse parvovirus
DNA sequencing
Amplified DNA fragments were purified using the QIAquick® PCR purification kit (Qiagen) using the protocol recommended by the manufacturer. Nucleotide sequences were determined by the dideoxy-chain termination method with the BigDye® Termination Cycle Sequencing v3.1 kit (Perkin-Elmer, Applied Biosystems, Foster, CA, USA) and confirmed with dideoxy-chain termination at the National Taiwan University College of Medicine. Sequence data were analysed with the EditSeqTM and SeqManTM components of the Lasergene package (DNASTAR). The sequences were compared with the published sequences of MVM and MPV.
Results
Specificity and sensitivity
The MVM/MPV/actin multiplex PCR assay was designed to simultaneously detect MPV, MVM and α-actin (housekeeping gene). Several rodent parvoviruses were screened to ensure the specificity of the multiplex PCR assay. The MVM (465 bp) and MPV (366 bp) fragments were amplified from MVM or MPV viral DNA, and the 153-bp α-actin fragment was amplified from the host's DNA (Figure 1a). The PCR products (465- and 366-bp fragments) were confirmed to be MVM and MPV DNA by DNA sequencing. In KRV, H-1, RPV, RMV and the negative tissue control, only the α-actin fragment (153 bp in size) was amplified. To determine the sensitivity of the multiplex PCR assay, various amounts of the MVM and MPV VP2 genes were amplified in the presence of MVM/MPV-free tissue DNA. The MVM/MPV/actin multiplex PCR assay simultaneously detected as few as 10 copies of MPV and 50 copies of MVM and the α-actin gene (Figure 1b).
(a) Specificity of the minute virus of mice (MVM)/mouse parvovirus (MPV)/actin multiplex PCR assay. MW: molecular weight marker; N: no template control; T: the MVM/MPV-negative tissue control; MPV: 102 copies of MPV DNA; MVM: 102 copies of MVM DNA; KRV: 105 copies of KRV DNA; RMV: 105 copies of RMV-1 DNA; RPV: 105 copies of RPV–NTU DNA; H-1: 105 copies of H-1 virus DNA. (b) Sensitivity of the MVM/MPV/actin multiplex PCR assay. Serial dilutions of MPV and MVM VP DNA were mixed with virus-negative tissue DNA. MW: molecular weight marker; N: no template control; T: MVM/MPV-negative tissue control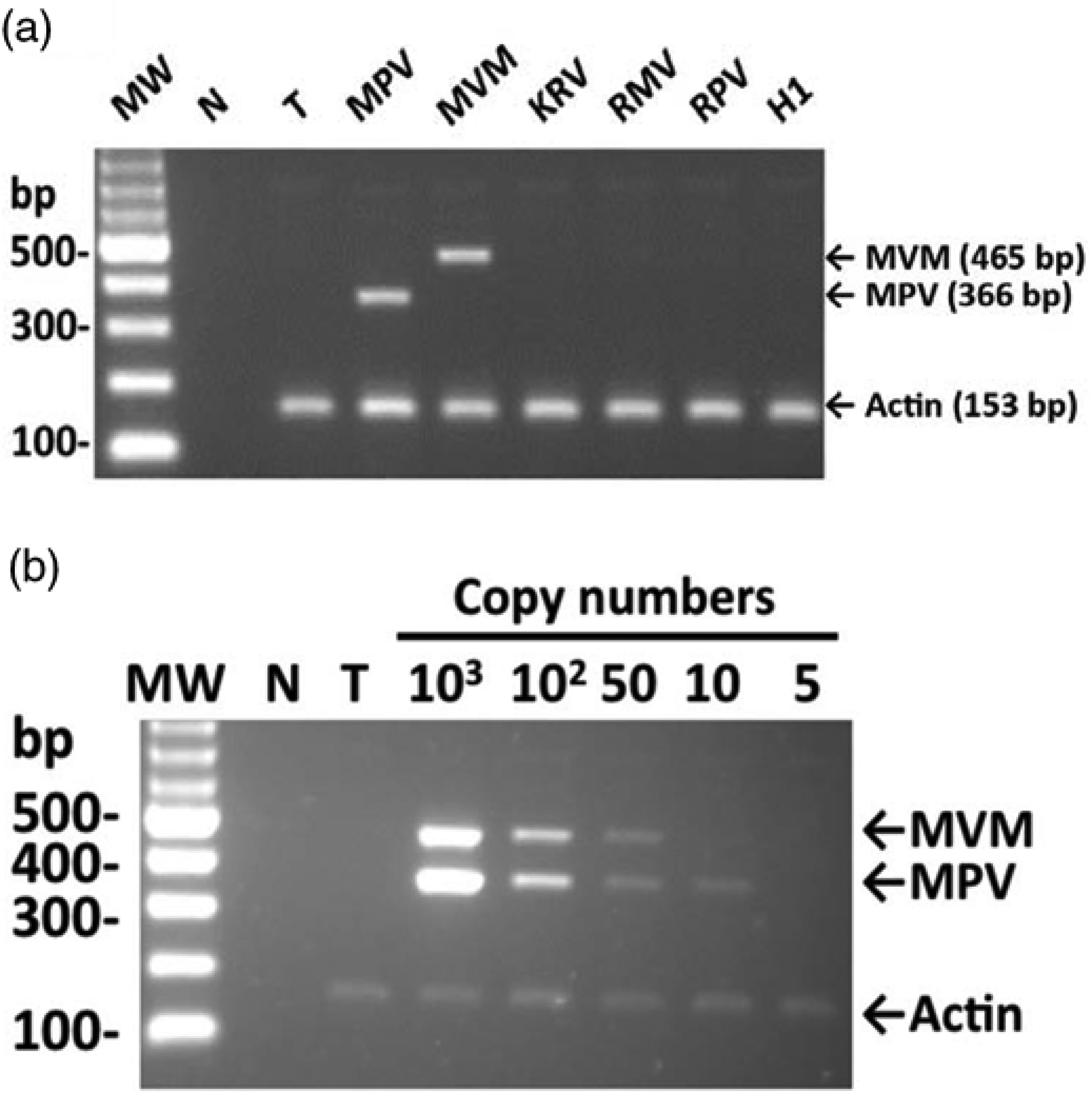
Detection of different amounts of MVM and MPV by the MVM/MPV/actin multiplex PCR assay
To evaluate the efficiency of amplification in the presence of co-infection by both viruses, serial dilutions of MVM and MPV DNA were mixed and tested. The results showed that in the presence of high amounts of MPV DNA (104 copies), the MVM amplicons could still be detected at as few as 50 copies of the original MVM template in the same PCR tube (Figure 2a). Similar amplification efficiency was observed in the reverse case (Figure 2b). In addition, a 153-bp actin DNA product was detected in every test assay except for the no template control (Figures 2a and 2b).
Detection of different combinations of minute virus of mice (MVM) and mouse parvovirus (MPV) by the MVM/MPV/actin multiplex PCR assay. (a) 104 copies of MPV DNA mixed with 104, 103, 102, 50 or 10 copies of MVM DNA. (b) 104 copies of MVM DNA mixed with 104, 103, 102, 50 or 10 copies of MPV DNA. MW: molecular weight marker; N: no template control; T: the MVM/MPV-negative tissue control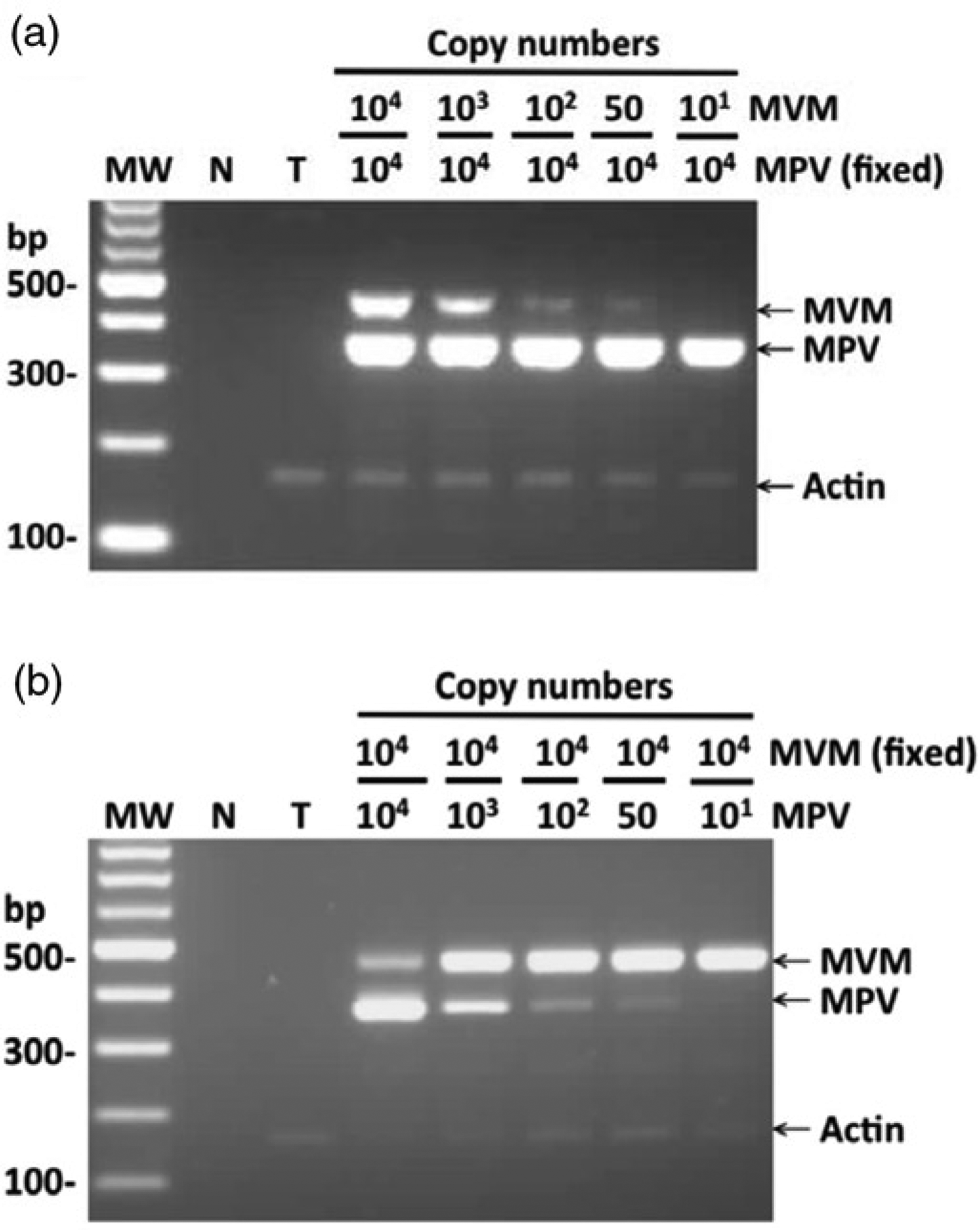
Surveillance of experimental mice and cell lines for MVM and MPV infections with the MVM/MPV/actin multiplex PCR assay
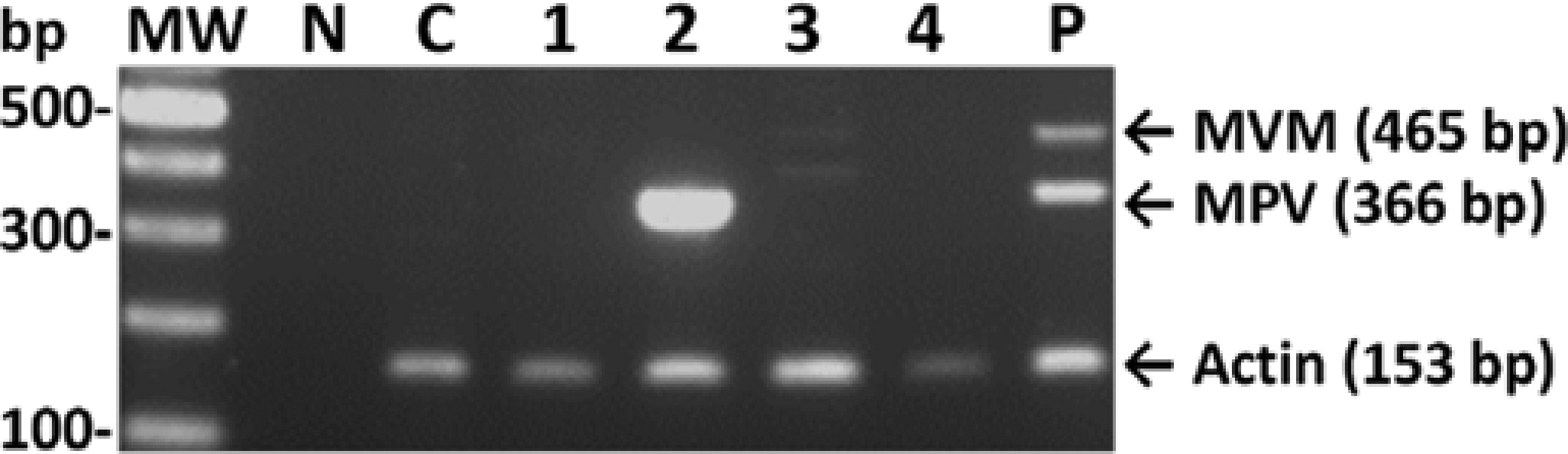
To determine the infection status of laboratory mice in Taiwan, 174 mice from seven facilities were tested using the MVM/MPV/actin multiplex PCR assay. Of the 174 samples screened, 20 mouse samples were found to be MPV positive, representing an infection rate of 11.5% (20/174) (Table 1). Sixteen MPV-positive samples were from the group of young adults, with a positive rate of 13.9% (16/115), and the other four positive samples were detected in the middle-aged adult group, with a positive rate of 11.4% (4/35) (Table 1). MPV DNA was not detected in any mice older than 16 weeks. The 20 positive samples were from three different laboratory mouse facilities, indicating that 42.9% of the total colony was infected. All 174 samples were MVM negative (Table 1, Figure 3). The amplified fragments from two MPV-positive samples from different units were selected for sequence analysis, and the sequences corresponded to the published sequences of MPV (data not shown). When the multiplex PCR assay was used to test 16 different cell lines and cultured cell samples, MPV DNA was only amplified from one cell line (Table 3, Figure 4), and MVM was not detected in any of the 16 cell samples screened.
Detection of minute virus of mice (MVM) and mouse parvovirus (MPV) in six laboratory mice by the MVM/MPV/actin multiplex PCR assay. MW: molecular weight marker; N: no template control; T: MVM/MPV-negative tissue control; lanes 1–6: tissue DNA extracted from the spleen samples of 6 different mice; P: 100 copies of MVM and MPV DNA mixed with negative tissue DNA Detection of minute virus of mice (MVM) and mouse parvovirus (MPV) in different cell cultures and cell lines by the MVM/MPV/actin multiplex PCR assay. MW: molecular weight marker; N: no template control; C: MVM/MPV-negative A9 cell control; lane 1: NB324K cells; lane 2: SPLC cells; lane 3: Raw cells; lane 4: BHK-21 cells; P: 100 copies of MVM and MPV DNA mixed with MVM/MPV-negative cellular DNA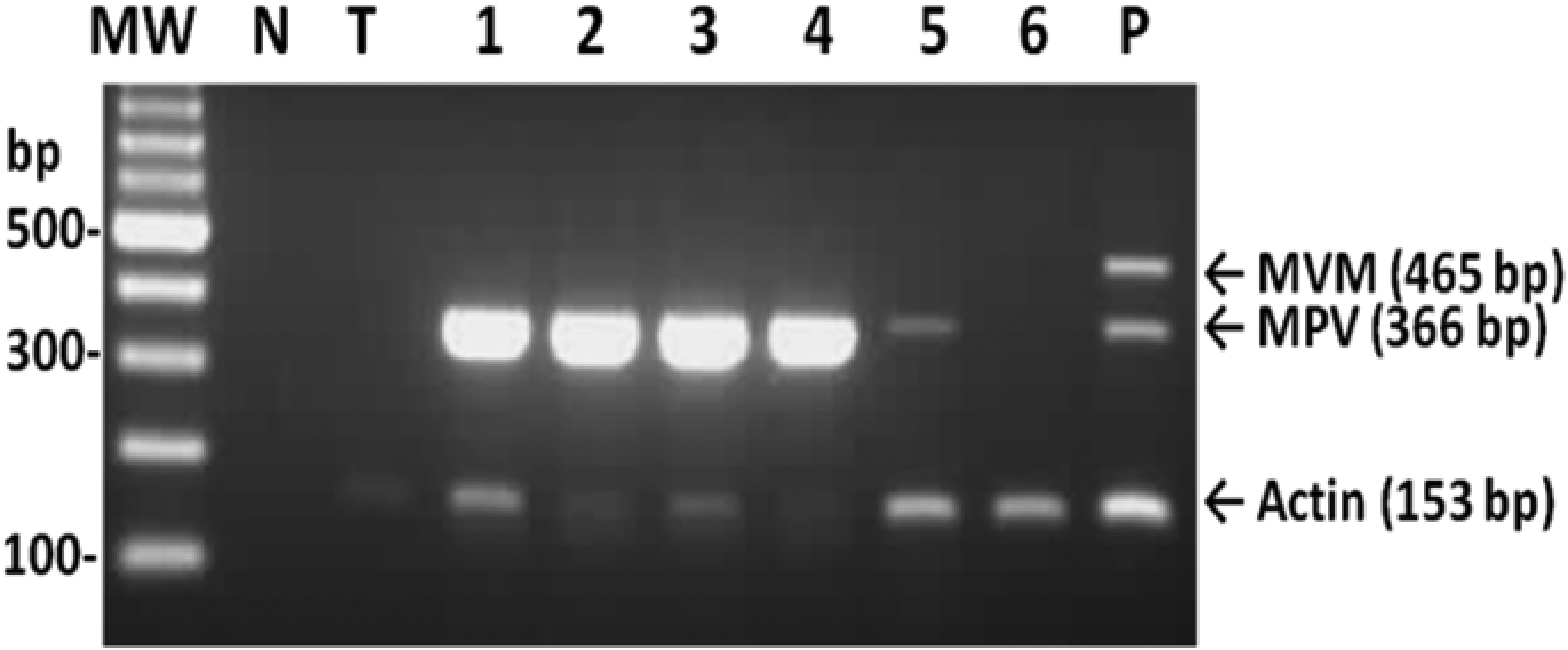
Discussion
In combination with a mouse housekeeping gene to monitor the interference of inhibition factors, the multiplex PCR assay established in this study can be used to simultaneously detect as few as 50 copies of both MVM and MPV. Compared to the detection limit of 30–100 copies for MVM, 11 the sensitivity of this three gene-targeted multiplex PCR assay is nearly equivalent to that of the single PCR assay. Nevertheless, multiplex PCR takes the same amount of time and effort but produces more data.
Infection with more than one parvovirus has been reported in various animal species, including dogs and cats.13,14 Although co-infection or superinfection of MVM and MPV has never been reported in mice, the possibility cannot be excluded. In the case of co-infections, the viral load of the two viruses can be similar or may vary greatly.15,16 Large differences in viral load present an obstacle to diagnosis because the virus with a lower titre may not be detected. The multiplex PCR developed here demonstrated the ability to diagnose co-infections with up to 200-fold differences in viral concentration. However, it remains to be determined how well this detection method can handle actual co-infections of mouse parvoviruses in mouse colonies.
The surveillance performed in our study revealed that the rate of MPV infection in mouse colonies in Taiwan is 11.5%, which is much higher than the rate of 1–3% reported in North America and Europe.17,18 This higher rate of MPV infection in Taiwan could result from a lack of routine monitoring for MPV before 2008 and difficulties in the complete elimination of infected transgenic colonies since that time. By contrast, MVM has been routinely monitored for two decades, and the MVM detection rate in our study is similar to that in North America and Europe (less than 0.5%). 15 The genetic background of mice and ages of exposure have important roles in the productive infections and the persistence of MVM or MPV in mice and should be taken into consideration while designing a health surveillance programme in animal facilities.19–23 Based on less productive viral shedding and/or lack of persistence of viral DNA in mouse tissues, NMRI mice seem to be unsuitable as sentinels for MVM PCR screening, nor are DBA/2 mice for MPV detection in PCR surveillance.20,22 In addition, ages of exposure to parvoviruses also affect the susceptibility of productive and/or persistent infections in mice.19,20,22 In our study, a total of 20 MPV-positive samples were all from mice of the young and the middle-aged adult groups with a positive rate of 13.3% (20/150), none from the mice older than 16 weeks old (0/24; positive rate: 0%) (Table 1). The data of age-related susceptibility in this research are consistent with previous findings.19,20,22 In this study, false-negative results may be possibly concluded for MVM or MPV infections in the facilities, while PCR samples were collected from mice of an older age. A larger survey on animals of different genetic background and proper age range will be necessary to determine the actual rates of MVM and MPV infections in research mouse colonies in Taiwan.
For the goal of 3Rs, the non-invasive sample collection has been applied to improve the diagnosis of infectious diseases, such as using a faecal sample with single PCR for both MVM and MPV.19,22,23 Although faecal samples were not as sensitive as haematopoietic tissue samples in the detection of the two viruses,19,22,23 designing a proper time frame for sample collections with the multiplex PCR assay may become practical. Therefore, further evaluations with the MVM/MPV/actin multiplex PCR in comparisons of (1) faecal and haematopoietic tissue samples, as well as (2) differences in mouse strains and ages are important.
In conclusion, the multiplex PCR assay developed in this study can be used to specifically and simultaneously detect MVM and MPV. This assay can be used to screen the infection status of mouse parvoviruses in biological materials and mice. Applying this multiplex PCR as a routine screening assay could also verify the possibility of co-infection with MVM and MPV and increase the understanding of these two viral infections in laboratory mouse colonies.
Footnotes
Acknowledgements
We thank Dr Ching-Fong Liao for providing MPV-1f-infected tissues, Dr Lela Riley for providing MVM, KRV, H-1 and RMV-1 viruses and Ms Chang for technical expertise. This research was supported by grant NSC95-2313-B-002–055 from the National Science Council, Taiwan and grant 99.B4.1.6 from the National Laboratory Animal Center, National Applied Research Laboratories, Taiwan.
