Abstract
Parvoviruses of mice, minute virus of mice (MVM) and mouse parvovirus (MPV), are challenging pathogens to eradicate from laboratory animal facilities. Due to the impediment on rodent-based research, recent studies have focused on the assessment of re-derivation techniques and parvoviral potential to induce persistent infections. Summarizing recent data, this review gives an overview on studies associated with parvoviral impact on research, diagnostic methods, parvoviral persistence and re-derivation techniques, demonstrating the complex nature of parvovirus infection in mice and unfolding the challenge of controlling parvovirus infections in laboratory animal facilities.
Keywords
Autonomous parvoviruses of mice, minute virus of mice (MVM) and mouse parvovirus (MPV), are major impediments to rodent-based research and quite challenging pathogens to eradicate from contemporary laboratory animal facilities. MVM and MPV are members of the genus Parvovirus, referring to small (18–26 nm), non-enveloped, single-stranded DNA viruses with a genome of approximately 5000 nucleotides.
Minute virus of mice
To date, four strains of MVM (MVMp, MVMi, MVMm and MVMc) have been described. The first strain was originally isolated by Crawford in 1966 from a preparation of adenovirus and, as the prototypic virus for the genus Parvovirus, designated MVMp. 1 The allotropic variant, the immunosuppressive strain MVMi, was isolated from a transplantable mouse lymphoma. 2 During the 1980s, the Cutter strain MVMc was isolated as a contaminant of BHK-21 cells. 3 Notably, these strains were all detected as contaminants of cell cultures. In contrast, MVMm (in reference to its isolation from mice obtained from the University of Missouri) has been isolated from naturally infected B-cell-deficient mice with a B-lymphocyte maturational defect. 4
Natural infection of mice with murine parvoviruses, including MVM infection, is generally asymptomatic. 5 However, several studies indicate that MVM infection in neonatal and immunosuppressed mice can induce runting, sudden or premature death, and disruption of haematopoiesis. In neonatal mice, infection with the immunosuppressive strain MVMi can result either in runting or in death, depending on virus dose and mouse strain. 6,7 Experimental MVMi infection was asymptomatic in C57BL/6 neonates, lethal with intestinal haemorrhage in DBA/2 neonates and lethal with renal papillary haemorrhage in BALB/c and C3H/He neonates. Furthermore, lethal infection due to myelosuppression and leukopenia occurred in Prkdcscid mice after experimental infection. 8,9 B-lymphocyte-deficient (Igh-6−/− ) mice naturally infected with MVMm displayed a runting syndrome with reduced fecundity, anaemia, leukopenia and premature death, indicating that MVM had infected haematopoietic progenitor cells in the spleen and bone marrow. 4 In addition, MVM has demonstrated pathogenic potential in hamsters and Mastomys after experimental infection. 10,11 However, serological surveys indicate that the mouse is the primary natural host. 12,13
MVMi primarily targets three cell types in mice: endothelium, lymphocytes and hepatic haematopoietic precursors. 6 Furthermore, susceptibility of mouse megakaryocytic and haematopoietic progenitors to MVMi has been demonstrated in vitro and in vivo. 8,9 In vitro, MVM variants demonstrate distinct cell tropisms with MVMp infecting cells of fibroblast origin and MVMi productively infecting erythropoietic progenitors and T-lymphocytes. 14,15 MVMp and MVMi are 97% homologous at the genomic level and serologically indistinguishable, but are reciprocally restricted for growth in each other's host cell type. 16,17 Minor structural alterations induced by changes at different amino acid loci confer the distinct cell tropisms of MVMp and MVMi. 18,19
MVM is the best characterized murine parvovirus regarding molecular biology and serves as a representative for related autonomous parvoviruses. 3,20,21 Viral capsids contain a linear non-segmented ssDNA genome usually of minus strand sense with short terminal palindromes at both the 5′- and 3′- end which are involved in viral replication. 22 After viral intake, mediated through dynamin-dependent endocytosis, the ssDNA of the virion is uncoated in the nucleus. 23 Nuclear replication is mediated through a modified rolling hairpin mechanism and occurs through monomer- and dimer-length duplex intermediates. 24,25 The ssDNA is converted into a double-stranded form, the replicative form (RF) DNA (parental RF synthesis). Amplification of the RF DNA proceeds semi-conservatively with RF DNA serving as the template for synthesis of viral mRNA and single-stranded progeny viral DNA.
The MVM genome (see Figure 1 for MVM genomic organization) contains several small and two large open reading frames, which are driven by promoters at map positions 4 and 38. The P4 transcripts encode two non-structural proteins (NS1 and NS2), which are highly conserved among rodent parvoviruses, and the P38 transcript encodes the two serotype specific viral capsid proteins, VP1 and VP2. Altogether, three mRNA species, R1, R2 and R3, are produced with R1 and R2 arising from the P4 promoter. 26,27 R1 encodes the non-structural protein NS1 which is required for viral DNA replication and transactivation of the P38 promoter. 28 NS2 is generated from R2 and required for virus replication, capsid assembly and nuclear egress of progeny virus. 28–31 R3 arises from the P38 promoter, encodes the viral capsid proteins VP1 and VP2 and produces, by proteolytic processing of VP2, a third structural protein (VP3), which is present only in infectious particles. 25,32,33 The MVM capsid is composed of a mixture of the viral proteins VP1, VP2 and VP3. 25 VP2 is the major capsid protein, although the presence of VP1 molecules is necessary to render a virion infectious. 34
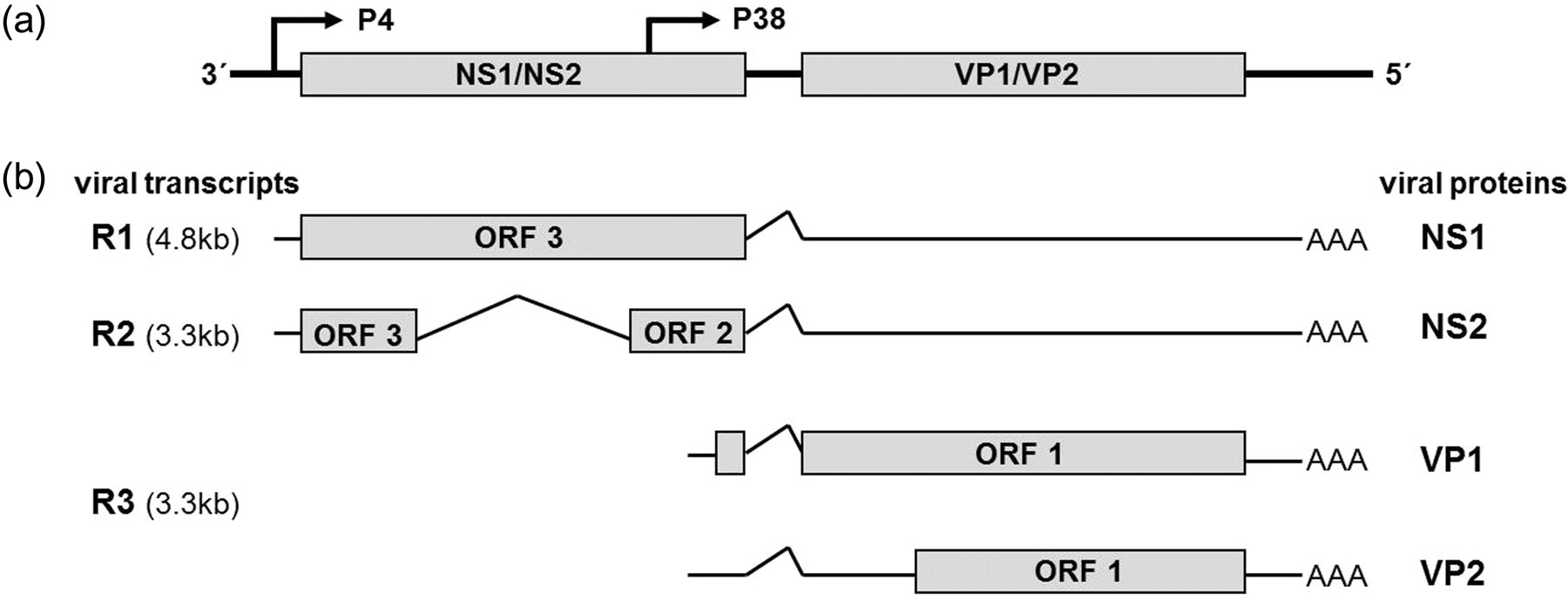
Genomic organization of the minute virus of mice (MVM) gene products. (a) Demonstration of the ssDNA genome including the two major open reading frames (ORFs) and the promoters P4 and P38. (b) Three mRNA species, denoted R1, R2 and R3, are transcribed (AAA indicates the polyA tail) from the genome. Boxes indicate ORF 1, 2 and 3 encoding viral proteins. Modified from Naeger et al. 31
Mouse parvovirus
So far, five strains of MPV (MPV-1, MPV-2, MPV-3, MPV-4 and MPV-5) have been described. MPV-1 was discovered as a contaminant of murine T-cell clones, not replicating well in currently available cell cultures. 35 Similar to MVM, the first strains of MPV were isolated from cell culture, although several field strains have now been detected. Several strains of MPV-1 (MPV-1a, MPV-1b, MPV-1c, MPV-1d, MPV-1e and MPV-UT) have been detected. 3,20,35–38 MPV-2 and MPV-3 were identified during a screen for parvoviruses in naturally infected mice. Interestingly, MPV-3 shows genetic similarity to Hamster parvovirus, suggesting that a cross species transmission had occurred, where the mouse had probably served as the natural host. 39 A very recent study by Christie et al. 40 provided evidence for the interspecies transmission of MPV-3. Additionally, MPV-4 (GenBank accession nos. FJ440683 and FJ445512) and MPV-5 (NC011618 and FJ440683) have been detected. 41
In contrast to MVM, no pathology in terms of clinical disease or histological lesions has been observed in mice experimentally or naturally infected with MPV. Even in newborn and immunocompromised animals infections are subclinical. 42,43
Prevalence
In general, parvoviruses are among the most common infectious agents of laboratory rodents, despite the establishment of hygienic barrier systems (Figure 2). 44–47 Contemporary reports on the prevalence of infectious agents in laboratory mice demonstrate that the prevalence of parvovirus infection varies (depending on the study) about 1–3% for North America and Europe. 45,48 Another study reported an increase in parvovirus prevalence in western Europe from 2% in the 1980s up to 8.8% for samples tested between 2000 and 2003. 49 An increased prevalence regarding parvoviruses was also reported by Carty et al. This study listed MPV, murine hepatitis virus (MHV) and MVM as the most frequently reported viral infections in mouse colonies indicating that considerable problems in the control of these pathogens exist. 44
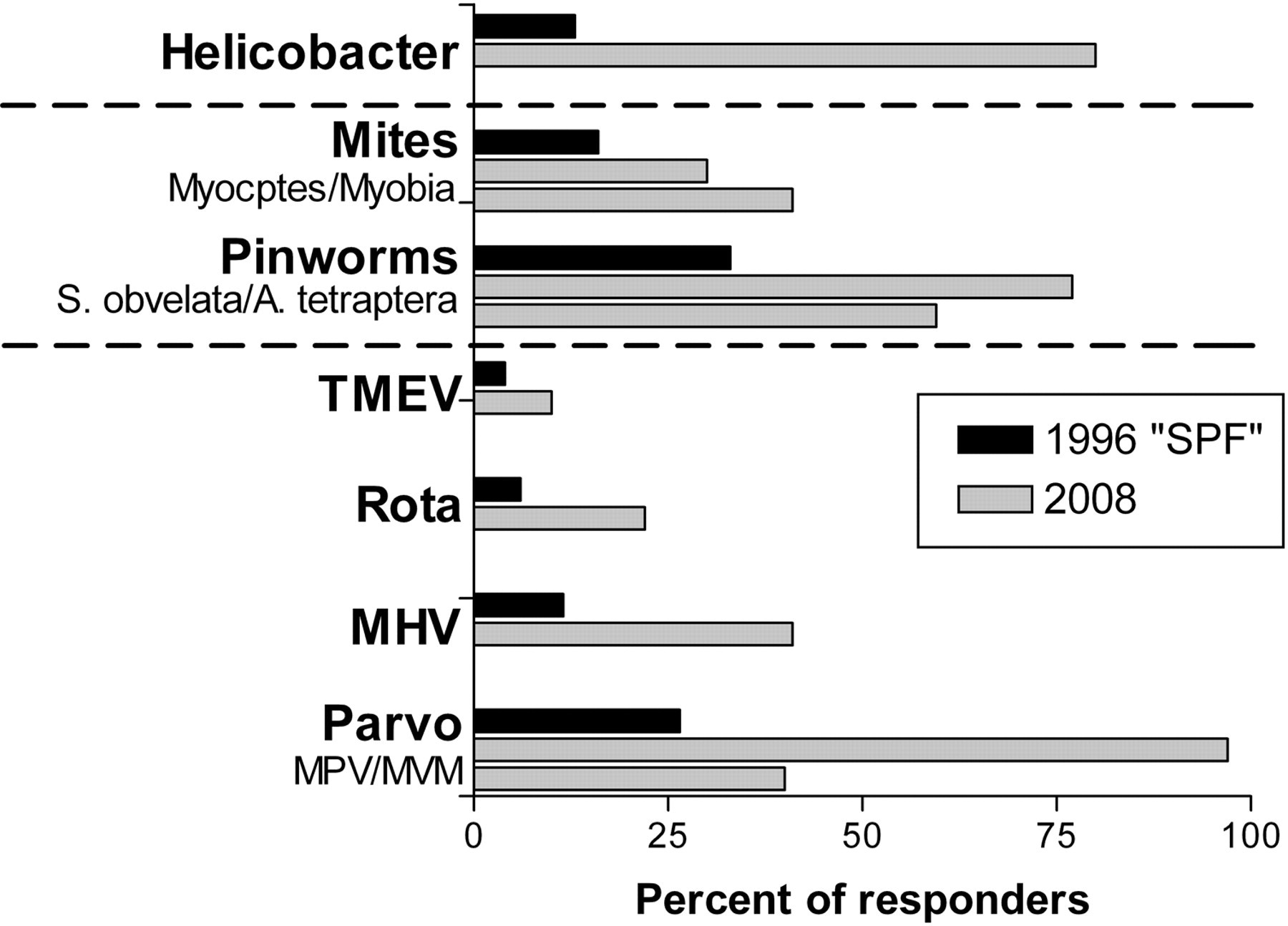
Minute virus of mice (MVM) and mouse parvovirus (MPV) are frequent viral pathogens in laboratory animal facilities (modified from Jacoby and Lindsey 47 and Carty 44 ). Although the percentage of sera testing positive for MVM and MPV is rather low in individual facilities, a high percentage of responding facilities reports the presence of parvovirus infection
According to our and others' experience, 50 the prevalence of parvovirus infection within a given colony is rather low. Therefore, from a statistical point of view, an infection can remain undetected for a very long period unless very high numbers of animals are tested. 51 For exclusion, all animals (or at least cages) of a colony have to be tested (own experience and Clifford and Watson). 50 As parvoviruses, though demonstrating a relatively low prevalence, are enzootic in experimental mice, they have to be considered as high-risk agents according to Pritchett-Corning et al. In this context, one might consider concentrating health monitoring resources for frequency of monitoring and sample sizes on such high-risk agents. 45 With regard to parvovirus, surveillance should rely on increased frequency and larger sample sizes. Additionally, apart from using a sentinel programme, the surveillance of colony mice is recommended.
Control and diagnostics
Several factors contribute to the problem of controlling murine parvoviral infections including the existence of different strains of MVM and MPV as well as factors associated with the host (e.g. genetic background, age, gender), factors associated with the biology of infection (e.g. shedding and persistence) and finally factors of concern when managing a facility (e.g. selection of sentinels, stability in the environment and risks associated with re-derivation) (Figure 3). As non-enveloped viruses, parvoviruses are remarkably resistant to conditions like heat, desiccation, and acidic and basic pH values demonstrating a high degree of environmental stability that aggravates eradication from mouse colonies and facilitates transmission among research facilities. 52,53 Probable ways of introduction into a facility are mice imported from experimental colonies, biological materials, and presumably food, 54 and of course feral mice. Therefore, quarantine or sanitation of incoming animals, testing of biological materials and sterilization of food by autoclaving or irradiation 50 (e.g. 50 kGy) should be applied.
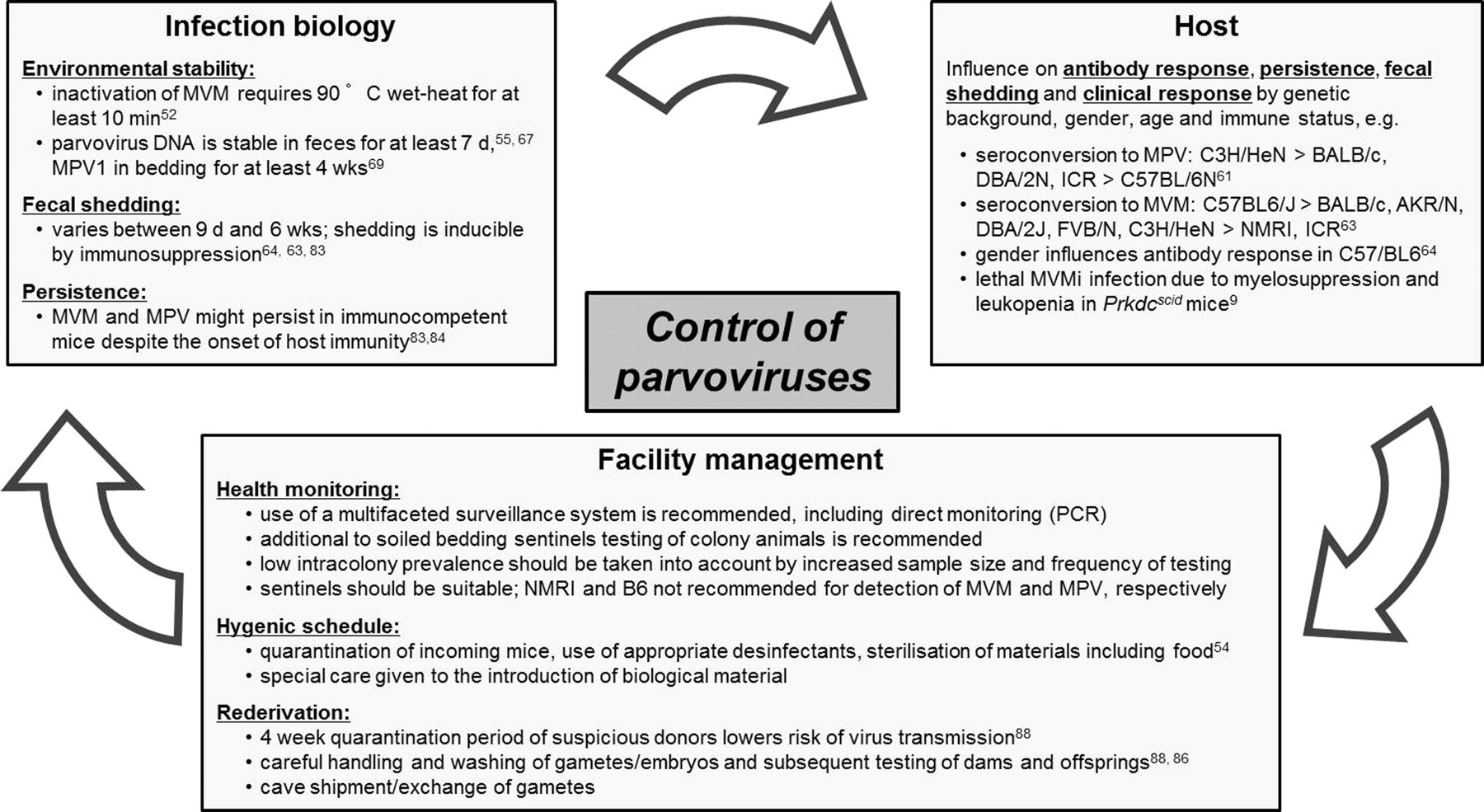
Factors to be considered when controlling murine parvoviral infection in laboratory animal facilities
Serology remains the most effective method of screening mouse populations for viral infections, although several previously published polymerase chain reaction (PCR) assays offer the opportunity to confirm serological results of parvoviral infection. 55–58
Notably, humoral immunity to MVM does not protect against MPV infection and vice versa. 59 Serological assays detect antibodies against both highly conserved non-structural proteins (NS) and virus-specific virion proteins (VP). See Table 1 for an overview on serological assays. Routine diagnostic methods are haemagglutination inhibition assay (HIA), indirect immunfluorescence assay (IFA), enzyme linked immunosorbent assay (ELISA) and multiplexed fluorometric immunoassay (MFIA), but only IFA and MFIA (depending on the design of beads) detect both types of antibodies simultaneously. The rNS1 ELISA is a generic rodent parvovirus ELISA using a recombinant NS1 protein as antigen, but IFA has been demonstrated to be more sensitive. 60,61 As infected cells serve as the antigen in IFA, this assay exhibits some degree of cross-reactivity with MPV and other closely related parvoviruses. In contrast, assays detecting antibodies against VP (such as the HIA and recombinant VP2 ELISA) are specific for the respective parvoviruses. 62 The HIA assay is less sensitive than IFA or ELISA, but differentiates among parvovirus serotypes due to haemagglutination mediated by viral capsid proteins.
Serological assays for parvoviral detection in mouse populations (modified from Shek R. Detecting and Controlling Rodent Parvoviruses. Charles River Laboratories Short Course, Strasbourg, 2003)
NS: non-structural; VP: virion protein
*Depending on the design of beads
When using serological methods for health surveillance, mouse strain-specific antibody responses to parvoviral infection should be considered.
For MVMi, antibody response varied among mouse strains, especially in contact exposed mice. 63 Seroconversion was detectable in all inoculated animals except some NMRI mice. In contrast, among contact-exposed animals, all C57BL6/J mice but only some BALB/c, AKR/N, DBA/2J, FVB/N and C3H/HeN mice seroconverted and no seroconversion was detected in NMRI and ICR mice by IFA. The finding that antibody production towards MVMi depends on the genetic background of mice has a major impact on the design of health-monitoring programmes as this indicates that MVM infection might remain undetected in animal facilities, especially when sentinel mice are used for surveillance. This inconsistency should be considered when interpreting results of serological tests and sentinel programmes. Additionally, titration studies with MVMi demonstrated that the dose to induce seroconversion was 10-fold higher in FVB/N mice than in B6/J mice. 63 Thus, negative results from low responders have to be treated with care and should be considered within sentinel programmes.
After experimental MPV-1 infection using a low viral dose, seroconversion occurred in C3H/HeN, to a lesser extent in BALB/c, DBA/2N and ICR mice, but was not detected in C57BL/6N mice. 61 This is of concern in laboratory animal facilities maintaining transgenic mice, as B6 mice provide the genetic background for the majority of genetically-modified mouse strains.
In the above-mentioned studies, investigation of strain-specific susceptibilities towards parvoviruses has yielded conflicting results concerning the B6 strain, whereas no seroconversion was detected after experimental infection of C57BL/6N mice with MPV-1, both inoculated and contact-exposed C57BL/6J mice showed a high antibody response to MVMi. This apparent inconsistency might be a consequence of the different B6 substrains used in the two studies or may be due to differences between MVMi and MPV-1.
Apart from the genetic background several other factors associated with the host can interfere with detection of parvoviral infection by serology. The age of exposure can play an essential role as data indicate that inoculation of older mice (12 weeks old) might not induce a productive infection. 61 A protective effect of maternal antibodies is inconclusive in MPV-1 infection, as DNA and antibodies were detected in some progeny of infected females at the age of six weeks. 37 In a recent study, Thomas et al. 64 demonstrated that gender influences the antibody response to MVMi in C57BL/6NCr and C57BL/6Tac mice, with males showing seroconversion to a greater extent. However, in another study using C57BL/6J mice all female mice seroconverted. 63 Together with the earlier cited findings these results might indicate that the B6/N and B6/J substrains differ in their susceptibility to parvovirus infection. 61
It has previously been discussed that the observed strain-specific antibody responses might be related to the predominant T-helper phenotype generated in response to MPV-1, 61,65 but this notion was not confirmed for MVMi infection. In another approach to fathom the reason behind the strain-specific antibody responses, the influence of the H2 haplotype on the antibody response was investigated. However, the H2q haplotype did not appear to be responsible for a low antibody response towards MVMi. 63
Recently, regarding the lack of seroconversion in some mouse strains as well as the possibility of persistent infections, more emphasis has been put on PCR as a diagnostic tool. However, although PCR is sensitive and specific, it requires access to tissues usually obtained during necropsy. In this context a recent study describing the detection of MVM and MPV by PCR of faecal samples provides the basis for antemortem detection of parvoviruses by PCR. 55 However, in this study mesenteric lymph nodes yielded a higher percentage of positive samples when analysed by PCR than faecal samples, thereby limiting this approach for general use in routine testing. However, further studies on the efficiency of PCR analysis of faecal samples compared with tissue testing might be required. Overall, several PCR assays valuable for confirming serological results or antemortem detection of parvovirus infection are available, an overview is given in Table 2. 56–58,66
Overview on PCR assays valuable for confirming serological results or antemortem detection of parvovirus infection in mice
NS: non-structural; VP: virion protein; PCR: polymerase chain reaction; MPV: mouse parvovirus; MVM: minute virus of mice
Other molecular diagnostics, e.g. immunohistochemistry, Western blot or in situ hybridization with strand-specific probes are valuable for direct virus detection, but not used in routine diagnostic.
Concerning health surveillance in animal facilities, genetic background, gender and age have to be taken into consideration when choosing sentinel mice. In this context, the use of parvovirus-sensitive mouse strains as sentinels (mouse strains reacting with a sufficient antibody production) has to be considered when testing for parvovirus infection. Thus, NMRI mice seem to be unsuitable for MVM detection as well as certain B6 substrains for MPV detection. Additionally, changing the sentinel strain at regular intervals might be helpful for detecting parvovirus contamination as well as other prevalent pathogens. With respect to the modern individually ventilated cage (IVC) maintenance systems where the use of sentinels is widely established for surveillance, the surveillance strategy has to be chosen carefully, whether to use sentinels derived directly from research animals or direct contact sentinels or sentinels exposed to soiled bedding. Notably, although MPV was transmitted to sentinels by soiled bedding transfer, proving soiled bedding transfer to be an effective method for detecting MPV under optimal conditions, the reliability of the method was dependent on the volume of soiled bedding transferred and the timing of the bedding transfer, i.e. the stage of infection at which the bedding was transferred. 67 Likewise, in a study by Compton et al. 68 the efficacy of the detection of MPV by soiled bedding transfer was dependent on the frequency and dilution of the soiled bedding transferred. As the dose of viral inoculum influences seroconversion rates, the amount of bedding transferred during sentinel programmes should be treated with special care, especially since recent data indicate that seroconversion is inconsistent in mice chronically exposed to low doses of MPV. Even advanced techniques such as the exposure of sentinels or filters to exhaust air are only of minor additional value for MPV infection. 68 According to Besselsen et al. 69 these findings suggest that sentinel-based detection of MPV occurs mainly when sufficient numbers of acutely infected mice are present. Therefore, to detect parvovirus infection in laboratory animal facilities current literature favours a multifaceted surveillance programme combining different detection systems such as soiled bedding transfer as well as the use of contact sentinels and direct monitoring. 64,67,68
Impact on research
Murine parvoviruses might affect a variety of studies with special regard to research areas such as immunology, transplantation, haematopoiesis and oncology. See Table 3 for an overview on in vitro and in vivo effects. MVM and MPV are common contaminants of transplantable tumours, cell lines, virus stocks and culture media. 70,71 Therefore, failure to establish long-term cell cultures from infected mice or a low incidence of tumour takes should alert researchers to the possibility of MVM contamination.
In vitro and in vivo effects demonstrated by murine parvoviruses
Immunomodulatory properties have been demonstrated in vitro and in vivo for MVM. MVMi has been isolated from an EL-4 lymphoma and found to be the mediator of the inhibitory activity displayed by these EL-4 ascites cells on allogeneic lymphocyte cultures, thus termed immunosuppressive strain of MVM. 2 Subsequently, inhibition of T-cell-mediated functions was demonstrated in vitro. MVMi was able to inhibit antigen-induced proliferation, generation of cytolytic T-cell activity and T-helper cell functions. 72 MPV modulated T-cell effector functions, potentiated tumour rejection and induced syngeneic graft rejection, suggesting that the virus affected peripheral tolerance mechanisms during infection. 73,74
Myelosuppression has been demonstrated for MVMi in vitro and in vivo. MVMi but not MVMp had myelosuppressive effects on the haematopoietic stem and committed progenitor cells, inhibiting the proliferative capacity of committed granulocyte–macrophage colony-forming unit, erythroid burst-forming unit (CFU-GM and BFU-E, respectively) and pluripotent (CFU-S) mouse haematopoietic progenitors in colony-forming assays. 15 In a subsequent study the myelosuppressive capacity of MVMi was demonstrated in vivo after infection of newborn BALB/c mice. 75 Fatal leukopenia and dysregulated erythropoiesis associated with depletion of primitive progenitors were the consequence of experimental MVMi infection in Prkdc scid mice. 9 Furthermore, MVMi infected and suppressed multipotent long-term re-populating lymphohaematopoietic stem cells of persistently infected Prkdc scid mice. 76 Finally, MVMi was shown to inhibit the proliferative capacity of megakaryocytic committed progenitors both in vitro and in vivo. 8
Apart from the immunomodulatory and myelosuppressive attributes which might impede rodent-based research, rodent parvoviruses are valued as excellent tools for virotherapy of cancer due to their strong natural oncosuppressive potential and low pathogenicity in humans. 77 Virus-based anticancer therapy involves the use of viruses either as replicating oncolytic agents or as recombinant vectors for gene transfer. The oncotropic and oncotoxic properties of rodent parvoviruses render them good candidates for both types of applications and are potent tools for cancer gene therapy purposes. 78 MVM suppressed Ehrlich ascites (EA) tumours in mice; co-injection of EA cells and MVM resulted in a pronounced inhibition of EA tumour formation. Additionally, mice surviving EA–MVM co-injection acquired long-term resistance to additional injection of EA cells. 79 Using a MVMp vector transducing the IP-10 chemokine, a metastatic haemangiosarcoma was suppressed in immunocompetent mice. 80
Persistence
Viral persistence is a common feature among autonomous parvoviruses, although the precise mechanisms of parvoviral persistence are not fully understood. Persistent infections have been described for several parvoviruses, including rodent parvoviruses such as Kilham rat virus (KRV). 81 KRV replication continued during persistent infection with host immunity reducing but not eliminating infection. 82
MVM was shown to persist in bone marrow haematopoietic stem cells of Prkdcscid mice. 9 Interestingly, Prkdcscid mice are persistently infected by both MVM and MPV. 9,37 However, until recently MVM was not thought to cause persistent infection in immunocompetent mice but characterized as an acute, self-limiting infection. 13 Recently, MVMi DNA was detected in spleens at 16 weeks post-exposure in some mouse strains (these animals were otherwise free of common murine pathogens), indicating that MVMi might indeed cause persistent infections. 63 The notion that MVMi persists in lymphoid tissues of immunocompetent mice despite the onset of host immunity was recently confirmed. 83 Viral mRNA was detected by reverse transcriptase (RT)-PCR in spleens and mesenteric lymph nodes at eight weeks post-inoculation in individually housed FVB/N mice despite seroconversion. The presence of replicating virus was confirmed using in situ RT-PCR. However, positive cells resembling macrophages and lymphocytes were detected to a lesser extent compared with 11 days post-inoculation and were mainly confined to the marginal zone of spleens. MPV infection was suspected to be persistent because transmission of infection occurred for up to six weeks and viral DNA was detected in C3H/HeN, BALB/c, ICR and C57Bl/6N mice at four weeks post-infection. 43,61 Subsequently, MPV-1-positive cells were detected in peripheral lymphoid tissues at nine weeks post-inoculation by in situ hybridization with dsDNA probes and homogenates of mesenteric lymph nodes remained infective for up to six weeks after infection with MPV-1. 42 Recently, Arc:Arc(s) mice experimentally inoculated at eight weeks of age were found to be persistently infected for 152 days after infection with MPV-1f. 84
Regarding persistence, the duration of viral shedding has to be considered as an important factor for pathogen surveillance in laboratory animal facilities. MVM faecal shedding occurred up to a period of six weeks in an enzootically-infected colony of SENCAR mice. 55 Here, DNA was still present in mesenteric lymph nodes in the majority of animals after 35 weeks. However, these data were obtained from an enzootically-infected colony that was positive for multiple pathogens; therefore co-infection with other pathogens as well as re-infection among cagemates might have interfered with viral shedding and persistence. Others identified viral shedding for only 9–12 days in faecal samples of B6 mice. 64 In FVB/N mice inoculated with MVMi, shedding occurred in the majority of mice for a maximum period of four weeks, irrespective of the age of infection. 63,83 Notably, an induction of viral shedding has been demonstrated in FVB/N mice when immunosuppression (by irradiation) was performed eight weeks after experimental infection with MVMi. 83
Faecal shedding of MPV occurred up to an age of 17 weeks in an enzootically-infected colony of SENCAR mice, but only lasted for a maximum of six weeks when mice were experimentally infected as infants. 43,55 Prior experimental infection with murine norovirus, enzootic in many specific pathogen free (SPF) mouse facilities, prolonged MPV faecal shedding by one week. 85 Neonatal Prkdcscid mice inoculated with MPV developed persistent faecal shedding. 37
Re-derivation
Embryo transfer (ET) is one of the most widely used techniques for re-derivation of infected mouse colonies. The possibility of parvoviral transmission during ET has been addressed recently as parvoviruses are associated with several risk-aggravating attributes including small size, affinity for mitotic active tissue and enhanced environmental stability.
Seroconversion was observed in ET recipients and offspring after the implantation of embryos contaminated in vitro with MVMp despite extensive washing of embryos. 86 On the contrary, virus-free seronegative pups were produced from murine embryos arising from IVF with spermatozoa exposed to MVMp in vitro. 87 These findings might indicate that the risk of viral transmission is more substantial through the female side. This hypothesis was supported by an in vivo approach where MVMi DNA was detected to a lesser extent in male than in female reproductive organs by realtime qPCR. 88 In this context the risk for transmission was determined to consist of a four-week minimum period as reproductive organs tested positive for MVMi DNA from day 5 post-inoculation up to day 30 post-inoculation. Even oocytes and sperm cells were found to be positive for MVMi DNA, but not two-cell embryos after in vivo infection and sufficient washing. In addition, oocytes tested positive for MVMi after in vitro incubation even after 10-fold washing, demonstrating that viral particles were not eliminated during this procedure. Furthermore, evidence was found that MVMi adhered firmly to the zona pellucida. 88 Together with the finding that 20 nm fluorescently labelled microspheres (similar in size to parvoviruses) penetrated into or crossed through the zona pellucida, these data demonstrate that MVMi is capable of adhering firmly to embryos. 86 MVMi might even cross the zona pellucida as has been shown for several other small viruses such as mengovirus and even other autonomous parvoviruses. 89–91
The successful ET re-derivation of C.B-17/Icr-Prkdcscid mice experimentally infected with MPV-1 has been reported. 92 However, although no productive infection was detected, antibody response occurred in recipients after ET when embryos were derived from infected females, and antibodies were also detected in their progeny, presumably from maternal transfer. Interestingly, this was not observed when MPV-positive male mice were mated to MPV-negative embryo donors. In endemically-infected mice, MPV-1 was detected in ovarian tissue, sperm cells, embryos and oocytes by PCR, corroborating the transmission of viral particles. 93
Recent studies indicate that careful washing is essential for successful re-derivation, but might not completely remove parvoviruses from oocytes, embryos and spermatozoa. When IVF is performed, gametes should be derived after an at least four-week quarantine period of the donor, as the percentage of reproductive organs testing positive for MVM decreased towards the end of a 30-day period after experimental inoculation of mice. 88 Concerning the extensive exchange of genetically-modified mice in recent years, special regard should be given to the fact that the shipment of gametes, which has been proposed as an alternative to sending live mice, may be a potential source for the introduction of parvoviral disease into animal facilities. However, ET can be evaluated as an adequate method for re-derivation of mouse strains comprising a manageable risk of transferring parvoviruses if certain precautions such as careful washing and accompanying testing are minded, while the exchange of gametes should be considered as a risk factor.
Conclusions
Recent studies indicate that several factors, including age, viral dose, co-infection with other pathogens, mouse strain and for some strains even gender, influence the rate of seroconversion to parvoviruses as well as the duration of viral shedding. Recently, evidence of MVMi and MPV-1 persistence in immunocompetent mice has been found. Furthermore, transmission of parvoviruses has to be considered a risk factor during re-derivation. These observations demonstrate the complex nature of parvovirus infection in mice and have to be considered in the design of health monitoring programmes for laboratory mouse populations as they may account for the relatively high prevalence of parvovirus infections in SPF colonies. Finally, this complexity might also be relevant to parvoviral infection in other species, including B19 infection in humans.
