Abstract
Laboratory animals have long since been used extensively in bioassays for prions in order to quantify, usually in terms of median infective doses [ID50], how infectious these pathogens are in vivo. The identification of aberrant prion protein as the main component and self-replicating principle of prions has given rise to alternative approaches for prion titration. Such approaches often use protein misfolding cyclic amplification (PMCA) for the cell-free biochemical measurement of prion-associated seeding activity, or cell assays for the titration of in vitro infectivity. However, median seeding and cell culture infective doses (SD50 and CCID50, respectively) of prions are neither formally congruent nor definitely representative for ID50 titres in animals and can be therefore only tentatively translated into the latter. This may potentially impede the acceptance and use of alternative methods to animal bioassays in prion research. Thus, we suggest performing PMCA and cell assays jointly, and to check whether these profoundly different test principles deliver consistent results in order to strengthen the reliability and credibility of prion ID50 assessments by in vitro methods. With regard to this rationale, we describe three pairs of PMCA and glial cell assays for different hamster-adapted prion agents (the frequently used 263K scrapie strain, and 22A-H scrapie and BSE-H). In addition, we report on the adaptation of quantitative PMCA to human variant Creutzfeldt–Jakob disease (vCJD) prions on steel wires for prion disinfection studies. Our rationale and methodology can be systematically extended to other types of prions and used to further reduce or replace prion bioassays in rodents.
Keywords
For decades, research into prions and the diseases they cause in animals and humans (e. g. scrapie, bovine spongiform encephalopathy [BSE], sporadic or variant Creutzfeldt–Jakob disease [sCJD or vCJD, respectively]) was largely dependent on animal experiments. 1 Only with the gradually unfolding molecular nature of prions, and the identification of aberrant prion protein (PrP) as their main component,1,2 it has also become possible to get a handle on these pathogens in vitro. Hence, a variety of biochemical methods that use pathological prion protein as a molecular surrogate marker for the qualitative or semiquantitative detection of prions has been established during the past few years.2,3
However, a direct appraisal of how infective prions are in vivo can still be done only by inoculation of sample material into animals and incubation time interval assays or endpoint titrations. 4 Both approaches rely on the transmission of a prion infection that eventually becomes evident by the onset of neurological symptoms. By contrast, the qualitative and quantitative detection of prion infectivity in vitro has become gradually feasible by cell culture approaches. 5 Yet, so far there are only a few cell assays available for this purpose, and their applicability is generally restricted because they mostly work with just one or a few of the multiple prion strains known to exist under laboratory and real-life conditions. Thus, animal bioassays are still being frequently used in prion research.
Prions consist essentially of an isoform of the host-encoded prion protein with a pathological, β-sheet rich folding and aggregation structure.1,6 Such prionforming conformers of PrP are referred to as PrPSc or PrPTSE (‘Sc’ and ‘TSE’ are acronyms for ‘scrapie’, and ‘transmissible spongiform encephalopathy’, respectively, with the latter being an alternative name for prion diseases).1,7 The replication of prions is thought to be mediated by a process that basically resembles the seeded growth of crystals. In this process, designated as nucleation-dependent polymerization,8,9 oligomers or polymers of PrPTSE act as nuclei (‘seeds’) that recruit cellular prion protein (PrPC) and incorporate it, in a misfolded form, into their growing amyloid-like aggregate structure. When PrPTSE aggregates break up into smaller units this leads to a multiplication of PrP particles with proteinaceous seeding activity and thereby causes further autocatalytic replication of the pathological protein state. According to this concept, prions are proteinaceous infectious particles whose self-replication is mediated by biochemical seeding activity, i.e. the property to convert normal protease-sensitive PrPC into misfolded, aggregated and usually proteinase K-resistant prion protein (PrPres).
Despite the great importance of animal bioassays in prion research they are time-consuming, expensive, rather restricted in throughput and potentially critical in both regulatory and ethical respects. Therefore, substantial efforts have been made in the search for alternative methods that would allow a reliable quantification of prion infectivity with reduced or no need for animal experiments. In addition to cell assays,10–12 such methods use cell-free biochemical approaches for the quantitative measurement of prion-associated seeding activity.13–15
Cell-based assays for the titration of prion infectivity
Due to substantial methodological advancements it has become possible to titrate the infectivity of certain murine scrapie prions (22L, RML) in quantitative cell-based assays using subcloned neuroblastoma (N2a) cells.10–12 Furthermore, RK13 cells transgenically expressing PrP from mouse, sheep or cervids were shown to allow the titration of RML prions, 16 a natural sheep scrapie isolate (PG127), 16 and chronic wasting disease agent, 17 respectively.
Biochemical assays for the measurement of prion-associated seeding activity
Prion replication by seeded PrP polymerization implicates the seeding activity of PrPTSE as an essential biochemical counterpart of biological prion infectivity. However, prion-associated seeding activity that transforms normal protease-sensitive PrPC into PrPres has become amenable to sensitive biochemical monitoring only by the introduction of protein misfolding cyclic amplification (PMCA) in the year 2001. 18 PMCA is a cyclic process that mimics, in an accelerated mode, nucleation-dependent PrP polymerization in the test tube. PMCA cycles basically consist of two phases. In the first phase, PrP seeds such as PrPTSE from animals or humans are incubated in normal brain homogenate containing an excess of PrPC to induce the growth of PrP oligomers or polymers. In the second phase, the sample is exposed to ultrasound. The ultrasonic treatment fragments grown PrP aggregates into smaller units, which in turn provides new seeding-active particles for further aggregate growth. Thus, with each PMCA cycle the number of seeds increases and accelerates the replication of the pathological protein state. 19 This effect may be enhanced by ‘serial PMCA’ in which reaction mixtures are periodically passaged into fresh normal brain homogenate after a certain number of PMCA cycles.20,21 Several different technical advancements of the PMCA technology, called quantitative PMCA and realtime quaking-induced conversion assay (RT-QuIC) now allow the direct titration of prion seeding activity in vitro.13–15,22
The introduction of PMCA and RT-QuIC was performed with hamster-adapted 263K scrapie agent that has long since been used as a laboratory prion strain for many purposes in basic and applied TSE research.13,14,18 When the biological infectivity and biochemical seeding activity of 263K scrapie prions in different sample materials were systematically compared, this empirically confirmed a consistent quantitative correlation between the biological infectivity and biochemical seeding activity.14,15
Rationale for the further reduction and replacement of prion titrations in animals by quantitative cell and PMCA assays
Quantitative prion bioassays in animals usually determine infectivity titres in terms of the median infective doses (ID50) that had been present in the inoculated sample material. One prion ID50 is the dose of prions that causes infection in 50% of inoculated animals. If such in vivo titration is to be reproduced in vitro this may be achieved best by a simultaneous titration of the median PMCA seeding dose (SD50) and the median cell culture infective dose (CCID50) in the test sample. In this context, one SD50 is the dose of seeding activity that converts PrPC into PrPres in 50% of PMCA samples, and one CCID50 is the dose of prions that causes infection (in terms of PrPres propagation) in 50% of inoculated cell cultures. Formally, neither SD50 nor CCID50 are congruent to ID50, and the reliability of PMCA and cell assays in terms of a correct ID50 determination cannot be taken for granted. However, based on empirically established quantitative correlations among SD50, CCID50 and ID50 in reference standards such as homogenized 263K scrapie hamster brain tissue (in the following referred to as ‘263K stock’), SD50 and CCID50 values detected in test samples can be tentatively translated into ID50 values. PMCA and cell assays represent profoundly different cell-free and cell-based test principles for the biochemical and biological titration, respectively, of prion activity in vitro. Therefore, if these assays independently deliver consistent ID50 assessments, this substantially backs up the overall test reliability as compared with titrations based on either PMCA or cell assays alone. Such internal consistency checks may mitigate reservations as to the reliability of in vitro alternatives to animal bioassays and thereby promote their acceptance and use in prion research. Accordingly, we suggest an integrated approach for the reduction and replacement of prion bioassay titrations in animals that includes both the measurement of seeding activity and the determination of cell culture infectivity by quantitative, sensitive and robust PMCA and cell assays, respectively.
With regard to this rationale, we report here on recent progress of our laboratory in the further development of PMCA and cell assays for different prion agents. Our present work builds on in vitro assays previously established by us for the qualitative biological detection of prion infectivity in glial cell cultures and the biochemical measurement of prion seeding activity by quantitative PMCA. 15 Firstly, we describe a glial cell assay that can be used for the quantitative biological titration of 263K scrapie infectivity in vitro, and its amendment by a simple quantitative PMCA assay for this prion strain. Secondly, we outline the adaptation of PMCA and cell assays to other hamster-adapted scrapie or BSE agents. Finally, we present a quantitative PMCA assay for human vCJD prions on steel wires that can be used in prion disinfection studies.
Animals, materials and methods
Prion agents from animals and humans
Hamster-adapted prion agents 263K scrapie, 22A-H scrapie and BSE-H were taken from brain sample stocks from our laboratory. These materials had been produced in previous studies by intracerebral inoculation, under Ketavet/Rompun anaesthesia, of outbred Syrian hamsters (mesocricetus auratus; strain: LVG Golden Syrian Hamster; Charles River, Sulzfeld, Germany) with hamster brain homogenates from clinically diseased donors as previously described. 23 These studies were performed in strict accordance with the European Convention for the Protection of Vertebrate Animals used for Experimental and other Scientific Purposes and the German Animal Welfare Act (Tierschutzgesetz). The protocols were reviewed and approved by the responsible Committee on the Ethics of Animal Experiments (‘Tierversuchskommission – Berlin’) affiliated at the Authority for Animal Protection in Berlin (‘Landesamt für Gesundheit und Soziales Berlin’, Berlin, Germany; see http://www.lageso.berlin.de; Permit Number G0085/00). Prion-infected hamsters had been regularly observed for clinical symptoms and were humanely euthanized by inhalation of CO2 upon the development of prion disease.
Human vCJD brain tissue (10% [w/v], i.e. 10–1 diluted, homogenate in 0.25 mol/L sucrose, reference code NHBY0/0003, in the following text referred to as ‘vCJD stock’) was kindly provided by the National Institute for Biological Standards and Control (Potters Bar, UK).
Use of normal hamsters and mice
Normal Syrian hamsters (for strain and supplier see above) were used as donors of brain tissue for the preparation of PrPC-containing substrate for PMCA of hamster-adapted prions and euthanized by exposure to CO2.
Brains from normal mice (species: Mus musculus; strain: 129; substrain: 129/Ola; transgene HuMM) transgenically expressing human prion protein homzygous for methionine at position 129 of the human PrP gene used to prepare the substrate for PMCA with vCJD prions were developed and kindly provided by the group of Jean Manson (Neurobiology Division of the Roslin Institute, University of Edinburgh, Edinburgh, UK). 24
For the preparation of glial cell cultures, normal neonatal Syrian hamsters (for strain and supplier see above) were used as donors of brain tissue and sacrificed 2–3 days after birth by decapitation. 15
Although not mandatory, we reported euthanasia of normally Syrian hamsters to the animal protection authority (Landesamt für Gesundheit und Soziales Berlin, Berlin, Germany; Registration Number T0220/07).
Preparation of brain homogenates
Brain homogenates from prion-infected hamsters, normal hamsters and normal HuMM-mice were prepared using previously published procedures. 15 Tissue homogenizations were performed in phosphate-buffered saline (PBS, pH 7.4) or conversion buffer (CB) for cell or PMCA assays, respectively.
Glial cell assays
Glial cell cultures used for the detection of 263K scrapie infectivity were prepared in culture flasks, infected and analysed as described previously. 15 For infection, these cultures were exposed to 1.0 × 10–7 or 1.0 × 10–8 g 263K stock. All other cell assays were based on the protocol by Pritzkow et al. 15 with modifications for the cryo-storage of cells and their cultivation in six-well plates.
Cryo-storage of glial cells
For cryo-storage, 3 × 106 cells were diluted in 1 mL 70% (v/v) Dulbecco's modified Eagle's medium containing 20% (v/v) fetal calf serum, and 10% (v/v) dimethyl sulfoxide (DMSO) per cryo-vial. Vials were cooled down overnight to –70°C using a freezing container with a cooling rate of –1°C/min. The next day, cryo-vials were transferred to liquid nitrogen for long-term storage.
Plate cultivation, infection, harvesting and analysis of glial cell cultures
After quick thawing of a cryo-vial, cells (1 mL) were washed with 9 mL growth medium (GM), resuspended, mechanically separated and cultivated for two days in 3 mL GM at a density of 1.5 × 104 cells/per well in six-well plates (8.96 cm2/well, TPP, Trasadingen, Switzerland).
Cell cultures were exposed to 1.0 × 10–3, 1.0 × 10–4 or 1.0 × 10–5 g BSE-H hamster brain tissue (‘BSE-H stock’), or to 1.0 × 10–5 g 22A-H scrapie hamster brain tissue (‘22A-H stock’), respectively, per plate well. Cultures for negative controls were exposed to 1.0 × 10–3 g normal hamster brain tissue. After three days of cultivation, the inoculum was removed and the cells were washed once with PBS.
Cultures were harvested at the indicated time points (3, 40, 42, 80, 100 or 122 days post initial exposure [DPE]). After washing with PBS, cells were detached with a cell scraper and collected in 1 mL PBS. Cells were pelleted by quick spin and resuspended in 50 μL PBS containing 1% (v/v) N-lauroylsarcosine (sarcosyl). Subsequent processing and analysis of harvested cells were performed as described previously, 15 and the Spearman–Kärber method was used for CCID50 titration. 25 The standard deviation of 263K CCID50 titration was determined on the basis of individual Spearman–Kärber analyses with four different sample sets.
Protein misfolding cyclic amplification
PMCA with hamster-adapted prions
PMCA with hamster-adapted prions was performed using a previously described procedure with specific adaptations. 15 In brief:
Only 10 μL samples of 10% (w/v) normal brain homogenate in conversion buffer containing 10–9, 10–10, 10–11 or 10–12 g 263K stock, 10–5 or 10–6 g 22A-H stock or 10–5 or 10–6 g BSE-H stock were mixed with 140 μL of 10% (w/v) normal brain homogenate in conversion buffer and glass beads, and subjected to PMCA. Approximately 30 μL (10 mg) of glass beads (diameter 0.5–0.75 mm; Roth, Germany) were filled into each reaction tube for PMCA with 263K, 22A-H and BSE-H. One PMCA round originally consisted of 24 cycles of 40 s sonications (190–220 W) followed by one hour incubation at 37°C.
However, for PMCA with 22A-H or BSE-H the following protocol modifications were applied as well: The concentration of ethylenediaminetetraacetic acid (EDTA) in PMCA batches was increased to 20 mmol/L, and one round of PMCA was adjusted to 12 cycles consisting of 40 s sonications followed by 2 h incubation at 37°C for BSE-H, and 4 h incubation for 22A-H, respectively.
Aliquots of 30 μL from PMCA batches collected prior to PMCA, or obtained after each round of PMCA, were processed and analysed as previously described. 15 SD50 titration of 263K stock was performed by using the method of Spearman–Kärber as previously described in the context of RT-QuIC.14,25 The standard deviation of 263K SD50 titration was determined on the basis of individual Spearman–Kärber analyses with 10 different sample sets.
PMCA with human vCJD prions on steel wires
The contamination with vCJD prions and PMCA processing of stainless steel wires were performed as described elsewhere with modifications: 15 For contamination, batches of 30 stainless steel wires (diameter 0.25 mm, length 5 mm) were incubated in 150 μL each of 10–2, 10–3, 10–4, 10–5, 10–6 diluted vCJD brain homogenate (vCJD stock) for 2 h. A 10% (w/v) brain homogenate from HuMM mice in PBS was used to similarly contaminate negative control wires, and as diluent for vCJD stock. Batches of 15 wires were subjected to PMCA in 150 μL 10% (w/v) HuMM mouse normal brain homogenate in conversion buffer as PrPC substrate and 30 μL glass beads. A total of nine serial PMCA rounds were performed. Each round consisted of 24 cycles of 40 s sonications (~210 W) followed by one hour incubation at 37°C. Upon completion of a PMCA round 75 μL of reaction mixtures were harvested and transferred into new reaction vials containing 75 μL of fresh 10% (w/v) HuMM mouse normal brain homogenate in conversion buffer and 30 μL glass beads. Aliquots of 15 μL from PMCA batches collected after each round of PMCA were subjected to PK digestion at 75 μg/mL PK and 55°C for 45 min for sodium dodecyl sulphate polyacrylamide gel electrophoresis (SDS-PAGE) and Western blotting.
SDS-PAGE and Western blotting
SDS-PAGE and Western blotting using the monoclonal anti-PrP antibody 3F4 for the detection of hamster and human PrP, 26 and the preparation of PrPres blot standards for Western blot analyses were performed as described elsewhere. 27
Results
Quantitative cell assay for the titration of 263K scrapie infectivity in vitro
As we reported recently, 15 primary glial cell cultures from Syrian hamsters can be infected with the 263K scrapie agent. Upon exposure to 263K scrapie prions such glial cell cultures showed an accumulation of seeding active PrPres. When we established this cell assay in a previous study, we observed that an inoculation with 2.5 × 10–5 g 263K stock resulted in a larger amount of detectable PrPres at 40 DPE than an inoculation with only 1.0 × 10–6 g 263K stock. 15 These findings prompted us to examine whether the glial cell assay exhibits a consistent dependency of PrPres formation from the infective dose in the inoculum. Therefore, we exposed cell cultures in the present study to lower amounts of infectivity.
Representative results from our previous analyses with 2.5 × 10–5 and 1.0 × 10–6 g 263K stock,
15
and from the cell assay now performed with 1.0 × 10–7 and 1.0 × 10–8 g 263K stock are displayed in Figure 1 (PrPres staining found at 3 DPE for 2.5 × 10–5 g 263K stock probably originated from the original inoculum that could not be washed off the cells). Aliquots each representing 3.8 μL of resuspended cell culture pellets were loaded onto the gels for Western blotting. We did not perform a normalization of the loaded material in terms of cell numbers or protein markers (e. g. actin) because the amount of cellular material in the cultures increased with the time period of cultivation (not shown). Thus, such normalization would have required a dilution of samples harvested at 40 DPE (or later) and thereby impaired the sensitivity of the assay.
Glial cell assay for in vitro endpoint titration of 263K scrapie infectivity. Western blot detection of PrPres, the PK-resistant core of misfolded PrP, at the indicated days post initial exposure (DPE) in glial cell cultures from hamsters that had been inoculated with normal hamster brain homogenate containing 0, 2.5 × 10–5, 1.0 × 10–6, 1.0 × 10–7, or 1.0 × 10–8 g 263K stock. Cells were cultivated for three days with these inocula. Subsequently, the inocula were removed, and the cells were washed and further cultivated until cell harvesting. Lane R, PrPres blot standard: PK-digested 263K stock corresponding to 5 × 10–7 g brain tissue. Lanes DPE, 3.8 μL aliquots from resuspended cell culture pellets harvested either immediately after removal of the inoculum (i.e. at 3 DPE), or at 40 DPE. Western blot results from cell cultures infected with 2.5 × 10–5 and 1.0 × 10–6 g 263K stock were reproduced under a creative commons licence from previously published work.15 PrPres: proteinase K-resistant prion protein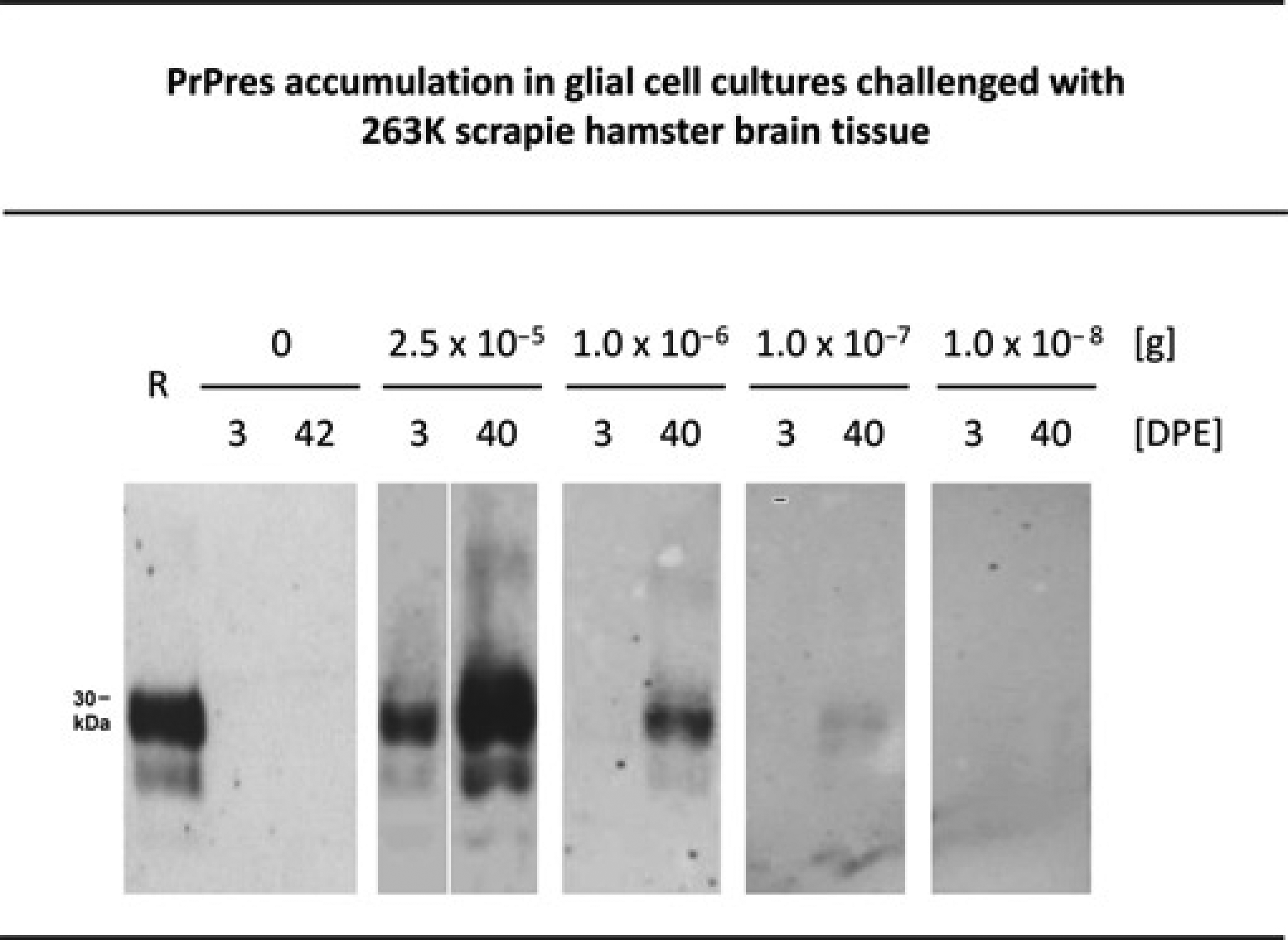
PrPres accumulation, i.e. infection, was detected in all, 50% or none of the cell cultures challenged with 2.5 × 10–5 g (n = 4) or 1.0 × 10–6 g (n = 4), 1.0 × 10–7 g (n = 4), or 1.0 × 10–8 g (n = 4) 263K stock, respectively. A titre analysis of these results performed in analogy to the method of Spearman–Kärber revealed that 1.0 × 10–7.0 g (standard deviation: 0.6 logarithmic10 units) of the 263K stock (from previous titrations in hamster bioassays known to carry about 1 × 102 intracerebral ID50) contained one CCID50 in the cell assay performed with a cultivation period of 40 DPE.25,28
Based on these results our cell assay can be used for the quantification of unknown infectivity titres in 263K scrapie samples by a simple standard procedure:
Test cell cultures are inoculated, at least in duplicate, with aliquots of serially tenfold 100–10–8 diluted samples of unknown 263K scrapie material. In parallel, reference cell cultures are similarly challenged with serial tenfold dilutions of 263K stock containing 1.0 × 10–6, 1.0 × 10–7 or 1.0 × 10–8 g 263K brain tissue (such internal assay calibration is required because the efficiency of PrPres production in infected cell cultures may theoretically vary between different assay batches). The dilutions of the unknown test material and of the 263K stock containing one CCID50 each are determined from the cell culture read-outs by the method of Spearman–Kärber. On this basis, the concentration of cell culture infectivity in the test material can be calculated and directly compared with the cell culture infectivity of the 263K stock. By using the correlation factor between CCID50 and ID50 of the 263K stock, cell culture infectivities of test samples are finally tentatively translated into ID50 values.
Cryo-storage and plate cultivation of glia
Our modified cell culture protocol allows the cryo-storage of ready-to-use glia for at least six months and their cultivation in six well plates. Due to these modifications glia isolated from a normal hamster brain are sufficient to produce about 300 individual glial cultures of 1.5 × 104 cells per well.
Cell assays for other hamster-adapted scrapie and BSE agents
In previous bioassay studies, 263K scrapie, 22A-H scrapie and BSE-H prions caused clinically fully developed prion diseases in Syrian hamsters at 83 ± 5, 206 ± 8 and 287± 28 days, respectively.
23
Despite the substantially prolonged incubation times of 22A-H and BSE-H prions as compared with 263K prions in the hamster bioassay, glial cell cultures could be infected by both of these agents (Figure 2). The efficiency of infection seemed to be similar for 263K and 22A-H scrapie after a challenge with 2.5 × 10–5 or 1.0 × 10–5 g 263K or 22A-H stocks, respectively (Figure 2a). By contrast, 1.0 × 10–5 and 1.0 × 10–4 g BSE-H stock failed to produce detectable infection of glial cultures at 42 DPE (Figure 2b). Therefore, we kept the glial cultures for longer cultivation periods of 100 and 122 days after exposure to BSE-H. Under these conditions, again, no cell culture infection could be detected after challenge with 1.0 × 10–5 g BSE-H stock. However, glial cells showed a dose-dependent PrPres accumulation at 100 and 122 DPE after exposure to 1.0 × 10–4 or 1.0 × 10–3 g BSE-H stock (Figure 2b). This indicated a lower sensitivity and highlighted the need for prolonged cultivation times of cell assays with BSE-H prions as compared with those with the 263K or 22A-H scrapie agents.
PrPres accumulation in glial cell cultures challenged with 22A-H scrapie or BSE-H prions. Western blot detection of PrPres at the indicated DPE in glial cell cultures from hamsters that had been inoculated with normal hamster brain homogenate containing 1.0 × 10–5 g 22A-H stock (a), or 0, 1.0 × 10–3, 1.0 × 10–4 or 1.0 × 10–5 g BSE-H stock (b). Cells were cultivated for three days with these inocula. Subsequently, the inocula were removed, and the cells were washed and further cultivated until cell harvesting. Lanes R, PrPres blot standard: PK-digested 263K stock corresponding to 5 × 10–7 g brain tissue. Lanes DPE, 3.8 μL aliquots from resuspended cell culture pellets harvested either immediately after removal of the inoculum (i.e. at 3 DPE), or later at the indicated DPE. PrPres: proteinase K-resistant prion protein; DPE: days post initial exposure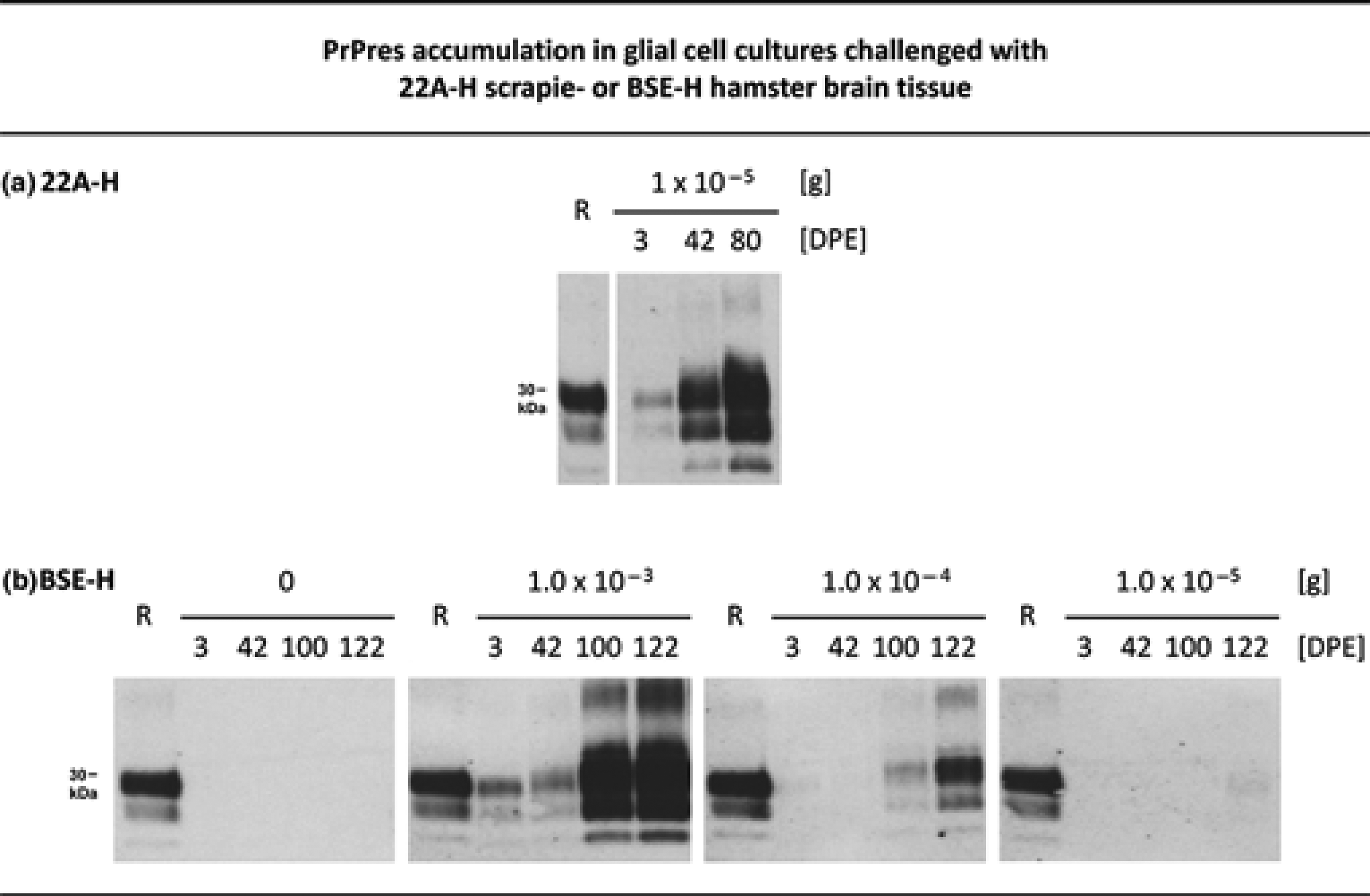
Quantitative PMCA assay for the titration of 263K scrapie seeding activity in vitro
In order to facilitate quantitative PMCA, we simplified a PMCA assay previously established in our laboratory,
15
and adapted it to rapid endpoint titration of prion seeding activity. Figure 3 shows that PrPres amplification could be detected after two rounds of PMCA when 1 × 10–11 g or larger amounts of 263K stock were used for seeding, while 1 × 10–12 g 263K stock produced negative results in this PMCA series. In unseeded PMCA batches no PrPres amplification was detected (not shown).
PMCA assay for in vitro endpoint titration of 263K scrapie-associated seeding activity. Western blot detection of PrPres after one or two rounds of PMCA seeded with 1 × 10–9, 1 × 10–10, 1 × 10–11 or 1 × 10–12 g 263K stock. Samples were run in duplicate, and sample lanes represent 4.2 μL aliquots from the respective PMCA batches. Lanes R, PrPres blot standard: PK-digested 263K stock corresponding to 5 × 10–7 g brain tissue each. The incubation time per PMCA cycle was one hour. PrPres: proteinase K-resistant prion protein; PMCA: protein misfolding cyclic amplification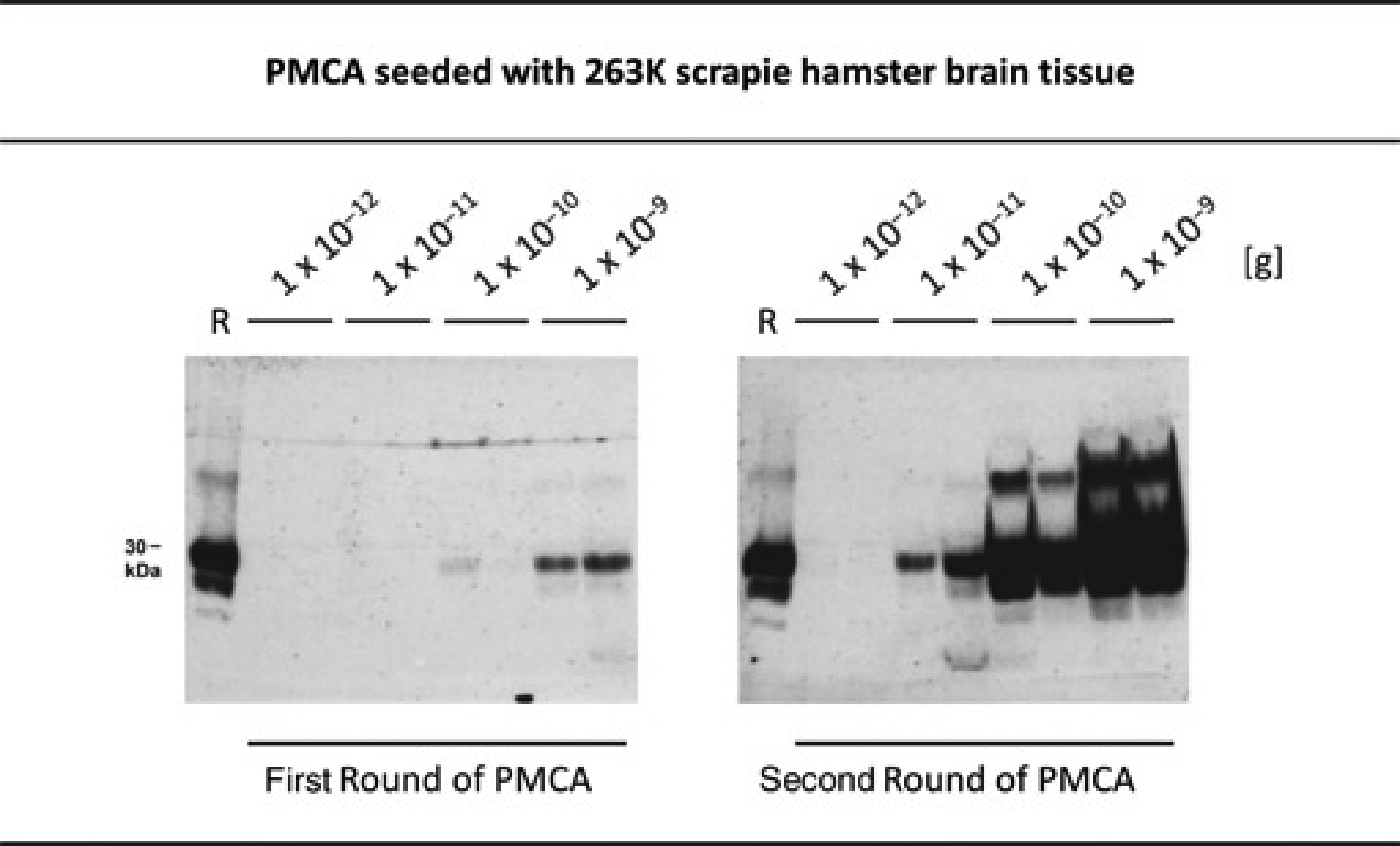
We used the data from these and similar PMCA experiments for an SD50 assessment. PMCA with 1 × 10–9, 1 × 10–10, 1 × 10–11 or 1 × 10–12 g 263K stock (n = 10 each) produced detectable PrPres amplification in 10/10, 10/10, 9/10 and 2/10 samples, respectively, and indicated that 1 × 10–13 g 263K stock would consistently deliver negative results after two rounds of PMCA. Spearman–Kärber analysis performed on this basis revealed that 1.0 × 10–11.6 g (standard deviation: 0.6 logarithmic 10 units) of the 263K stock (from previous titrations in hamster bioassays estimated to carry about 3 × 10–3 intracerebral ID50) contained one SD50 in our PMCA assay after two amplification rounds.
In the light of these results we suggest the following simple standard procedure for the endpoint titration of unknown SD50 titres in 263K scrapie samples by quantitative PMCA:
Test PMCA batches are seeded, at least in duplicate, with aliquots of serially tenfold 100–10–12 diluted samples of unknown 263K scrapie material. In parallel, reference PMCA batches for internal assay calibration are seeded with 1.0 × 10–10, 1.0 × 10–11, 1.0 × 10–12 and 1.0 × 10–13 g 263K stock. The dilutions of the unknown test material and of the 263K stock that contain one SD50 each are calculated from the Western blot read-outs after two PMCA rounds by the method of Spearman–Kärber. On this basis, the concentration of seeding activity in the test material can be concluded and directly compared with that in the 263K stock. By using the correlation factor between SD50 and ID50 of the 263K stock, the seeding activities of test samples are tentatively translated into ID50 values.
PMCA assays for other hamster-adapted scrapie and BSE agents
The highly efficient PMCA protocol established for the 263K scrapie agent was subsequently adapted to 22A-H and BSE-H prions. For this purpose, we systematically examined the influence of the incubation time between the sonication steps, and of the concentration of EDTA in the conversion buffer, on the efficiency of PMCA with these TSE agents. This revealed that a robust amplification of PrPres could be achieved when both the EDTA concentration was elevated to 20 mmol/L, and the incubation times were increased to 4 or 2 h for 22A-H- and BSE-H prions, respectively (Figure 4). We found that 1.0 × 10–5 and 1.0 × 10–6 g of 22A-H and BSE-H stock effectively seeded, in a dose-dependent manner, the propagation of PrPres by PMCA.
PMCA with 22A-H scrapie or BSE-H prions. Western blot detection of PrPres after zero, one, two, three or four rounds of PMCA seeded with 1.0 × 10–5 or 1.0 × 10–6 g 22A-H stock (a), or with the same amounts of BSE-H stock (b). Sample lanes represent 4.2 μL-aliquots from PMCA batches. Lanes R, PrPres blot standard: PK-digested 22A-H (a) or BSE-H (b) stock corresponding to 5 × 10–7g or 1 × 10–6 g brain tissue, respectively. The incubation time per PMCA cycle was 4 h for 22A-H prions in (a), and 2 h for BSE-H prions in (b). PMCA: protein misfolding cyclic amplification; PrPres: proteinase K-resistant prion protein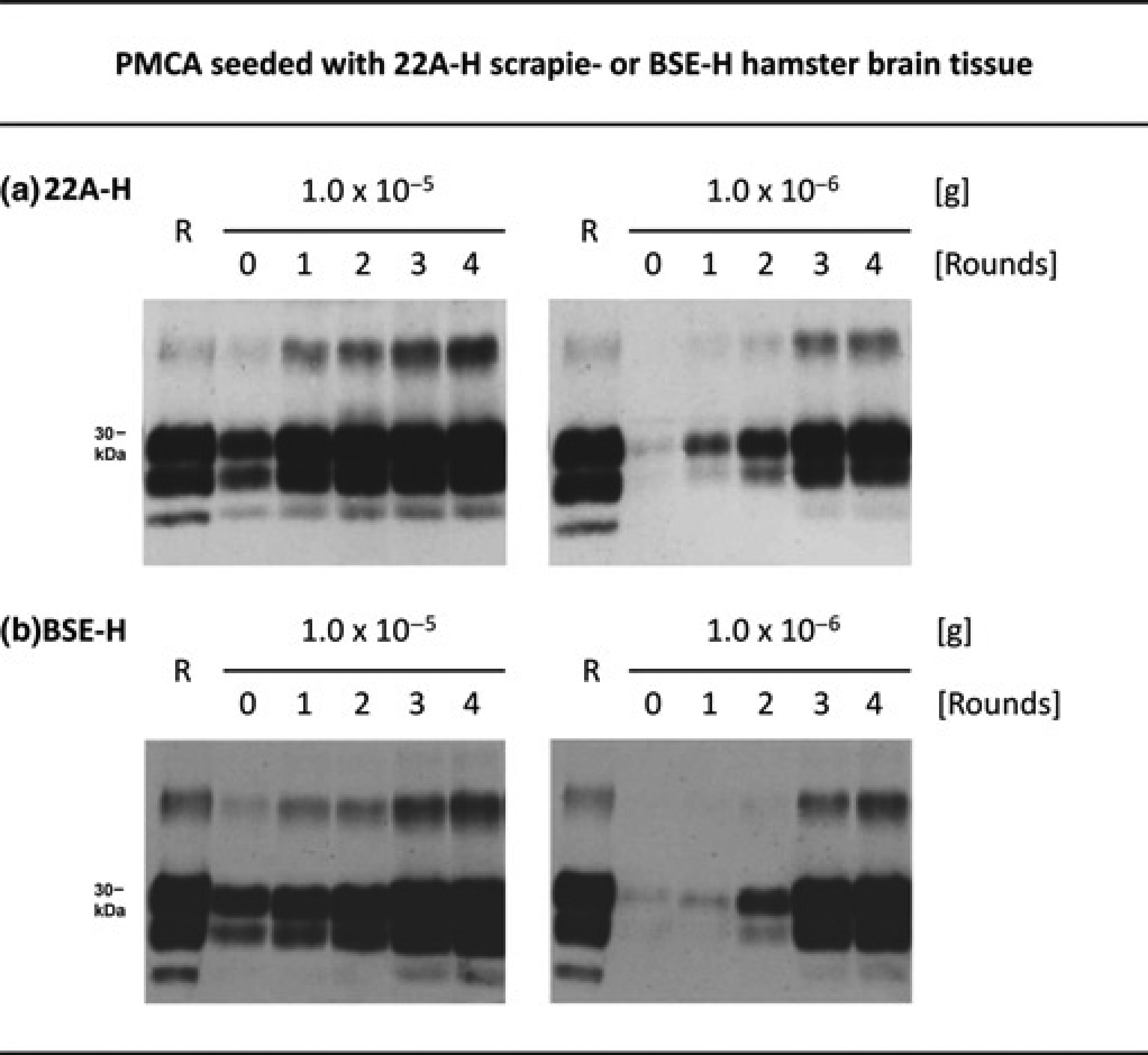
Quantitative PMCA assay for the titration of vCJD seeding activity on steel wires
A further aim of our work was to adapt to human vCJD prions a previously described quantitative PMCA assay for the monitoring of 263K scrapie disinfection on surrogates for medical instruments. 15 For this purpose, steel wires were contaminated with different amounts of vCJD prions, and brain homogenate from transgenic HuMM mice served as the PMCA substrate.
As shown in Figure 5, PrPres amplification showed a consistent dependency from the dilution of the vCJD stock that has been used for the contamination of wires. Amplified PrPres could be detected after 5–8 PMCA rounds with wires that had been exposed to serially tenfold 10–2–10–5 diluted vCJD stock, respectively. By contrast, wires that had been coated with 10–6 diluted vCJD stock, or negative control wires not coated with vCJD stock, did not produce detectable PrPres amplification after up to nine rounds of PMCA. A preliminary Spearman–Kärber analysis of our findings from the two independent PMCA assessments performed so far indicated that wires contaminated with 10–5.5 diluted vCJD stock would carry one SD50 in our PMCA assay. Thus, we suggest the following procedure for the endpoint titration of vCJD SD50 values on re-processed steel wires in disinfection studies:
PMCA with vCJD prions on steel wires. Western blot detection of PrPres after PMCA seeded with steel wires that had been contaminated with the indicated dilutions of vCJD stock. Lanes R, PrPres blot standard: PK-digested 263K stock corresponding to 5 × 10–7 g brain tissue. Numbered lanes 1–9 represent 4.2 μL aliquots from PMCA batches sampled after one, two, three, four, five, six, seven, eight or nine rounds of amplification. Negative control wires were subjected to PMCA in normal HuMM mouse brain homogenate without prior contamination with vCJD stock. M: molecular mass indicator. PMCA: protein misfolding cyclic amplification; PrPres: proteinase K-resistant prion protein; vCJD: variant Creutzfeldt–Jakob disease
Test steel wires are incubated in 10–1 diluted vCJD stock (and thereby contaminated with about 3 × 104 SD50). The test wires are then exposed to different disinfectants, and subsequently subjected to nine rounds of PMCA. For internal assay calibration reference wires are contaminated with 10–4, 10–5 or 10–6 diluted vCJD stock and subjected to PMCA without disinfection. The residual seeding activity remaining on the surface of re-processed test wires can be calculated from the PMCA read-outs by Spearman–Kärber analysis. By comparing the initial and residual seeding activities on test wires, the reduction factors achieved by different disinfection methods can be subsequently concluded over a range of slightly more than 4 logs.
Discussion
Quantitative cell assay for the titration of 263K scrapie infectivity in vitro
To the best of our knowledge, this report presents for the first time a cell-based assay for the in vitro titration of 263K scrapie infectivity. Our cell assay for 263K scrapie titration is still about 100-fold less sensitive than bioassays in hamsters (one CCID50 corresponds to 100 ID50) and has a measuring range of 7 logs of infectivity. However, recently Arellano-Anaya et al. 16 reported that the sensitivity of assays using transgenic RK13 cells for the detection of ovine or mouse scrapie prions was 100-fold increased by two successive rounds of infection. If the sensitivity of our cell assay could be similarly enhanced by this approach, it would be on par with the hamster bioassay.
Only two other cell culture models were previously found to be infectible with hamster scrapie prions.29,30 However, these cell assays were not used for the in vitro titration of scrapie infectivity and refer to a different hamster-adapted scrapie isolate, i.e. Sc237.31,32 While Sc237 and 263K prions have the same origin, 33 they were ultimately obtained in distinct sets of passages and may thus differ in their properties. Findings by Kimberlin and Walker, 34 as well as new evidence recently presented at the Prion 2012 Conference in Amsterdam,35,36 indicate that Sc237 and 263K are distinct hamster-adapted prion agents, and that other than the cloned 263K strain, the Sc237 isolate contains different substrains.
Cryo-storage and plate cultivation of glia allow efficient use of normal hamster brain tissue in the cell assay
By the cryo-storage of ready-to-use glia cells and their cultivation in six well plates donor hamster brains can be better used for the preparation and supply of glia cells, and more cultures can be processed simultaneously, than with our previous cell culture protocol. Three hundred cell cultures (each to be kept in an individual plate well) can be produced from the stock of glia harvested from one neonatal hamster brain.
For the titration of 263K scrapie infectivity in an unknown sample by hamster incubation time interval bioassays, usually five hamsters have to be inoculated (endpoint titrations would still require a substantially larger number of animals). In the cell assay described above for 263K scrapie prions, 24 glial cultures derived from normal hamster brain tissue and tiny amounts of 263K stock are required to determine the CCID50 of an unknown specimen in duplicate. If the internal assay calibration is performed in duplicate with 10–6, 10–7 or 10–8 g 263K stock, one brain from a scrapie hamster (weighing about 1 g) provides sufficient reference tissue for the titration of about 5 × 105 unknown samples in the cell assay. At the same time, the 300 cell cultures available from one normal hamster brain allow the determination of the CCID50 of 12 263K scrapie specimens in vitro. The titration of this number of samples in hamster incubation time interval bioassays would require inoculation of 60 animals. Thus, one normal hamster brain and slightly more than 2 × 10–6 g of a scrapie hamster brain optimally utilized in glial cell assays are theoretically sufficient to replace 263K scrapie bioassay titrations in 60 hamsters.
Suitability of glial cell assays for infectivity titration of other hamster-adapted scrapie and BSE agents
In additional experiments, we found that glial cell cultures were also susceptible to infection with 22A-H and BSE-H prions. Infection of primary cells or cell lines with these laboratory TSE agents has not yet been reported to the best of our knowledge.
We observed a similar efficiency of cell infection by 263K and 22A-H prions. For the BSE-H agent we found a lower, yet dose-dependent efficiency of infection that required higher doses of inoculum and a prolongation of the cultivation period. The threshold of infecting BSE-H or 22A-H stock still causing detectable PrPres accumulation in glial cell cultures within a set number of days remains to be determined individually. With this information glial cell cultures can be directly used for the CCID50 titration of 22A-H and BSE-H prions in a similar way as described for the 263K scrapie agent. When further pursuing this approach it should also be established whether the sensitivity of 22A-H and BSE-H cell assays can be increased by successive rounds of infection.
Quantitative PMCA assay for the titration of 263K scrapie seeding activity in vitro
In order to facilitate the practical use of quantitative PMCA we report here an approach for the rapid endpoint titration of 263K scrapie seeding activity by only two rounds of PMCA and simple Spearman–Kärber analysis of the assay read-outs. The rationale for such endpoint titration of seeding activity was previously described by Wilham et al. 14 in the context of RT-QuIC. Our method for the titration of 263K scrapie seeding activity by quantitative PMCA is about 300-fold more sensitive than bioassays in hamsters (one SD50 corresponds to about 3 × 10–3 intracerebral ID50) and able to measure a range of seeding activity of more than 11 logs. With respect to the intrinsic variation of prion bioassay titrations our observed SD50/ID50 ratio is in good accordance with recently reported data from a study by Makarava et al. 22 who found that one PMCA SD50 of their 263K stock corresponded to about 5–6 × 10–3 intracerebral ID50. In this context it has to be noted that the 263K stock used by Makarava et al. showed about tenfold higher titres of both ID50 and SD50 in hamster and PMCA assays, respectively, than our 263K stock.
In our two-round PMCA assay, 68 test and reference PMCA batches, 34 of which each require an amount of about 150 or 120 μL 10% (w/v) normal hamster brain homogenate as PrPC substrate in the first and second PMCA round, respectively, have to be processed for the determination of the SD50 of an unknown specimen in duplicate. In this assay, one normal hamster brain and slightly more than 2 × 10–10 g 263K stock are required to determine the 263K scrapie seeding activity of one unknown sample. A conventional hamster bioassay, in contrast, would usually require five animals for the sample's titration.
Suitability of PMCA assays for seeding activity titration of other hamster-adapted scrapie and BSE agents
We have been able to establish in vitro assays for the seeding activity of BSE-H and 22A-H prions that can be combined with a testing in the glial cell cultures described above. We are not aware of previous reports describing similar serial PMCA procedures for BSE-H or 22A-H. However, the feasibility of PMCA with 22A-H prions has been previously established by Ayers et al. who demonstrated the amplification of 22A-H-associated PrPres after one round of PMCA. 37
Although our PMCA assays for BSE-H and 22A-H prions are less sensitive than the PMCA assay for 263K scrapie agent, in principle, they can be similarly used for an endpoint titration of unknown SD50 titres. For this purpose only the minimum amount of BSE-H or 22A-H stock that is just sufficient to induce detectable PrPres amplification after a set number of PMCA rounds remains to be determined individually. On this basis, the range of test sample dilutions suitable for SD50 endpoint titration and the reference amounts of BSE-H and 22A-H stocks needed for assay calibration can be easily adjusted in analogy to quantitative PMCA with the 263K scrapie agent.
Quantitative PMCA assay for the titration of vCJD seeding activity on steel wires
Prions rank among the most tolerant pathogens in hierarchical scales of resistance to disinfection and place particularly high demands to the re-processing of medical devices. 38 So far, bioassays in animals provide the gold standard for monitoring the disinfection of prions.39,40
Recently, we were able to establish the proteinaceous seeding activity of 263K scrapie-associated PrPTSE as a highly sensitive quantitative indicator for the disinfection of 263K scrapie prions on steel wires used as surrogates for medical devices. 15 However, the tolerance to individual disinfection methods may vary between distinct prion strains. 41 We have therefore suggested devising PMCA protocols for the sensitive quantification in vitro of sCJD- and vCJD-associated seeding activity in disinfection studies. In this report we describe, to the best of our knowledge for the first time, such an assay for vCJD prions on steel wires.
There are no rodent models commonly available that would allow the sensitive titration of vCJD prions over a broad range of infectivity. Mouse strains frequently used for the detection of vCJD infectivity show long incubation periods upon primary passage of the agent from humans as reported for RIII (~300–400 days) and C57BL6 mice. 42 In a most recent vCJD inactivation study, Fernie et al. 43 observed incubation times of about 670 days in RIII mice that had been challenged with 10–4 dilutions of vCJD brain tissue. The range of infectivity measurement in the RIII mice of these studies was 3 logs.
As compared with such studies in RIII mice our PMCA assay would be substantially less time-consuming and more sensitive, since it allows the monitoring of seeding activity reductions on steel wires of more than 4 logs. So far we have performed PMCA with vCJD-contaminated steel wires for up to nine rounds in two independent test runs. A further increase in the number of rounds might improve the sensitivity of the assay and allow the detection of lower amounts of seeding activity. Thus, our SD50 estimate of vCJD-contaminated steel wires still requires further validation.
According to our rationale, we are currently trying to establish a complementing cell assay for the in vitro titration of vCJD infectivity. Based on our findings with hamsters, glial cells from RIII mice would provide obvious candidates for this purpose.
Conclusion and outlook
In this paper, we present a rationale and methodology for the progressive reduction and eventual replacement of prion bioassay titrations in laboratory rodents. This approach will also facilitate studies on the anti-prion effectiveness of (re-)processing procedures for medical devices and biological products, and help to further harness prions as test agents for the development of broad-range disinfectants and as bio-indicators for sterilization processes. 41
However, in order to definitely prove that combinations of PMCA and cell assays provide an effective or even superior alternative to bioassays in laboratory rodents, prion titre estimates from such jointly performed in vitro assays remain to be further validated by actual ID50 data from – preferably completed – in vivo studies.
Footnotes
Acknowledgements
We are grateful to Marion Joncic and Patrizia Reckwald for excellent technical assistance.
