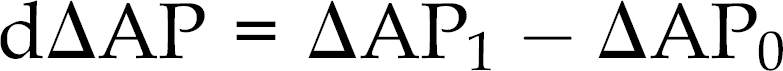Abstract
This study was designed to evaluate the oscillometric technique for intermittent non-invasive measurement of arterial pressure (AP) in the anaesthetized pig. In six pigs under sevoflurane anaesthesia, intermittent (every 2 minutes) oscillometric measurements of AP made at a forelimb (FL), a hindlimb (HL) and the tail base were compared with simultaneous direct AP measurements made at the aortic arch (AA) over a wide pressure range (30–140 mmHg of mean AP). All oscillometric measurements strongly correlated (Pearson correlation coefficient >0.837, P < 0.001) with direct AP measurements, with those made at the FL exhibiting the highest correlation. On the basis of consistency of difference between oscillometric and AA pressure readings at graded pressure levels, measurements made at the tail base provided reliable prediction of systolic AP, at the FL of diastolic AP and at both the tail base and the FL of mean AP. In conclusion, oscillometric measurements at the FL and the tail base offer reliable prediction of AP in the anaesthetized pig.
Intraoperative measurement of arterial pressure (AP) provides useful information for the determination of cardiovascular function adequacy during anaesthesia and the estimation of anaesthesia depth. The pig serves as an excellent model for human cardiovascular research due to similarities in anatomy and physiology with humans. In pig models, intra-arterial AP measurement is currently the method of choice in order to obtain accurate and reliable readings. Arterial access is usually achieved by catheterization of large neck or femoral arteries, such as common carotid and femoral arteries. 1 However, when surgical cut-down techniques are applied, manipulations of the vessel to avoid blood leakage from the catheterization site may increase the risk of postoperative complications due to ligation of the artery.2–4 Distal ligation of the artery immediately after cannulation and proximal ligation following withdrawal of the catheter are usually preferred over a demanding surgical repair of the vessel. 1 Percutaneous catheterization techniques may also be used to reduce trauma, but these should be performed by experienced personnel as the vessel cannot be seen directly.
Non-invasive methods for intraoperative intermittent AP measurement offer a safe, less demanding, cost-effective and practical alternative, as long as they provide accurate and reliable readings. Furthermore, they contribute to the enhancement of animal welfare during experimentation since they cause no trauma, pain or suffering. The oscillometric technique has been shown to reliably predict AP during anaesthesia in the dog,5,6 the cat 7 and the rabbit. 8 To date, there is evidence that the tail cuff oscillometric technique can be used to obtain reliable AP measurements in the conscious pig 9 and the minipig. 10
The aim of the present study was to evaluate the accuracy and reliability of the oscillometric technique for intermittent non-invasive AP measurement in the anaesthetized pig. For this reason, AP measurements using pressure cuffs placed at various anatomical sites were compared with simultaneous intra-arterial pressure readings.
Animals
Six Landrace pigs (Sus scrofa domestica), four months of age, weighing 30–35 kg were used in the study. The animals were purchased from a certified pig breeder. They were housed individually under controlled environmental conditions (20–22°C room temperature, 40–60% relative humidity, 12 hour photoperiod) in slatted-floor stainless steel kennels which allowed them to move around and lie down with outstretched legs. A plastic ball was provided for environmental enrichment. The facilities were in accordance with Directive 86/609/EEC. The pigs were fed twice a day (8:00 and 15:00) with 1 kg of commercially available standard pig pellets (EL.VI.Z., Xanthi, Greece) and tap water ad libitum. They were allowed a 14-day acclimatization period before the beginning of the study.
Materials and Methods
Experimental design
All animals were subjected to general anaesthesia during which AP was intermittently measured oscillometrically using an inflatable cuff placed around a forelimb (FL), a hindlimb (HL) and the tail base. Oscillometric measurements were compared with simultaneous intra-arterial pressure readings from the aortic arch (AA), considered as ‘true’ pressure, at a range between 30 and 140 mmHg of mean AP. Oscillometric AP measurements were evaluated on the basis of (a) their correlation with simultaneous AA pressure readings, (b) their difference from simultaneous AA pressure readings (ΔAP) and (c) the deviation of oscillometric–AA pressure difference at graded AA pressure levels from the oscillometric–AA pressure difference at a pre-set baseline AA pressure level (dΔAP); this was calculated using the following equation:
where ΔAP1: oscillometric–AA pressure difference at a specific AA pressure level.
ΔAP0: oscillometric–AA pressure difference at a pre-set baseline AA pressure level.
Baseline AA pressure levels were set at 80–90 mmHg for systolic pressure, at 50–60 mmHg for diastolic pressure and at 70–80 mmHg for mean pressure. The area under the curve (AUC) was calculated for comparison between techniques. The experimental protocol was approved by the Animal Care and Ethics Committee of the local Veterinary Service since it was in compliance with Directive 86/609/EEC.
Animal preparation
All animals studied were healthy as assessed by clinical, biochemical and haematological examinations. After deprivation of food for 24 hours and water for 12 hours, general anaesthesia was induced by intramuscular injection of a xylazine (2 mg/kg)–atropine (0.05 mg/kg)–ketamine (33 mg/kg) mixture. The animals were then endotracheally intubated and connected to an anaesthetic machine–mechanical ventilator (Avance/Datex Ohmeda, Madison, WI, USA). Initial settings of the ventilator (12 breaths/min and 300 mL tidal volume) were adjusted, if needed, to maintain end tidal CO2 (ETCO2) between 35 and 45 mmHg as measured automatically in the expired gas. The fraction of inspired O2 (FiO2) was set at 100%. Anaesthesia was maintained by administration of sevoflurane (2–3% in oxygen). The animals were placed at dorsal recumbency on a heated-surface operation table with their shoulder, elbow, hip and stifle joints at flexion. A temperature probe was inserted intrarectally to monitor core temperature; this was maintained between 38 and 39°C throughout the experiment. A jugular vein was catheterized after surgical preparation of the vessel (according to the procedure described below) to facilitate intravenous infusion of agents.
Intra-arterial AP measurement
Intra-arterial AP measurements were made after having catheterized the AA through the common carotid artery ipsilateral to the cannulated jugular vein. Catheterization was performed aseptically following surgical preparation of the artery. The latter was exposed after paramedian incision over the jugular furrow and dissection of the fascial plane on the dorsal surface of the sternocephalic muscle. The common carotid artery was then isolated with vessel loops and catheterized with a 16G peripherally inserted central catheter (PICCO Arrow/reading, PA, USA). The tip of the catheter was forwarded at a fixed length, determined in pilot autopsies in pigs of the same weight, in order to reach the aortic arch, also guided by pressure wave recording. The free end of the catheter was connected to a pressure monitoring set (Pressure monitoring set/Edwards Lifesciences, Irvinen, CA, USA) which consisted of a 150 cm long extension line made of material proper for pressure monitoring, two three-way stopcocks, a transducer and a pressurized common infusion line connected to a bag with heparinized saline. The system was zeroed at heart level. AP (systolic, diastolic and mean) was monitored using the S/5 Compact Critical Care Monitor (Datex-Ohmeda).
Oscillometric AP measurement
Pressure cuffs were placed circumferentially around (a) the middle of the forearm with the artery indicator arrow dorsomedially over the radial artery (FL cuff), (b) the middle of the tibia with the artery indicator arrow dorsomedially over the medial saphenus artery (HL cuff) and (c) the base of the tail with the artery indicator arrow ventrally over the coccygeal artery (tail cuff). Commercially available human child cuffs (7.5 cm in width × 29 cm in length, Datex Ohmeda) were placed around the FL and the HL, and a human infant cuff (4.5 cm in width × 24.5 cm in length, Datex Ohmeda) around the base of the tail. The width of the cuffs accounted for approximately 35% of the FL and HL circumference (child cuff) and 50% of that of the tail base (infant cuff). Measurements were made automatically every two minutes. The maximum inflation pressure was 280 mmHg. The AP readings were videotaped and later analysed. In order to evaluate the oscillometric technique over a wide range of APs, including extreme blood pressures, AP was either increased or decreased by intravenous infusion of noradrenaline (Noradren sol. inf. 8 mg/4 mL/DEMO ABEE, Athens, Greece) solution (0.032% in normal saline) or sodium nitroprusside (Nitriate powder injection 50 mg/vial/Serb-Labo, Paris, France) solution (0.1% in normal saline), respectively. Noradrenaline and sodium nitroprusside were infused at incremental rates in order to achieve the desired AP levels. All pigs were normotensive at the beginning of the study and were provoked to achieve incremental mean AP levels as high as 140 mmHg and then decremental levels as low as 30 mmHg.
Statistical analysis
Linear regression analysis was performed between oscillometric and simultaneous intra-arterial pressure readings. Mean differences between oscillometric and AA pressure measurements for various AA pressure levels were subjected to repeated measures analysis of variance (repeated measures ANOVA) followed by the least significant difference test for multiple comparisons of means among different AA pressure levels. The AUC was calculated using the trapezoidal rule 11 to assess the overall deviation of oscillometric measurements from AA pressure for each cuff site tested. A probability of P < 0.05 was considered statistically significant.
Results
Heart rate remained within normal range (70–100 beats per minute) with the exception of tachycardia (>200 beats per minute) noted at extreme AA pressure levels (>130 mmHg of mean AP).
Correlation of oscillometric with AA pressure measurements
Oscillometric readings strongly correlated with AA pressure readings. Measurements made at the FL exhibited the strongest correlation compared with those made at the HL and the tail base for systolic, diastolic and mean pressures (Table 1). Regression equations of oscillometric against AA readings are presented in Table 2.
Correlation coefficients (Pearson) of oscillometric against direct aortic arch pressure measurements (n = 158) in anaesthetized pigs (P < 0.001)
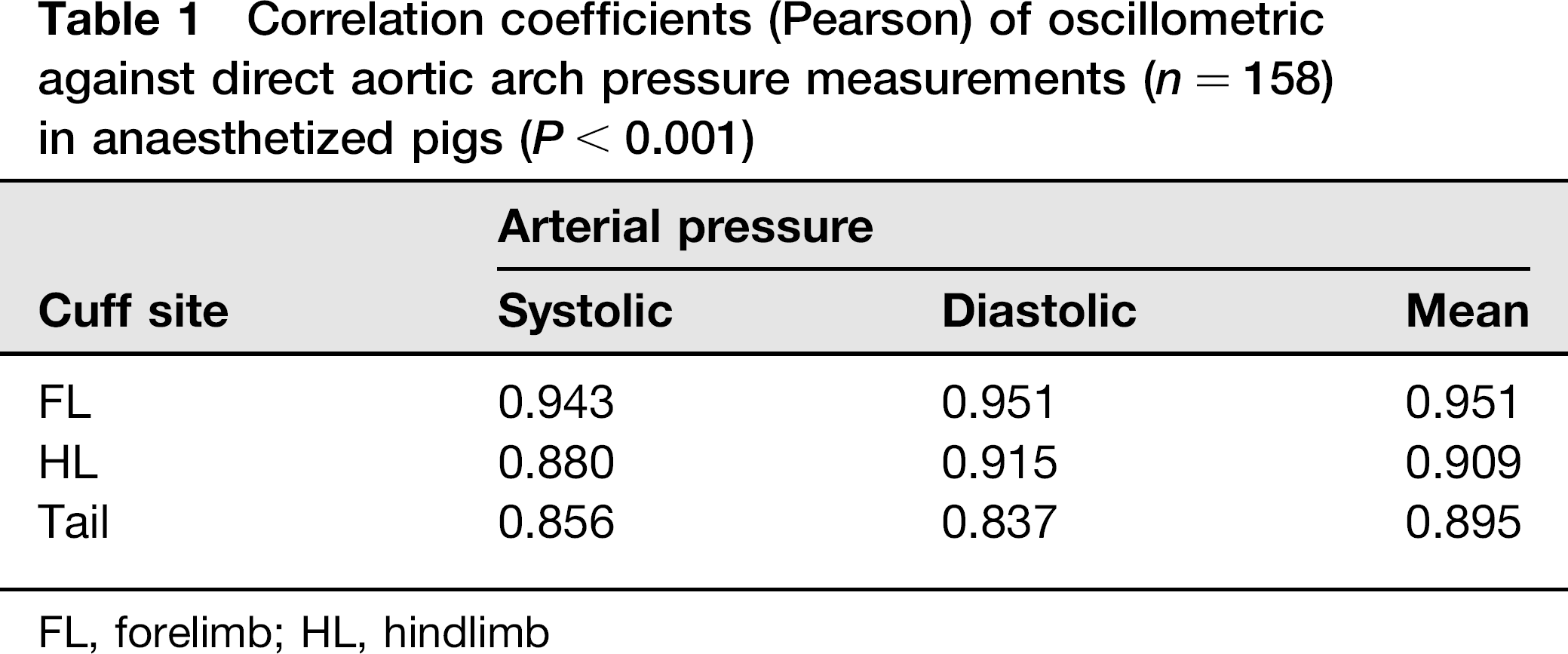
FL, forelimb; HL, hindlimb
Regression equations of oscillometric against aortic arch pressure measurements (n = 158) in anaesthetized pigs
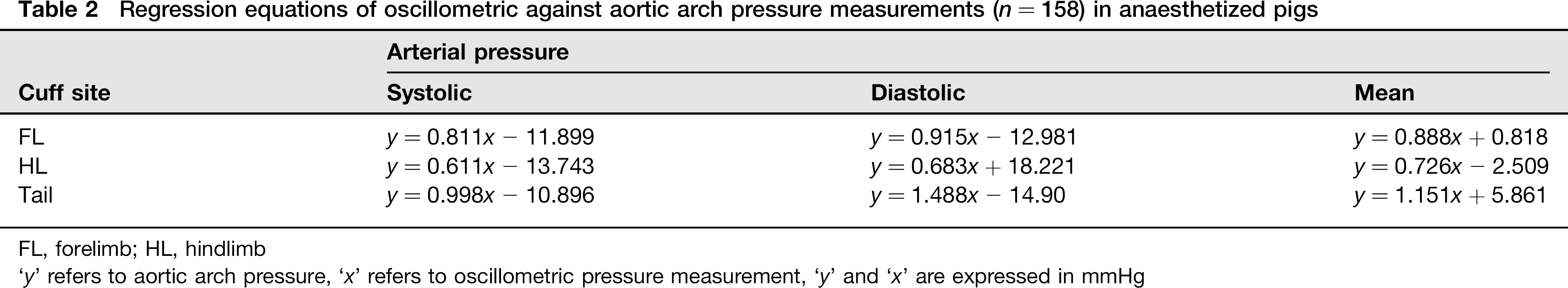
FL, forelimb; HL, hindlimb
‘y’ refers to aortic arch pressure, ‘x’ refers to oscillometric pressure measurement, ‘y’ and ‘x’ are expressed in mmHg
Difference of oscillometric from AA pressure measurements
Systolic AP
All oscillometric AP measurements overestimated AA systolic pressure. Tail base measurements exhibited the lowest overall deviation from AA pressure compared with those made at the FL and the HL (AUCs; 117.5 mmHg2 for the tail base, 407.9 mmHg2 for the FL, 925.7 mmHg2 for the HL). As AA pressure increased, deviation from AA pressure was (a) consistent for the tail base measurements, (b) fairly consistent for the FL measurements with a plateau noted between 70 and 150 mm of AA pressure and (c) progressively incremental for the HL measurements with a plateau noted between 60 and 110 mmHg of AA pressure (Figure 1).
Difference of oscillometric from simultaneous direct (AA pressure) measurements in anaesthetized pigs. a,b,c,d – For each cuff site, means that do not share a common letter differ significantly (P < 0.05). AA, aortic arch; OSC, oscillometric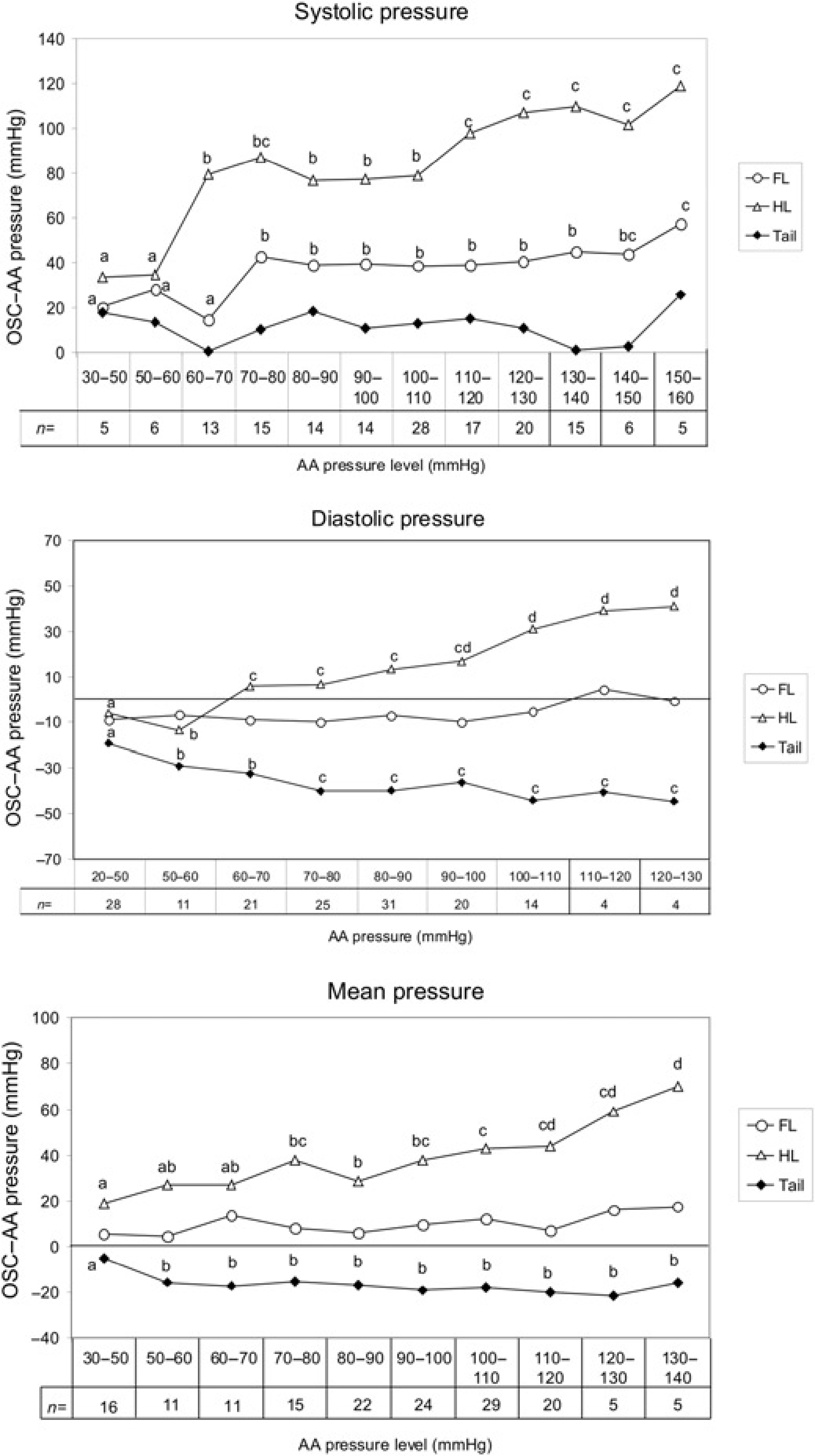
Diastolic AP
Measurements made at the HL overestimated AA diastolic pressure, while those made at the FL and the tail base underestimated it. FL measurements exhibited the lowest overall deviation from AA pressure compared with the other cuff sites tested (AUCs; 52.2 mmHg2 for the FL, 143.7 mmHg2 for the HL and 293.6 mmHg2 for the tail base). As AA pressure increased, deviation from AA pressure was (a) consistent for the FL measurements, (b) progressively incremental for the HL measurements with a plateau noted between 60 and 100 Hg pressure and (c) progressively decremental for the tail base measurements with a plateau noted between 70 and 130 mmHg of AA pressure (Figure 1).
Mean AP
Measurements made at the FL and the HL overestimated AA mean pressure, while those made at the tail base underestimated it. FL measurements exhibited the lowest overall deviation from AA pressure readings compared with the other cuff sites tested (AUCs; 88.2 mmHg2 for the FL, 155.2 mmHg2 for the tail base and 348.4 mmHg2 for the HL). As AA pressure increased, deviation from AA pressure was (a) consistent for the FL and the tail base measurements, while (b) progressively incremental for the HL measurements with a plateau noted between 50 and 100 mmHg of AA mean pressure (Figure 1).
Deviation of oscillometric–AA pressure difference
The overall deviation of oscillometric–AA pressure difference from the baseline oscillometric – AA pressure difference was lowest for the FL measurements compared with the other cuff sites tested for systolic, diastolic and mean pressures (Figure 2).
Deviation of oscillometric–AA pressure difference and AUCs in anaesthetized pigs. Dashed lines denote baseline AA pressure levels set at 80–90 mmHg for systolic AP, 50–60 mmHg for diastolic AP and 70–80 mmHg for AA mean AP. AA, aortic arch; AP, arterial pressure; OSC, oscillometric; AUC, area under the curve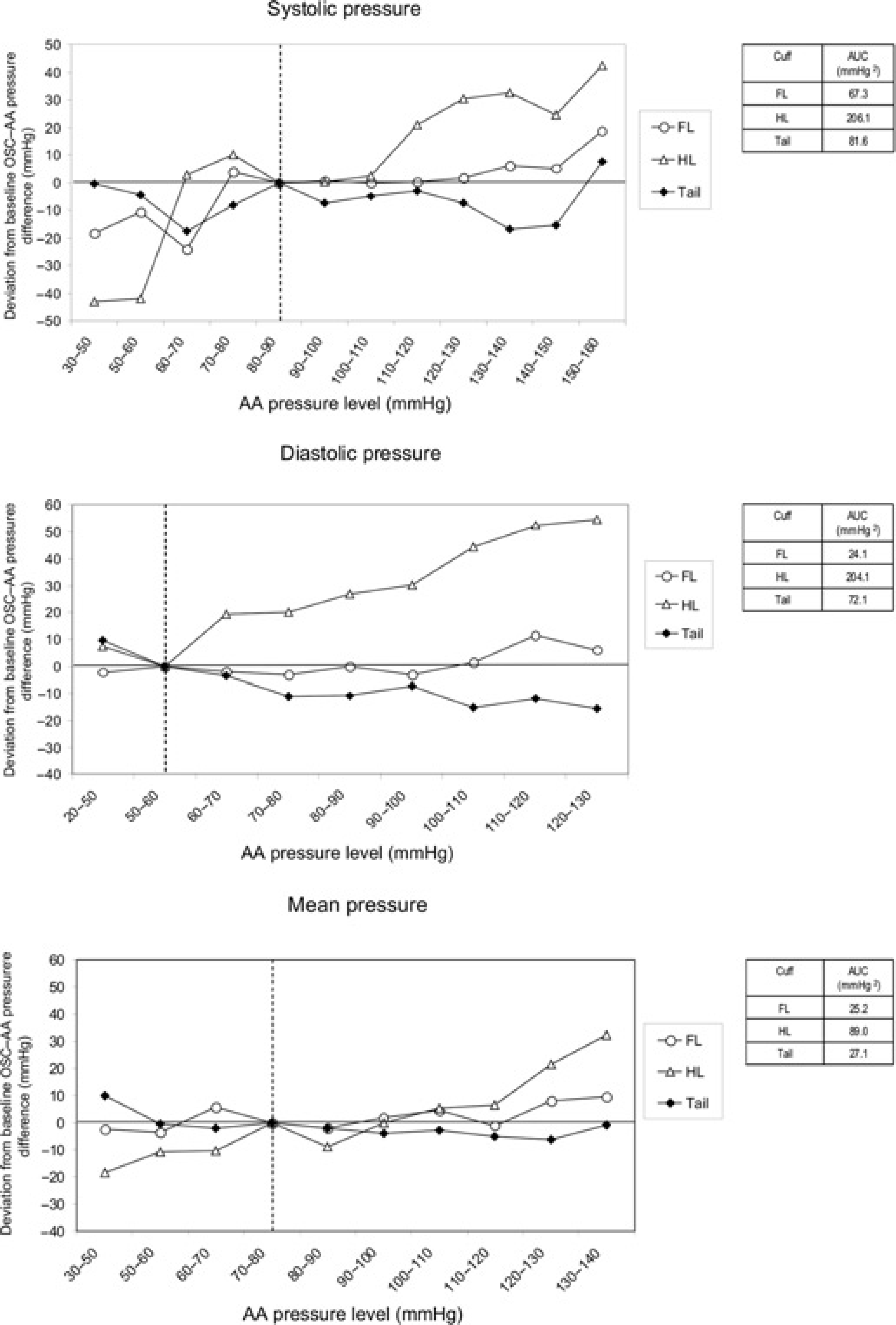
Discussion
The present study evaluated and compared the oscillometric technique used for intermittent AP measurement at the FL, the HL and the tail base in the anaesthetized pig against simultaneous direct AP readings. All oscillometric readings exhibited strong correlation with intra-arterial pressure readings. Oscillometric measurements made at the tail base provided reliable prediction of ‘true’ systolic AP, at the FL of diastolic AP and at both the FL and the tail base of mean AP. The results of the present study are useful in cases where estimating blood pressure changes is more important than monitoring the exact central AP which would necessitate the performance of invasive surgery just for the purpose of direct AP measurement. Therefore, they contribute to minimizing unnecessary pain and suffering of the animals during experimentation.
Direct AP measurements made at the AA were used as reference measurements. The AA is the curved portion of the aorta, between its ascending and its descending part, and represents a large vessel close to the heart which is not subjected to vasoconstriction, therefore providing a reliable site for accurate AP measurement.
Oscillometric AP measurements were conducted in anaesthetized pigs at dorsal recumbency by placing pressure cuffs at anatomical sites offering access to the radial (FL cuff), medial saphenus (HL cuff) and coccygeal arteries (tail cuff). The oscillometric technique makes use of an inflatable cuff placed around a limb or the base of the tail occluding blood flow at the underlying artery. A sensor on the cuff measures the magnitude of oscillations caused by blood pressure pulses as the cuff is deflating. A monitor measures the oscillation with the greatest amplitude which represents mean pressure, while it calculates systolic and diastolic pressures according to certain algorithms that differ from one model of device to another.
Factors that may affect the accuracy of oscillometric measurements include the ratio of cuff width to limb circumference, the horizontal and vertical distance of the vessel from the heart, core temperature deviations, extreme heart rates (<40 or >200) and extreme pressure deviations (hypo- or hypertension). 12 It has been estimated in humans as well as in animals that the width of the cuff should account approximately for 40% of the limb circumference to provide accurate prediction of AP. 13 In the current study, the cuff used for measurements at the FL and the HL corresponded to approximately 35% of the limb circumference, while the one used at the tail to 50% of the tail base circumference. Measurements were made in animals at dorsal recumbency keeping their shoulder, elbow, hip and stifle joints at flexion, thus minimizing the vertical distance of the FL and HL cuffs, respectively, from the heart. During anaesthesia, core temperature was maintained within normal limits to avoid hypo- or hyperthermia. Heart rate remained within normal range, with the exception of tachycardia (>200 beats per minute) noted at high AA pressure. According to linear regression analysis, measurements made at all cuff sites strongly correlated with AA pressure, with those made at the FL exhibiting the highest correlation. Depending on the cuff site tested, pressure was either over- or underestimated. A discrepancy between the appropriate cuff size and the size of the cuffs that were actually used may have contributed to the inaccuracy of oscillometric measurements. Therefore, an overestimation of pressure by FL and HL measurements could be attributed to a narrow cuff, while an underestimation by tail base measurements to a wide cuff. A close approximation of ‘true’ pressure, though, can be obtained by entering oscillometric AP data in regression equations since there was a strong correlation between the variables tested. Nevertheless, validation of oscillometric measurements based on regression analysis has been questioned since it may mask large individual differences or even systematic differences between methods. 14
We also assessed the difference of oscillometric from direct pressure readings over a wide range of graded pressure levels which provided further justification of the reliability of the technique. The difference remained consistent, even at extreme pressures, for systolic pressure when measurements were made at the tail base, for diastolic pressure at the FL and for mean pressure at both the tail base and the FL. On the other hand, the HL did not prove to be a reliable cuff site since oscillometric measurement difference was consistent only at a narrow pressure window and deviation from AA pressure increased at high pressure levels. The fact that peripheral arteries are subjected to vasoconstriction at high pressures, in contrast to large central arteries such as the AA, may constitute a source of error in oscillometric measurements.
A comparative estimation of reliability between cuff sites was possible by calculating the deviation of oscillometric from direct pressure difference at various AA pressure levels from that at a pre-set baseline AA pressure level. Based on this criterion, measurements made at both the FL and the tail base proved to provide the least variable difference from AA mean pressure, with those made at the FL being superior for systolic and diastolic pressures assessment. According to previous animal studies, the tail base has been the proposed site for oscillometric AP measurement in the conscious pig 9 and the minipig. 10
In experimental protocols involving anaesthesia, not only accurate measurements but also a trend in AP in relation to a baseline value may provide useful information on cardiovascular function and anaesthesia depth. In conclusion, the present study showed that oscillometric measurement of AP performed at either the FL or the tail base can reliably predict ‘true’ AP in the anaesthetized pig offering a scientifically sound and practical alternative to direct pressure measurement for successful detection of trends in AP. The application of this method contributes to the ‘refinement’ of experimental procedures, according to the principle of ‘3Rs’, by reducing unnecessary suffering of animals.