Abstract
Endotracheal intubation is often necessary for positive pressure ventilation of rats during open thoracic surgery. Since endotracheal intubation in rats is technically difficult and is associated with numerous complications, many techniques using various devices have been described in the scientific literature. In this study, we compared the effectiveness of airway management of a home-made supraglottic airway device (SAD), which is cheap to fabricate and easy to place with that of an endotracheal intubation tube in enflurane-anaesthetized rats. Twenty male Sprague-Dawley rats (200–300 g) were randomly assigned to two equal groups for positive pressure mechanical ventilation using either the SAD or an endotracheal intubation tube. The carotid artery of each rat was cannulated for continuous blood pressure measurements and obtaining blood samples for determination of oxygen tension, carbon dioxide tension, and blood acidity before, during and after SAD placement or endotracheal intubation. Proper placement of the SAD was confirmed by observing chest wall movements that coincided with the operation of the mechanical ventilator. No complications and adverse events were encountered in the rats in which the SAD was placed, during SAD placement and immediate removal, during their mechanical ventilation through the SAD, and one week after SAD removal. From the results of blood gas analyses, we conclude that anaesthetized rats can be successfully ventilated using an SAD for open thoracic surgery.
Endotracheal intubation is often necessary for the mechanical ventilation of anaesthetized rats during open thoracic surgery. In rats, endotracheal intubation is technically difficult because of its narrow oropharynx and the small size of its oral cavity. That no prefabricated intubation systems are available, many methods of endotracheal intubation in rats using various instruments or selfdesigned devices have been described in the scientific literature. These methods include blind orotracheal intubation 1 or the use of the blade of an infant laryngoscope and a fibreoptic light guide for direct visualization of the larynx or transillumination of the larynx.2–8 However, endotracheal intubation in rats using such instruments and/or devices can often result in intubation failure because injuries to and bleeding in the oral cavity and oropharynx, and respiratory arrest due to laryngeal spasm.
The face mask is an alternative to endotracheal intubation for anaesthesia and airway support because its use eliminates the difficulties and potential injuries of endotracheal intubation. Although hypercapnia and low partial pressures of oxygen can occur with their use, several reports on its successful use for artificially ventilating rabbits and rodents have been published.9,10
The laryngeal mask airway (LMA) is another alternative device for anaesthesia, airway support and positive pressure ventilation without endotracheal intubation in humans. 11 The LMA is a supraglottic airway device (SAD) that was developed in 1981 and was designed to fill the gap between endotracheal intubation and face masks. In humans, the LMA is inserted blindly into the pharynx, and is used when an endotracheal intubation is difficult or has failed. The LMA has many advantages: it is easy to correctly place even when its placement is done by a relatively inexperienced person, and it does not stimulate the airway because no contact between the device and the vocal cords and tracheal mucosa occurs. 12 There are now several reports in the veterinary and medical literature which describe the LMA as an effective alternative method to endotracheal intubation in cats which require anaesthesia under spontaneous or positive ventilation, 13 and for the instillation of surfactant into the trachea of newborn pigs. 14
In view of the advantages of an LMA, we modified the design of the human LMA in order to fabricate an SAD for use in rats when (a) airway support is required in a procedure, such as open thoracic surgery, and (b) endotracheal intubation or instillation is not required or possible and a face mask cannot be used. In this report, we describe the results of a study in which we compared the effectiveness of airway management of this home-made SAD to that of an endotracheal intubation tube in enflurane-anaesthetized rats.
Materials and Methods
The maintenance and welfare of the rats complied with the Korean national legislation on the use of animals for experimental and other scientific purposes (Article 14, Animal Protection Act). The study and all its procedures were reviewed and approved by the Animal Care and Use Committee of the Inje University.
Animals and equipment
The study comprised 20 male Sprague-Dawley rats (200–300 g), which were purchased from Charles River Laboratories (Seoul, Korea) and acclimated for at least two weeks in the Animal Care Facility of the Paik Institute for Clinical Research. During this acclimation period, the rats were housed in open-top plastic cages (two rats per cage) under controlled environmental conditions: 22 ± 2°C ambient temperature; 50% relative humidity; an air exchange rate of 12–15 changes/hour and a light : dark cycle of 12 : 12 hours with lights on at 06 : 00. The rats had free access to tap water and a pelleted rat chow (Cargill Agri Purina, Inc., Seongnam, Gyeonggi, Korea), which contained 20% crude protein, 4.5% crude fat, 6% crude fibre and 12.15 MJ/kg net energy. The health status of each rat was daily assessed by a physical examination and weighing. Before the start of the experiment, the rats were maintained for one week in open-top plastic cages (one rat/cage) under the identical environmental conditions.
The SAD comprised a diagonally cut soft rubber section and plastic tubing of a disposable blood transfusion set (BD, Suzhou, China) (Figures 1a and d). The endotracheal intubation tube comprised a 16G intravenous catheter (45 mm [length], 1.77 mm [inner diameter]; BD Angiocath Plus, Busan, Korea), which was inserted into the trachea by passing it through the lumen of an 18G epidural needle (BD PerisafeTM, USA).
8
Required equipment and assembly of the supraglottic airway device (SAD) and its placement. The parts of a disposable blood transfusion set that are required to assemble the SAD (a), the diagonally cut soft rubber section and plastic tubing of a disposable blood transfusion set (b), the cut rubber sections and plastic tubing shapes (c). The cut rubber section in the SAD which is used for covering the laryngeal opening (arrow in d). The SAD is connected to a Y connector (arrow head) in the breathing circuit with an operating ventilator (d). The handling of the animal for SAD placement is shown in (e). The tongue is pulled cranially and ventrally until resistance is felt. The SAD is then inserted into the mouth with the cut rubber section pointed ventrally (e). The larynx, epiglottis (arrow head) and oesophagus (arrow) in the posterior oral cavity (f). One dead rat from the SAD group was used to photograph the oral cavity and larynx (arrow head) before (g) and after placing the SAD, and the correct placement of SAD (arrow) over larynx (arrow head) in the oral cavity (h). Placement of the SAD for artificial ventilation in an anaesthetized rat (i)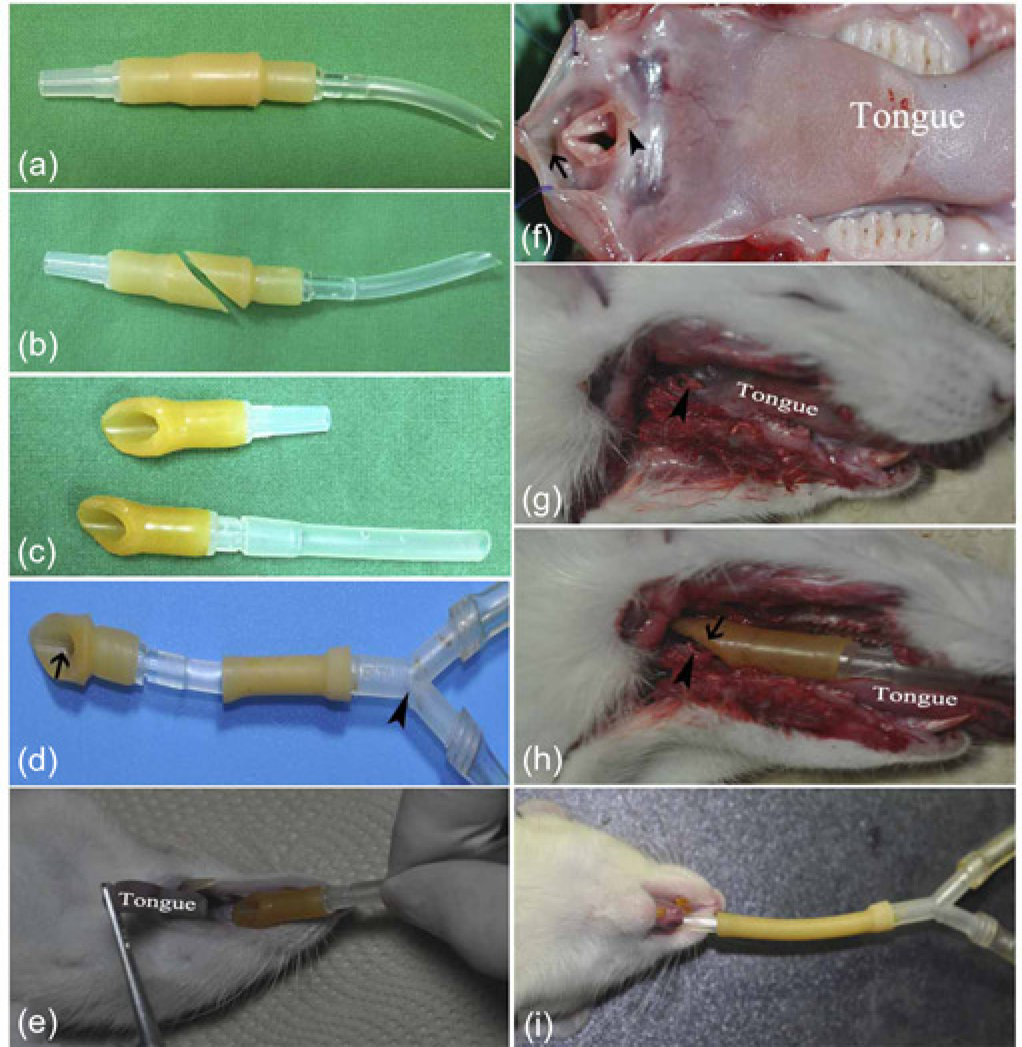
Anaesthesia and hemodynamic monitoring
All experiments were performed between 10:00 and 17:00. On the day of the experiment, each rat was first weighed and then placed in a 5-L acrylic chamber into which oxygen was delivered at a flow rate of 5 L/minute for five minutes before anaesthetic induction. Anaesthesia was then induced by delivering 5% enflurane (Alyrane, Baxter, USA) in 5 L/minute oxygen into the chamber until the rat was recumbent and the eyelid closure reflex was lost. The rat was then removed from the chamber, and transferred to an operating table where it was placed in the supine position in front of the barrel of a 60-cm3 syringe (Baxter, Deerfield, IL, USA), which was used as a nose cone. The face of each rat was snugly placed into the syringe's barrel, whose other end was connected to an enflurane vaporizer (Vapor 19.1; Drager, Germany) using the plastic tubing of an intravenous administration set. Anaesthesia of the rats was maintained by 3–5% enflurane in 2 L/minute oxygen. The carotid artery of the spontaneously breathing enflurane-anaesthetized rats was cannulated with a PE35 polyethylene catheter (BD, Japan), which was sterilized before use by ethylene oxide gas, for arterial blood pressure measurements and obtaining arterial blood samples. The artery was cannulated under aseptic surgical conditions. Arterial blood pressure was measured continuously through carotid artery catheter by a Bridge Amp pressure transducer (www.adinstruments.com, Australia) whose output was recorded and analysed using LabChart® software (version 7 for Windows). The body temperature of the anaesthetized rats was maintained between 36.5 and 37.5°C using a heating pad (SOFTRON, TMS-201, Beijing, China).
SAD placement insertion and endotracheal intubation
The 20 anaesthetized rats were randomly assigned to two equal groups for positive pressure mechanical ventilation: The SAD group was ventilated through the SAD and the ETD group was ventilated through an endotracheal intubation tube. For airway management, the SAD or the endotracheal intubation device was connected to a rodent ventilator (Model 683; Harvard Apparatus, Holliston, MA, USA) whose rate and tidal volume was set as 60 breaths/minute and 8 mL/kg according to the rat's body weight. For SAD placement, the mouth of each anaesthetized rat was opened and the tongue was then pulled cranially and ventrally until resistance was felt. The SAD was then inserted into the mouth with the cut opening of the rubber pointed ventrally (Figures 1d and e) until resistance was felt (Figures 1e, h, and i). The ETD group underwent blind endotracheal intubation using a previously described method. 8
The duration of each experiment on each enflurane-anaesthetized rat (3–5% enflurane) was one hour and comprised two phases – a 30-minute phase of spontaneous breathing and a 30-minute phase of artificial breathing during which the enflurane-anaesthetized rat was attached to a rodent ventilator. The operational settings of the ventilator were 8 mL/kg (tidal volume), 60 breaths/minute (rate) and 1.0 (fraction of inspired oxygen [FiO2]), in accordance with a previously published report. 10 SAD placement or endotracheal intubation was assumed to have failed when the chest wall movements did not coincide with the inspiratory–expiratory cycle of the ventilator, 10 minutes after placing the device. SAD placement or endotracheal intubation was successful in all rats as evidenced by chest wall movements that coincided with the inspiratory–expiratory cycle of the ventilator.
Haemodynamic and respiratory parameter measurement
Mean arterial blood pressure (MABP) was measured in the spontaneously breathing enflurane-anaesthetized rats immediately before SAD placement or endotracheal intubation, during SAD placement or endotracheal intubation, and in the mechanically ventilated enflurane-anaesthetized rats, 10 minutes after SAD placement or endotracheal intubation. The continuous MABP data that were collected over each minute at these three time points were averaged, and this average MABP was used in the data analysis. Arterial blood samples (0.3 mL) from each rat were collected through the carotid artery catheter before and after SAD placement or endotracheal intubation for the determination of arterial oxygen tension (PaO2), arterial carbon dioxide tension (PaCO2) and arterial blood acidity (pH) using a blood gas analyser (RAPIDLAB®1265, Siemens Sudbury, UK). At the end of the experiment, the arterial catheter was removed, the carotid artery was ligated, the wound was closed using 5-0 silk, the enflurane anaesthesia was discontinued and the rat was allowed to recover. When the movements of the rat began to be active, the intubation device or the SAD was removed and the rat was returned to its cage in the animal care facility. After their return to the facility, the postprocedural monitoring comprised subcutaneous buprenorphine injections (0.025 mg/kg) every 12 hours for two days, and daily clinical examinations and weighing for seven days. All rats were humanely killed by placing them in the anaesthetic chamber and exposing them to an overdose of enflurane, two weeks after completion of the experiment.
Data analysis
Data are presented as mean±standard deviation. Data from the two groups of rats were compared using a Student's t-test. The level of statistical significance was set at 5% (P < 0.05).
Results
None of the rats died during the experiment and all rats recovered from the procedure and experiment.
The results of the arterial blood gas analysis and arterial blood pressures before and after SAD placement or endotracheal intubation in the enflurane-anaesthetized rats are presented in Table 1. All values of PaCO2 (35–45 mmHg), PaO2 (300–500 mmHg) and pH (7.35–7.45) of the rats in the SAD and ETD groups were within their normal ranges during artificial ventilation. Moreover, no significant differences in these values were found between the two groups. The PaCO2 values of the spontaneously breathing enflurane-anaesthetized rats before SAD placement or endotracheal intubation were higher than those in the mechanically ventilated enflurane-anaesthetized rats, 10 minutes after SAD placement or endotracheal intubation. In contrast, the blood pH values before SAD placement or endotracheal intubation were significantly lower (P < 0.05) than those found after SAD placement or endotracheal intubation. Although MABP rose significantly during SAD placement or endotracheal intubation, no statistically significant differences in both the absolute MABP and the pressure increment were found between the two groups. The time taken to place the SAD was significantly shorter (P < 0.05) than that taken for endotracheal intubation (Table 1). One week after completion of the experiment, all rats in the two groups were healthy with normal activity.
Changes of arterial blood gas tensions, pH and mean arterial blood pressures
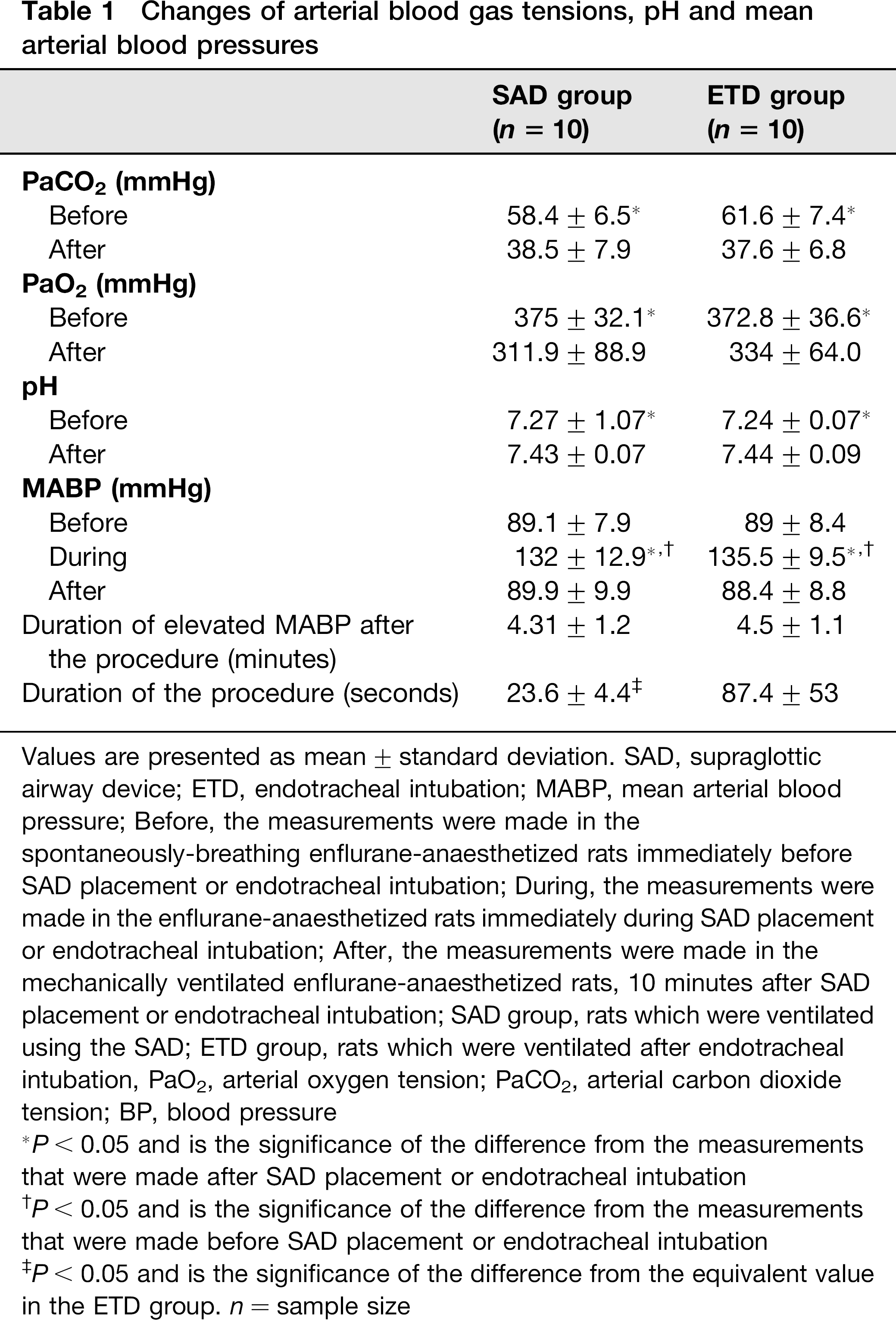
Values are presented as mean±standard deviation. SAD, supraglottic airway device; ETD, endotracheal intubation; MABP, mean arterial blood pressure; Before, the measurements were made in the spontaneously-breathing enflurane-anaesthetized rats immediately before SAD placement or endotracheal intubation; During, the measurements were made in the enflurane-anaesthetized rats immediately during SAD placement or endotracheal intubation; After, the measurements were made in the mechanically ventilated enflurane-anaesthetized rats, 10 minutes after SAD placement or endotracheal intubation; SAD group, rats which were ventilated using the SAD; ETD group, rats which were ventilated after endotracheal intubation, PaO2, arterial oxygen tension; PaCO2, arterial carbon dioxide tension; BP, blood pressure
P < 0.05 and is the significance of the difference from the measurements that were made after SAD placement or endotracheal intubation
P < 0.05 and is the significance of the difference from the measurements that were made before SAD placement or endotracheal intubation
P < 0.05 and is the significance of the difference from the equivalent value in the ETD group. n = sample size
Removal of the SAD or endotracheal tube at the end of experiment was not associated with any adverse effects, such as laryngospasm. However, we found occult blood in the endotracheal tube of two rats. We attribute the presence of this blood to injury and bleeding in the oral cavity and pharynx that were caused by the tip of the 18G epidural needle that was used as a guide. No blood or excess mucous was found on the SAD after its removal. No necropsies on the rats were done because all the rats in the two groups recovered from the procedure and experiment. However, one dead rat from the SAD group was used to photograph the oral cavity and larynx before and after placing the SAD (Figures 1f and h).
Discussion
In this study, we found that the PaCO2 levels were high and the blood pH values were low before SAD placement or endotracheal intubation in the spontaneously breathing enflurane-anaesthetized rats, and we attribute these findings to be due to enflurane induced-hypoventilation. We also found that the PaO2 levels in the spontaneously breathing rats were significantly higher than those in the mechanically ventilated rats. The obvious explanation for this finding is that the SAD and endotracheal tube interfered with pulmonary oxygenation. Since it is known that mechanical ventilation increases intrapulmonary shunting, 15 we posit that intrapulmonary shunts were created when mechanical ventilation was started in the spontaneously breathing rats.
We also found that the MABP rose when the rat's tongue was pulled out of the mouth for SAD placement or endotracheal intubation. We also found that the rises in MABP and the durations of the MABP elevation were the same in the SAD and ETD groups, despite the time for SAD placement being substantially shorter than that required for endotracheal intubation. These results suggest that pulling of the rat's tongue triggers an autonomic reflex which causes the MABP to rise. Interestingly, the MABP remained elevated in some rats in the ETD group for more than five minutes after they had been endotracheally intubated. We posit that this prolonged rise in the MABP was probably caused by tissue irritation by the endotracheal tube when it was passed through the larynx into the trachea.
In a previous study that was done by our group, 8 we found that some rats died of airway obstruction immediately after removal of the endotracheal tube. Airway obstruction following removal of an endotracheal tube is thought to be caused by laryngospasm, excessive mucous secretion or glottic oedema. Although several methods of endotracheal intubation in rodents are described in the scientific literature, some of these methods require the use of expensive instruments2–4 and/or user training.1,5,7,16 Complications, which are sometimes very difficult to overcome, such as laryngospasm, pharyngeal oedema, local tissue or glottic trauma, and mucous obstruction of the endotracheal tube, can occur during endotracheal intubation. Moreover, repeated attempts to intubate the same animal can increase the frequency of the complications. 17
Face masks can also be used for artificially ventilating of rats. In their report, Yuan et al. 10 wrote that their rodent face mask is easy to construct, simple to use, the components are readily available and the operator needs no specific technical training. They also commented that its major disadvantage was that some of the anaesthetic gas mixture that is delivered through a face mask enters the oesophagus. Although this gas will increase intra-abdominal pressure as the ventilation time increases, the gas will does not alter spontaneous respiration and other activities of the rat, and abdominal pressure usually returns to normal within six hours, postoperatively.
The use of an SAD is another method for artificially ventilating rodents. According to the results of our arterial blood gas analyses, we showed that our SAD was as efficient in maintaining mechanical ventilation as endotracheal intubation. Accordingly, we propose that the use of an SAD is more advantageous than endotracheal intubation because its use eliminates the difficulties and complications of endotracheal intubation. For example, the placement of an SAD causes less irritation to the larynx than an endotracheal tube and does not irritate the trachea. Incorrect placement of the SAD can be easily detected and immediately corrected by repositioning. If the SAD is incorrect placed over the oesophageal entrance, the incorrect placement can be detected by abdominal wall movements that do not coincide with the inspiration and expiration phases of the mechanical ventilator. Should such movement be seen, the SAD can be repositioned in the pharynx. After SAD placement, gas leakage through the mouth, which can be detected by cheek wall movements, can be easily corrected by increasing tidal volume of the mechanical ventilator. However, endotracheal intubation is still required for intratracheal or intrabronchial operations, such as intracheal or intrabronchial instillation of drugs 18 or bronchoalveolar lavage. 19 The main disadvantage of an SAD in rodents is the same as that described for laryngeal masks in humans: 20 an SAD may be unable to completely protect the airway against aspiration of gastric contents.
In summary, we describe the assembly and use of a simple, easy and cheap to fabricate SAD to successfully ventilate an anaesthetized rat. Since placement of an SAD does not require any specialized equipment and its placement is non-injurious to the rat, its operator does not require prior training in order learn how to use the SAD.
