Abstract
Conflict research is rife with ethical issues, and the field is increasingly reflecting on how to best address these. Recent debates in political science have mainly focused on ethics in practice, leaving questions of procedural ethics to the side. But procedural ethics are important: they are increasingly required across all areas of research, they are the bedrock of institutional approaches to regulating ethics, and they shape ideas about what constitutes ethical research practice. This article introduces the Research Ethics Governance dataset, the first globally comprehensive dataset of national-level ethics regulations. The dataset provides a picture of the status of research ethics regulations and how they pertain to conflict research. While 87% of countries have requirements for ethical review, only 25% extend those regulations to the social sciences. Of countries with no evidence of requirements, nearly half are classed as fragile or conflict-affected states. The data will be useful for scholars concerned with questions of research ethics, as well as those seeking to study the politics of this regulatory structure and its implications for knowledge production.
Keywords
Before beginning research, a familiar experience for scholars is securing ethical approval from an institutional review board (IRB) – also known as research ethics committees (RECs), human subjects research committees (HSRCs) and related monikers. Ethical review is especially salient for conflict research, which often entails significant and unique risks (Campbell, 2017; Cronin-Furman and Lake, 2018; Mazurana et al., 2013). At the same time, ethical review processes can be complex, contradictory and highly bureaucratic, not to mention time-consuming and costly – even more so for research that crosses geographic and disciplinary borders (Desposato, 2015; Fisher, 2021; Yanow and Schwartz-Shea, 2016). Despite the widely recognized importance of ethical regulations for social science and conflict research, we currently know strikingly little about them beyond a handful of Global North countries.
In this article, we introduce the Research Ethics Governance (REG) dataset, a new and original dataset that offers a systematic and global view of national-level research ethics regulations as of 2020. REG is a cross-sectional dataset that documents the year that countries first adopted ethical regulations; some key characteristics of ethics regulations; and whether, when and how they apply to social science research. The dataset complements existing studies on research ethics in peace and conflict studies, which mainly focus on ethics in practice, by describing the state of procedural ethics – or the process of seeking formal approval for human subject research (Guillemin and Gillam, 2004).
While procedural ethics are often the subject of critique, they are nonetheless widely recognized as an important element of how ethics is understood, implemented and enforced. They are increasingly reinscribed not only by national-level requirements and universities, but also by funding and publication requirements. REG focuses on national-level regulations, and in this sense, does not account for how ethical regulations are implemented, often through ethics committees based at universities, hospitals, or even private organizations. Nonetheless, national-level regulations provide a useful initial picture of the status of procedural ethics: they set out the minimum standard required by the state, often specifying operating procedures for ethics committees, providing the legal basis for their functioning, and stipulating the guiding principles that those committees implement.
The findings have significant implications for scholars of peace and conflict, and for social scientists more broadly. For the former, of the minority of countries worldwide for which we identified no requirement for ethics regulations, nearly half were found in the world’s most conflict-affected or institutionally fragile states as of 2020 (see Tables 1 and 2). In these contexts, studied populations are characterized by unique vulnerabilities stemming from complex and intersecting crises of livelihoods, health and security. Beyond heightened risk to researchers, their teams and respondents, scholars have reflected that in such contexts, power relations may be particularly uneven, amplifying concerns about research tourism and exploitation (Cronin-Furman and Lake, 2018). For social scientists, where ethics regulations exist, they are almost exclusively concerned with biomedical and clinical research, with no adaptations to account for the ethical conundrums particular to the social sciences (nor conflict research). Moreover, in most countries, central governments maintain an important role in granting ethical approval, making ethical review a potential source of government oversight or censure. REG also finds that some countries monetize ethical review, creating complex and potentially perverse incentives that can make ethical review a bottleneck to scholarly activity. Taken together, the study reaffirms the growing role of procedural ethics for the social sciences and sets the groundwork for a new research agenda on procedural ethics in these fields.
Countries with no evidence of ethical review framework as of 2020.

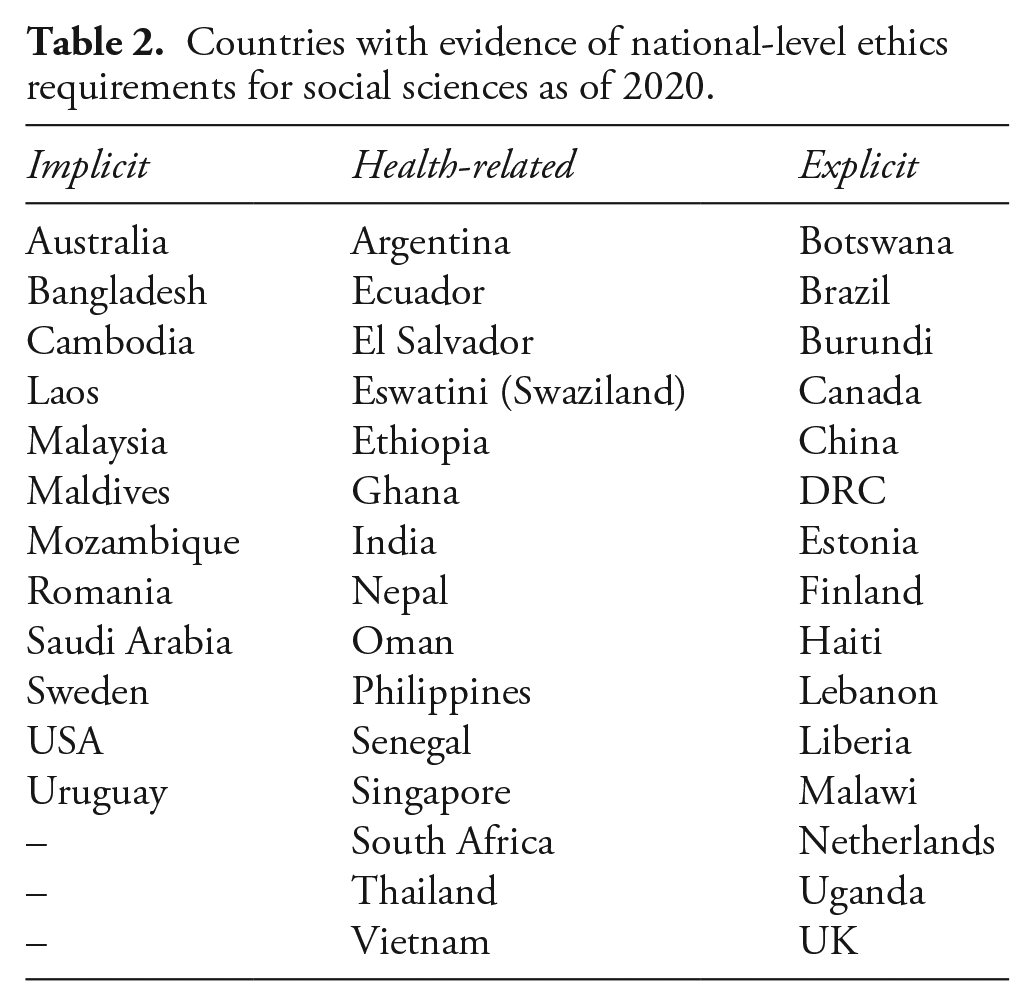
Countries with evidence of national-level ethics requirements for social sciences as of 2020.
In the following sections, we first justify the need for and relevance of REG. Second, we discuss the dataset, its construction and its limitations. We then present findings and key implications, concluding with pathways for future research. Several appendices provide further details on data collection and the construction and operationalization of key variables (Online appendix A), the codebook (Online appendix B), coding decisions for difficult or borderline cases (Online appendix C) and sources used for each country (Online appendix D). We expect the dataset to be of interest to scholars who wish to think critically about research ethics and their regulation, as well as those seeking a practical starting point to understand ethical requirements in diverse field sites.
Why a dataset on research ethics regulations?
The need for this dataset derives from competing observations. First, there is a clear demand among social scientists to raise the bar for ethical research practice. In recent years, scholars have raised ethical concerns for studies ranging from randomized experiments (Desposato, 2015; Humphreys, 2015) to archival research (Subotić, 2021) and even desk research (Hoover Green and Cohen, 2021). Scholars note that ethical concerns are heightened in fragile states (Cronin-Furman and Lake, 2018) and conflict contexts (Malejacq and Mukhopadhyay, 2016; Mazurana et al., 2013).
Second, research ethics regulations – the main formal process for ensuring ethical research practice – have consistently been criticized as poorly suited to identify and address the ethical issues that social scientists face. This is often attributed to their biomedical origins and orientation, assuming a controlled research environment, clear power relations between researcher and subject, and an experimental and positivist approach to research (Bhattacharya, 2014; Michelson, 2016; Yanow and Schwartz-Shea, 2016).
Such concerns are especially pronounced for peace and conflict research, which typically occurs across borders and is characterized by complex and changing dynamics of positionality, power and relationality. For example, securing prior ethical approval sits uncomfortably with the unpredictable nature of fieldwork (Fujii, 2015) and the potential for once-innocuous data to become sensitive, especially in unstable political contexts (Knott, 2019). Often embedded in national-level regulations and institutions, ethical review can create particularly tricky situations in conflict-affected and politically repressive contexts, whether related to manipulation or politicization, the effect of raising the profile of politically vulnerable groups to state authorities, or the potential that respondents will interpret state permission as evidence that researchers are affiliated with or reporting to the state (Fisher, 2021; Glasius et al., 2018). Studying difficult topics may demand confrontation, even if it creates discomfort and upsets research participants (Subotić, 2021). And personal safety sometimes cuts against impartiality, for instance for female researchers working in violent and misogynistic contexts (Krause, 2021).
Beyond conflict research, ethical review has attracted its share of criticism from social scientists; implementing bodies (ethics committees or IRBs) have been shown, repeatedly and in diverse contexts, to protect the interests of the institutions within which they are embedded, for instance prioritizing the university’s reputation and avoiding legal liability (Guillemin et al., 2012: 42; Lincoln and Tierney, 2004). More mundanely, ethical review can impose delays that make research untenable (Fisher, 2021; Mamotte and Wassenaar, 2009; Ralefala et al., 2018). And the common preference for signed informed consent and anonymization of participants can create its own harms, for instance in contexts where visibility rather than anonymity is central to security (Lederach, 2021: 464), or where signing a document is associated with giving away rights rather than gaining protections (Wynn and Israel, 2018).
These two imperatives – a demand to raise ethical standards, alongside widely held views that existing regulations are poorly suited to the needs of social scientists, more so peace and conflict scholars – points to a significant and pressing need to better understand existing ethical regulations. While one might wish to ask ‘What should we do?’, we cannot begin to answer this question without first taking stock of the current state of institutional ethical review around the world.
Until now, we simply lacked the data to do this. Existing databases including ClinRegs, Health Research Web, European Network of Research Ethics Committees (EUREC) and the Research Ethics Guidelines International Online Navigation Map (REGION) emphasize practical information that a northern-based researcher might need to abide by ethics regulations at home or abroad; they are concerned with biomedical research and include only a subset of countries. Scholarship mainly focuses on Global North contexts, especially the USA and the UK (Babb, 2020; Hedgecoe, 2020; Schrag, 2010; Stark, 2011), biomedical research (Krugeret al., 2014; Nwabueze, 2013), ethics-in-practice (Cronin-Furman and Lake, 2018; Fujii, 2012; Malejacq and Mukhopadhyay, 2016; Shesterinina, 2019; Wood, 2006), or country case studies (Mamotte and Wassenaar, 2009; Ralefala et al., 2018; Woldu, 2019). Ours is the first study that offers a comprehensive picture of national-level research ethics regulations and their relevance for social sciences and conflict research.
Data sources, missing data and other limitations
REG is a cross-sectional, global dataset, based on over 800 source documents and coded between January and July 2020. The dataset relies on publicly available information collected via desk-based online searches. We prioritized original legislation and guidelines, as well as policy plans, institutional brochures and government websites. For countries with limited governmental data, we used secondary sources, including newspaper articles, journal articles and reports from international organizations, policy institutions and NGOs. We also used regional and international research ethics databases to identify primary sources and triangulate findings.
REG focuses on national-level regulations for several reasons. First, even while ethical requirements for the social sciences are implemented through contingent constellations of state and non-state regulators, their origins and logic can be traced to states’ adoption of ethics standards for biomedical and clinical research. Second, national regulations provide the legal framework that allows ethics committees to function, often specifying the structure they must take and the principles they should implement. Finally, subnational review processes – e.g. those at universities, hospitals, or ad hoc community panels – are difficult to trace reliably and consistently, especially across contexts. Though our focus on national-level policy offers a partial picture, it has the benefits of setting out the minimum standard by country, offering consistency across cases, and providing a broad picture of the regulatory landscape within which implementing bodies function.
Our variables and coding are based on inductive and grounded analysis. We first conducted an initial round of research that described ethical review processes in several countries. From this, we determined the variables we would collect. After conducting a first round of descriptive coding, we refined our variable selection and transformed descriptive variables into binary variables. Subsequent rounds of coding refined the data to ensure consistency, and to create composite variables such as the degree to which review is centralized.
We note several challenges and limitations. First, as further detailed in Online appendix A, some official sources were not available in the languages spoken by the research team (English, French, Spanish, Portuguese and German). This was especially the case for European, North African and Asian countries. We mitigated this by using secondary sources and automated translation (Google Translate) and supplementing with English-language sources, which allowed us to collect reliable data on almost all countries.
Second, our methodology relies on online evidence of ethical review structures. Some countries, particularly those that have experienced conflict over many years, have less robust Internet presence. Nonetheless, a virtual footprint is a helpful indication of presence, as well as the reach and formality of ethical requirements and processes.
Another challenge was missing or contradictory data. Online appendix C provides a detailed log of boundary cases and justifications for coding decisions. Finally, we note that research ethics regulation is a rapidly developing field; users are advised to validate individual datapoints.
Variables and findings
REG includes 17 original variables describing national-level regulations for research ethics in 197 countries. 1 In this section, we discuss key variables and findings, focusing on two questions. First, we examine the status of ethical review frameworks globally, offering a comprehensive picture of whether and how countries regulate research ethics. Second, we examine if and how these regulations extend to the social sciences.
Ethical review framework
This binary variable describes whether a country has any national-level framework to regulate research ethics. We use a flexible definition of ‘ethical review framework’, including any national-level agency or instrument that stipulates ethical review. While it is possible for such requirements to lapse, we did not identify any cases where ethical review institutions or rules were dissolved after adoption. Three countries (Georgia, Nicaragua, the Solomon Islands) are coded as missing due to conflicting evidence such that we were unable to conclude whether any structure, policy or regulation exists.
As of 2020, we were unable to find evidence of national-level requirements for ethical review in 23 countries (12%). 2 These are mainly conflict-affected, politically closed, or small (island) nations.
Regions with the least ethical regulation include Oceania and the Caribbean (where we found no evidence of ethical review requirements in 43% and 27% of countries, respectively). Following are the Middle East and North Africa (20%), Africa (10%) and Asia (7%).
Year of establishment (in general and for social sciences)
REG codes the year when a country’s central government first adopted any requirement or process to regulate research ethics, as well as the year these were extended to the social sciences, if applicable (Figure 1).
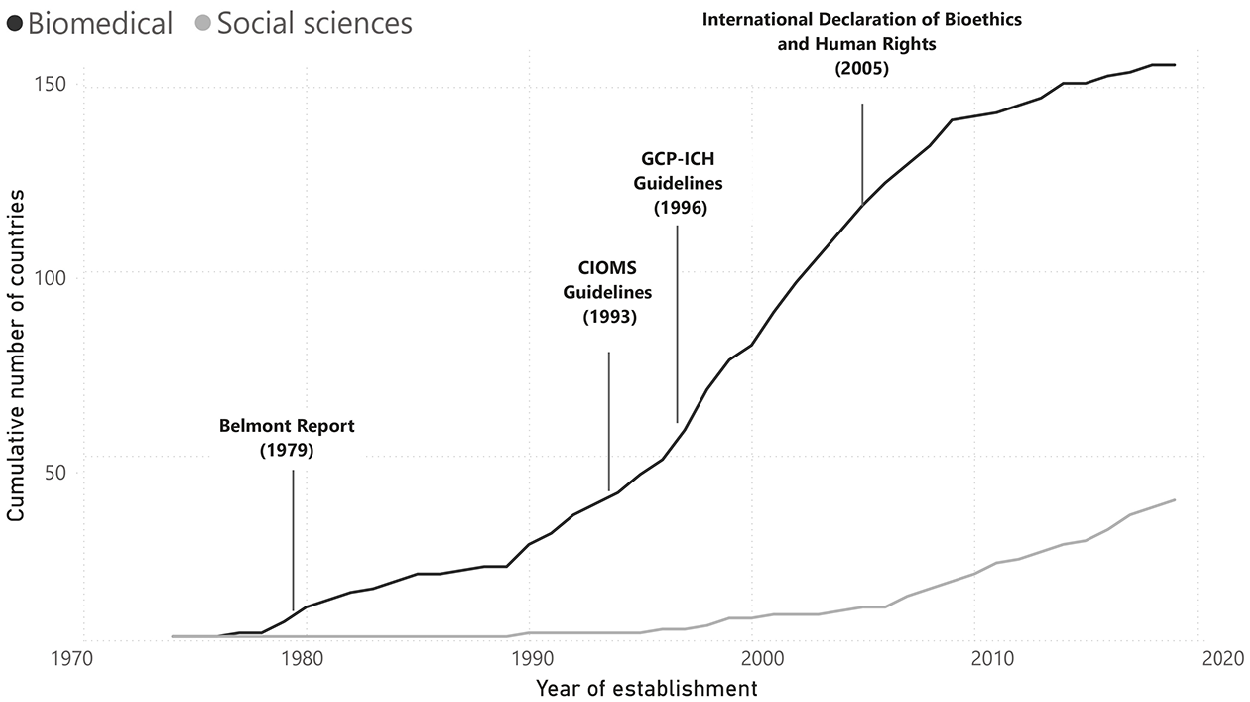
The global spread of national-level ethics regulations.
The requirement for prior ethical review by committee was formally established in the United States in 1974 with the Belmont Report, following the now well-known biomedical ethical scandals in the 1960s–70s in which scientists experimented on people without their knowledge or consent (see e.g. Schrag, 2010; Stark, 2011; see e.g. Babb, 2020). Since then, we observe a gradual increase in the number of countries with national-level requirements that follow this model. Increasingly, these rules are being extended to the social sciences. For example, Uruguay adopted its first regulations in 1998, which were only applicable to clinical trials; in 2019, these were extended to apply to all research. The adoption of ethics regulations accompanies key international conventions and guidelines on research ethics. These are focused on biomedical and clinical research; none speak in depth about the social sciences or ethical issues particular to them.
Ethical review of the social sciences
The variable ‘social science review’ specifies requirements for ethical review of social science research. We coded this variable generously, seeking to capture any requirement to regulate social sciences specified in national-level guidance, including references to methods employed by social scientists (e.g. surveys or interviews), or any social science discipline or area of study (e.g. cultural or behavioural research) (see Online appendix A for details). We find that national-level regulations focus predominantly on biomedical research and clinical trials: only 21% (or 42 countries) extend regulation to the social sciences. However, for 12 countries that obligation is implicit (applying to all human subject research); for another 15 these obligations extend only to health-related social research. We found only 15 countries (8%) worldwide that explicitly require the ethical regulation of social sciences research.
To understand if ethical review is tailored to the social sciences and how regulations are practically extended to them, we developed additional variables documenting whether the country has any guidelines or institutions specific to social science research. We might anticipate that if regulators saw the social sciences as an important area for regulation, they would develop guidance to deal with its particularities. However, such tailoring is unusual. Instead, countries that regulate social sciences typically use institutions and guidelines designed for biomedical or clinical research. For example, 31% of all countries that extend ethical review to the social sciences (13 out of 42) provide guidelines that explicitly reference social sciences. 3 The rest either use the same guidelines as for biomedical sciences (29% or 12 countries) or do not provide guidance at all (40% or 17 countries). We identified only six countries worldwide that have social science-specific institutions at a national level. 4 These statistics show that, in the main, countries do not treat the social sciences as a distinct object of regulation, suggesting they are not concerned with them, or perhaps are not aware or convinced of their particularities.
We combined the variables described above into a scalar variable to capture the comprehensiveness of ethics regulations in the social sciences. While the way in which ethics regulations are formally extended to the social sciences is not a good proxy for the rigour of review, combining these variables provides a clearer picture. At the lowest level we include countries that only regulate the social sciences implicitly; at the highest level, we include those that explicitly regulate the social sciences, and also have guidelines or institutions tailored to the social sciences (22%). 5 This variable presented several boundary cases, detailed in Online appendix C (for instance, while the USA and Australia implicitly regulate the social sciences, the comprehensiveness of their guidelines led us to include them in the highest level).
Centralization of ethical review
REG also describes the structure of ethical review, focusing on the extent to which it is centralized or decentralized (on paper). In centralized systems, the national level fully controls ethical review; in decentralized states, the national level has virtually no formal control, and review is instead conducted at subnational institutions such as hospitals and universities, private organizations, or subnational government agencies (e.g. in federal systems).
Globally, ethics review structures are more often centralized than decentralized. Nearly 70% of countries for which we have data have quasi-centralized or centralized structures (48% and 22% respectively). In comparison, only 30% of countries have mixed (11%), quasi-decentralized (14%), or decentralized (5%) structures. Purely decentralized structures are rare, mainly including the federal systems of Germany and Canada, where ethical review competences are delegated to states (Länder and regions respectively). Other cases include countries where a national ethical review process is in place, but it is not directly linked to the government. Quasi-centralized and centralized structures are especially popular in Africa and the Caribbean, where they account for 92% and 88% of countries respectively.
In many countries, decentralized agencies are themselves beholden to regional or national regulation. For instance, the United States has a ‘quasi-decentralized’ structure, in which IRBs are established subnationally (i.e. by research centres, private businesses, universities, hospitals) and are subject to both state and federal regulations – the former being legally mandated and the latter being a condition to receive federal funding. Sarah Babb (2020) describes this as a product of the US ‘workaround state’, where federal responsibilities are outsourced to subnational actors. In other countries, the central government retains greater authority. For instance, Finland has a ‘quasi-centralized’ structure, where regional RECs conduct review and are overseen by a central REC that decides on appeals (Figure 2).
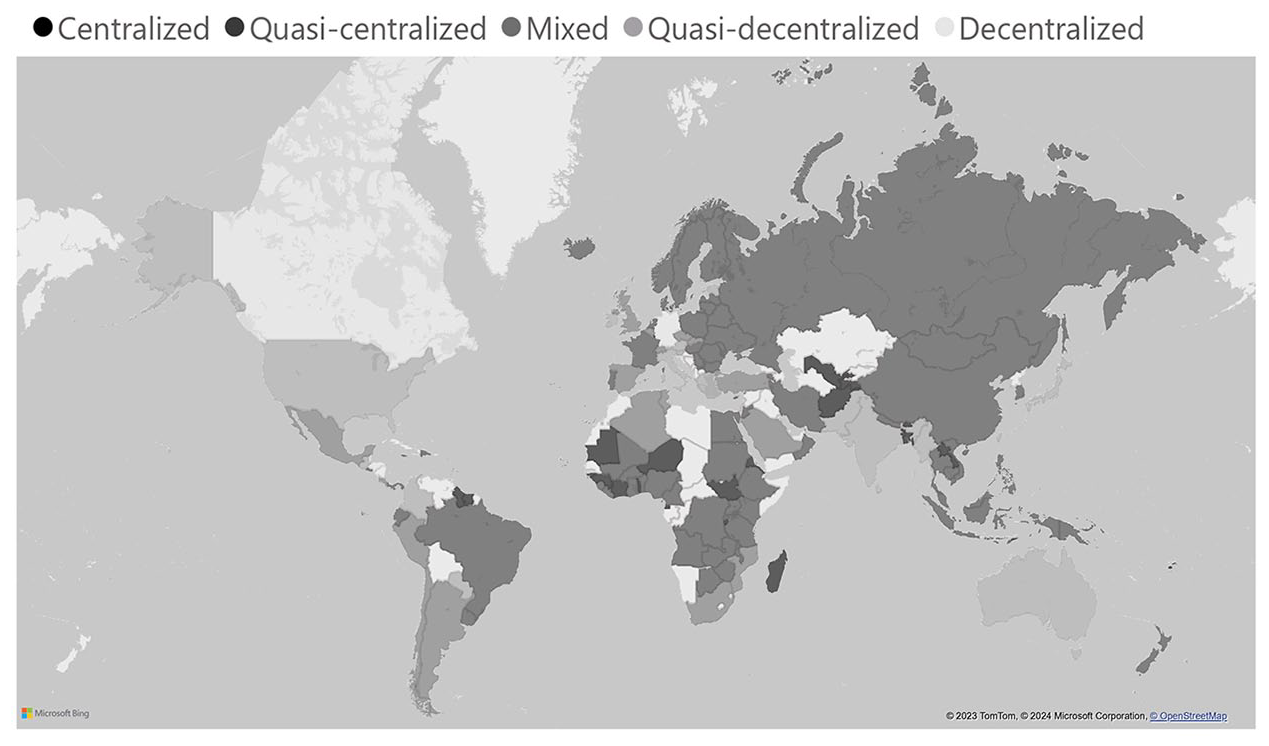
Structure of ethical review globally.
This extent of centralization is counter-intuitive at first glance – why would governments take on the responsibility and cost of providing ethical review, rather than outsourcing this task? The overwhelming popularity of centralized and quasi-centralized structures suggests that at least some governments view research ethics as an area worthy of control and direct oversight. Ethics regulation may be an avenue to broadly influence scientific inquiry, or keep tabs on ‘delicate and often sensitive’ research, including on sexual behaviour or governance (see e.g. Rwabukwali, 2007: 117). Ethical review may also constitute an important checkbox to attract foreign research, as suggested by scholars who link the adoption of RECs to the internationalization of clinical trials (see e.g. Jacobs and Tinnerholm Ljungberg, 2021). More benignly, in countries with centralized governance structures, ethical review may be integrated into existing governance structures, and thus be centralized by default – though we note this can still have knock-on effects of remaining under the purview of the central state. These hypotheses need not contradict, but instead could be reinforcing; they likely are borne out different depending on country context.
Fees and foreigner-specific rules
To probe whether the adoption of ethics review requirements is linked to research internationalization, we included dichotomous variables that describe if there are rules specific to foreign researchers and whether ethics committees can charge fees for ethical review. For countries with data, we find that 22% of countries (24 countries) have rules for foreign researchers. These generally refer to research led by foreigners, but this can be ambiguous. For instance, in Brazil, rules apply to research ‘coordinated from abroad’ or ‘with foreign participation’. However, research is not considered foreign if the researcher is affiliated with a Brazilian institution, or if the research is conducted by a multinational company with headquarters in Brazil. In Indonesia, any research conducted by foreign universities, foreign ‘research and development institutes’ or foreign individuals is considered ‘foreign’.
All countries that specify rules for foreigners are in the Global South, mainly in Africa (10), Oceania (8) and Asia (4). Substantively, most of these rules set higher fees for review, e.g. El Salvador, Ghana, Kenya, Laos and Nepal. Others include requirements for special approvals or the obligation to submit foreign research proposals to a specific authority for ethical review. For example, in Brazil, foreign research must undergo additional scrutiny by the central ethics committee (CONEP) after going through regular ethical review. In Indonesia, foreign research must receive special approval and a research permit.
The variable ‘fees’ indicates whether it is permissible to charge for review. We find that though some Global North countries explicitly allow fees for ethical review, it is most common in Asia and Africa, pointing to the monetization of ethical review and perhaps (foreign-funded and conducted) research more broadly (Figure 3).
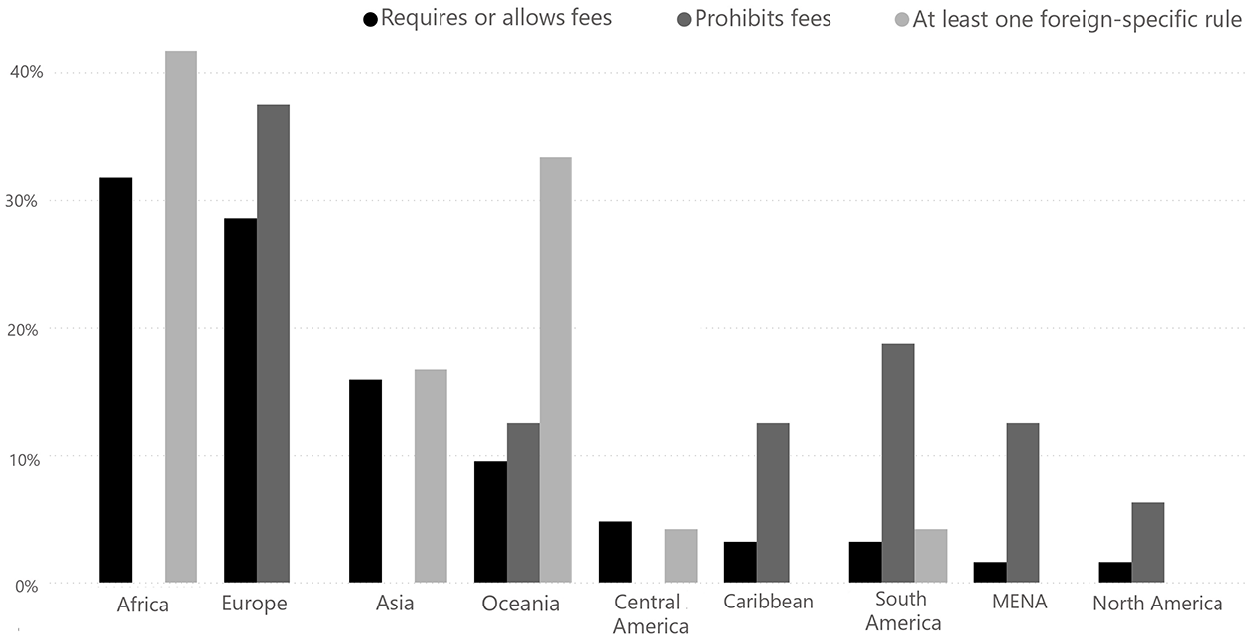
Fees and foreigner-specific rules.
This analysis suggests a political economy of human subject research, wherein lower-income countries may attract research due to real or perceived lower barriers (whether legal or ethical) to conducting research. This has been well elaborated for medical research and clinical trials (Nwabueze, 2013), and social scientists have made similar speculations (Cronin-Furman and Lake, 2018; Desai and Tapscott, 2015; Teele, 2014). Regional variation suggests countries may seek to use research ethics regulation for various purposes depending on both domestic and international considerations.
Application to conflict-affected and institutionally fragile contexts
Combining REG with data on conflict affectedness and institutional fragility reveals notable variation, though the small-N and large degree of missingness mandates a cautious interpretation. Of the few countries with no evidence of ethics regulations, 48% were conflict-affected or institutionally fragile per the World Bank’s 2020 classifications (see Tables 3 and 4; World Bank, 2023: 4). In these countries, researchers may follow regulations of other nations (i.e. if they are based in a country with ethical review requirements for research conducted abroad, such as the USA or UK), and/or they may adopt ad hoc approaches to procedural ethics, perhaps foregoing local review or voluntarily identifying a non-state option. While national structures of ethical review may not be desirable regulatory solutions, it may nonetheless be concerning that procedural ethics is at its most fragmented, foreign and inconsistent in the world’s most conflict-affected and institutionally fragile contexts.
High- and medium-intensity conflict (World Bank 2020 classifications).

High institutional and social fragility (World Bank 2020 classifications).
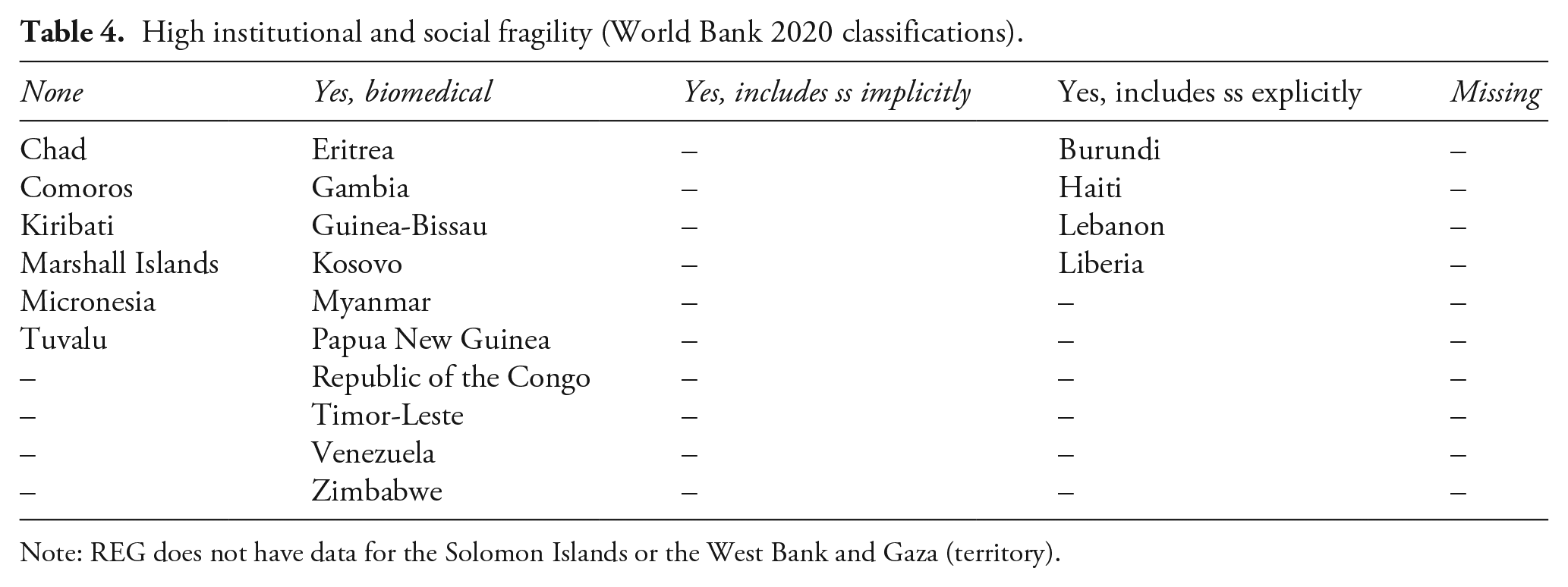
Note: REG does not have data for the Solomon Islands or the West Bank and Gaza (territory).
REG also shows that conflict-affected and fragile countries are especially likely to be centralized, with 88% having either centralized or quasi-centralized systems, compared to 67% of countries not classed as conflict-affected or fragile. Where we have data, conflict-affected countries allow for charging fees across the board (100%, or eight countries).
Discussion and outlook
Conflict scholars have demonstrated a growing concern with ethics in practice, increasingly shared by political scientists and scholars of international relations. But how should we, as a scholarly community, relate these concerns to questions of procedural ethics? Thus far, these questions have largely been left to university administrators, journal editors and funding agencies, whose interests and norms arguably are not consistently aligned with those of researchers, research subjects, nor sometimes even the various publics whom research is meant to benefit. Moreover, REG demonstrates that, globally speaking, ethical regulations are expanding their remit, presenting a new and pressing demand for disciplinary perspectives on how to engage with procedural ethics in our professional practice.
Requirements for ethical review are now almost universal, present in at least 87% of countries. However, even while scholars may view ethical review as an important step in the research process, our data affirms that countries worldwide have adopted versions of a regulatory structure designed for biomedical and clinical research, rather than the types of ethical considerations raised by social scientists and conflict researchers. REG also highlights the prominent role of national governments in ethical regulation, with central governments maintaining significant influence in nearly 70% of countries. Paired with research that finds that ethics committees tend to produce ‘site-specific prejudices as a matter of organizational design’ (Stark, 2011: 165), the findings detail an institutional design prone to conflicts of interest, especially for politically contentious or sensitive studies. Finally, REG highlights patterns in the monetization of ethical review. Charging fees may be justifiable in under-resourced institutions, where income supports the work of committee members and ethics administrators. However, in instances where ethical review is both costly and required, it can create undue obstacles for unfunded (often domestic) scholars. These patterns appear to be accentuated in conflict-affected and institutionally fragile settings, with disproportionately centralized structures that when they mandate the regulation of social sciences, do so explicitly and without specialized guidance.
REG has both applied and academic relevance. It may be a useful starting point and instructional tool to better understand ethics requirements in countries worldwide, including Online appendix D, which lists resources on each country. A future initiative could continue to update this resource, as ethics regulation is a quickly developing area. From a scholarly standpoint, the dataset complements scholarship on ethics-in-practice, opening a new research agenda on the regulation of ethics for social scientists and conflict researchers, which to this point has been mainly the purview of biomedical scholars (and to a lesser extent, sociologists and lawyers).
The dataset points to future research that can deepen and nuance our understanding of procedural ethics from a global standpoint – their form, substance and implementing bodies – as well as if and how this impacts social science and conflict research in practice. For example, additional data could help flesh out understanding of the regulatory environment, including which ministries grant ethical approval; if national ethical review systems are open to foreigners, whether there are additional requirements for local collaboration or research visas, and if review requirements are linked to particular funders; as well as data on the types of studies reviewed in practice (e.g. discipline, funding source, or method). Conflict scholars may also wish to explore how procedural ethics is treated in contexts without formal requirements for such, and whether or not it would be desirable to develop norms around this. REG also highlights the need for further investigation of how ethics committees interpret and apply regulations, as well as how social science scholars navigate them, especially outside of Global North settings. Such studies can elucidate whether current (often biomedical) models restrict the quality of social sciences, and the unanticipated consequences of fortifying such requirements – for example, on Global South or unfunded research, and on knowledge production.
Footnotes
Acknowledgements
The authors thank colleagues for constructive feedback on various iterations of this article: Sarah Babb, Susanna Campbell, Deval Desai, Ben Martill, Nicholas Rush Smith, Peregrine Schwartz-Shea and Shaina Western. We also thank Hoda Khaled Salah Awwad for research assistance.
Data replication
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research was funded by the Swiss National Science Foundation PZ00P1_185971 and is a product of the Governing Research Ethics project.
Notes
REBECCA TAPSCOTT, PhD in International Relations (The Fletcher School, Tufts University, 2017); Lecturer, University of York; book: Arbitrary States: Social Control and Modern Authoritarianism in Museveni’s Uganda (Oxford University Press, 2021).
DANIEL RINCÓN MACHÓN, MA in Development Studies (Geneva Graduate Institute 2021); PhD Candidate, Development Studies, University of Cambridge.
