Abstract
Peroxisomes in the human hepatoblastoma cell line HepG2 exhibit a high degree of plasticity. Whereas in confluent cultures they appear as small (0.1-0.3 μm) spherical particles, they undergo dramatic changes, forming elongated tubules measuring up to 5 μm on separation of cells and cultivation at low density. We recently showed that several growth factors, including nerve growth factor (NGF), induce the formation of tubular peroxisomes and that this induction is sensitive to K 252b, a specific tyrosine kinase inhibitor, suggesting the involvement of this signal transduction pathway. Because tyrosine kinase is also involved in signal transduction via the reactive oxygen species (ROS), we have analyzed in this study the effects of UV irradiation, H2O2, and oxygen on tubulation of peroxisomes. UVC irradiation induced a significant increase in formation of tubular peroxisomes (40-50% of cells) and this effect was dose-dependently inhibited by pretreatment with N-acetyl cysteine, confirming the involvement of ROS in the UV effect. Furthermore, H2O2 also directly induced the tubulation of peroxisomes, although to a lesser extent. Finally, cultivation under hypoxic conditions (1.5% O2) drastically reduced the inducing effect of fetal calf serum on tubulation of peroxisomes, suggesting the involvement of oxygen-mediated signaling. Taken together, our observations indicate that ROS induce the tubulation of peroxisomes in HepG2 cells. Because peroxisomes harbor most of the enzymes for catabolism of ROS, the tubulation and expansion of the peroxisome compartment could have a cell rescue function against the destructive effects of ROS.
Keywords
T
Peroxisomes in confluent cultures of human-derived HepG2 cells are typically small spherical particles (0.2-0.3 μm) resembling those observed in a variety of other cell types in culture (Schrader et al. 1994). Dramatic alterations of peroxisomal morphology, as evidenced by the formation of elongated tubular forms measuring up to several μm, were noted recently (Schrader et al. 1996). Moreover, we found that the formation of tubular peroxisomes was cell density-dependent and was induced in serum-free cultures by specific growth factors (e.g., nerve growth factor, NGF) and by polyunsaturated fatty acids (PUFA), particularly by arachidonic acid, suggesting the involvement of intracellular signaling (Schrader et al. 1998). Because tubular peroxisomes are transient structures that divide by fission, giving rise to several spherical particles, their formation is interpreted as an early sign of rapid expansion of the peroxisomal compartment associated with cell growth and differentiation (Schrader et al. 1998).
Ultraviolet irradiation of cells elicits a complex cellular response comprising the induction of expression of several genes, including those of growth factors (Fornace 1992; Huang and Adamson 1995). In addition, it induces the formation of ROS, such as hydrogen peroxide, superoxide radical, and the singlet oxygen (Tyrrell 1991). Because both the growth factors and ROS have a significant impact on the peroxisomal compartment, we have investigated the effect of UV irradiation on peroxisomes of HepG2 cells. The results indicate that UV exposure and, more directly, hydrogen peroxide induce the formation of tubular peroxisomes. Moreover, the induction of tubular peroxisomes by fetal calf serum also appears to be dependent on oxygen, suggesting the participation of oxygen radicals as intracellular second messengers (Finkel 1998) in this process.
Materials and Methods
Cell Culture
HepG2 cells (Aden et al. 1979; Knowles et al. 1980) were obtained from the American Type Culture Collection (ATCC; Rockville, MD) and were routinely cultured in minimal Eagle's medium (α-MEM) containing 2 g/liter sodium bicarbonate, 2 mM glutamine, 100 U/ml penicillin, 100 μg/ml streptomycin, and 10% fetal calf serum (FCS) (all from Biochrom KG; Berlin, Germany) at 37C in a humidified atmosphere of 5% CO2 and 95% air (normoxic conditions). For experiments, the cells were washed with serum-free DMEM/N1 [Dulbecco's modified Eagle's medium (DMEM; Gibco, Grand Island, NY); N1 = 0.25% BSA; 6.25 × 10−8 M transferrin; 8.3 × 10−7 M insulin; 3 × 10−8 M selenium; 2 × 10−8 M progesteron; 1 × 10−4 M putrescine (all from Sigma Immunochemicals; Munich, Germany); 50 μg/ml penicillin; 50 μg/ml streptomycin; 100 μg/ml neomycin; according to Bottenstein et al. (1980)] and detached by incubating with a trypsin/EDTA solution [0.125%/0.05% (w/v); Biochrom KG] in PBS for 5 min at 37C. The cells were harvested with DMEM/N1 containing 1 mg/ml soybean trypsin inhibitor (Serva; Heidelberg, Germany) and centrifuged at 70 × g for 5 min (Beckman TJ-6). The cell pellet was then resuspended in DMEM/N1. Cells were plated at a defined density of 2 × 105 cells/ml on glass coverslips in 35-mm culture dishes (Greiner; Frickenhausen, Germany). The cells were routinely prepared for immunofluorescence after 24 hr when the tubular peroxisomes reached their maximum (Schrader et al. 1996, 1998).
Indirect Immunofluorescence
HepG2 cells were processed as described (Schrader et al. 1994, 1995). Briefly, cells were fixed for 20 min with 4% paraformaldehyde in PBS, pH 7.4, and treated for 10 min each with 0.2% Triton X-100 and 1% glycine in PBS. Then they were incubated for 1 hr with the primary antibody [polyclonal rabbit anti-rat acyl-CoA oxidase, as characterized previously (Beier et al. 1988)]. The immune complexes were detected with dichlorotriazinylaminofluorescein (DTAF)-conjugated goat anti-rabbit IgG (Dianova; Hamburg, Germany). Samples were examined in a Zeiss Axiophot and photographed on Kodak TMY film. For quantitative evaluations, 100 cells per coverslip were examined and categorized as cells with either tubular, spherical, or rod-shaped peroxisomes, as described recently (Schrader et al. 1998). The data were analyzed statistically in comparison with the appropriate controls using Student's t-test.
UV Irradiation
Four hours after the cells were sown, the DMEM/N1 medium was removed and saved (to be added again to the same cultures after UV exposure) and the dishes (with lids removed) were exposed to 40 J/m2 of UVC (∼2 sec) in a UV-Crosslinker, Model 2400 (Stratagene; San Diego, CA) according to Huang and Adamson (1995). The energy of the UV light (using 254-nm bulbs) was precisely controlled by the crosslinker. After exposure, the cells were grown in the same medium as before UV irradiation for 20 hr. For control cultures, the same procedure was followed but UV treatment was omitted. Cells were then processed for immunofluorescence for detection of peroxisomes.
Treatment with NAC, OAG, BIM, and Hydrogen Peroxide
One hour before UV irradiation the cells were pretreated with the antioxidant N-acetyl-cysteine (NAC). NAC (Sigma) was dissolved in DMEM/N1 immediately before use. Some cultures were also treated with 1-oleolyl-2-acetyl-sn-glycerol (OAG; 0.2-10 μm), an activator of PKC, or with bisindolylmaleimide I (BIM; 0.01-1 μm), a PKC inhibitor, 1 hr before UV irradiation. Hydrogen peroxide was obtained as a 30% solution (Sigma) and diluted to 200 or 400 μM (final concentration in the culture medium) immediately before use.
Hypoxia/Hyperoxia
For exposure to hypoxia (hyperoxia), HepG2 cells grown to confluence under normoxic conditions (95% air) were seeded in DMEM/N1 or DMEM/N1/10%FCS and immediately transferred into a thermostatized (37C) acrylic plastic chamber (volume about 8 liters) that was flushed with sterile-filtered (filter size 0.22 μm) humidified gas composed of various concentrations of O2 (1.5%, 3%, 40%), 5% CO2, and N2 for 24 hr. After an initial flushing with the appropriate gas at a higher rate for 5 min, the gas flow was kept constant at a rate of about 0.2 liter/min during the incubation. The PO2 and PCO2 in the atmosphere of the acrylic plastic chamber and in the culture medium supernatant was checked with a blood gas analyzer (Model 278; Corning, Corning, NY). After 24 hr the cells and appropriate normoxic controls were fixed and processed for immunofluorescence.
Results
UV Irradiation Induces Tubular Peroxisomes via ROS
As shown previously (Schrader et al. 1996), in the presence of FCS elongated tubular peroxisomes can be induced in HepG2 cells in a cell density-dependent manner (Figure 3A). Under routine culture conditions (MEM/10% FCS; 2 × 105 cells/ml), approximately 40-50% of the cells contain tubular peroxisomes after 24 hr (Figure 2). The formation of tubular peroxisomes requires specific serum factors and/or fatty acids (Schrader et al. 1998) and is almost completely inhibited under serum-free (DMEM/N1) culture conditions (Figures 1A and 2).
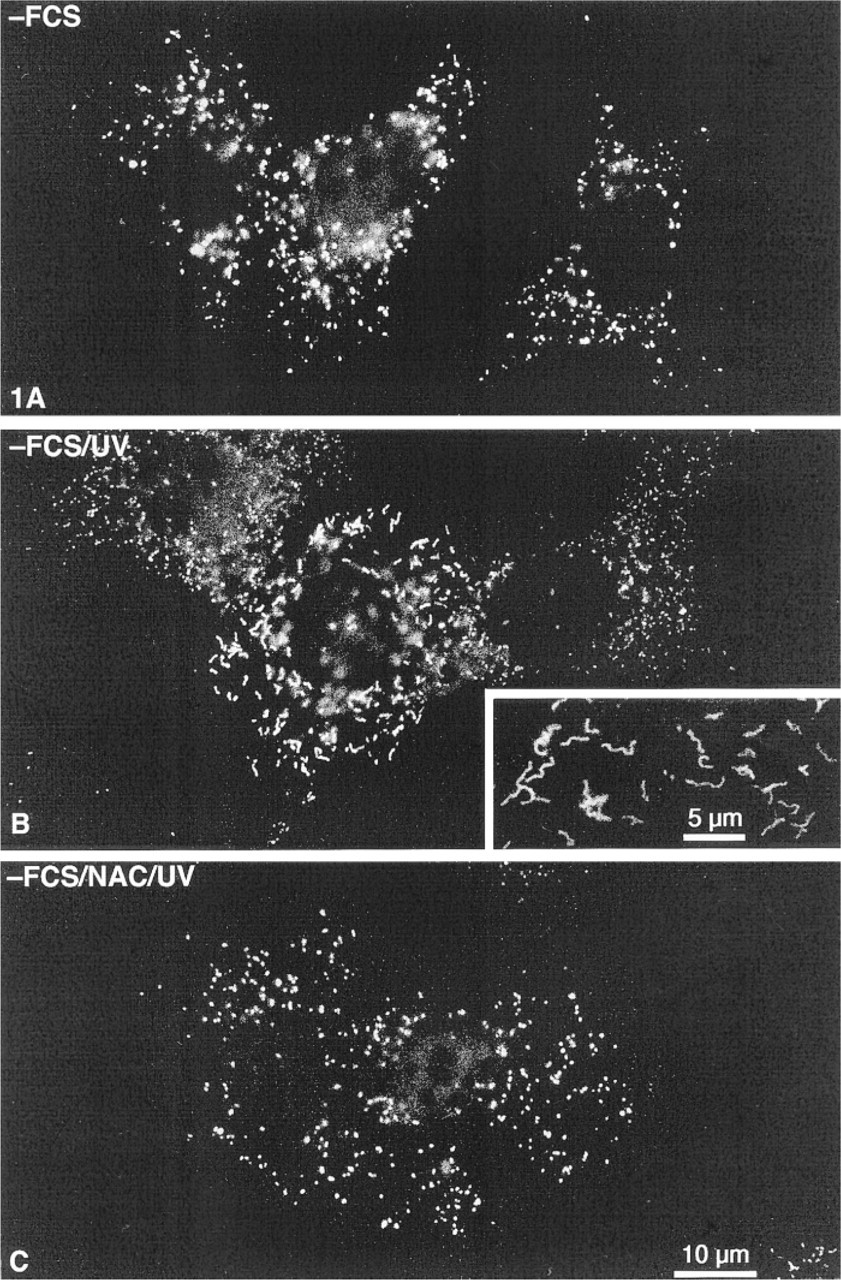
Immunofluorescence with an antibody to peroxisomal acyl-CoA oxidase. HepG2 cells were cultured in serum-free DMEM/N1 medium and treated as indicated. (
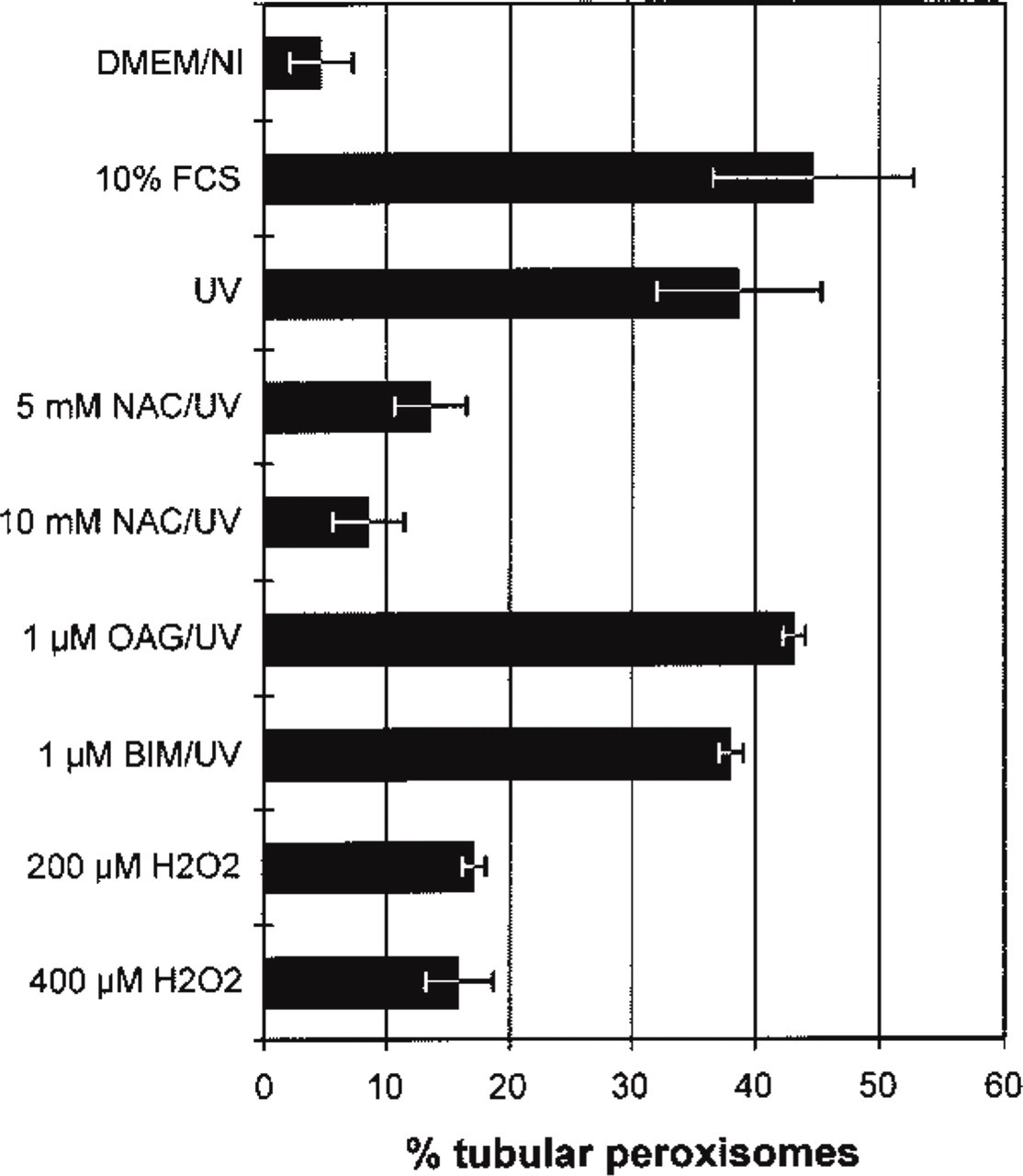
Quantitative analysis of cells with tubular peroxisomes. HepG2 cells were seeded in serum-free (DMEM/N1) medium, pretreated as indicated and then exposed to UV light and processed by immunofluorescence for detection of peroxisomes. The percentage of cells with tubular peroxisomes was calculated. Whereas in the serum-free medium only 5% of cells contain tubular peroxisomes, more than 45% contain such structures in the presence of fetal calf serum (FCS). Exposure to UV in the absence of serum induces formation of tubular peroxisomes in almost 40% of cells (UV). Pretreatment with the antioxidant N-acetyl cysteine (NAC) blocks the stimulatory effect of UV irradiation on the formation of tubular peroxisomes in a dose-dependent manner. The PKC-activator 1-oleolyl-2-acetyl-sn-glycerol (OAG) and the PKC inhibitor bisindolylmaleimide I (BIM) showed little effect on the UV-induced formation of tubular peroxisomes. Hydrogen peroxide (H2O2) also induced, to a lesser extent, the formation of tubular peroxisomes.
To determine whether dynamic changes of the peroxisomal compartment and the formation of elongated tubular peroxisomes can also be induced by oxidative stress, HepG2 cells grown at low density under serum-free conditions were exposed to a 40 J/m2 dose of UVC radiation and analyzed by immunofluorescence 24 hr after UV treatment. Control cells were cultured either in serum-free DMEM/N1 medium or in the presence of 10% FCS. As shown in Figure 2, in response to UV irradiation tubular peroxisomes were induced in the absence of FCS to a level comparable to that of 10% FCS (Figures 1B and 2). To test whether ROS are involved in the initiation of UV-induced tubular organelles, we applied NAC, which can be rapidly converted into reduced glutathione (GSH) after uptake by cells, thus serving as an intracellular reducing agent (Meister 1991). As shown in Figures 1C and 2, the induction of tubular peroxisomes by UV was blocked by preincubation with NAC in a dose-dependent manner. NAC alone, however, had no effect on peroxisomes (not shown). To test further the effects of other ROS, cells were treated with H2O2. Tubular peroxisomes could also be induced by H2O2, but to a lesser extent and not in a dose-dependent manner. Furthermore, the induction of tubular peroxisomes by UV irradiation appeared to be independent of PKC. Neither treatment with OAG, a potent activator of PKC, nor with BIM, an inhibitor of PKC, showed significant effects on the formation of tubular peroxisomes after UV irradiation. It should be mentioned that the tubular structure of mitochondria, as revealed in immunofluorescence with an antibody to Mn-SOD, was not altered under the same serum-free culture conditions or by UV irradiation (not shown).
Formation of Tubular Peroxisomes in HepG2 Cells Depends on Oxygen Partial Pressure
In another set of experiments, HepG2 cells were seeded in serum-free DMEM/N1 or in DMEM/N1/10% FCS and cultured for 24 hr under normoxic (controls), hypoxic (1.5%, 3% O2), or hyperoxic (40% O2) conditions (Figures 3 and 4). Forty percent of HepG2 cells grown in the presence of 10% FCS in normoxia showed elongated tubular peroxisomes extending over several μm (Figure 3A), whereas hypoxia inhibited their formation in a dose-dependent manner (Figure 4). Almost all peroxisomes under hypoxic conditions were spherical, although FCS was present in the medium (Figure 3B). Furthermore, under hypoxia the cells appeared to be enlarged and more flattened. In contrast, hyperoxia (40% O2) had no significant effect on the formation of tubular peroxisomes. Hyperoxia was neither stimulatory on the formation of tubular peroxisomes in cells grown under serum-free conditions nor inhibitory on cells grown in the presence of 10% FCS (Figure 4). Similar to the UV effect, the tubular structure of mitochondria was not altered with changing oxygen concentrations (not shown).
Discussion
Formation of Tubular Peroxisomes Is Mediated by the Tyrosine Kinase Signal Transduction Pathway
This study has shown that UV irradiation of HepG2 cells under serum-free culture conditions induces the formation of tubular peroxisomes in 40-50% of cells. Therefore, the inducing effect of UV appears to be comparable to the combined effects of various proteins and lipids present in FCS (Schrader et al. 1998). We recently found that tubular peroxisomes could be induced by supplementation of serum-free culture media with specific growth factors and polyunsaturated fatty acids (PUFA), both of which are involved in intracellular signaling (Schrader et al. 1998). Nerve growth factor (NGF) was the most potent among those tested, and its effect appeared to be mediated by tyrosine kinase because an inhibitor of this signal transduction pathway, i.e., K 252b, blocked the NGF effect completely. That observation has great relevance for the present study because tyrosine kinase activity has also been implicated in cellular responses to ROS (for review see Finkel 1998). Extracellular administration of H2O2 activates the mitogen-activated protein kinase (MAPK) as well as the c-Jun amino-terminal kinase (Stevenson et al. 1994; Sundaresan et al. 1995). Finkel (1998) has suggested that the burst-like activation of tyrosine kinase by ROS could be due to transient inactivation of protein phosphatases, most of which contain a functionally important cysteine residue in their active sites. Activation of kinase is inhibited by pretreatment with N-acetyl cysteine (NAC) (Lo et al. 1996; Cui and Douglas 1997). This antioxidant also elicited an inhibitory effect on the UV-induced formation of tubular peroxisomes, thus supporting the role of ROS and tyrosine kinase in this induction process.
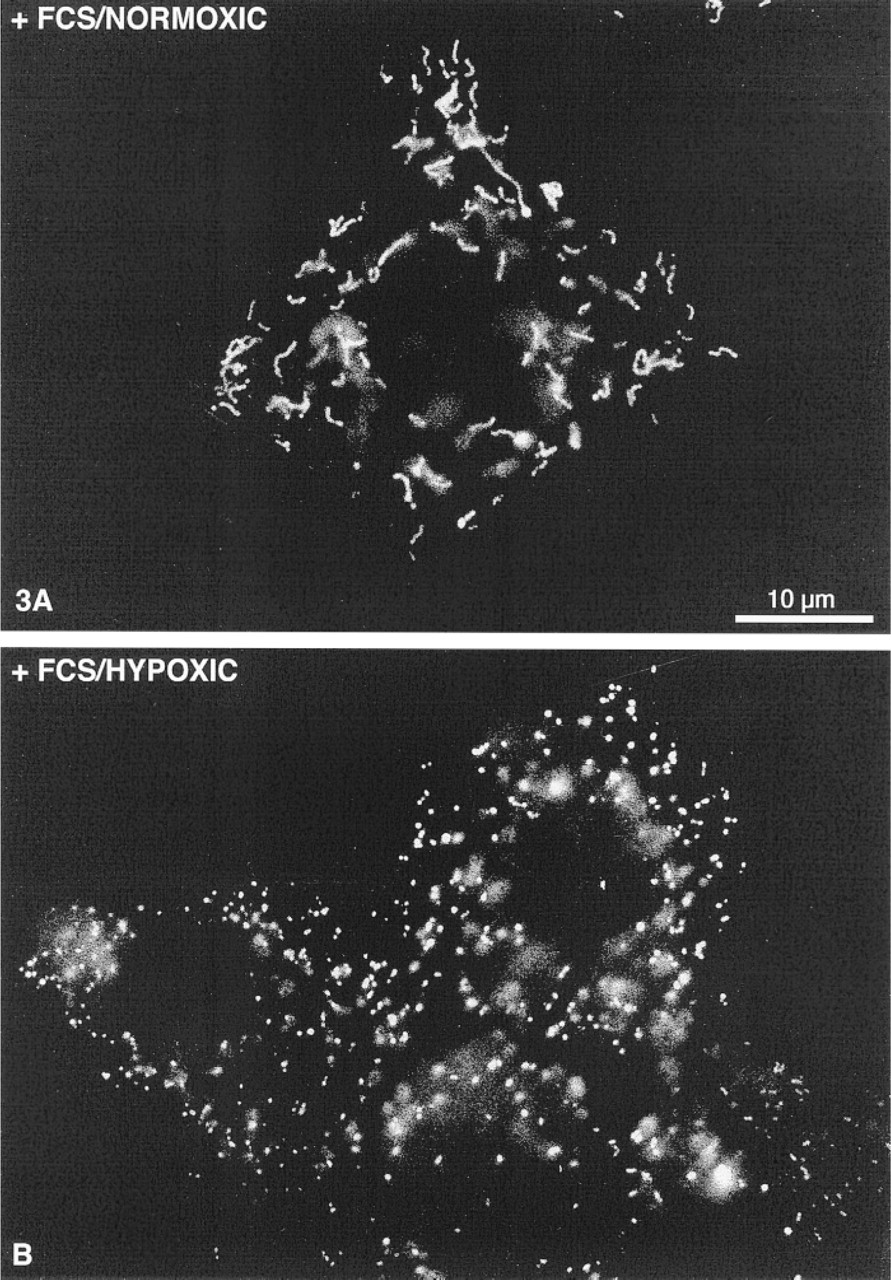
HepG2 cells stained as indicated in Figure 1. (
On the other hand, our previous studies with activators and inhibitors of protein kinase C (PKC) showed no effects on tubulation of peroxisomes (Schrader et al. 1998). The observations with OAG and BIM in this study confirm that the UV induction of tubular peroxisomes also does not operate through the PKC.
UV irradiation stimulates the upregulation of a wide range of gene products (Fornace 1992; RadlerPohl et al. 1993; Huang and Adamson 1995). Prominent among them are the transcription factors c-Fos, all members of the Jun family, p53, c-Myc, and NFκKB. Other activated genes include those for growth factors and cytokines, such as bFGF, αINF, IL-1, TNFα, and signal intermediaries, such as PKC, c-Ras, and many others (reviewed by Fornace 1992). The precise mechanisms for the generation of a signal from the extracellular incidence of UV and the gene response remain largely unknown. Tyrosine kinases such as c-Src have been implicated (Devary et al. 1992; Sachsenmaier et al. 1994a,b; Schieven et al. 1994; Coffer et al. 1995), and several groups have noted that the growth factor signal pathway has elements and components in common with the irradiation pathway (Datta et al. 1992). Recently, Huang et al. (1996) have shown that ROS as second messengers activate growth factor receptors in the UV response.
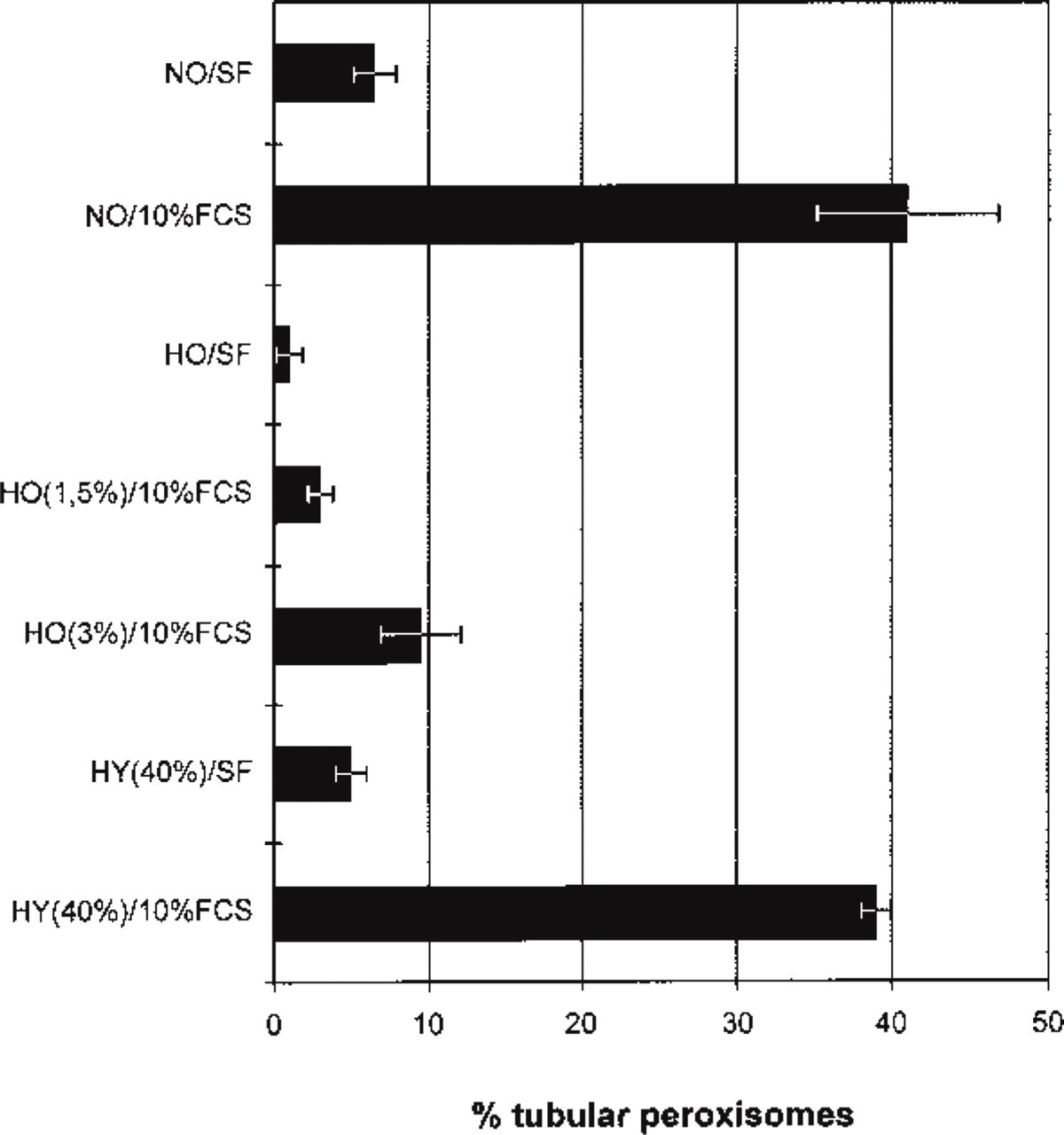
Quantitative analysis of the effects of hypoxia (HO) and hyperoxia (HY) on the formation of tubular peroxisomes in HepG2 cells cultured in serum-free DMEM/N1 medium (SF) or in the presence of 10% FCS. Whereas under normoxic (NO) conditions the formation of tubular peroxisomes is induced by the addition of serum (NO/10% FCS), this response is completely blocked under hypoxic conditions (HO) in a dose-dependent manner. Hyperoxia (40% O2) does not appear to have a significant effect on the induction of tubular peroxisomes.
Formation of Tubular Peroxisomes Requires Oxygen
An important observation of the present study was the demonstration of oxygen-dependence of the induction of tubular peroxisomes by FCS (Figure 4). This suggests that the peroxisomal response to various (growth) factors and PUFAs in FCS is also mediated by ROS. A number of studies in recent years have shown that ligand stimulation of nonphagocytic cells results in an increase in intracellular oxygen species. Therefore, the participation of ROS as intermediates of response to growth factors acting through the tyrosine kinase pathway is well documented (Lo and Cruz 1995; Sundaresan et al. 1995; Bae et al. 1997). Moreover, the involvement of NADPH oxidase in the response of renal epithelial cells to arachidonic acid, which also induces tubular peroxisomes, was recently demonstrated (Cui and Douglas 1997). Taking together all these findings, it appears that all stimuli and agents that induce the formation of tubular peroxisomes, i.e., UV, growth factors, and arachidonic acid, all operate via ROS that activate the tyrosine kinase pathway.
The Peroxisomal Response May Be Central to Cellular Rescue from ROS
We showed recently (Schrader et al. 1996, 1998) that tubular peroxisomes are highly dynamic, transient structures in the process of rapid expansion of the peroxisomal compartment. They appear temporarily in association with cell growth and differentiation in HepG2 cells and are involved in the generation of spherical peroxisomes by fission (Schrader et al. 1996). Furthermore, peroxisomes play an important role in lipid metabolism and contain a variety of enzymes involved in the metabolism of ROS: catalase, superoxide dismutase, and glutathione peroxidase (Singh 1996). Therefore, the induction of tubular peroxisomes, as an indication of the expansion of the peroxisomal compartment, appears to be a logical response to the oxidative stress. Surprisingly, there have been very few studies on the response of mammalian peroxisomes to oxidative stress. As mentioned above, Singh and associates have analyzed the alterations of peroxisomes in several models associated with this medically important process. Their results in the model of renal ischemia and reperfusion of rat kidney (Gulati et al. 1992) indicate that, despite cell damage due to ROS, the peroxisomal compartment exhibits signs of regeneration (Singh and Gulati 1995) similar to the response observed in regenerating rat liver (Lüers et al. 1993). Along the same line, the oxidative stress induced by endotoxin treatment in rat liver caused a significant elevation of the antioxidative peroxisomal enzymes SOD and glutathione peroxidase (Dhaunsi et al. 1993). In cultured skin fibroblasts, Kremser et al. (1995) compared the sensitivity of the peroxisomal lipid β-oxidation system to ROS with that of the mitochondrial system and noted that peroxisomes were more sensitive than mitochondria. This is consistent with our observations reported here, indicating the lack of mitochondrial alterations to UV irradiation in comparison to peroxisomes. The central role of peroxisomes in defense against ROS is also clearly demonstrated by the high sensitivity of mutant CHO cell lines (Zoeller et al. 1988) and cells from patients with peroxisomal biogenesis defects to UV irradiation, which is partly due to the low levels of plasmalogens in those cells (Hoefler et al. 1991; Kremser et al. 1995). We have shown that the tubulation of peroxisomes in HepG2 cells seeded at low density and their subsequent disappearance by fission correlated well with the level of mRNA for 70-kD peroxisomal membrane protein (Schrader et al. 1998). Further studies are required to analyze the alterations of peroxisomal enzymes involved in defense against ROS in association with the induction of tubular peroxisomes. The model of UV induction of such tubular peroxisomes presented here provides an excellent experimental approach for such future studies.
Footnotes
Acknowledgements
Supported by grants from the Deutsche Forschungsgemeinschaft (DFG) through SFB 352 (project C 7) and SFB 601 (project B 1).
The technical assistance of Gabriele Krämer and Inge Frommer is gratefully acknowledged. The support of Markus Grabenbauer in digital processing of micrographs is appreciated. The activator and inhibitor of PKC (OAG and BIM) were a generous gift from Dr M. Radloff (University of Hamburg, Germany). We are also grateful to Prof Alfred Völkl and Dr Eveline Baumgart for helpful discussions.
