Abstract
A recent study by Morgan et al. on the mechanism of the heating antigen retrieval (AR) has raised an interesting issue concerning calcium-induced modification of protein conformation demonstrated by immunohistochemistry (IHC). The current study is based on calcium-induced modification of thrombospondin (TSP) and Ki-67, as demonstrated by IHC using seven monoclonal antibodies (MAbs) to TSP and an MAb MIB1. Experiments were carried out on frozen tissue sections of bladder carcinoma and lymph node. Frozen sections were incubated with solutions of 50 mM CaCl2 and/or 10 mM EDTA at 4C overnight before formalin or acetone fixation for TSP and Ki-67, respectively. Sections were then fixed in 10% neutral buffered formalin or acetone before immunostaining. Seven MAbs to TSP, named Ab1 to 7 representing clone numbers of A4.1, D4.6, C6.7, A6.1, B5.2, A2.5, and HB8432, respectively, and MIB1 were utilized as primary antibodies. ABC was used as the detection system and AEC as the chromogen for immunohistochemical staining. An extracellular immunostaining pattern represented a positive result for TSP, and nuclear staining for MIB1. Frozen sections preincubated in 50 mM CaCl2 overnight at 4C showed significant loss of staining and/or altered staining pattern for six of the seven antibodies to TSP and MIB1 compared to positive controls not exposed to CaCl2. Lack of immunostaining of TSP and MIB1 attributable to exposure to CaCl2 could be partially recovered by incubating the frozen sections in EDTA. Calcium-induced modification of protein structure was demonstrated more than 10 years ago on the basis of immunochemical techniques. In this study, similar calcium-induced modification of protein was detectable by IHC in frozen tissue sections, suggesting that calcium-induced modification of protein structure may occur independently of fixation-induced modification. The fact that calcium binding may affect IHC staining is not surprising in view of the fact that antibody/antigen interactions are protein structure-dependent. However, in this experiment the change occurred before and independent of formalin fixation and does not necessarily imply a role for calcium in AR. There may be a valuable role for the use of chemical modification in visualization of protein structure changes in tissue sections by IHC.
Keywords
R
We have also studied the effect on antigenicity in the presence of CaCl2, with the following observations. (a) Similar elimination of staining for MIB1 could be obtained by using CaCl2 as the AR solution. (b) However, not all antibodies were affected, e.g., staining for keratin remained unchanged after heating with CaCl2. (c) Similar satisfactory results of AR-IHC can be obtained by using different buffer solutions, as compared to citrate buffer (i.e., in the absence of a chelating agent), as long as the pH is at an appropriate value (Taylor et al. 1994,1996; Shi et al. 1995,1997). These findings suggest that the calcium-induced effect may not be sufficient to explain the loss of antigenicity and restoration of staining by AR. However, the studies of Morgan and co-workers on the mechanism of the AR technique do raise an interesting issue concerning calcium-induced modifications of protein conformation that exercise an effect on IHC staining.
Our recent study of different conditions of AR to achieve optimal staining of thrombospondin (TSP) stimulated a search of the literature regarding biochemical studies of TSP. We found that a biochemical study of monoclonal antibodies that recognized calcium-dependent structures of TSP was published more than 10 years ago (Dixit et al. 1986), and we also found other more recent studies of calcium-induced effects on protein conformation (Wakabayashi et al. 1986; Wilson 1991). With this concept of calcium-induced alterations in the structure of TSP in mind, we tested the effect of treatment with calcium chloride on antigen-antibody reactions as manifested by immunohistochemical staining with seven MAbs to TSP, and Ki-67. These included two important antibodies, A6.1 and D4.6, that were used by Dixit et al. for their immunochemical study, which demonstrated that these antibodies may recognize calcium-dependent structures of TSP (Dixit et al. 1986). The specific aims of our study were (a) calcium-induced modification of protein, exemplified by using TSP and Ki-67, as demonstrated by IHC, and (b) calcium-induced modification of protein independent of formalin-induced modification (crosslinking) of protein.
Materials and Methods
Tissues
Fresh frozen tissue sections of transitional cell carcinoma of bladder and lymph node were obtained from files at the Norris Cancer Hospital and Cancer Research Institute, University of Southern California School of Medicine. Frozen tissue was immediately embedded in OCT compound (Miles Laboratories; Elkhart, IN) and stored at −70C. Frozen sections were cut at 5–7 μm with a cryostat and mounted on commercially provided charged slides (Fisher Scientific; Pittsburgh, PA). Slides were stored at −70C before use.
Antibodies
Seven MAbs to thrombospondin were provided by NeoMarkers (Fremont, CA). Table 1 indicates the clone numbers of the seven MAbs, designated TSP Ab-1 to Ab-7. Originally, Ab-1 to Ab-6 were documented by Dixit et al. (1985a,b, 1986), and the Ab-7 was reported by Jaffe et al. (1983). The specificity of all antibodies has been confirmed by the Western blot method. Known characteristics of the epitopes for TSP Ab-1 to Ab-6 are listed in Table 1. The MAb to Ki-67 (MIB1) was purchased from Immunotech (Westbrook, ME).
The avidin-biotin system (ABC kits; Vector Laboratories, Burlingame, CA) was used as the detection reagent. Other chemical reagents, including the chromogen (3-amino-9-ethylcarbazole, AEC), were purchased from Sigma (St Louis, MO).
Calcium-induced Modification of TSP and Ki-67 Conformation
Frozen tissue sections of bladder carcinoma and lymph node were used to test calcium-induced modification of TSP and Ki-67 conformation using the following experiments:
1. Frozen sections previously unfixed were incubated with 50 mM CaCl2 (pH 7.1) at 4C overnight, washed in PBS, then fixed in 10% neutral buffered formalin (NBF) for 10 min.
2. Frozen sections previously unfixed were incubated with 10 mM EDTA solution (pH 5.0) at 4C overnight, washed in PBS, then fixed in 10% NBF for 10 min.
3. Experiments 1 and 2 were performed sequentially, i.e., frozen sections previously unfixed were incubated with 20 mM EDTA solution after overnight incubation with 50 mM CaCl2, washed in PBS, then fixed in 10% NBF for 10 min.
Immunohistochemical staining results of seven monoclonal antibodies a

aAbs, antibodies; frozen, frozen tissue section; N, N-terminal; C, C-terminal.
bA few tumor cells showed cytoplasmic staining in addition to extracellular staining pattern.
In all experiments, frozen sections without incubation of CaCl2, overnight incubation with normal horse serum (pH 6.9), distilled water (pH 6.8), and PBS (pH 7.4), followed by fixation in 10% NBF, were used as controls. All seven MAbs to TSP and MIB1 were used as primary antibodies for immunohistochemical staining in all three experiments, appropriate titers for optimal staining having been established using the positive control (untreated) sections. A titer of 1:100 was satisfactory in each case.
Immunohistochemical Staining Method
The ABC detection system was used for immunostaining according to the manufacturer's protocol. The incubation time for primary antibody TSP and MIB1 was overnight at room temperature. After incubation with primary antibody, staining was followed by ABC detection system using biotinylated anti-mouse immunoglobulin at a dilution of 1:200 and avidin-biotin peroxidase macromolecular complex at 1:100, with an incubation time of 30 min for each step. A wash of 10 min using PBS was added in between each step. AEC was used as chromogen. Finally, the slides were counterstained with hematoxylin and mounted in aqueous mounting medium for examination.
Negative Control
All immunohistochemical staining experiments were performed with negative control slides, including replacing the primary antibody with PBS or normal serum.
Evaluation
Positive staining of TSP and MIB1 was characterized by an extracellular staining pattern (Figures 1A and 1D) and nuclear staining (Figures 1E, 1G, and 1H), respectively. The intensity of positive immunostaining was graded as + + + +, + + +, + +, +, or − for extremely strong, strong, moderate, weak, and negative staining, respectively. For grading MIB1, the nuclear positive staining was also compared between each group based on both the population of positive cells and the intensity of staining in the germinal center of lymph nodes. To compare the influence of different chemical conditions, a semiquantitative estimation of the positive area of each section was compared with a frozen section without pretreatment, which was defined as 100%, and was rendered as a percentage. i.e., total positive staining area of tissue section with pretreatment/total TSP-positive staining area of tissue section without pretreatment = %. This estimation of percentage was performed independently by three authors (SRS, YS, and ST).
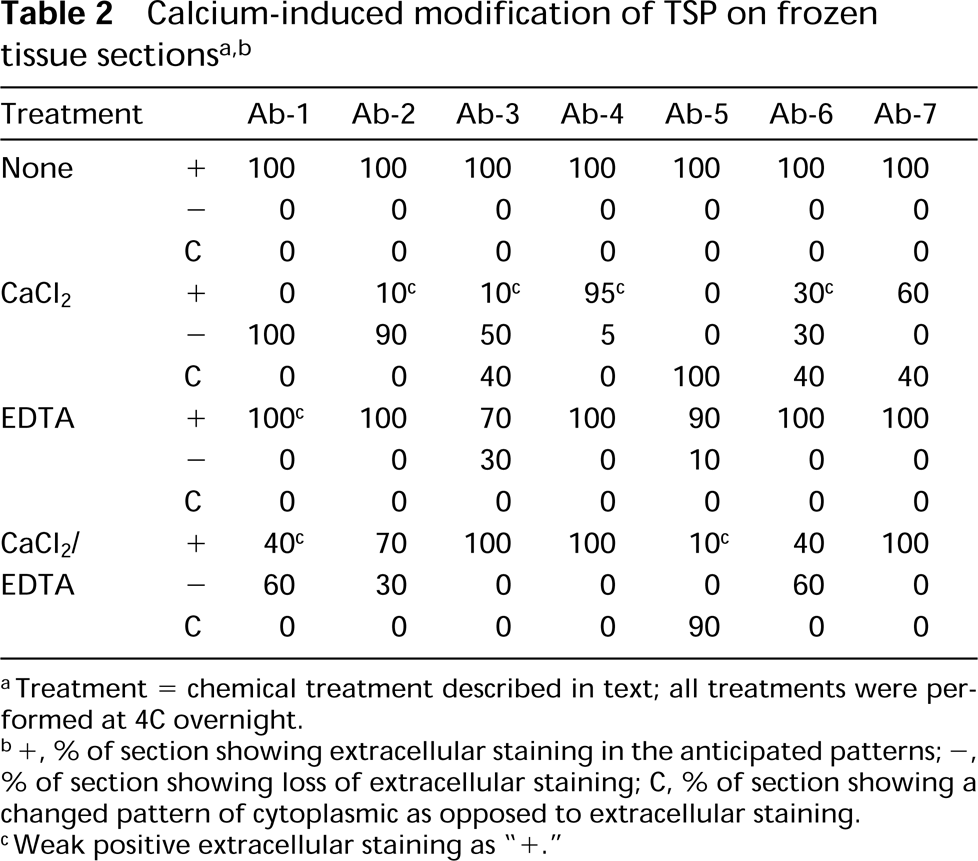
aTreatment = chemical treatment described in text; all treatments were performed at 4C overnight.
b+, % of section showing extracellular staining in the anticipated patterns; −, % of section showing loss of extracellular staining; C, % of section showing a changed pattern of cytoplasmic as opposed to extracellular staining.
cWeak positive extracellular staining as “+.”
Results
Table 1 summarizes immunohistochemical staining results of seven MAbs to TSP in frozen tissue sections. All antibodies gave positive results on frozen sections, although some sections showed weaker but still clearly positive staining (Table 2). The immunostaining pattern was similar among the seven MAbs, i.e., a predominantly extracellular staining pattern (Figures 1A and 1D). Rare tumor cells showed cytoplasmic staining of TSP with Abs-2, −5, and −7 in frozen sections. Nuclear staining was observed as background focally, appearing in both scattered normal and cancer cells using antibodies TSP Ab-3 and Ab-5.
The results of immunohistochemical staining of seven MAbs to TSP on frozen tissue sections after different chemical treatments before formalin fixation are summarized in Table 2. In general, treatment with CaCl2 had a significant effect on the immunohistochemical staining for TSP. Except for antibodies Ab-4 and Ab-7, which showed slight or moderate reduction of extracellular staining pattern (5% and 40%, respectively), all other antibodies yielded either negative staining results and/or an altered staining pattern, such as cytoplasmic staining (Figures 1B and 1C). Interestingly, these calcium-induced modifications of TSP immunostaining patterns could be either partially or completely reversed by secondary incubation with EDTA (Table 2; Figure 1D). Incubation with EDTA in the absence of prior CaCl2 treatment did not cause a significant change in the TSP immunostaining pattern. The immunostaining results with MIB1 were similar to those of TSP after various treatments (Table 3; Figures 1E-1H). In contrast to TSP, incubation in EDTA yielded a slight decrease of staining intensity for MIB1 (Figure 1G). EDTA incubation could retrieve only 50% of the MIB1 staining intensity that was induced by calcium (Figure 1H). All positive control slides that were not incubated with CaCl2 yielded positive staining for TSP and MIB1. Negative control slides that were incubated with PBS or normal serum instead of the primary antibodies (TSP and MIB1) showed absence of staining with a clean background.
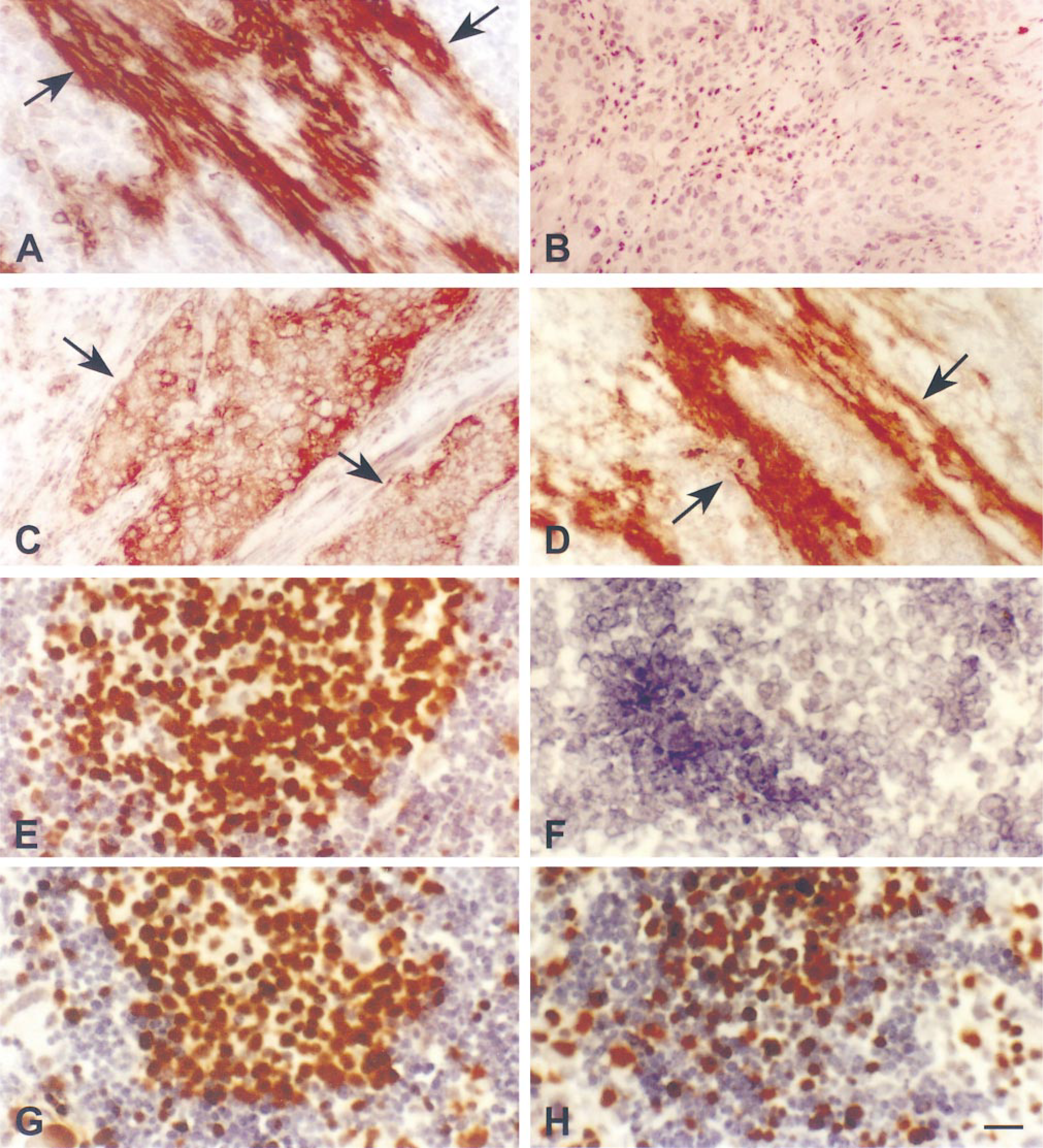
Immunohistochemical staining results of MAb Ab-3 to TSP on frozen tissue sections of bladder cancer, showing the effects of pretreatment with CaCl2 (A-D) and MAb MIB1 on frozen sections of lymph node showing similar effects of CaCl2 (E-H). (A) Frozen section fixed in 10% NBF for 10 min, showing strong TSP labeling in an extracellular pattern (arrows): no pretreatment (positive control):(B) Frozen section incubated with 50 mM CaCl2 at 4C overnight, then fixed in 10% NBF. A negative staining result was demonstrated in 50% of total area in the section (refer to Table 2). (C) Frozen section incubated with CaCl2 at 4C overnight, then fixed in 10% NBF. An altered pattern of staining showing cytoplasmic staining of cancer cells (arrows) was observed in 40% of total area in the tissue section for Ab-3. Such an altered pattern was demonstrated in 4/7 antibodies (refer to Table 2). (D) Frozen tissue section incubated with 20 mM EDTA after overnight incubation with 50 mM CaCl2 and fixed in 10% NBF for 10 min. Restoration of extracellular positive staining (arrows) was demonstrated. Bar = 50 μm. (E) Frozen tissue section of lymph node was incubated in distilled water at 4C overnight, then fixed in acetone, showing strong MIB1 nuclear staining of germinal center. (F) Frozen section incubated with 50 mM CaCl2 at 4C overnight, then fixed in acetone, showing negative result. (G) Frozen section incubated with 20 mM EDTA at 4C overnight, then fixed in acetone, showing slightly decreased nuclear staining. (H) Frozen section incubated with 20 mM EDTA after overnight incubation with 50 mM CaCl2 at 4C and fixed in acetone, showing partial restoration of nuclear staining of MIB1. AEC was used as chromogen; hematoxylin was used as the counterstain. Bar = 25 μm.
Discussion
It has been verified by our experiments that calcium-induced modification of protein can be demonstrated by IHC (Figure 1; Tables 2 and 3) and that it is independent of formalin-induced crosslinking. Our results are confirmed by control groups, i.e., only calcium chloride can induce the reduced or altered IHC staining among reagents tested in our study but this is not caused by the pH of the solution, because the pH of 50 mM CaCl2 solution is similar to those of control groups. The fact that the calcium-induced modification can be reversed by sequential incubation with EDTA (Figure 1D and 1H) may also support our conclusions.
The relation of structure to function for thrombospondin has been studied over the past decade using MAbs specific for different domains (Dixit et al. 1985a,b, 1986; Galvin et al. 1985; Prater et al. 1991). During the course of these studies, it was recognized that the ability of some MAbs to recognize their corresponding antigens (epitopes) appears to be calcium-dependent under certain conditions. A proposed mechanism involves binding of calcium that induces a conformational modification, resulting in either a diminished antigen-antibody recognition effect, as for TSP (Dixit et al. 1986; Wilson 1991), or the reverse effect, as seen with protein C (Wakabayashi et al. 1986). On the basis of immunohistochemical studies using fresh tissue sections, we demonstrated that results similar to those obtained by these earlier biochemical studies were also evident by IHC. The same MAbs tested by Dixit et al. (1986), including A6.1 (Ab-4) and D4.6 (Ab-2), were tested. Some variations were observed between the purified, calcium-replete TSP protein and the formalin-fixed frozen tissue section samples used in this study. Nevertheless, the major conclusion remains that calcium-induced modification of TSP structure yields a reduced antigen-antibody reaction for many antibodies to TSP (Table 2; Figure 1). Dixit et al. (1986) postulated that removal of calcium by chelation induced significant modification of the TSP conformation, described as an unrolling or unraveling of the large domains, changes that may expose epitopes necessary for the binding of certain MAbs. Among the seven MAbs tested, the effect of calcium-induced modification of TSP structures differed for different antibodies, as indicated in Table 2. The different patterns and degrees of TSP immunostaining after calcium-induced modification may be an indication of variable epitope mapping of TSP that can be recognized by the monoclonal antibodies under investigation (Dixit et al. 1986; Wilson 1991). Calcium-induced modification of Ki-67 may behave in a similar fashion as TSP, although the subsequent EDTA-incubation could not retrieve calcium-induced negative nuclear staining of MIB1 completely (Table 3; Figure 1H).
It is likely that not all antigens or epitopes will be influenced by calcium in the same manner as TSP. Indeed, Wakabayashi et al. (1986) demonstrated an effect opposite to that described for TSP. They found that detectable antigen-antibody immunoreactivity of protein C is manifest only in the presence of calcium. Their experiments used three MAbs to protein C, a vitamin K-dependent enzyme involved in blood coagulation, and were based on immunochemical techniques. Both the TSP effect and the protein C effect are consistent with the notion that antigen-antibody recognition is protein conformation-dependent (Davies et al. 1988; Wilson 1991). Therefore, it is not surprising to see a decreased intensity of immunostaining for TSP and other proteins after the tissue has exposed to calcium salts. This effect is particularly evident when high-temperature heating occurs in the presence of CaCl2, as has been previously documented (Morgan et al. 1994,1997). Our present study suggests that the calcium-induced modification of protein structure may occur independently of any conformational change in protein that is fixation-induced. This conclusion is supported by our findings that the intensity of immunostaining in the control group not pretreated with CaCl2 before formalin or acetone fixation was much greater than in those cases pretreated with CaCl2 (Tables 2 and 3; Figures 1A and 1E).
In earlier studies we reported that boiling formalin-fixed, paraffin-embedded tissue sections in 50 mM CaCl2 did not influence the immunoreactivity of keratin. However, under the same conditions the immunoreactivity of MAbs MIB1 and p53-1801 was significantly reduced (Shi et al. 1997). Clearly, calcium binding does influence the immunoreactivity of some epitopes, but equally clearly others are unaffected. As a result, calcium release, allowing conformational changes to a more immunoreactive state, cannot be advanced as the mechanism of action of AR in all instances. The original hypothesis of heat-induced modification of protein structure represents a more likely explanation overall (Shi et al. 1997).
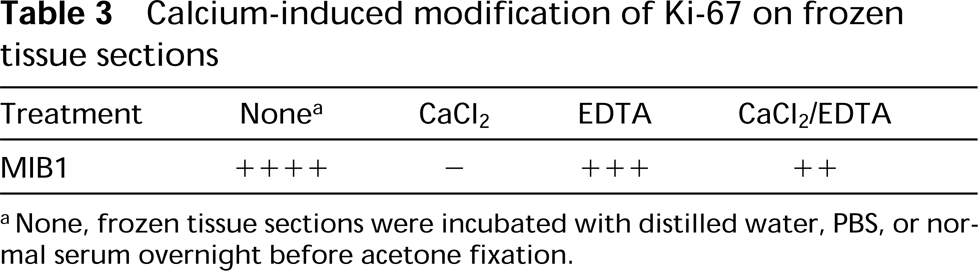
Calcium-induced modification of Ki-67 on frozen tissue sections
aNone, frozen tissue sections were incubated with distilled water, PBS, or normal serum overnight before acetone fixation.
Hypothesis: Detection of Molecular Alteration of Protein Based on Chemical Modification
The study of calcium-induced modification of protein structure has provided food for thought. Morgan et al. (1994) performed an interesting experiment using the calcium heating method that may provide a basis for further studies of the molecular alteration of protein structure as detected by IHC. An immunohistochemical approach for detection of the chemical modification of a protein may serve as a bridge between orthodox morphology and molecular biology (Figure 2). For example, if chemical modification of a protein structure results in significant effects on certain antigen-antibody reactions, then these effects may be detected by IHC. In this instance, IHC serves as a method to detect structural alteration of a protein (“target structure” in Figure 2) that has been modified by a chemical reagent (“chemical modification solution, CMS” in Figure 2) with a resultant change in antigenic structure (“target antigen” in Figure 2).
A review of the literature reveals several publications (Olcott and Fraenkel-Conrat 1947; Atassi 1975; Barlow et al. 1986; Wilson 1991; Wong 1991) supporting the hypothesis that immunohistochemical methods can be employed to detect modifications of protein structure, both those occurring during the course of cell metabolism and those induced by external agents. The prototype for applying chemical modification methods to the study of proteins was developed more than half a century ago, when biochemists studied the structure-function relationship of proteins after reaction with various chemical agents (Olcott and Fraenkel-Conrat 1947; Wong 1991). Development of orthodox histochemistry early in this century also contributed valuable theory and technique, with the design of chemical reactions that could be visualized by microscopy. Lillie and co-workers (1958, 1976) summarized a variety of methods utilizing the blockade of hydroxyl and amine groups of protein that proved to be valuable in protein and cell recognition, and provided examples of some critical principles of chemical modification. Other reactions were designed for detection of, e.g., tryptophan and histidine. Furthermore, histochemical methods have been used in combination with immunohistochemistry to detect citrulline-containing proteins as products of endogenous peptidylarginine deiminase activity (Asaga and Senshu 1993). The blocking effects of acetylation and nitrosation of tissue amines in histochemistry have been extended to blockade of the antigen-antibody reaction, exemplified by immunohistochemical detection of lysozyme in Paneth cells and histiocytes of human small intestine (Montero and Segura 1989).
Although today's morphologist is equipped with a variety of microscopes characterized by high resolution, along with modern histochemical techniques, there remains a gulf between morphology and modern molecular biology. IHC and ISH effectively allow the performance of molecular pathology at the tissue section level to detect protein (equivalent of Western blots) and RNA or DNA (Northern or Southern blots). Even subtle alterations in protein structure may on occasion be detectable by orthodox IHC, enabling a pathologist to recognize molecular alterations of protein by regular light microscopy (e.g., wild-type vs mutant p53 “naturally occurring” chemical modification). However, for slight changes of molecular structure, it may be difficult or impossible to develop a specific antibody to the altered protein. For example, the tumor suppressor gene Rb has different subtypes based on slight molecular alterations (phosphorylated vs nonphosphorylated proteins) that are not distinguishable by antibodies available to date. In theory, it may be positive to distinguish phosphorylated and nonphosphorylated versions by inducing further chemical modification of OH groups on serine or threonine in Rb because phosphorylation occurs in the G1, S, and G2/M transition (Riley et al. 1994). Antibodies may then detect these more major chemical modifications. With this approach, it may be possible to detect minor changes in protein structure by a chemical modification approach that would allow recognition by light microscopy of the formation of different molecular structures that are not directly detectable. Such an approach to the identification of subtle molecular changes could lead to a new approach to molecular morphology, with potential applications for cancer diagnosis and research.
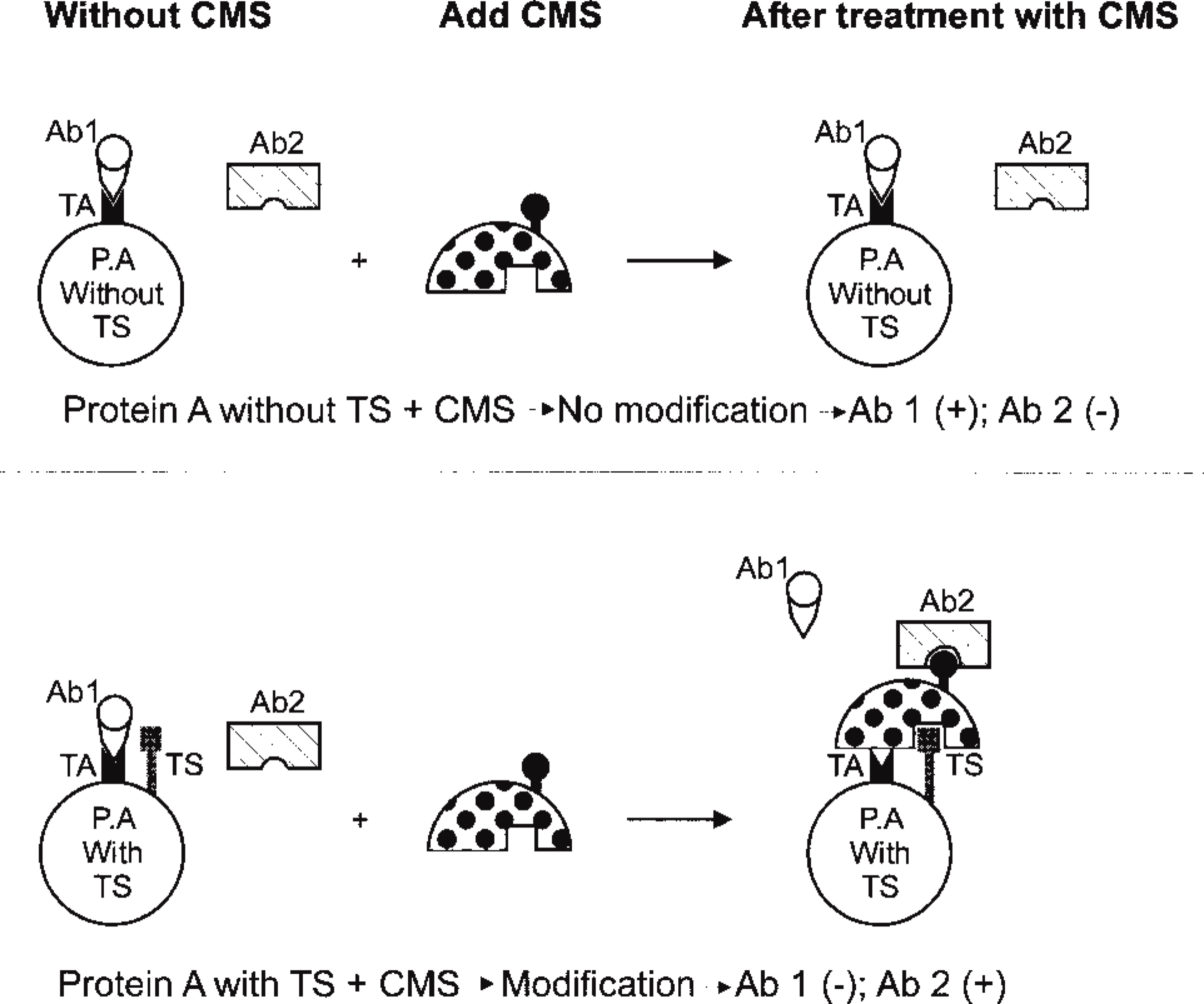
Schematic of the potential approach for detection of molecular alteration of protein using the antigen-antibody reaction as a probe, based on modification of protein conformation. (Top) Protein A (P.A), which is lack of target structure (TS). Therefore, no alteration exists after treatment with a chemical modification solution (CMS), resulting in positive immunoreaction of antibody 1 (Ab 1) binding to the target antigen (TA), but negative of antibody 2 (Ab 2), since no TA is available for Ab 2. (Bottom) CMS-treated protein A which has TS. It may have two broad effects: (a) to abolish reactivity for Ab 1 or (b) to create a new epitope (hapten effect) such that a previously unreactive antibody will bind to the new epitope.
Footnotes
Acknowledgements
Supported by the University Pathology Associates of the University of Southern California, by an NIH grant CA7090303, and by the American Cancer Society (ACS IN-21-31).
The authors appreciated Dr Randall B. Widelitz for preparing Figures 1 and ![]() , Dr Syed Ashraf Imam for his kind help in microphotography, Dr Atul K. Tandon for providing seven monoclonal antibodies to TSP for testing, and Ms Florence Miyagawa's help in preparing this manuscript.
, Dr Syed Ashraf Imam for his kind help in microphotography, Dr Atul K. Tandon for providing seven monoclonal antibodies to TSP for testing, and Ms Florence Miyagawa's help in preparing this manuscript.
