Abstract
Hair follicle development, growth (anagen), and regression (catagen) largely result from bidirectional epithelial-mesenchymal interactions whose molecular basis is still unclear. Because adhesion molecules are critically involved in pattern formation and because the fundamental importance of neural cell adhesion molecule (NCAM) for feather development has been demonstrated, we studied the protein expression patterns of NCAM during hair follicle development and regression in the C57BL/6 mouse model. During murine hair follicle development, NCAM immunoreactivity (IR) was first detected on epithelial hair placodes and later on selected keratinocytes in the distal outer root sheath. Mesenchymal NCAM immunoreactivity (IR) was noted on fibroblasts of the future dermal papilla (DP) and the perifollicular connective tissue sheath. Fetal hair follicle elongation coincided with strong, ubiquitous dermal NCAM IR, which remained strong until the follicles entered into their first neonatal catagen. At this time, the strong interfollicular dermal NCAM IR decreased substantially. During consecutive hair cycles, mesenchymal NCAM IR was seen exclusively on DP and perifollicular connective tissue sheath fibroblasts and on the trailing cells of regressing catagen hair follicles. These highly restricted and developmentally controlled expression patterns suggest an important role for NCAM in hair follicle topobiology during morphogenesis and cyclic remodeling of this miniorgan.
T
For several reasons, neural cell adhesion molecule (NCAM) is a particularly interesting candidate as a key adhesion molecule in hair follicle topobiology (Chuong et al. 1993; Kaplan and Holbrook 1994; Müller-Röver and Paus in press). NCAM is a widely distributed and abundant cell surface receptor belonging to the immunoglobulin superfamily, which mediates predominantly homophilic binding and is encoded by a single gene, yet is expressed in several alternatively spliced forms (Cunningham et al. 1987; Small et al. 1988; Brummendorf and Rathjen 1996; Walsh and Doherty 1996). NCAM is expressed in specific spatiotemporal patterns in many tissues during embryogenesis (Chuong and Edelman 1985; Crossin et al. 1985) and is a key determinant of neuron-Schwann cell and neuron-extracellular matrix interactions during neuronal growth, development, sprouting, remodeling, and regeneration (Brackenbury 1988; Martini and Schachner 1988; Martini 1994; Botchkarev et al. 1997a; Saffell et al. 1997). NCAM-deficient mice exhibit a reduction in brain weight and a decline in size of the olfactory bulb (Cremer et al. 1994; Tomasiewicz et al. 1993). NCAM may be crucial for pattern formation because the activity of its promotor is controlled by HOX and PAX gene products in vitro (Jones et al. 1993; Edelman and Jones 1995; Holst et al. 1997) which play a pivotal role in controlling pattern formation and specify the attributes of entire body regions (Lewin 1997; Müller 1997).
During feather follicle morphogenesis, NCAM-mediated cell-cell interactions are crucial to the appropriate formation of the dermal condensations that are the avian analogue of the developing mammalian DP. The application of anti-NCAM antibodies during feather follicle morphogenesis in vitro induced a highly irregular shape of the dermal condensations (Jiang and Chuong 1992). NCAM expression was also found in the DP, the perifollicular connective tissue sheath, and the outer root sheath (ORS) of fetal tylotrich hair follicles and adolescent pelage hair follicles of mice (Chuong et al. 1991; Vielkind et al. 1995; Hardy and Vielkind 1996; Combates et al. 1997) and on the DP and perifollicular connective tissue sheath of developing human hair follicles (Kaplan and Holbrook 1994).
Given the puzzling differences in NCAM immunoreactivity patterns that have been described in skin (Chuong et al. 1991; Kaplan and Holbrook 1994; Vielkind et al. 1995; Hardy and Vielkind 1996), we were interested to learn whether these differences reflect developmental differences in the NCAM expression patterns of fetal, neonatal, and adolescent skin. These types of skin were therefore analyzed separately, using the well-defined C57BL/6 mouse model (Chase 1954; Straile et al. 1961; Paus et al. 1990,1994a; Paus 1996). In addition, we focused on defining the follicular NCAM expression during hair follicle regression (catagen), which had not been previously characterized in detail (Combates et al. 1997).
Materials and Methods
Animal Models and Tissue Collection
Syngenic C57BL/6 mice (female, 1-21-day-old neonatal or 6-9-week-old; Charles River, Sulzfeld, Germany) were housed in community cages, with 12-hr light periods, at the Charità animal facilities, Virchow Campus, Berlin, and were fed water and mouse chow ad libitum. Active hair growth (anagen) was induced in the back skin of adolescent mice in the telogen phase of the hair cycle (as judged from their homogeneously pink back skin color) by application of a melted wax/resin mixture under general anesthesia as described (Paus et al. 1990). Early anagen and all eight stages of catagen development (Straile et al. 1961; Paus et al. 1994a) were studied, using at least five mice per time point: anagen IV (5 days a.d.), and catagen (the latter developed spontaneously 17, 18, or 19 days a.d.). Preliminary experiments had revealed a distinct NCAM expression pattern during neonatal hair follicle development (Müller-Röver et al. 1996) compared to fetal hair follicle morphogenesis (Vielkind et al. 1995), even though the same antibody (clone H28-123-16; Boehringer, Mannheim, Germany) and mouse strain (C57BL/6J) were used. Therefore, skin sections of C57BL/6J embryos harvested at the estimated gestational age (E) days E16 and E18, and skin sections of neonatal mice harvested on postpartum (P) days P1-P6, P8, P12, P19, and P21 were studied to compare NCAM expression during fetal and neonatal hair follicle development and during the spontaneous initiation of hair follicle cycling, which begins in murine back skin with the first synchronized entry of hair follicles into catagen (P17-19).
The neck region of the back skin was harvested parallel to the vertebral line to obtain longitudinal sections through the hair follicles from one defined site as described (Paus et al. 1994b), frozen in liquid nitrogen, and covered with embedding medium.
Immunohistochemistry
Ten-μm sections were used to analyze fetal skin and 8-μm sections were used for neonatal and adolescent skin. The sections were incubated with a monoclonal rat anti-mouse anti-body (clone H28-123-16; Boehringer) against the extracellular domain of NCAM specifically binding to NCAM 120, 140, and 180 (Hirn et al. 1981). Incubation with nonimmune rat Ig in the same concentration as the primary antibody was used as a negative control. Comparisons of the observed IR patterns with published IR patterns of NCAM (Crossin et al. 1985; Chuong et al. 1991; Kaplan and Holbrook 1994; Vielkind et al. 1995; Hardy and Vielkind 1996; Vielkind and Hardy 1996; Combates et al. 1997) were employed as internal positive and negative controls.
Antigen-antibody complexes were visualized by both immunofluorescence (IF) and avidin-biotin-complex (ABC) labeling. For the ABC labeling, cryostat sections were incubated with the primary antibody for 1 hr at room temperature (RT) (1:400), followed by an incubation with biotin-conjugated F(ab)2 fragments of a goat anti-rat IgG (Jackson Immunoresearch, Bar Harbor, ME; 1: 200, 30 min, RT). Skin sections were then incubated with ABC complex (Vecta-Stain Kit; Vector Laboratories, Burlingame, CA) for 45 min at RT. The alkaline phosphatase color reaction was developed as described (Handjiski et al. 1994; Paus et al. 1994b). IF labeling was performed as described (Botchkarev et al. 1997a). The cryostat sections were incubated with the primary antibody overnight at RT (1:50), followed by an incubation with tetramethylrhodamine-isothiocyanate (TRITC)-conjugated F(ab)2 fragments of a goat anti-rat IgG (Jackson Immunoresearch;1:200, 30 min, 37C).
Immunomorphometry
The immunoreactivity (IR) pattern was scrutinized by studying at least 20 microscopic fields or hair follicles per mouse, and five mice were assessed per stage of hair follicle morphogenesis and cycling. Thus, we examined at least 100 microscopic fields or hair follicles derived from at least five different mice per stage of hair follicle morphogenesis and cycling. The most consistently recognizable specific IR patterns were recorded and summarized in schematic representations of murine hair follicle morphogenesis and cycling that had been computer-generated (Paus et al. 1997).
Because we have previously described hair cycle-dependent NCAM expression patterns in selected skin nerve fibers and Schwann cells (Botchkarev et al. 1997a), analysis of the neuronal NCAM expression patterns was excluded from the present study.
Results
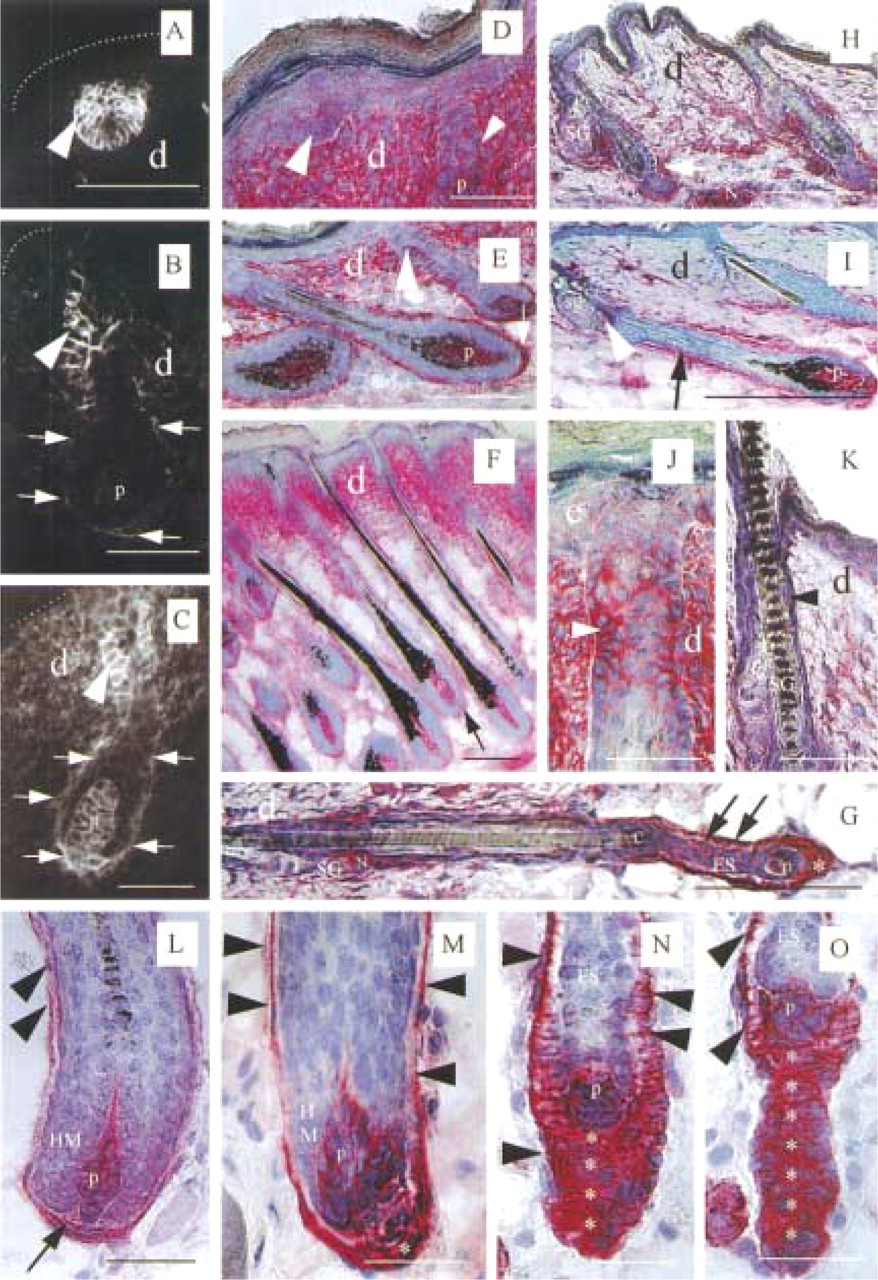
The expression patterns of NCAM in skin epithelium and mesenchyme during fetal and neonatal hair follicle morphogenesis and during later hair follicle cycling were strikingly different and appeared to be subject to stringent developmental controls. Figure 1 shows representative NCAM IR patterns during various stages of hair follicle morphogenesis, regression and consecutive cycling, which are summarized schematically in Figure 2.
NCAM Expression During Fetal Hair
Follicle Morphogenesis
In late fetal skin at E16, no cutaneous NCAM IR was found during Stage 1 of fetal tylotrich hair follicle morphogenesis in C57BL/6 mice. The first epithelial NCAM IR was detected in the hair placode epithelium of fetal back skin during Stage 2 (Figure 1A). No (or only extremely weak) mesenchymal NCAM IR was detected on the underlying dermal fibroblast condensations. At E18 (Stages 3-5), NCAM IR became restricted to a selected group of keratinocytes of the developing ORS, and the developing DP and the fibroblasts of the distal perifollicular connective tissue sheath showed increasing NCAM IR. Intermediate NCAM IR was found throughout the dermis (Stage 4; Figure 1B). NCAM IR remained strong throughout hair follicle development (Stages 5-8) on keratinocytes in the peri- and suprainfundibular region of the ORS (Figures 1B and 2). During Stages 5-8, the dermal skin compartment showed increased homogeneous NCAM IR compared to the preceding stages, which was particularly accentuated on DP fibroblasts (Figures 1C and 2). Throughout fetal hair follicle development, heterogeneous NCAM staining patterns were noted on the arrector pili muscle, but no NCAM IR was noted in the sebaceous gland (Figures 1H and 1I).
NCAM Expression During Neonatal Hair
Follicle Morphogenesis
Comparing fetal (Figures 1A, 1B, and 2, Stages 1-5, left) and neonatal hair follicle development (Figures 1C-1F and 2, Stages 1-5, right) in mice, we found much more intense NCAM IR in neonatal skin compared to fetal skin, yet largely the same follicular NCAM IR patterns, whereas a significant change in the dermal expression patterns of NCAM was noted. In contrast to hair follicle development in fetal skin, NCAM was strongly and homogeneously expressed throughout the dermis during neonatal hair follicle morphogenesis (Figures 1C-1F and 2, Stages 1–5, right). Neonatal hair follicle elongation (Stages 5–8) was associated with strongly pronounced NCAM IR on the proximal perifollicular connective tissue sheath and on DP fibroblasts (Figures 1C, 1E, and 1F). Similar to the findings in fetal skin, strong NCAM IR was noted on selected keratinocytes in the distal isthmus region of the developing ORS during Stages 3 and 4 (Figure 1D) and in the peri- and suprainfundibular ORS during Stages 5-8 (Figures 1C and 1E), while heterogeneous NCAM IR patterns were noted on the arrector pili muscle. Again, sebocytes did not display any NCAM IR.
NCAM Expression During the First Neonatal Catagen and the Beginning of Hair Follicle Cycling
During the first neonatal hair follicle regression (catagen) (Figure 1G), the strong interfollicular dermal NCAM IR declined dramatically (Figures 1G-1I). The regressing epithelial strand showed intermediate NCAM IR (Figure 1G), and the fibroblasts of the proximal perifollicular connective tissue sheath, as well as the trailing mesenchymal cells, displayed intense NCAM IR (Figure 1G). During the initiation of the first neonatal hair cycle (Figure 1H), interfollicular dermal NCAM IR was very weak compared to neonatal hair follicle morphogenesis. This dermal NCAM expression pattern was maintained throughout consecutive hair cycles (Figure 1I). In contrast to the interfollicular dermis, the perifollicular connective tissue sheath and DP fibroblasts of the first anagen I hair follicles showed intense NCAM IR (Figure 1H).
NCAM IR of periinfundibular ORS keratinocytes was observed to be particularly pronounced on tylotrich hair follicles (Figure 1J) in a strikingly hair cycle-dependent manner. Intense periinfundibular ORS NCAM IR has been found throughout neonatal hair follicle morphogenesis (Figure 1J) and middle anagen (Figure 1I), which decreased strongly during catagen (Figure 1K).
NCAM Expression During Catagen in Adolescent Skin
In contrast to fetal and neonatal hair follicle morphogenesis, during hair follicle regression in the skin of adolescent mice (Figures 1L-1O) mesenchymal NCAM IR was seen exclusively on DP and perifollicular connective tissue sheath fibroblasts and on the trailing cells of the regressing epithelial hair bulbs. Also in contrast to hair follicle morphogenesis, epithelial NCAM IR was seen on hair matrix keratinocytes during early catagen (Figure 1L) and on keratinocytes of the epithelial strand (Figures 1N and 1O).
NCAM expression during fetal (
During late anagen VI and catagen I-II, the perifollicular connective tissue sheath was restricted to one layer of NCAM+ fibroblasts (Figure 1L). When the DP was moving upwards, the DP and the developing “tail” of trailing connective tissue sheath fibroblasts displayed very intense NCAM IR (Figures 1L-1O). With progressing catagen, the NCAM IR found on all hair matrix keratinocytes (Figures 1L and 1M) became restricted to one layer of basement membraneassociated keratinocytes during late catagen (Figures 1N and 1O). Heterogeneous NCAM IR patterns were detected on the arrector pili muscle throughout the entire hair cycle.
In summary, during fetal hair follicle morphogenesis, we noted a switch of NCAM expression from the hair placode epithelium to a ubiquitous and homogeneous NCAM expression pattern throughout the skin mesenchyme, with maximal NCAM expression by DP and perifollicular connective tissue sheath fibroblasts. During the first neonatal catagen, this interfollicular dermal NCAM IR declined dramatically. During the consecutive hair cycles, mesenchymal NCAM became highly restricted to the DP and the perifollicular connective tissue sheath. During neonatal and adolescent catagen, the trailing cells of the regressing epithelial hair bulb expressed the most intense NCAM IR.
Discussion
In this report we present a profile of the developmentally controlled expression of NCAM on distinct fibroblast populations during fetal and neonatal morphogenesis of the skin and hair follicles, and define NCAM expression during the first neonatal catagen and the beginning of hair follicle cycling (Figure 2). In addition, we show that NCAM is also expressed in the cutaneous mesenchymal epithelium of murine skin, i.e., on selected keratinocytes of the hair placode and the developing hair bulb, as well as on ORS keratinocytes of anagen hair follicles and hair matrix keratinocytes of early catagen hair follicles (Figure 1).
Important previous studies in murine skin have focused mainly on the fetal development of tylotrich follicles (Vielkind et al. 1995; Hardy and Vielkind 1996; Vielkind and Hardy 1996) and on the telogen-anagen transformation of pelage hair follicles in adolescent skin (Combates et al. 1997). The present study corroborates the cutaneous NCAM expression patterns reported for tylotrich hair follicle development (Vielkind et al. 1995; Hardy and Vielkind 1996; Vielkind and Hardy 1996) (Figure 2, Stages 1-5, left) and compares these expression patterns with those seen during neonatal morphogenesis of pelage hair follicles (Figure 2, Stages 1-5, right), which represent the large majority of murine back skin hair follicles. In addition, we compare these findings with NCAM expression patterns during the first neonatal catagen, the beginning of postnatal hair follicle cycling (Figures 1G-1H and 2) and all stages of adolescent catagen (Figures 1L-1O), which had not been previously studied (Combates et al. 1997).
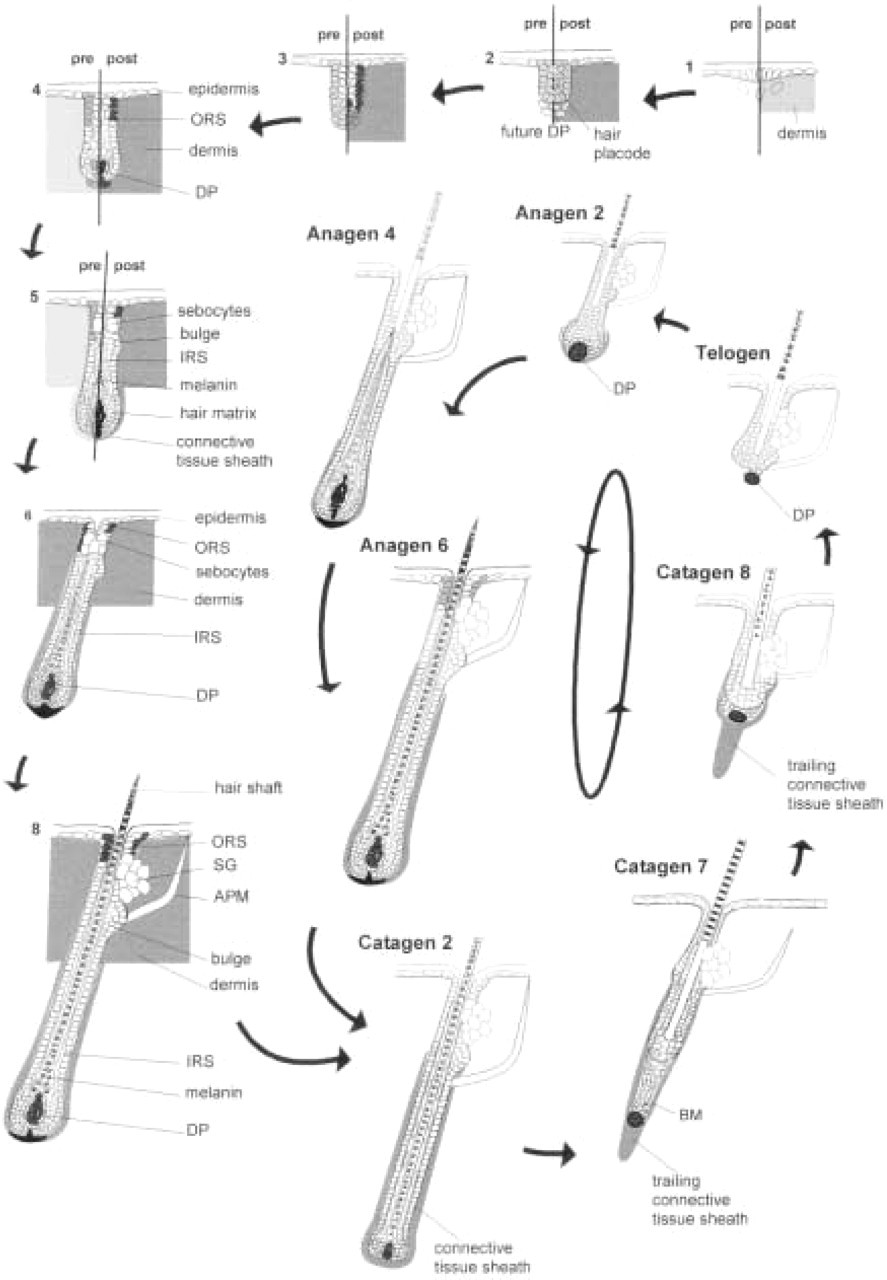
Schematic representation of NCAM expression during fetal and neonatal hair follicle morphogenesis, regression and the first postnatal anagen. Figures divided by lines contrast the different NCAM expression of interfollicular dermal fibroblasts during fetal and neonatal hair follicle morphogenesis. pre, prenatal; post, postnatal. During fetal hair follicle development (Stages 1–5, left), NCAM expression undergoes a dramatic switch from epithelial expression [hair placode (Stage 2) and distal outer root sheath (ORS) (Stages 4–5)] to ubiquitous dermal expression (Stages 4–5), which is most prominent in the dermal papilla (DP) (Stages 3–5). During neonatal development of pelage hair follicles (Stages 1–8, right), the entire hair placode displays NCAM immunoreactivity (IR), and the distal ORS shows even stronger NCAM IR compared to fetal hair follicle development. The previously ubiquitous dermal NCAM IR (Stages 5–8) becomes more accentuated on peri- and intrafollicular fibroblasts, most prominently in the DP (Stages 5–8). The interfollicular dermal NCAM IR dramatically declines during the first neonatal catagen (catagen 2–8). During the consecutive hair follicle cycles, strong NCAM expression is restricted to the fibroblasts of the DP and perifollicular connective tissue sheath fibroblasts and to the trailing cells of the regressing proximal catagen hair follicle. Dark gray indicates strong NCAM IR; light gray intermediate NCAM IR. The gray box represents a homogeneously NCAM+ dermis. Numbers indicate the developmental stages according to Paus et al. (1997). Telogen and anagen staining results according to Combates et al. (1997) and Müller-Röver and Paus (unpublished data; cf. Müller-Röver and Paus in press). APM, arrector pili muscle; DP, dermal papilla; IRS, inner root sheath; ORS, outer root sheath; SG, sebaceous gland.
Comparing fetal (Figures 1A, 1B, and 2, Stages 1-5, left) and neonatal hair follicle development in mice (Figures 1C-1F and 2, Stages 1-5, right), we found much more intense NCAM IR in neonatal skin compared to fetal skin, i.e., a significant change in the dermal expression patterns of NCAM was noted. In contrast to hair follicle development in fetal skin, NCAM was strongly and homogeneously expressed throughout the dermis during neonatal hair follicle morphogenesis (Figures 1C-1F and 2, Stages 1-5, right). However, the follicular NCAM IR patterns were largely the same.
This peak of interfollicular dermal NCAM expression is seen only throughout the neonatal period of pelage hair follicle development (Figures 1D-1F). During fetal human hair follicle morphogenesis (Kaplan and Holbrook 1994), NCAM is initially present in the entire dermis but becomes subsequently weaker in the interfollicular dermis, and becomes pronounced on fibroblasts of the dermal condensation/DP and perifollicular connective tissue sheath (Kaplan and Holbrook 1994). This expression pattern corresponds well to that seen during neonatal murine hair follicle development (Figures 1C and 1D-1F), during which strong interfollicular NCAM IR becomes progressively accentuated on proximal perifollicular connective tissue sheath and DP fibroblasts (Figures 1C and 1D-1F). This raises the possibility that the NCAM expression pattern in fetal human skin reflects developmental programs that are evolutionarily conserved between humans and mice. This encourages one to exploit the C57BL/6 mouse model as a tool for dissecting the functional significance of NCAM during human hair follicle morphogenesis.
Because we have recently demonstrated strong expression of neurotrophins and their receptors during hair follicle morphogenesis and catagen development (Botchkarev et al. 1997b), the strong, developmentally controlled mesenchymal NCAM expression in neonatal (Figures 1C-1F) and catagen skin (Figures 1M-1O) may reflect the presence of an NCAM-upregulating neurotrophin or cytokine milieu associated with perinatal and neonatal hair follicle morphogenesis.
The functional importance of NCAM for skin appendage formation was first established by analyzing the development of feather follicles in chicken skin explant cultures (Chuong et al. 1991,1993; Jiang and Chuong 1992; Chuong 1993). In this experimental system, application of anti-NCAM antibodies during feather follicle morphogenesis induced a highly irregular shape of the dermal condensations (Jiang and Chuong 1992). The morphogenesis of feather and hair appears to share many developmental controls and follows similar developmental principles (Chuong 1993; Chuong et al. 1991), and the NCAM expression patterns in feather and hair follicles show some similarities. This raises the question of whether NCAM can be utilized as a key downstream effector molecule (Edelman 1992) for the stringently controlled bidirectional epithelial-mesenchymal induction processes during hair follicle development (Hardy 1992; Müller- Röver and Paus in press).
As discussed elsewhere in greater detail (Müller-Röver and Paus in press), NCAM may segregate and differentiate cutaneous cell populations into morphogen-elaborating cell collectives, which thereby attain novel functional properties compared to neighboring cell collectives. NCAM may be a key adhesion molecule for keratinocyte segregation in the developing hair bud (Figure 1A) and for segregation and differentiation of connective tissue sheath and DP fibroblasts (Figures 1C, 1E, 1F, 1H, and 1I), which have the unique capacity to induce hair growth (Jahoda and Reynolds 1996). Because NCAM is a crucial factor for activating the fibroblast growth factor receptors on neuronal cells (Saffell et al. 1997), NCAM expression on DP and perifollicular connective tissue sheath fibroblasts may also be employed to induce a higher susceptibility of these mesenchymal cells to morphogens and/or to inductive signals released by the proximal hair follicle epithelium.
