Abstract
We developed a simple method employing the use of flat-embedding techniques on thick frozen sections which allows correlation of light and electron microscopic immunohistochemistry. This method has been particularly useful in visualization of pancreas sections, an adaptation especially important because this tissue is not amenable to conventional vibratome sectioning. In this study we demonstrate the use of this technique to examine the same tissue section at the light and the electron microscopic level while maintaining morphology.
Keywords
I
Until now, most electron microscopic (EM) techniques for visualizing the pancreas have utilized cells and islet co-culture, followed by pre-embedding immunohistochemistry (IHC) and thin sectioning of cell pellets (Hamaguchi and Leiter 1990; Hayakawa et al. 1991; Nagata and Yoon 1992), or postembedding IHC of ultrathin frozen sections from tissue fragments embedded in Lowicryl (Mayer and Bendayan 1997). With these techniques it is not possible to correlate structures seen at the light and EM level within the tissue and they are therefore less useful in examining disease processes.
Flat-embedding techniques were originally developed for Golgi electron microscopy (Somogyi 1978) and adapted for high-resolution immunohistochemistry in brain (Leranth and Nitsch 1994). Routinely, 50-60-μm sections are cut on a vibratome, free-floated in buffer, immunostained, and then flat-embedded on liquid release- coated slides (EMS; Fort Washington, PA) and coverslips. These procedures enable easy handling of large sections with minimal loss of potentially valuable material while preserving excellent ultrastructure, thus facilitating light and electron microscopic examination of specifically stained areas. Unfortunately, a limitation of this technique is that soft or fatty tisssues cannot be cut (even if kept in place with gelatin) on a vibratome. More recently, a flat-embedding technique utilizing thin paraffin sections has been described (Hirabayashi and Yamada 1998). Although correlative light and electron microscopic detection of intracellular antigens is possible, this technique lacks the sensitivity to detect cell surface antigens. Therefore, a modification of these techniques using thick frozen sections has been devised which retains the ease of handling tissue sections, detection of cell surface and intracellular antigens, excellent morphology, and correlation of light and immunoelectron microscopy in the same section.
Materials and Methods
Tissue Preparation
Adoptive transfer experiments were performed in which 6-8-week-old NOD/Caj mice were irradiated and CD8 isletspecific cloned T-cells bearing a T-cell receptor (TCR) using the Vβb 6 gene segment were transferred into the mice
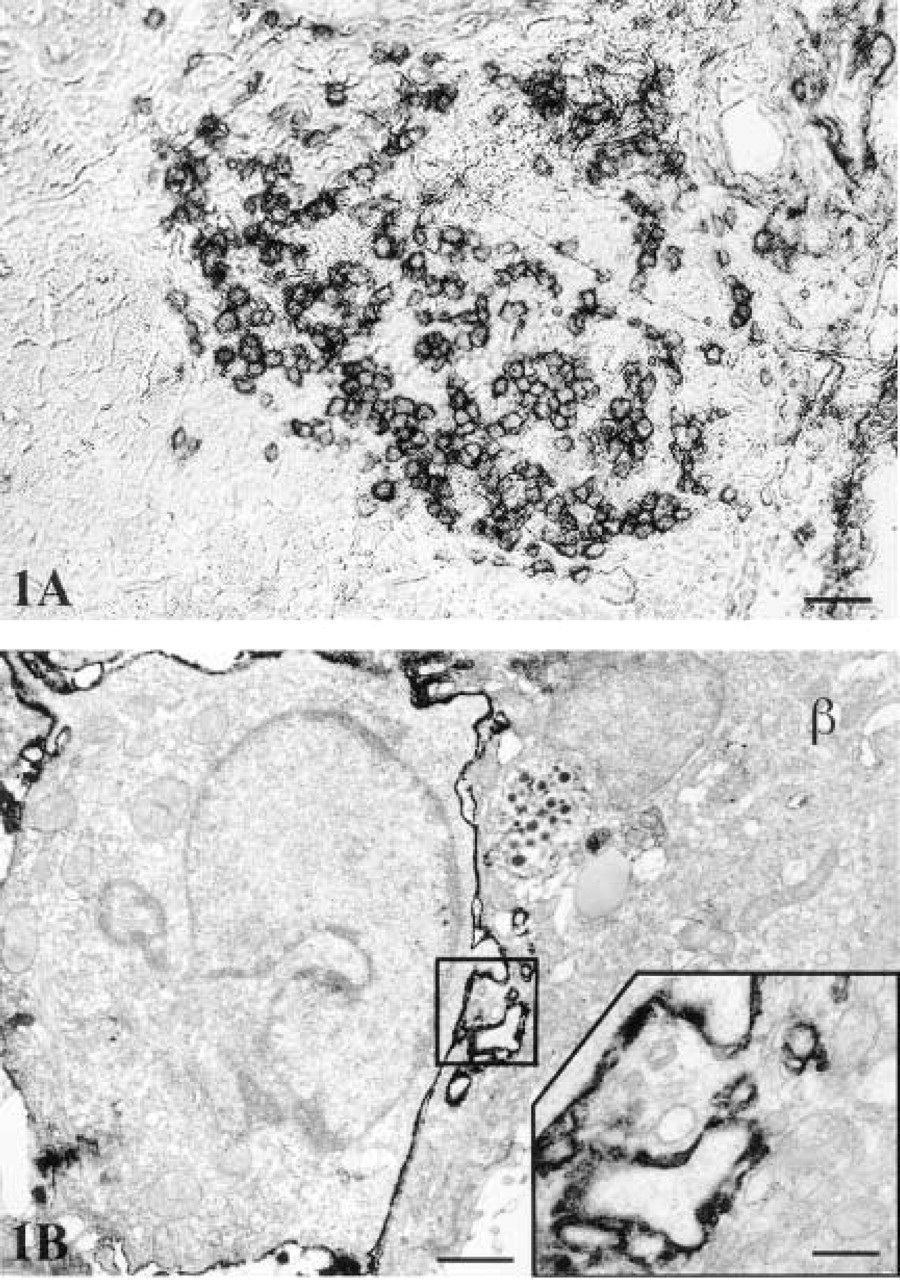
Light (
Immunohistochemistry
Thick (20-25-μm) sections were cut in a cryostat and affixed to Thermanox plastic coverslips (EMS). Immunostaining was performed as previously described (Wong et al. 1996). Briefly, tissue sections were blocked in 3% bovine serum albumin (BSA) solution containing 0.01% Triton X-100 (Pierce; Rockford, IL) in 0.1 M PB. Sections were incubated for 90 min at room temperature (RT) with biotinylated anti-CD8 monoclonal antibody (TIB 105) as primary, diluted in 1% BSA in 0.1 M PB-0.01% Triton X-100 (to demonstrate surface staining of T-cells) or guinea pig antiinsulin polyclonal antibody (Biogenics; San Ramon, CA) as primary for intracellular staining of subcellular particles in pancreatic β-cells. After washing with 0.1 M PB, the sections stained with the anti-insulin antibody were incubated with biotinylated anti-rabbit IgG for 30 min at RT. The sections were further washed with 0.1 M Tris buffer, pH 7.5 and both sets of sections were incubated with streptavidin-HRP (Zymed; San Francisco, CA) for 30 min at RT. The tissuebound peroxidase was visualized with a mixture of 0.5 mg/ml diaminobenzidine (DAB) (Sigma; St Louis, MO) and 100 mg/ml nickel ammonium sulfate (Sigma). The sections were kept at 4C overnight in 2% paraformaldehyde in 0.1 M PB. The coverslips were placed, wet-mounted, on a clean microscope slide and the sections photographed under light microscopy.
Tissue Preparation for EM
The sections were then washed, postosmicated (1% OsO4 in 0.1 M PB for 15 min), dehydrated through increasing concentrations of ethanol (the 70% ETOH contained 1% uranyl acetate), cleared in propylene oxide (PO), and placed in a mixture of PO-Epon 812, 50?50 v/v (EMS). Coverslips were again placed on glass slides for microscopic visualization of islets that were to be ultrasectioned for electron microscopy. Once these areas were identified, as in Figure 2A, the sections were first covered with a drop of 100% Epon 812 and then a gelatin capsule filled with Epon 812 was positioned over the areas of interest. Usually more than one capsule was placed on the section. After polymerization in a 60C oven for 48 hr, the coverslips were removed and the blocks photographed as in Figure 1A for further correlation. Ultrathin sections were cut on a Reichert-Jung Ultracut E (Leica; Deerfield, IL). Grids were observed in a Philips CM10 electron microscope. Further contrasting with lead citrate was unnecessary.
Results
The results demonstrated that this method allows the correlation of both surface and intracellular stain in the sections under light and electron microscopy. The CD8 coreceptor is a cell surface marker. At the light microscopic level, the immunoperoxidase reaction product was ringed in appearance and was associated with the cell membrane of the T-cells (Figure 1A). Correlative EM examination confirmed that the staining of the CD8 co-receptor is indeed specific for the surface membrane of the cloned T-cells and is not found on the pancreatic β-cells (Figure 1B). Pseudopodia from the T-cells were routinely seen extending into the β-cells in the late phases of cytotoxicity (Figure 1B).
In contrast, insulin is an intracellular marker that is present in the secretory granules of pancreatic β-cells. The immunoreaction for insulin at the LM level appeared as patchy cytoplasmic staining (Figure 2A). Electron microscopic observations of the same tissue showed the subcellular localization of insulin to be specific for the secretory granules (Figure 2B).
The ultrastructural preservation was excellent in all of the preparations, as evidenced by completely intact intracellular organelles such as endoplasmic reticulum, lysosomes, and mitochondria, and by undamaged cellular and nuclear membranes (Figures 1 and 2). The tissue antigenicity was also well preserved, which is clearly demonstrated by the highly specific and intense immunoreaction for both antibodies and particularly for the cell surface marker.
Discussion
This method is an adaptation of standard techniques that allows examinination of the same frozen section at the light and electron microscopic levels. This is important because it enables investigators to examine soft tissues, such as the pancreas, in the natural history of disease or after manipulations. The ability to look at cellular interactions and changes in surface and intracellular markers is very useful in the investigation of pathogenic processes.
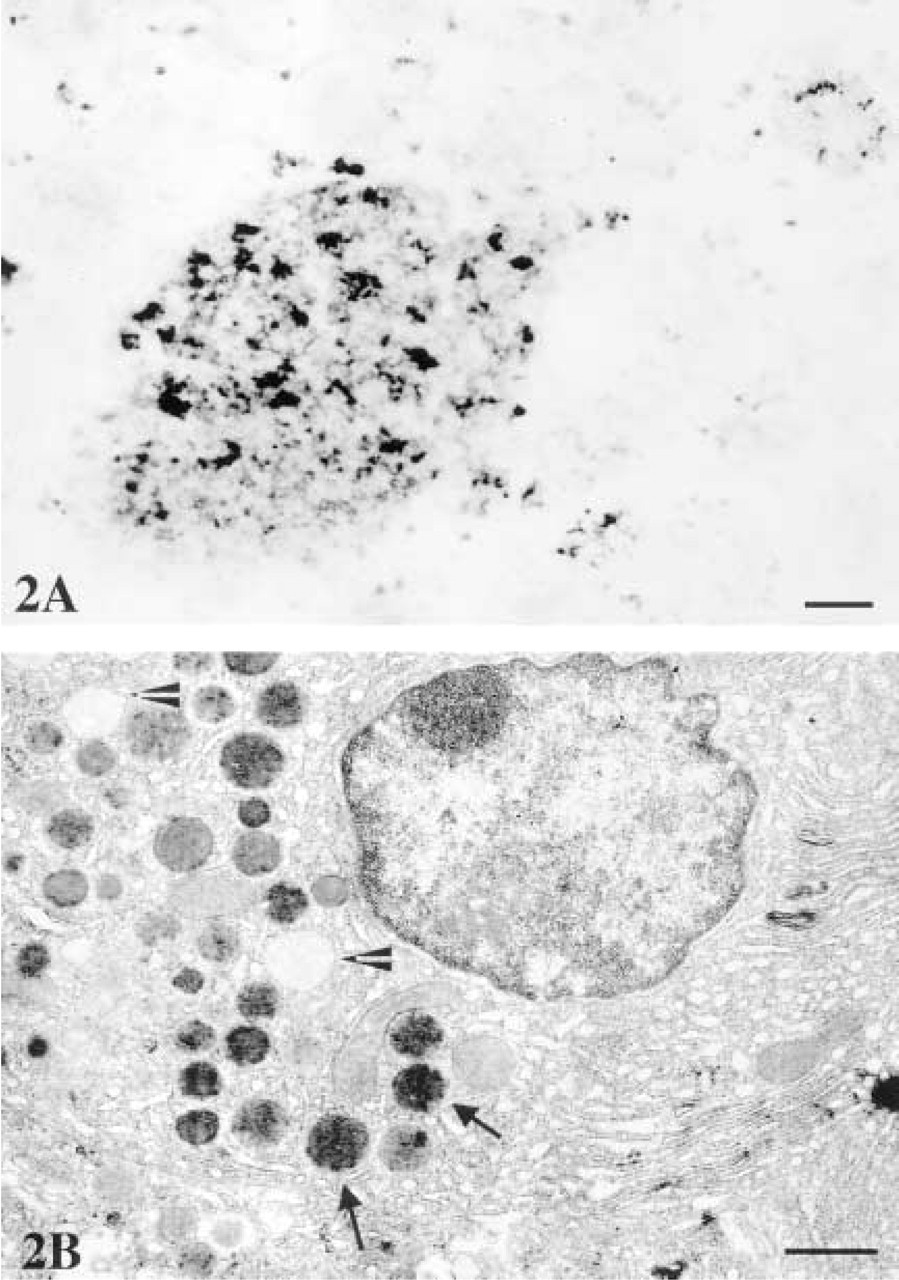
Light (
Essentially, the conditions of tissue preparation are those used for light microscopy, which reduces the time required at the initial stages. Although the frozen sections are thick, this technique allows clear visualization of immunostaining and the ability to verify the specificity of the antibody reaction before postosmication and embedding for EM. In addition, LM photographs can be taken either before or after EM polymerization, and thus the immunohistochemical results can be correlated at the light and electron microscopic levels for both membrane and intracellular staining. Furthermore, the ultrastructural preservation of the tissue is maintained. Fixation of tissue before immunostaining helps retain antigens and preserve morphology. Sucrose infusion and embedding of tissue in an isopentane bath on dry ice aids in reduction of empty spaces around cells caused by intracellular ice crystal formation.
An alternative procedure to use of the vibratome for thick sections is the sectioning of free-floating frozen tissue with a freezing microtome or cryostat (Cote et al. 1993). This was initially attempted for the pancreas using various fixatives. However, we found that whichever fixative was used, once the sections were floated in buffer the tissue fragmented, destroying all morphology. The use of frozen sections does not require the same degree of harsh fixation necessary for vibratome sections, which can be detrimental to the visualization of cell surface antigens. Although Triton-X 100 is not normally recommended for EM preparations, here the low concentration, combined with the use of milder fixatives, allows retention of antigenicity, along with increased antibody penetration, without adversely affecting the ultrastructure.
Major advantages of the use of Thermanox plastic cover slips in this method are that the tissue adheres throughout the entire procedure, there is good visualization by light microscopy, and the tissue is flatembedded on the coverslip itself. After polymerization the coverslip can be easily removed. A similar approach has been used by Yagi et al. (1995), in which frozen sections from the pancreas were placed on glass slides and visualized at LM and EM levels. However, in that study the tissue was removed from the glass slide using ether and re-embedded for electron microscopy, a procedure no longer necessary when our technique is used.
We believe that the modifications that we have made in this study advance our ability to correlate immunohistochemistry at the LM and EM level while maintaining good morphology and definition of ultrastructure at the EM level. This approach will be generally important in the analysis of cellular interactions and the localization of surface and intracellular antigens after immunostaining in soft tissue.
Footnotes
Acknowledgments
Supported by a Career Development Award from the Juvenile Diabetes Foundation International to FSW and the National Institutes of Health grant PO1-DK53015 to CAJ. CAJ is an investigator of the Howard Hughes Medical Institute.
We gratefully acknowledge the assistance of Hannah Janeway in the preparation of this manuscript.
