Abstract
Immunocytochemical localization of immediate early gene proteins, such as Fos, provides a powerful tool with which to demonstrate activated neuronal populations in response to specific stimuli. In contrast to studies using rat brain tissue that consistently show good Fos detection with a variety of antisera, studies using brain tissue from other species yield variable Fos detection. This may be partly due to differences in Fos protein sequences among species or to perfusion and fixation methods. To determine the ability of various Fos antisera to detect neuronal activation in nonhuman primate tissue, we tested nine Fos antisera and compared these antibodies under conditions of intense or physiological stimulation. Monkey brain tissue was either perfused and postfixed with 4% paraformaldehyde or perfused with 4% paraformaldehyde and postfixed with 2.5% acrolein in 4% paraformaldehyde. In rat tissue, stained for comparison, several antisera resulted in good to excellent Fos detection. However, few antisera tested in monkey tissue resulted in excellent Fos staining. We demonstrate that detection of Fos in monkey brain tissue perfused with 4% paraformaldehyde can be improved by postfixation in a dilute acrolein solution. Our findings emphasize the importance of choosing appropriate antisera and perfusion–fixation procedures to optimize Fos detection in nonhuman primate tissue.
Keywords
I
In addition to Fos, there are other chemically similar proteins. These Fos-related antigens, or FRAs, are formed by different immediate-early genes but share sequence homology with Fos protein. Staining with antisera generated against the M-peptide region of Fos has revealed widespread immunopositive neuronal staining in brains of control animals, in contrast to staining with an N-terminally directed Fos antiserum, which has shown little or no baseline Fos expression (Dragunow and Faull 1990; Jacobson et al. 1990). Therefore, certain Fos antibodies can conceivably detect Fos and FRAs, and this crossreactivity may partly account for the differences in Fos detection of various antisera.
In contrast to the many studies of Fos detection under intense stimulatory conditions, few studies have measured Fos activation under physiological levels of stimulation (Rea 1989; Rusak et al. 1990; Lee et al. 1990,1992; Hoffman et al. 1991,1993a,b). These studies have demonstrated that certain physiological stimuli can induce Fos expression in appropriate brain regions. Recent studies have demonstrated that, under other physiological conditions, a scattered distribution of less intensely activated neurons is found in specific regions of the central nervous system, suggesting that the use of highly sensitive antisera may be necessary to detect neuronal activation at these low levels of Fos induction (Herdegen et al. 1993,1995).
In addition to the antisera used, Fos staining can be markedly influenced by the type of fixative used during tissue preparation (Sharp et al. 1991; Hoffman et al. 1992,1993b). Buffered 4% paraformaldehyde has been widely used by investigators measuring Fos immunoreactivity under a variety of stimulatory conditions, including seizures (Morgan et al. 1987), water deprivation (Sagar et al. 1988), acute hyperosmolality (Giovannelli et al. 1990; Sharp et al. 1991; Oldfield et al. 1991,1994; Larsen and Mikkelson 1995), electrical or noxious stimulation (Hunt et al. 1987; Sagar et al. 1988; Ceccatelli et al. 1989; Bullit et al. 1992), brain injury (Dragunow and Robertson 1988), and growth factor or hormone administration (Curran and Morgan 1985; Hoffman et al. 1990; McKinley et al. 1992; Wu et al. 1995). Using this fixative, tissue morphology was generally well preserved and protein crosslinking did not appear to hinder antisera penetration.
The primary goal of the present study was to identify Fos antisera that would be most effective in detecting Fos expression in monkey brain tissue, for future use in experiments examining physiological changes in neuronal activation. To further improve Fos staining, the addition of acrolein to the fixation perfusate has been employed by several investigators (Hoffman et al. 1991,1992,1993b). However, the task of perfusing larger animals, such as nonhuman primates, with an acrolein solution is potentially hazardous because it can expose the investigators to large amounts of this highly toxic substance. Considering this limitation, in the present study we examined the effect of postfixation in a dilute acrolein solution for potential enhancement of Fos immunocytochemical staining.
Materials and Methods
Animals
One adult female cynomolgus monkey (Macaca fascicularis; 4.6 kg), two juvenile male Rhesus monkeys (Macaca mulatta; 3.5 and 4.9 kg), and three adult male Sprague–Dawley rats (250–300 g) were used in this study. This experiment was conducted within NIH guidelines for animal research and was approved by the Institutional Animal Care and Use Committee of the University of Pittsburgh.
Catheterization
Monkeys that received an IV injection of cholecystokinin before sacrifice had either a femoral or a subclavian venous catheter implanted using sterile surgical techniques, before use in the experimental protocol. After implantation, the free end of the catheter was tunneled
Sacrifice
Seventy-five minutes before sacrifice, a bolus injection of cholecystokinin (CCK, sulfated octapeptide; Research Plus, Bayonne, NJ; 15 mg/kg
Tissue Preparation Protocols
Rats. The ability of various antisera to stain Fos in rat tissue was used to validate our immunohistochemical staining technique, because many other investigators have detected Fos in paraformaldehyde-fixed rat tissue using a wide variety of antisera (Dragunow and Faull 1990; Sharp et al. 1991; Hoffman et al. 1993b). Rats were deeply anesthetized with sodium pentobarbital (100 mg/kg
Monkeys
Protocol I. Monkeys were anesthetized with ketamine hydrochloride (10 mg/kg
Protocol II. A second protocol was used to assess whether fixation with acrolein would enhance Fos staining in monkey brain tissue. Rather than perfusing with an acrolein-paraformaldehyde solution, as in the rat, we minimized investigator exposure to acrolein by immersing blocked monkey brain tissue in a postperfusion fixative of 2.5% acrolein + 4% paraformaldehyde–KPBS solution for 2 hr at 25C before immersion in fresh 4% paraformaldehyde overnight at 25C. For all monkeys used in Protocols I and II, 30-mm-thick sections of the hypothalamus were cut on a freezing microtome and stored in cryoprotectant for at least 1 week before staining.
Immunohistochemical Reagents. Perfusion reagents included paraformaldehyde (#P6148; Sigma Chemical, St Louis, MO), acrolein (#00016; Polysciences) and sodium nitrite (#S2252; Sigma). Reagents used in the immunohistochemical processing included bovine serum albumin (#A7030; Sigma), normal donkey serum (#017-000-121; Jackson ImmunoResearch Laboratories, West Grove, PA), sodium acetate (#S8750; Sigma), sodium borohydride (#S9125; Sigma), Triton X-100 (#T6976; Sigma), secondary antibodies and Peroxidase Vectastain Elite ABC kits from Vector Labs (Burlingame, CA) with 3,3'diaminobenzidine (#32750, Fluka, Lake Ronkonkoma, NY) and nickel sulfate (#N4882; Sigma) used in the chromogen solution. We used the following Fos antibodies for these experiments: Cambridge Research Bio-chemicals, #02019/2059 (Wilmington, DE); Oncogene Sciences, #40890207, #21930201, #3119B, #3117, #2627 (Cambridge, MA); and Santa Cruz Biotechnology, #4-SC52C192 (Santa Cruz, CA). The Niman antibody (#4114E10) was a gift from Dr. Henry Niman (University of Pittsburgh, PA). The Larsen antibody (#94012) was a gift from Dr. Philip J. Larsen (Panum Institute, Copenhagen, Denmark).
Immunohistochemical Processing.
The immunocytochemical procedure was described previously by Hoffman et al. (1992), although antibody concentrations and duration of exposure to chromogen varied depending on the primary antibody. Briefly, tissue sections were rinsed with KPBS to remove residual fixative and cryoprotectant. Tissues that had been fixed or postfixed with acrolein were incubated in 1% sodium borohydride in KPBS for 20 min to neutralize the acrolein, then rinsed thoroughly with KPBS. All tissue sections were then incubated for 48 hr at 4C in primary antiserum, appropriately diluted in KPBS containing 0.4% Triton X-100. These initial steps were modified for tissue stained with Larsen antibody (#94012; Rinaman et al. in press). Specifically, tissue was rinsed with KPBS, incubated in a solution of 0.5% sodium borohydride in KPBS for 30 min, then rinsed thoroughly with KPBS. Tissue was then incubated for 10 min in 0.3% hydrogen peroxide in KPBS and rinsed thoroughly with KPBS. The final modification included a blocking step for this bovine serum albumin-conjugated antibody. Tissue was incubated in 1% bovine serum albumin, 0.3% Triton X-100 in KPBS for 30 min, then placed in primary antiserum appropriately diluted in the 1% bovine serum albumin, 0.3% Triton X-100, and 1% normal donkey serum in KPBS for 48 hr at 4C. After these initial modifications, all tissue received similar treatment for the remaining immunohistochemical steps.
All tissue sections were removed from primary antiserum, rinsed in KPBS, and then placed in biotinylated secondary antibody (anti-rabbit/mouse/sheep/donkey IgG, depending upon the species in which the primary antibody was raised; Vector Labs; diluted in KPBS + 0.4% Triton X-100 at 1:600) for 1 hr. Sections were rinsed in KPBS and incubated for 1 hr with avidin–biotinylated peroxidase complex (45 ml of both Solution A and B in 10 ml of KPBS + 0.4% Triton X-100; Vectastain Elite ABC Peroxidase #PK6100; Vector Labs). After incubation, tissue was rinsed in 0.175 M sodium acetate. The Fos antibody–peroxidase complex was stained with a solution of nickel sulfate (0.250 g), 3,3'-diaminobenzidine (0.002 g), and hydrogen peroxide (8.3 ml of 3%) in 10 ml of 0.175 M sodium acetate buffer, with the reaction proceeding for 5–60 min. Controls for the specificity of the antisera consisted of incubation of tissue in anti-sera that had been preabsorbed with antigen which blocked all staining (Hoffman et al. 1990).
Immunohistochemical Analyses.
The first goal of this project was to determine the ability of various Fos antisera to detect neuronal activation under intense stimulatory conditions (i.e., after a bolus injection of CCK in the monkey or hypertonic saline in the rat). We have substantial experience with Fos induction in the hypothalamic magnocellular system under these conditions and we used this neuronal system as a basis to compare the staining abilities of the various Fos antibodies. Tissue sections containing the supraoptic (SON) and paraventricular (PVN) nuclei were stained to analyze Fos induction in magnocellular neurons (4–6 sections/nucleus in monkeys, 2–3 sections/nucleus in rats). To analyze Fos induction in the PVN and SON and the presence of nonspecific binding, we used a semiquantitative approach based on the following criteria: excellent staining (+ + +), reflecting staining of many Fos-positive cells with very dark, distinct nuclear staining and minimal tissue discoloration or background staining; good staining (++), reflecting staining of many Fos-positive cells with variable intensity, distinct nuclear staining and minimal or low background staining; fair staining (+), reflecting a preponderance of Fos-positive cells showing only light nuclear staining with an increase in background nonspecific staining; poor staining (-), reflecting staining of very few lightly stained Fos-positive cells, only at the highest concentrations of antibody, along with high background staining. Antibodies with “excellent” or “good” staining detected many Fos-positive cells, whereas antibodies with “fair” or “poor” staining detected fewer numbers of cells in these nuclei. Background staining included nonspecific staining around tissue edges, adjacent to blood vessels, and around the third ventricle.
A second goal was to determine the ability of various Fos antisera to detect Fos expression under nonstimulated conditions. We initially analyzed tissue sections containing the PVN and SON from nonstimulated rats and monkeys but observed no Fos staining in these regions. We then analyzed tissue sections that contained the suprachiasmatic nucleus, lateral septum, cingulate cortex, posterior hypothalamus, and the paraventricular nucleus of the thalamus, because these areas typically exhibited minimal Fos expression under nonstimulated conditions.
Results
Protocol I
Rats. Fos staining was detected, in tissue from
Comparison of cFos immunocytochemical staining using two different perfusion fixatives in monkey and rat hypothalamic tissuea
para, paraformaldehyde; PF, postfixed; K = 1000; NT = not tested; staining: + + +, excellent; + +, good; +, fair; -, poor.
Note. Monkey: Fos reactivity was determined in response to
Background staining refers to the non-nuclear staining that includes cytoplasmic staining and nonspecific staining adjacent to blood vessels and at tissue edges.
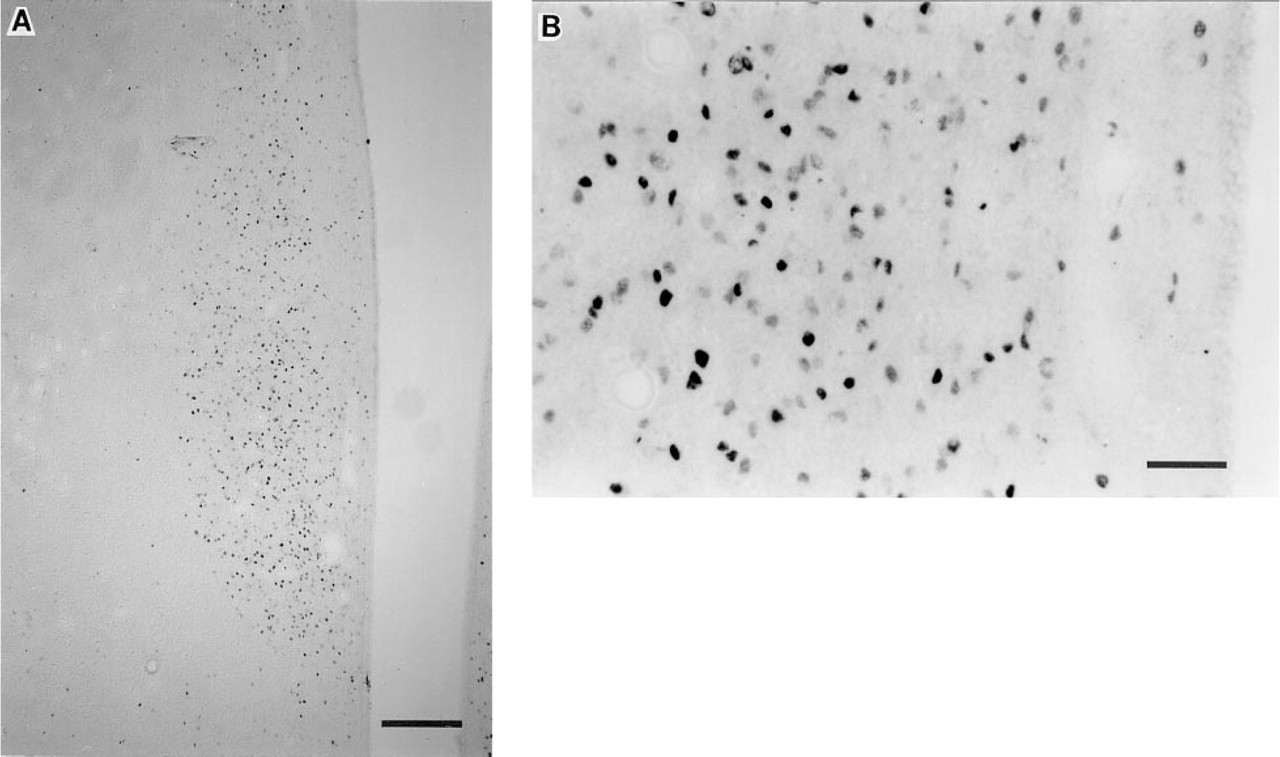
Monkeys. In paraformaldehyde-fixed tissue, excellent Fos staining was observed with LA (#94012), although some background staining was observed at the tissue edges and along several circumventricular organs (Figures 1A and 1B). Three OS antisera (#40890207, #2627, and #3117) resulted in good Fos staining, with low to moderate background staining at the tissue edges and adjacent to the third ventricle. At lower concentrations of these antisera there was less nonspecific binding but also lighter nuclear Fos staining. Fair Fos staining, with moderate to high nonspecific binding, was detected using another OS antibody (#3119B) and the SC antibody. The SC antibody resulted in Fos staining that was of variable intensity, and widespread background staining made it difficult to distinguish the lighter Fos-stained nuclei. Several antisera, including the OS-21930201, NM, and CRB, showed little or no detection of Fos. Even at high concentrations of these antisera, few Fos-positive cells were detectable and most nuclear staining was very pale. The intense nonspecific binding that was seen in rat tissue with the CRB antibody was also observed in monkey tissue (Figures 2A and 2B). As was the case with rat tissue, for antibodies that stained only lightly for Fos and showed more intense background staining, it was difficult to distinguish distinct Fos-positive nuclei from nonspecific binding.
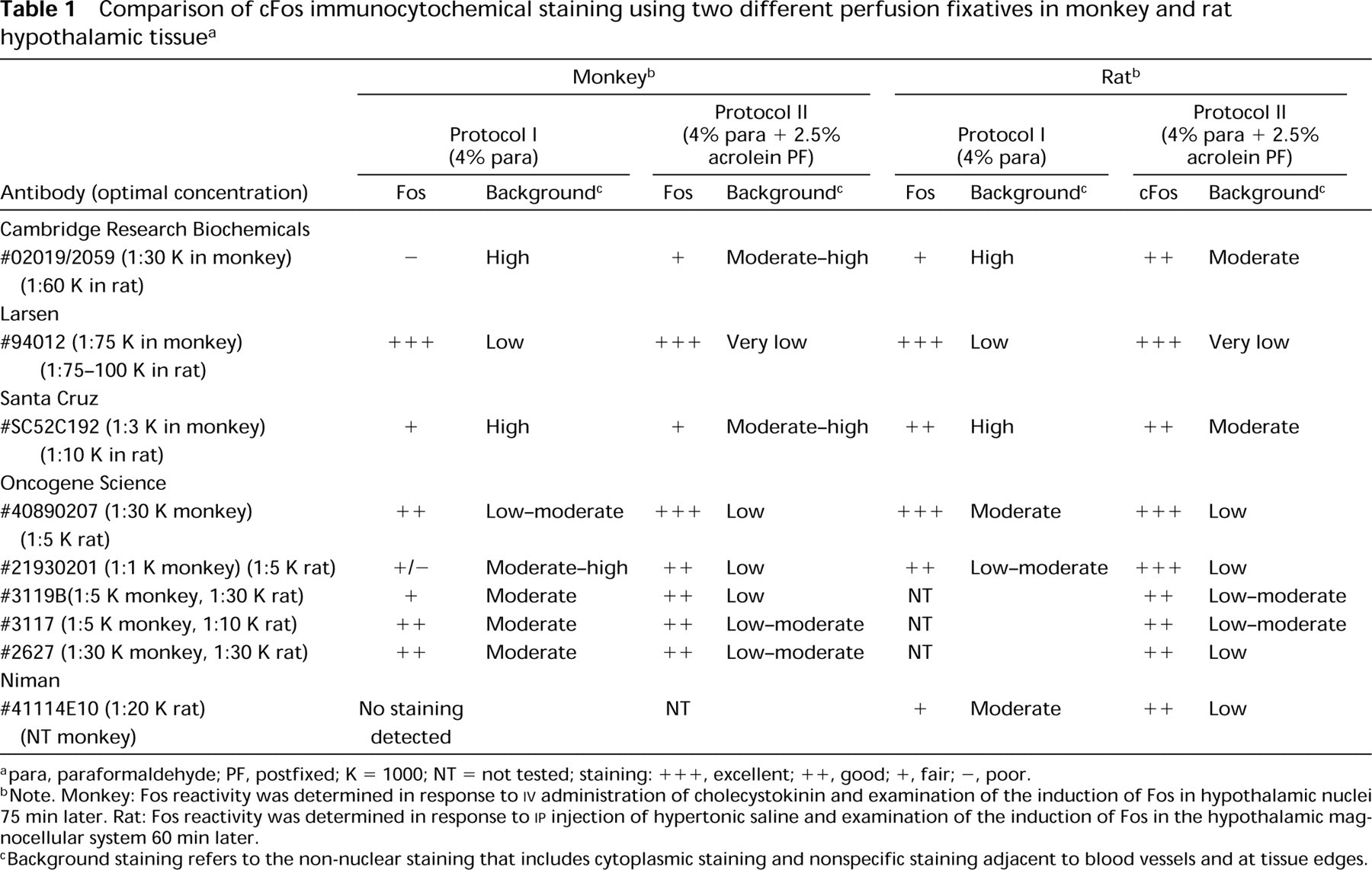
Induction of Fos expression in neurons within the paraventricular (PVN) nucleus of an adult female cynomolgus monkey after systemic injection of CCK (15 mg/kg). Paraformaldehyde-fixed tissue sections are stained with Larsen Fos antibody (#94012; concentration 1:75,000). (
In paraformaldehyde-fixed tissue from the nonstimulated monkey, no Fos expression was found in the PVN and SON. Subsequent analysis of sections containing the suprachiasmatic nucleus (SCN), lateral septum, cingulate cortex, posterior hypothalamus, and the paraventricular nucleus of the thalamus revealed only minimal Fos expression in the nonstimulated condition. With the LA antibody (#94012), fair Fos staining was observed scattered in neurons in these hypothalamic areas, with slight background at tissue edges. After prolonged exposure to the chromogen, only one other antiserum (Oncogene Science #40890207) detected this specific Fos pattern under Protocol I conditions, but this staining procedure also resulted in increased background staining.
Protocol II
Rats. Perfusing rats with 2.5% acrolein–4% para-formaldehyde improved the Fos staining for most antisera and reduced the nonspecific binding that had been present with Protocol I. Excellent staining was observed with LA antibody (#94012), two OS antibodies (#40890207, #21930201), and the SC antibody. For these antibodies, Fos staining was noticeably darker than was observed with Protocol I. Three other OS antibodies, not tested in paraformaldehyde-fixed rat tissue with Protocol I, were tested in Protocol II and resulted in good Fos staining, with low to moderate background staining. The SC antibody still showed moderate nonspecific staining after the addition of acrolein, unlike the OS antisera, whose staining markedly improved after acrolein treatment. In contrast to the poor specific staining of the NM and CRB antisera with Protocol I, acrolein markedly improved the staining abilities of these two antibodies and reduced the level of background staining. With the NM and CRB antisera, nonspecific staining and nuclear Fos staining were diminished by acrolein treatment as well. This was particularly evident at the lower antisera concentrations.
Induction of Fos expression from adjacent paraformaldehyde-fixed tissue sections within the PVN of the same monkey, as previously described in Figure 1. Micrographs depict sections stained with Cambridge Research Biochemical (CRB) Fos antibody (#02019/2059; concentration 1:15,000). (
In acrolein–paraformaldehyde-fixed tissue from the nonstimulated rat, no Fos expression was found in the PVN and SON. Subsequent analysis of sections containing the SCN, lateral septum, cingulate cortex, posterior hypothalamus and the paraventricular nucleus of the thalamus revealed neurons with low Fos staining intensity. These neurons were detected with all antisera tested and were visibly weaker than the Fos induced by the hypertonic saline or other exogenous stimuli. The various antibodies differed markedly in the level of background staining in these areas of endogenous Fos expression. With the LA-94012 antibody, good Fos staining was observed in these brain regions, with minimal background staining. The OS antibody (OS-21930201) resulted in good Fos staining in these neural regions, but with intense nonspecific binding around the tissue edges, neuropil and adjacent to the third ventricle. Both the SC and NM antisera showed fair Fos staining in only some of these neural regions, with a range of nonspecific binding intensities. The SC antiserum resulted in high background staining, whereas tissue stained with the NM antiserum showed moderate levels of background staining. With the CRB antibody, even at high concentrations, little or no detection of Fos was observed. With the SC, NM, and CRB antisera, substantial loss of specific Fos staining and nonspecific binding were observed at lower concentrations of antisera.
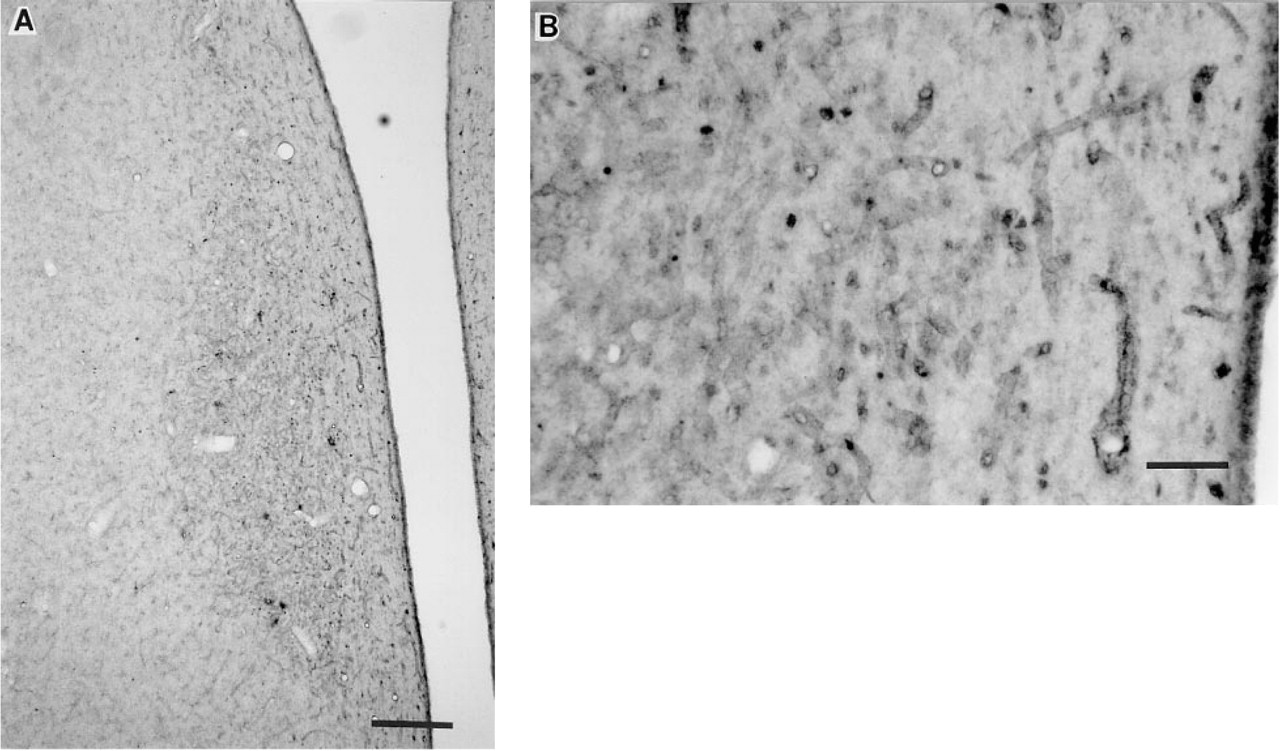
Monkeys. Postfixation with acrolein improved the ability of several antisera (LA-94012, OS-40890207, OS-21930201, OS-3119B, and CRB) to detect Fos and to reduce background staining. Darker, more prominently stained nuclei were readily visible in many areas throughout the hypothalamus, reducing the required exposure time with the chromogen solution. Tissue that was postfixed with acrolein showed a considerable reduction in nonspecific staining, allowing detection of Fos staining in areas in which Fos staining had been difficult to assess with Protocol I. Excellent staining was observed with the LA antibody (#94012) (Figures 3A and 3B) and two OS antibodies (#40890207, #21930201). There was a dramatic contrast between the darkly stained nuclei and the faint background staining with LA-94012 and OS-40890207, whereas the other OS antiserum (#21930201) resulted in moderate nonspecific binding, particularly at tissue edges and in the median eminence. The ability of OS-3117B to detect Fos improved, and nonspecific binding was markedly reduced with the acrolein postfixation. The staining ability of two OS antisera (#3117 and #2627) and the SC antiserum was not improved with acrolein postfixation, although nonspecific binding was reduced for these antisera. CRB antiserum staining was markedly improved and nonspecific binding was reduced with acrolein postfixation (Figures 4A and 4B). Nevertheless, nonspecific staining was still prevalent in tissue stained with both the SC and CRB antisera, and background staining was further diminished at lower concentrations of antisera. Two-hr immersion of tissue in 2.5% acrolein + 4% paraformaldehyde did not result in tissue damage or loss of Fos antigenicity.
Using Protocol II we tested one of the most effective Fos antisera (LA-94012) in tissue from the nonstimulated monkey and observed no Fos expression in the PVN and SON. However, good Fos staining was observed in the SCN, lateral septum, cingulate cortex, posterior hypothalamus, and paraventricular nucleus of the thalamus, with minimal background staining.
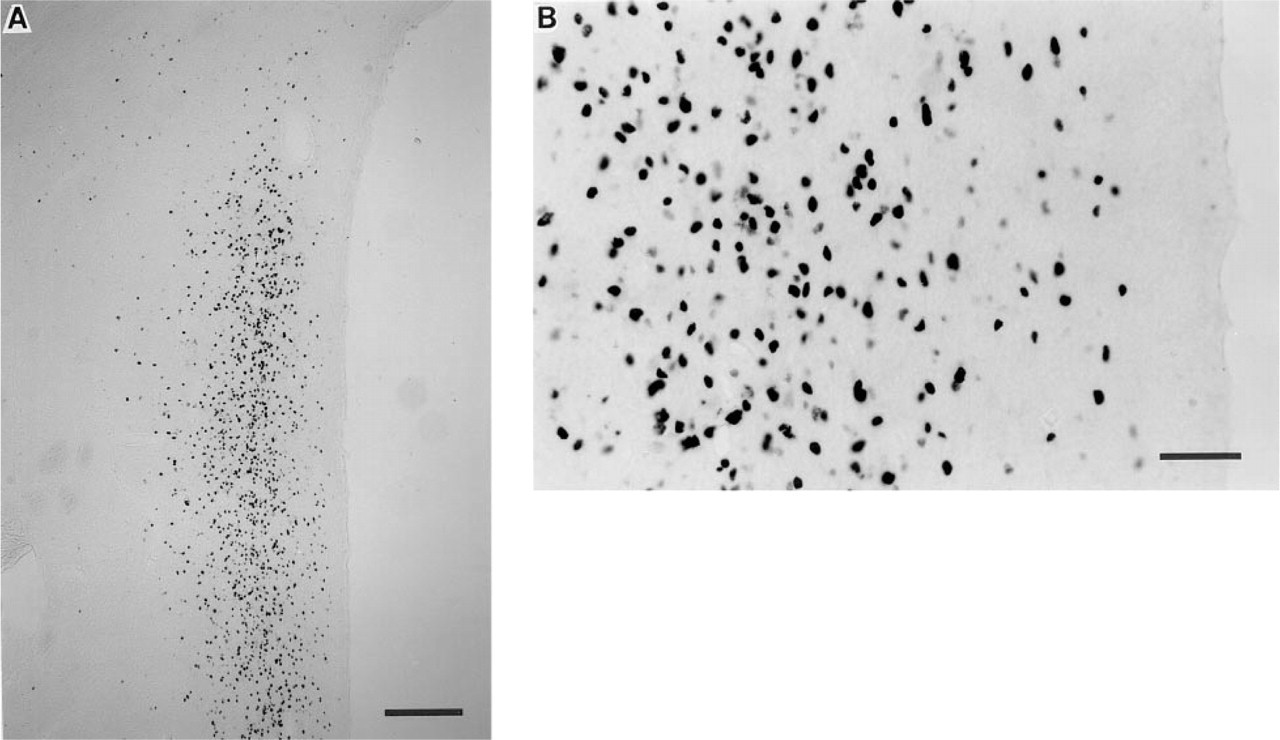
Induction of Fos expression in neurons within the PVN of a juvenile male Rhesus monkey after systemic injection of CCK (15 mg/ kg). Micrographs depict acrolein-postfixed sections stained with Larsen Fos antibody (#94012; concentration 1:75,000). (
Discussion
In this study we have shown that there is substantial variation in the staining abilities of the various Fos antisera that were tested. We observed staining that ranged from “excellent” detection of many intensely stained cells, with little background staining, to “poor” detection of a few lightly stained neurons, with high levels of nonspecific binding, which in some cases obscured detection of specific staining. We consistently detected “good” to “excellent” Fos staining using the LA-94012 antiserum, and several OS antisera (i.e., #40890207, #21930201, #2627), in both rat and monkey brain tissue. The SC antibody also showed excellent staining with rat tissue. The efficiency of virtually all antisera tested in rat tissue perfused with 4% paraformaldehyde–KPBS was consistent with several Fos immunocytochemical studies in rodents (Hoffman et al. 1992,1993b; Sharp et al. 1993; Hughes and Dragunow 1995), as well as in several other species including the mouse, hamster, vole, sheep, and chicken (Berriman et al. 1992; Wu et al. 1992; Moenter et al. 1993; Kirkpatrick et al. 1994; McCabe and Horn 1994; Grosse et al. 1995; Kollack–Walker and Newman 1995; Schneider et al. 1995). In contrast, the same Fos antisera tested in monkey brain tissue that had been perfused in 4% paraformaldehyde–KPBS resulted in less consistent Fos staining, which was often difficult to discriminate from background staining. Several other investigators have previously reported variable Fos staining with low to moderate levels of nonspecific staining in nonhuman primate tissue fixed in 4% paraformaldehyde (Verbalis et al. 1993; Witkin et al. 1994; Michael et al. 1996). Studies in human central nervous system (CNS) tissue are quite rare but also have reported inconsistent detection of Fos expression. In a study of patients after neurosurgical trauma, Dragunow et al. (1990) reported that resectioned brain tissue revealed distinct nuclear Fos immunoreactivity, with moderate nonspecific staining, in tissue specimens that had been submerged in 4% paraformaldehyde within 3 hr after surgical removal. These authors used an antibody directed against the M-peptide region of Fos rather than the N-terminus, and therefore may have detected FRAs in addition to Fos. In contrast, a postmortem study of Alzheimer's disease patients and nondiseased subjects of similar ages observed intense specific nuclear Fos staining, along with a preponderance of non-neuronal staining within glial cells and neuropil in many CNS regions (Zhang et al. 1992). Moreover, no Fos staining was detected in the brainstem of any specimen. It was postulated that the widespread staining of glial cells and neuropil, usually not seen in rodent brain tissue, may have been caused by the delay between death and fixation of the brain tissue (in 4% paraformaldehyde), which ranged from 4 to 21 hr. However, it is difficult to speculate about the cause of the variable staining observed in these studies, because many factors may have contributed to the atypical Fos staining (e.g., brain injury associated with death, disease state, or aging; extended interval between death and fixation). The reason for less consistent staining ability of Fos antisera in the primate and nonhuman primate brain tissue remains unknown, but less than optimal staining results may be related to the presence of structural proteins which, if remaining unbound, may interfere with the specific binding of the antisera. The improved staining abilities of several Fos antisera used in acrolein-postfixed monkey brain tissue partially supports this speculation.
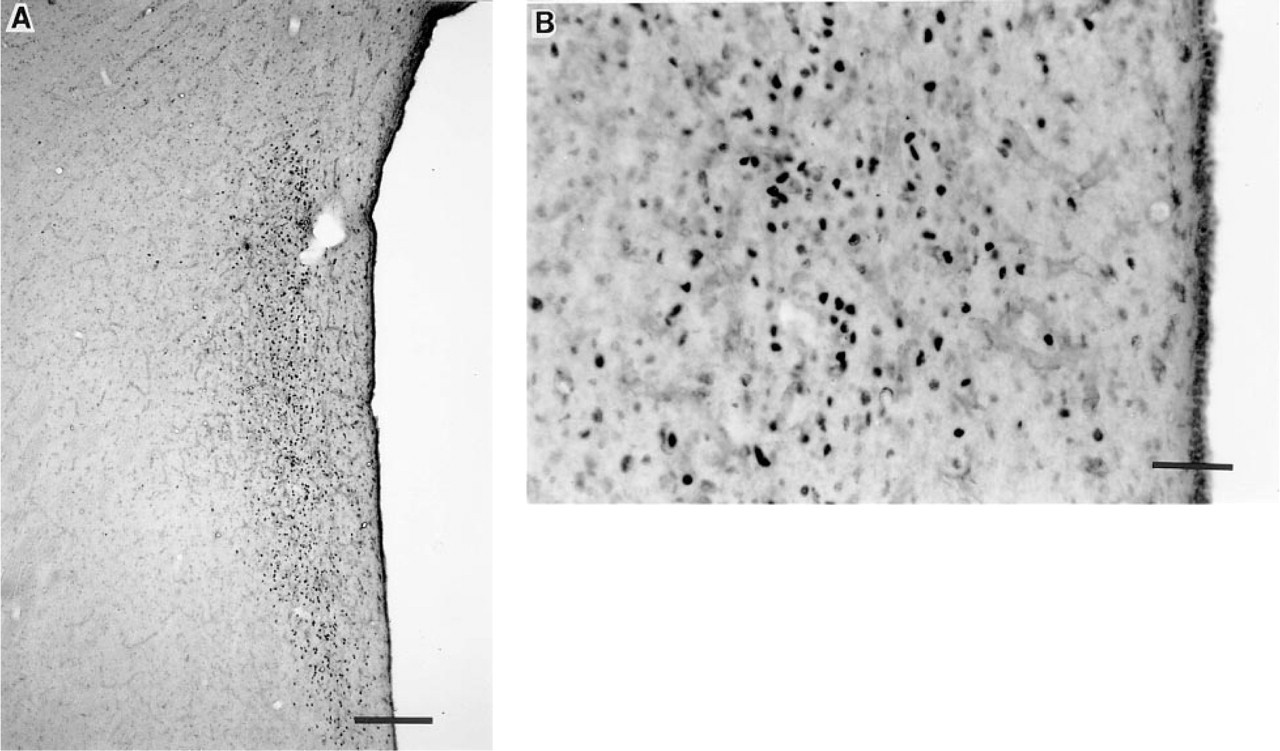
Induction of Fos expression from adjacent acrolein-postfixed tissue sections within the PVN of the same monkey, as previously described in Figure 3. Micrographs depict sections stained with CRB Fos antibody (#02019/2059; concentration 1:15,000). (
An earlier report concerning the use of acrolein indicated that this highly reactive monoaldehyde rapidly penetrates and stabilizes proteins in fixed tissue by binding with the free amino terminals, and that in dilute concentrations during short intervals of fixation acrolein can preserve morphology of the tissue without destroying immunoreactivity (King 1983). We previously observed an overall enhancement of Fos detection in rat brain tissue perfused with a solution of 2.5% acrolein in buffered 4% paraformaldehyde–KPBS using several available Fos antibodies (Hoffman et al. 1992). Many of these antisera produce relatively good staining with moderate amounts of background staining in paraformaldehyde-fixed tissue, but with the addition of acrolein to the perfusate they produce more distinct nuclear staining with less background. In the present study we established a short-duration postfixation step with acrolein to minimize the potentially hazardous exposure to this toxic fixative. As in rat tissue, detection of Fos protein in nonhuman primate CNS tissue can be improved by addition of acrolein, with an increased number of stained neurons as well as a substantial reduction in background staining.
In contrast to the intense Fos induction observed in many neurons in response to specific stimuli, there is generally little Fos expression under nonstimulated conditions. Attempts to analyze brain regions such as the SCN, cingulate cortex, lateral septum, and the paraventricular nucleus of the thalamus, in which basal Fos has reportedly been observed (Rea 1989; Rusak et al. 1990; Hoffman et al. 1991,1993a,b; Lee et al. 1992; Herdegen et al. 1993,1995), remains a difficult process because under nonstimulated conditions Fos staining is usually light and may be easily obscured by nonspecific binding. In this study under nonstimulated conditions, Fos detection was observed in monkey brain tissue with only those antisera that had resulted in dark nuclear Fos staining under stimulated conditions. Under nonstimulated conditions, basal Fos expression was significantly lighter, with increased background staining even in tissue postfixed in an acrolein solution. Our observations suggest that even under conditions in which antisera and fixative have been chosen to optimize the quality of Fos immunoreactivity, there still may be difficulty in detecting Fos expression in nonhuman primate tissue under physiological conditions.
In the present study we tested antisera that were available to us from various companies. Many of these companies have now produced other Fos antibodies, which may not necessarily behave in the same manner as the antisera presented here. Therefore, it is important to test more than one antibody produced by a single company if the initial results were less than optimal.
In summary, our findings emphasize the importance of choosing appropriate antisera and perfusion–fixation procedures to optimize Fos detection in non-human primate tissue.
Footnotes
Acknowledgements
Supported by NIH grants HD20789, HD26888, HD08610, and NS09561.
We would like to acknowledge the generous gifts of anti-sera essential for our studies from Drs Philip Larsen and Henry Niman, and Dr Linda Rinaman for her advice and assistance.
