Abstract
Disappearance of E-cadherin is a milestone for epithelial-mesenchymal transition (EMT), found both in carcinomas and in some fibrotic diseases. We have studied the mechanisms of EMT in oral squamous cell carcinoma (SCC) cells isolated from primary tumor (43A) and its recurrent tumor (43B). Whereas the cells from primary carcinoma displayed a typical phenotype of squamous epithelial cells including E-cadherin and laminin-332 (laminin-5), cells from recurrent tumor expressed characteristics of dedifferentiated, EMT-experienced tumors. 43B cells expressed E-cadherin repressors ZEB-1/δEF1 and especially ZEB-2/SIP1, which therefore appear as candidates for endogenous EMT in these cells. Differences between endogenous and exogenous EMT were assessed by transfecting 43A cells with SNAIL cDNA. SNAIL-transfected cells showed complete EMT phenotype with fibroblastoid appearance, vimentin filaments, E-cadherin/N-cadherin switch, lack of hemidesmosomes and, as a new feature of EMT, lack of laminin-332 synthesis. Upregulation of ZEB-1 and ZEB-2 was evident in these cells, suggesting that SNAIL can regulate these E-cadherin repressors. New monoclonal antibodies against SNAIL showed nuclear immunoreactivity not only in the SNAIL-transfected cells but also in carcinoma cells lacking production of Lm-332 and showing signs of EMT. These results suggest that changes in the epithelial cell differentiation program and EMT in SCC cells can result from the interplay among several E-cadherin repressors; however, SNAIL alone is able to accomplish a complete EMT.
P
Laminins (Lm), the main components of BMs, comprise at least 15 different trimers. Lm-332, previously known as laminin-5, consists of α3, β3, and γ2 chains. Lm-332 is present in most epithelial BMs (Patarroyo et al. 2002; Miner and Yurchenco 2004; Aumailley et al. 2005) and is produced and deposited by most carcinoma cells. Special attention has been devoted to Lm-332 because it has been suggested to play an active role in the invasion of carcinoma cells and may portray a poor prognosis of SCCs (for reviews, see Katayama and Sekiguchi 2004; Miyazaki 2006; Ziober et al. 2006). Carcinoma cells have been suggested to secrete unprocessed Lm γ2 chain or Lm β3γ2 chain dimer, which may contribute to aggressive behavior (Koshikawa et al. 1999; Yamamoto et al. 2001). The processing of α3 and γ2 chains of Lm-332 has been proposed to modulate the migration of carcinoma cells (Patarroyo et al. 2002; Katayama and Sekiguchi 2004; Miyazaki 2006). Expression of Lm γ2 chain has been detected in oral SCCs (Ono et al. 1999; Katoh et al. 2002; Lim et al. 2004). Former studies were usually accomplished with monoclonal antibodies (MAbs) against only the γ2 chain. Distribution of α3 and β3 chains, as well as the role of Lm-332 trimer, remains undefined.
Previous studies have shown that the progression of SCCs takes place by generation of cells with several phenotypic changes leading to an invasive phenotype (Kramer et al. 2005). Although there are reports on the effects of SNAIL on continuous cell lines with diverse genotypes, our aim was to study the progression of oral SCCs using newly established cell lines from primary and recurrent tumors that share similar genetic backgrounds. We also aimed to characterize the expression of E-cadherin repressors in oral SCC cells and to investigate the effects of SNAIL overexpression on SCC cells. For this study, we raised new MAbs against SNAIL, studied several markers of epithelial-mesenchymal transition, and focused on changes in the expression of typical epithelial features, laminin-332, and hemidesmosomal proteins.
Materials and Methods
Permission for the use of patient samples was given by the Joint Ethics Committee of the University of Turku and Turku University Central Hospital, Turku, Finland. Informed consent was obtained from the patients. The Animal Experimentation Committee of the University of Helsinki approved the animal protocols.
Establishment of Oral SCC Cell Lines and Cell Culture
UT-SCC-43A (43A) was established from a primary tumor of a 75-year-old female who had an ulceration in the mandibular gingiva and an ipsilateral metastasis. Histology of the T4N1M0 tumor showed a moderately to well-differentiated SCC. 43A cell line was established from a biopsy using methods described (Takebayashi et al. 2000). The patient was treated with radiotherapy followed by surgery. UT-SCC-43B (43B) cell line was established from the resected recurrent tumor. In Balb/cnu/nu mice (Harlan; Horst, The Netherlands), 43A cells slowly induced a single solid subcutaneous tumor, whereas 43B cells rapidly developed multiple subcutaneous tumors throughout the body (Takkunen M, et al., unpublished data).
43A cells were first studied at passage 4 and the phenotypic properties were maintained for over 50 passages. 43B cells were first studied at passage 3 and further after 15 to 30 passages. BxPC-3, AsPC-1, HPAC, and PANC-1 pancreatic carcinoma cells were obtained from American Type Culture Collection (Manassas, VA). All cells were maintained in RPMI 1640 medium (Sigma; St Louis, MO) with 10% fetal calf serum and antibiotics. For a set of experiments, 43A and 43B cells were cultured in defined serum-free keratinocyte medium (KGM-1; PromoCell, Heidelberg, Germany), and PANC-1 cells were exposed to proteasome inhibitor MG132 (10 μM; Sigma) for indicated time periods.
Immunofluorescence Microscopy
Cells were grown on coverslips and fixed in methanol at −20C. Primary antibodies were applied for 1 hr followed by Alexa Fluor-488 or Alexa Fluor-568 conjugates (Molecular Probes; Eugene, OR) for 30 min. Hoechst 33,258 (DAPI; Riedel-de Haën AG, Seelze-Hanover, Germany) was used to detect the nuclei in a set of double-staining experiments. Table 1 shows the antibodies used in the experiments. Specimens were studied with a Leica Aristoplan microscope, and Leica TCS SP2 system (Leica Microsystems AG; Wetzlar, Germany) was used in confocal microscopy with argon excitation line 488 nm and HCX PL APO CS 63 × 1.40 numerical aperture oil-immersion objective. Image stacks were collected through the specimen using a standardized 120-nm z-sampling density. Selected image stacks were further subjected to deconvolution and restoration using theoretical point spread function and iterative maximum likelihood estimation algorithm (Scientific Volume Imaging BV; Hilversum, The Netherlands).
Antibodies and antisera used a
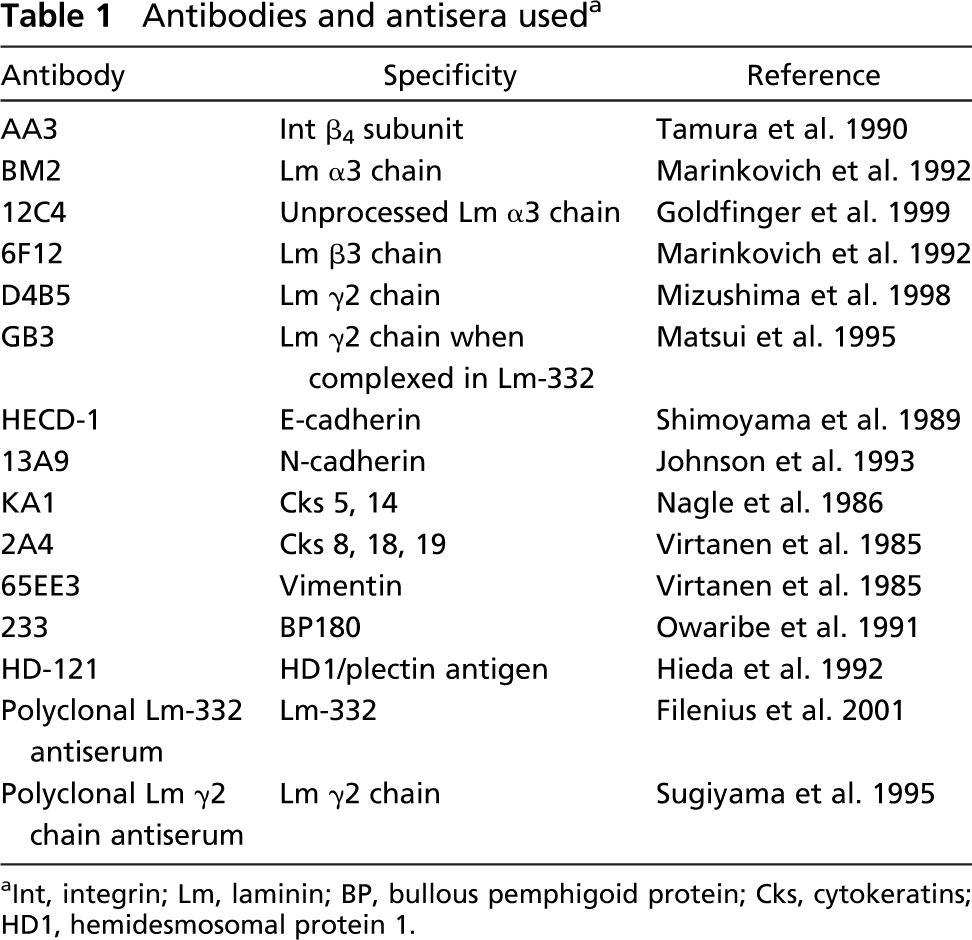
Int, integrin; Lm, laminin; BP, bullous pemphigoid protein; Cks, cytokeratins; HD1, hemidesmosomal protein 1.
Immunoprecipitation and Western Blot Analysis
Immunoprecipitations with [35S]methionine-labeled (Amersham Biosciences; Uppsala, Sweden) cells were prepared from culture medium and extracellular matrix (ECM) material enriched with 0.5% deoxycholate in 50 mM Tris-HCl (pH 7.4). Antibodies were prebound with GammaBind-Sepharose beads (Amersham Biosciences). Alternatively, ammonium sulfate precipitation was used to recover proteins from culture medium. SDS-PAGE followed Laemmli's procedure with 5-8% reducing gels. For Western blots, specimens were transferred onto nitrocellulose membranes by standard methods. Equal loading was verified with Amido Black (Sigma) stainings or with MAb to β-tubulin (a kind gift from Stephen Blose; Cold Spring Harbor Laboratory, Cold Spring Harbor, NY).
Northern Blot Analysis
Northern blot and total RNA isolation followed standard methods (Tani et al. 1997). Poly-A-RNAs were enriched using Dynabeads Oligo (dT)25-beads (Dynal Biotech; Oslo, Norway). cDNA probes were detected with DIG High Prime DNA Labeling and Detection Starter Kit II (Roche; Mannheim, Germany). cDNA probes for Lm α3 (702 bp) (Ryan et al. 1994), Lm γ2 (534 bp) (Airenne et al. 1996), human SNAIL (500 bp) (Batlle et al. 2000), and human SLUG (800 bp) (Domínguez et al. 2003) were used. Full-length cDNAs of murine ZEB-1 (3.5 kb) and human ZEB-2 (3.6 kb) were from Tom Genetta (Children's Hospital, Philadelphia, PA) and Antonio Postigo (Washington University School of Medicine, St Louis, MO), respectively.
Stable Transfections
For transfection experiments, 43A cells were manually cloned by picking single cells under microscopic control. Full-length, hemagglutinin-tagged cDNA of murine SNAIL (Batlle et al. 2000) was transfected to 43A cells with JetPei reagent (Qbiogene; Carlsbad, CA). Transfection efficiency was monitored with MAb to hemagglutinin (Roche). G418 (Sigma) was used for selection (43A-SNA; 200 μg/ml). All experiments were performed with at least five SNAIL-transfected clones as well as with uncloned cells.
MAbs to SNAIL
MAbs against murine SNAIL were raised as partially described recently (Francí et al. 2006). Briefly, female Balb/c mice (Harlan) were immunized SC with 1-2 μg of GST-murine-SNAIL fusion protein in ImmunEasy adjuvant (Qiagen; Hilden, Germany). After three immunizations, tail veins were injected with the soluble antigen, and hybridomas were produced with X63-Ag8.653 myeloma cells. Hybridomas were screened with ELISA using the purified fusion protein and with immunostainings using 43A, 43B, and stably transfected 43A-SNA cells produced in this study. Reactivity of the hybridomas was confirmed with immunoblots of 43 cell lines and permanent SNAIL transfectants, and a 32-kDa polypeptide was found only in the transfected cells (see Results). Hybridomas 173CE2 (IgG2a) and 173EC3 (IgG1) were cloned and cultured as above. Antibodies were purified with GammaBindPlus-Sepharose beads (Amersham Biosciences). Preabsorption of the purified MAbs with GST-tagged recombinant Snail protein abolished the immunoreactions (not shown). Immunostainings of SLUG-transfected cells showed no cross-reaction with SLUG, and mapping of the epitope indicated that MAb EC3 reacts with 1-82 amino acid sequence of the SNAIL protein (Francí et al. 2006).
Preparation of Nuclear Extracts From PANC-1 Cells
Cells were collected by centrifugation, resuspended in ice-cold homogenization buffer (10 mM HEPES-KOH, pH 7.9, 1.5 mM MgCl2, 10 mM KCl, 0.5 mM dithiothreitol, 0.5 mM phenylmethylsulfonyl fluoride), incubated for 10 min on ice, and centrifuged. The pellet was resuspended (0.5% Triton X-100, 50 mM Tris-HCl, pH 7.9) and homogenized with a Dounce homogenizer.
Results
Morphology, Cytoskeletal, and Cell-Cell Adhesion Proteins in 43A and 43B Cells
43A cells grew as epithelioid cell islands, whereas 43B cells showed a more scattered organization with sparse cell-cell contacts (Figures 1A and 1B). Phalloidin labeling showed actin bundles encircling 43A cells (Figure 1C), whereas typical actin stress fibers were found in 43B cells (Figure 1D). 43A cells showed a strong cytokeratin network consisting of cytokeratins (Cks) 8, 18, and 19, typical for simple epithelial cells (Figure 1E) and Cks 5 and 14 (not shown). 43B cells showed a variable, focal immunoreactivity for Cks 8, 18, and 19, and no immunoreactivity for Cks 5 and 14 (not shown). 43A cells presented only occasionally cytoplasmic foci of vimentin without filament formation (Figure 1G), whereas more organized, fibrillar cytoplasmic vimentin filaments were found in 43B cells (Figure 1H).
In accordance with the epithelial morphology, only E-cadherin was detected at the cell-cell junctions in 43A cells (Figure 2A), and only N-cadherin was detected in 43B cells. Western blots confirmed abundant E-cadherin in 43A cells (Figure 2C) with only a weak band corresponding to N-cadherin. In contrast, a prominent band for N-cadherin was detected in 43B cells with no evidence of the presence of E-cadherin. Furthermore, when the cells were cultured in KGM-1 medium, which is used to promote the growth of keratinocytes over non-epithelial cells, 43A cells proliferated and retained all squamous epithelial cell characteristics, whereas 43B cells ceased to proliferate and died within a week (not shown).
Lm-332 and Hemidesmosomal Proteins in 43A and 43B Cells
Lm-332 and hemidesmosomes are typical for all normal squamous epithelial and many carcinoma tissues (Patarroyo et al. 2002; Katayama and Sekiguchi 2004; Miner and Yurchenco 2004). We therefore analyzed the expression of Lm-332 and hemidesmosomal proteins in these cells. 43A cells showed a cell substratumconfined immunoreactivity for deposited Lm-332 (Figure 3A) and for Lm γ2 chain (Figure 3C). 43B cells showed at early passages (p3-6) cytoplasmic Lm-332 (Figure 3B), γ2 (Figure 3E), and β3 chains, whereas only γ2 chain was found in late passage cells (not shown). MAb GB3 against γ2 chain, reacting only when complexed in Lm-332, showed γ2 localization in a typical cell substratum-associated manner in 43A cells (Figure 3D), whereas no immunoreactivity was found in 43B cells (Figure 3F). Among hemidesmosomal proteins, integrin (Int) β4 immunoreactivity was found in a typical granular pattern with numerous hole-like structures (arrows in Figure 3G), generating a “Swiss cheese”-like appearance (Spinardi et al. 1995). Int β4 distribution closely corresponded to that of other hemidesmosomal proteins, HD1/plectin and BP180 (Figure 3G, insets). In 43B cells, only streaks of Int β4 subunit immunoreactivity were found (Figure 3H), HD1/plectin was associated with cytoplasmic fibrils (Figure 3H, inset), and BP180 was absent (not shown). These results suggest that 43A cells synthesize and deposit all Lm-332 chains and, unlike 43B cells, express hemidesmosomal complexes. Early passage 43B cells synthesize and secrete but do not deposit Lm β3γ2 dimer, and late passage 43B cells only synthesize Lm γ2 chain but do not secrete it.
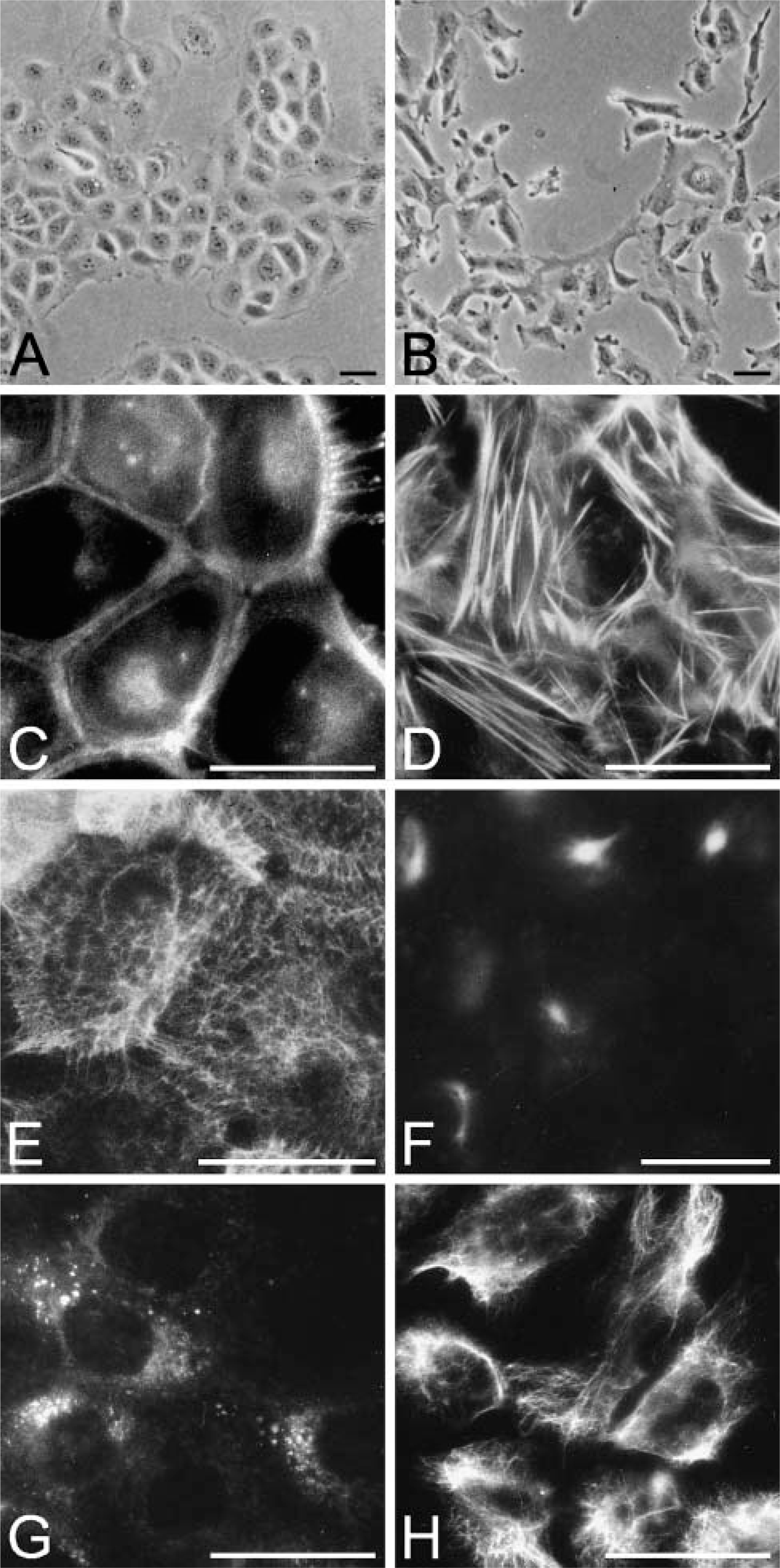
Phase-contrast microscopy of 43A and 43B cells and organization of F-actin and intermediate filaments. Subconfluent 43A cells present an epithelioid appearance (
Production of Lm-332 was next studied by immunoprecipitation (Figure 4A). Culture medium of 43A cells showed (Figure 4A, Lanes 1-3) 165-kDa polypeptide processed α3î chain, 145-kDa β3 chain, 155-kDa unprocessed γ2 chain, and 105-kDa processed γ2î chain (the processed forms are indicated as α3î and γ2î in text and in Figure 4). Instead, early passage 43B culture medium (Figure 4A, Lanes 4-6) contained γ2 and β3 chains, whereas α3 chain was absent. No Lm-332 chains were found in the culture medium of late passage 43B cells (not shown). However, the 43B cell lysate contained unprocessed γ2 chain (Figure 4A, Lanes 7 and 8). Furthermore, 43A culture medium showed α3î chain together with γ2, β3, and γ2î chains, but no unprocessed α3 chain (Figure 4B, Lanes 9 and 10). In detergent-resistant ECM material, the unprocessed 190-kDa α3 chain was also found with the above-mentioned chains, which was confirmed with MAb specific to unprocessed α3 chain (Figure 4B, Lanes 11 and 12). These results suggest that 43A cells synthesize and secrete Lm-332 containing unprocessed and processed α3 and γ2 chains together with unprocessed β3 chain. Instead, early passage 43B cells secreted Lm β3γ2 dimer, but late passage cells expressed only cytoplasmic γ2 chain.
Northern blots of 43A cells confirmed the synthesis of Lm α3 and Lm γ2 transcripts, whereas under the same exposure conditions neither α3 nor γ2 was observed in late passage 43B cells (Figure 4C, Lanes 1 and 2). Only after a prolonged exposure, γ2 mRNA could be faintly detected (Figure 4C, Lane 2), correlating with the finding that the late passage cells could not secrete it.
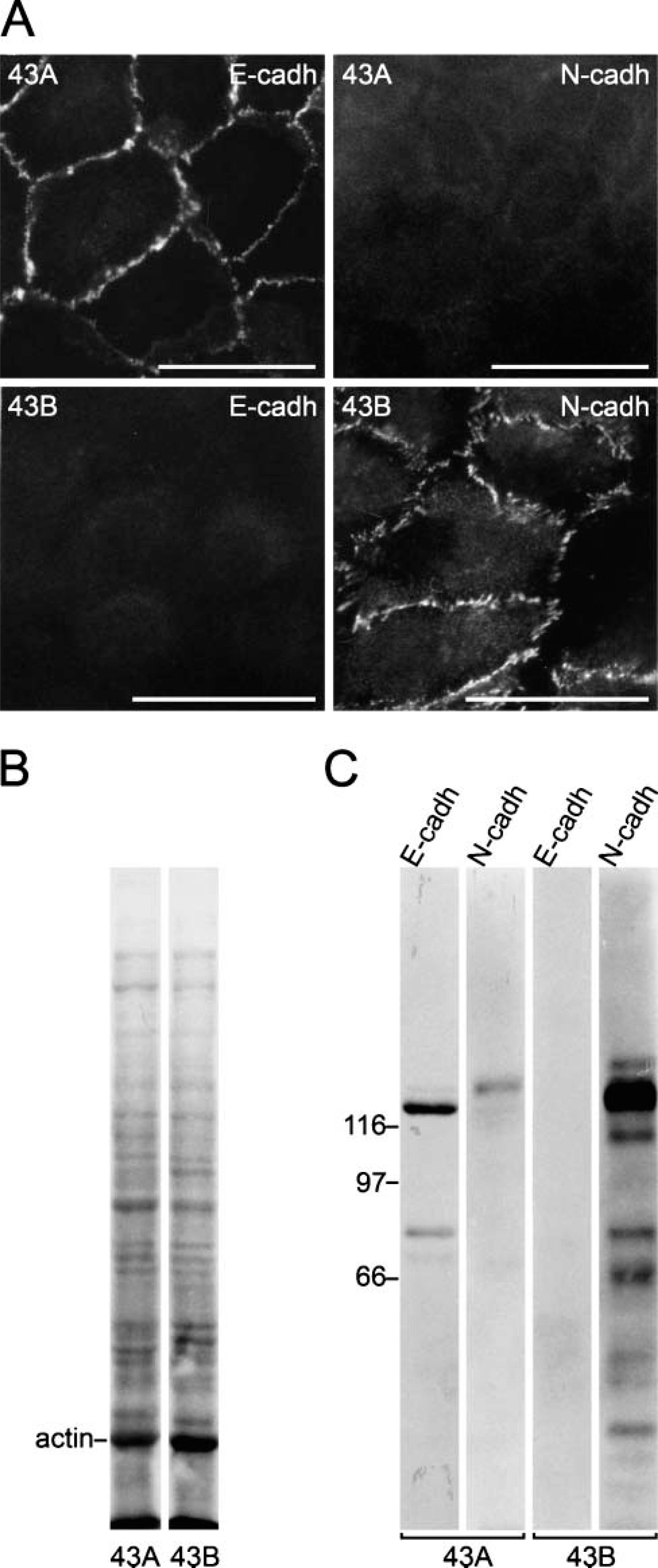
Expression of E- and N-cadherin in 43A and 43B cells. 43A cells present an even distribution of E-cadherin at cell-cell junctional areas (
Induction of EMT by Overexpression of SNAIL in 43A Cells
As reviewed above, SNAIL is able to induce EMT in a variety of carcinoma cells and also in oral SCC cells. To determine its relevance for 43A cells, cells were stably transfected with cDNA of murine SNAIL. Efficiency of SNAIL transfections was determined by double stainings with DAPI and newly raised MAbs against murine SNAIL (Figure 5). SNAIL was detected in the nuclei of interphase 43A-SNA cells, whereas parental 43A cells were negative. In Western blots, MAbs reacted with a 32-kDa polypeptide in 43A-SNA cells, whereas 43A and 43B cells remained negative (Figure 5B).
As a consequence of SNAIL transfection, 43A-SNA cells acquired a fibroblastoid appearance and abundant actin stress fibers. Immunostainings and Western blots showed prominent vimentin fibers; only traces of Cks 8, 18, and 19 (Figure 6); and lack of Cks 5 and 14 (not shown). 43A-SNA cells lacked E-cadherin but expressed N-cadherin at their occasional cell-cell junctions. 43A-SNA cells did not express Lm-332 chains as detected with MAb against Lm γ2 chain (Figure 6A) and with ammonium sulfate immunoprecipitation with polyclonal antibody against Lm-332 (Figure 6B). Furthermore, 43A-SNA cells were negative for Int α6β4 (not shown). These results show that overexpression of SNAIL in SCC cells leads to an extensive loss of epithelial phenotype characteristics and to a complete EMT. These changes exceed those found in endogenous EMT of 43B cells.
Expression of E-cadherin Repressors in SCC Cells
The above results suggesting features of EMT in 43B and 43A-SNA cells led us to study the E-cadherin repressors ZEB-1, ZEB-2, SLUG, and SNAIL in these cells. Northern blots of 43A cells showed a weak band corresponding to ZEB-1, whereas no ZEB-2 was detected (Figure 7). In 43B cells, ZEB-1 and ZEB-2 were present. SLUG was expressed in all cell lines, whereas SNAIL was not detected in either 43A or 43B cells. These results suggest that endogenous EMT that occurred in 43B cells may be as a result of expression of transcription factors ZEB-1 and ZEB-2. 43A-SNA cells showed prominent expression of SNAIL mRNA as expected but also transcripts for ZEB-1, ZEB-2, and SLUG (Figure 7). We found, however, that our SLUG probe also detects SNAIL in 43A-SNA cells, which can be distinguished by its larger size. These results suggest that SNAIL can upregulate other E-cadherin repressors, namely, ZEB-1 and ZEB-2, as they were either absent (ZEB-2) or synthesized only at very low levels (ZEB-1) in the non-transfected cell line 43A. These results also indicate that SLUG is not a major contributor in the progression of EMT in 43 cells.
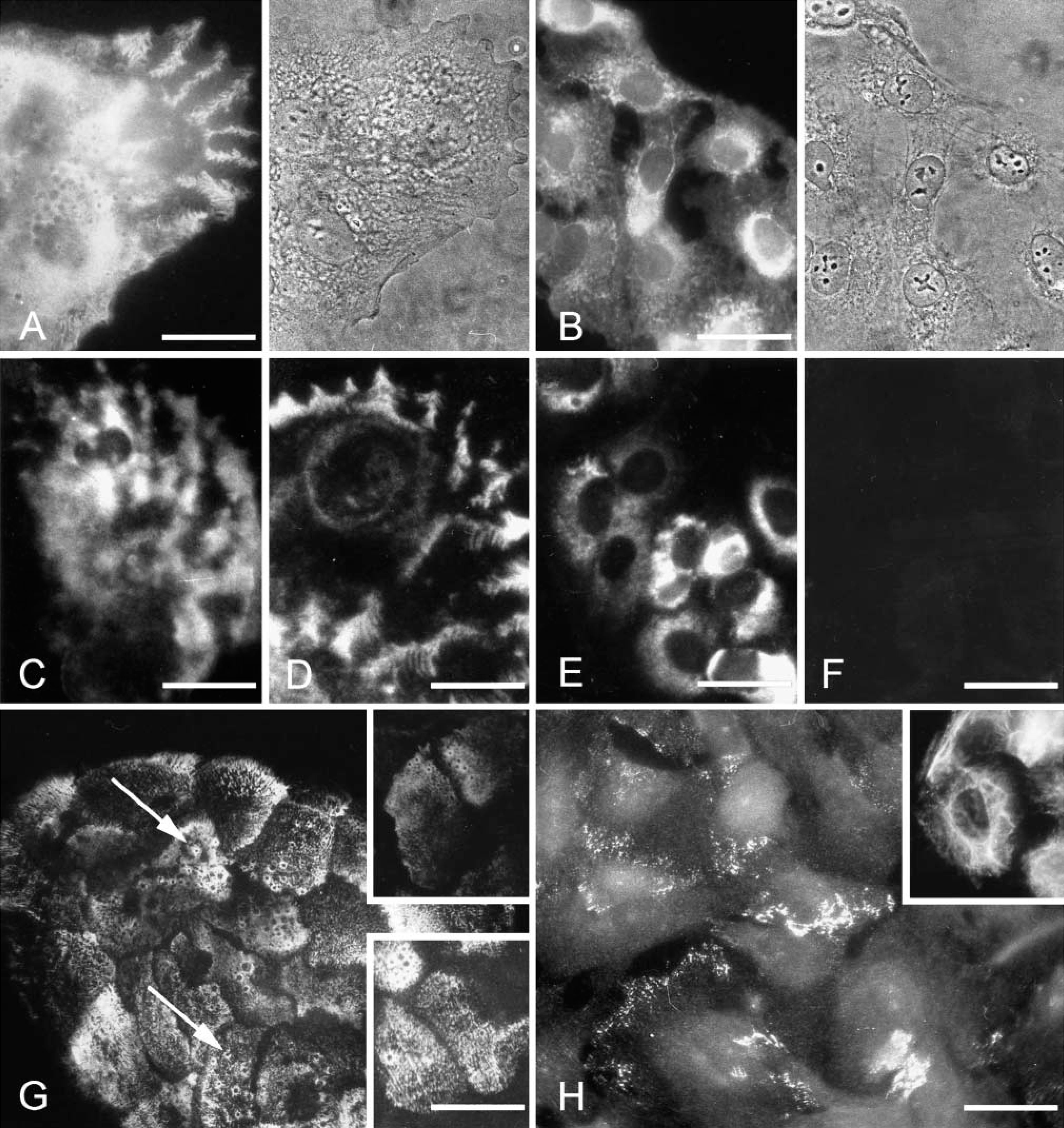
Distribution of laminin-5 and integrin (Int) β4 subunit in 43A and 43B cells. 43A cells show a typical cell substratum-confined immunoreactivity for Lm-332 (
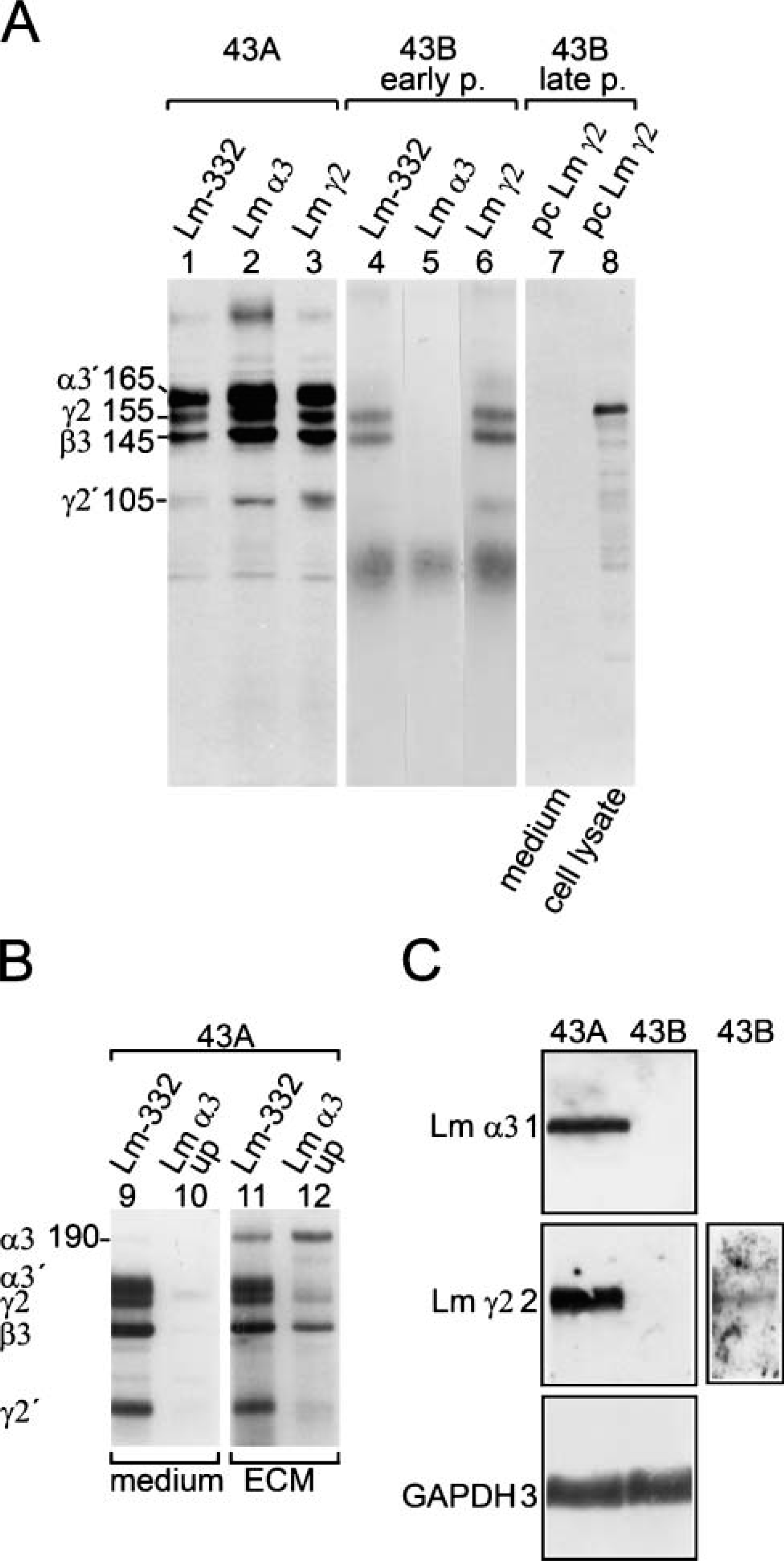
Production of the chains of Lm-332 by 43A and 43B carcinoma cells. Processed forms of Lm-332 chains are indicated by prime. (
Next, we studied whether SNAIL was expressed in carcinoma cell lines showing features of EMT. Pancreatic carcinoma cells AsPC-1, BxPC-3, and HPAC have been reported to secrete all chains of Lm-332 and express hemidesmosomal proteins, whereas PANC-1 pancreatic carcinoma cells lack the synthesis and expression of Lm-332 and express only low levels of Int β4 (Tani et al. 1997; Katayama et al. 2003). Among these cell lines only PANC-1 showed immunoreactivity for SNAIL (Figure 8) and signs of EMT (N-cadherin instead of E-cadherin, lack of hemidesmosomal proteins; not shown), although intensity of endogenous SNAIL was variable and weaker than in SNAIL-overexpressing cells. However, Western blots of PANC-1 nuclear extracts clearly showed the presence of SNAIL (Figure 8C). We then exposed PANC-1 cells to MG132, a proteasome inhibitor that stops degradation of SNAIL (Zhou et al. 2004). Western blots of proteasome inhibitortreated whole-cell lysates showed a clear increase in SNAIL protein after 1, 3, and 5 hr (Figure 8G).
Discussion
Results of the present study suggest that expression of E-cadherin repressors correlates with progression of EMT in oral SCC cells and, further, that overexpression of SNAIL may lead to upregulation of other transcriptional E-cadherin repressors and to a complete EMT. Recent results with invading carcinoma cells have indicated that disturbed secretion of Lm-332 (laminin-5) chains, including expression of γ2 chain, are hallmarks of malignancy for many carcinomas (Patarroyo et al. 2002; Katayama and Sekiguchi 2004; Miyazaki 2006). Present results show that EMT of oral SCC cells is clearly accompanied by a progressive decrease in secretion of Lm-332 chains, which has not been previously reported.
We first studied expression of cadherins in 43A and 43B cells. Several studies have implicated that loss of E-cadherin and neoexpression of N-cadherin could serve as markers for EMT both in vitro and in vivo (Behrens 1999; Tran et al. 1999; Tomita et al. 2000). Loss of E-cadherin in oral SCCs has been shown to correlate with a poor patient prognosis (Chow et al. 2001; Diniz-Freitas et al. 2006). Also, N-cadherin as well as vimentin intermediate filaments have been reported in oral SCCs (de Araujo et al. 1993; Islam et al. 2000; Taki et al. 2006). Our results showed that in association with increasing vimentin content and decreasing content of Cks and hemidesmosomal proteins, 43B cells acquired N-cadherin expression and lost E-cadherin expression. These alterations were accompanied by changes from typical epithelial cell morphology into a more mesenchymal phenotype.
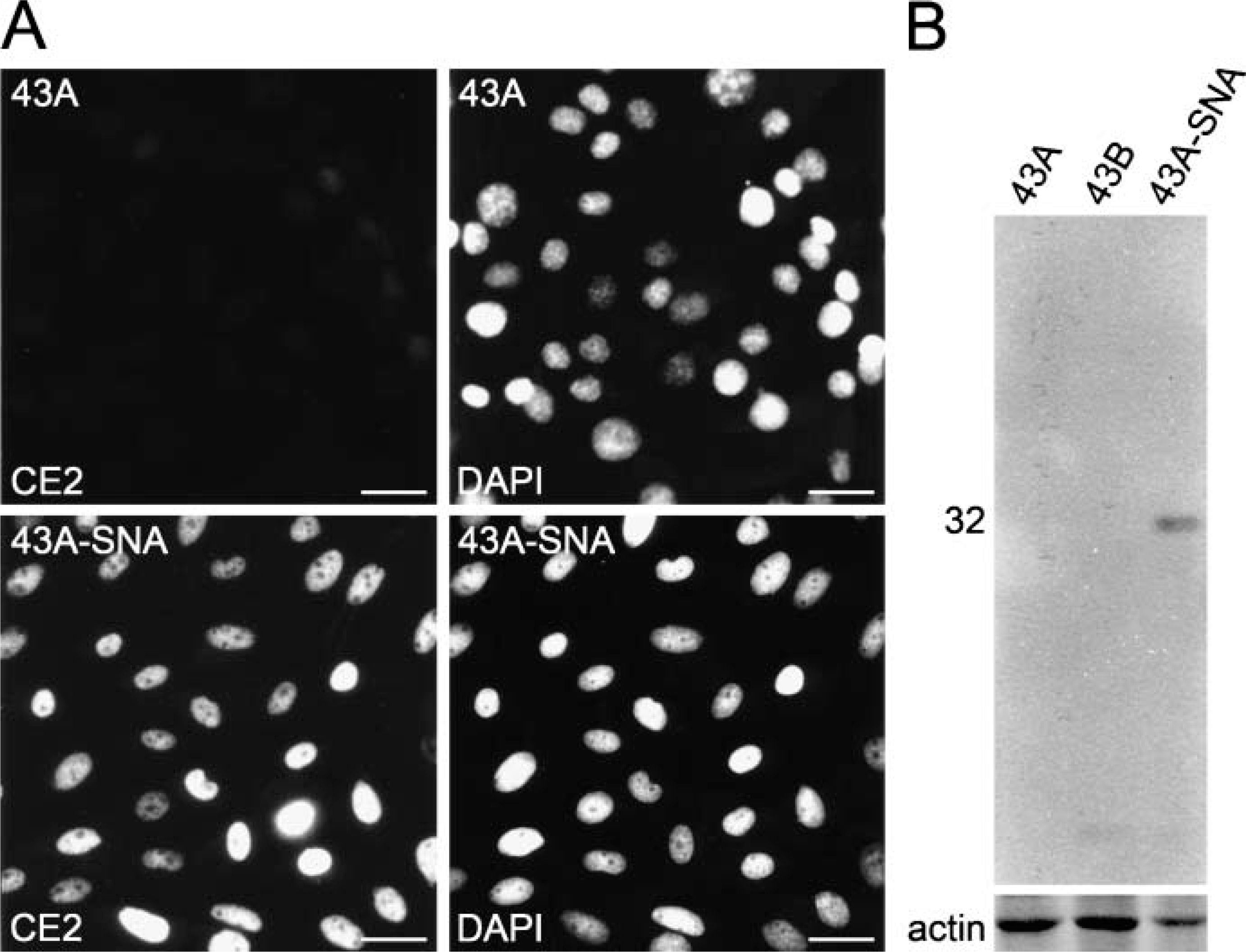
Expression of SNAIL in 43A, 43B, and 43A-SNA cells. (
We then studied expression of E-cadherin repressors in 43A and 43B cells. Northern blots showed that both cell types expressed SLUG; 43A cells expressed low levels of ZEB-1, whereas 43B cells strongly expressed both ZEB-1 and ZEB-2. Neither of the cell types expressed SNAIL. Our results on 43B cells suggest that ZEB-2 especially may induce morphological changes and produce EMT, including loss of E-cadherin and upregulation of N-cadherin. Supporting our findings, ZEB-2 was recently suggested to have an inverse correlation with E-cadherin in oral SCCs (Maeda et al. 2005).
As ZEB-1 and ZEB-2 expression produced an endogenous EMT in our cell system, we transfected 43A cells with the cDNA of SNAIL to investigate differences between endogenous and exogenous EMT. SNAIL has been reported to induce EMT in several cell systems (Batlle et al. 2000; Cano et al. 2000; De Craene et al. 2005a). Our results show that SNAIL transfection augmented ZEB-1 and ZEB-2 levels and induced disappearance of E-cadherin as well as emergence of N-cadherin. Results concerning upregulation of ZEB-1 and ZEB-2 by SNAIL are in line with results from other cell models (Guaita et al. 2002; Taki et al. 2006). Our results suggest that E-cadherin repressors may differentially contribute to EMT of SCC cells, ZEB-1 and ZEB-2 being involved regardless of SNAIL. SNAIL-overexpressing cells lacked Cks, Lm-332 expression, and all hemidesmosomal proteins and were morphologically indistinguishable from cultured fibroblasts. In this respect, endogenous EMT in 43B cells led to attenuated fibroblastoid characteristics when compared with SNAIL-induced EMT.
Int α6β4, a major component of hemidesmosomes, has been suggested to contribute to the invasion of breast carcinoma cells through the PI3-K/Akt pathway (Shaw et al. 1997). SNAIL is induced by the same pathway (Grille et al. 2003), thus providing a possible functional link between proteins. Studies on undifferentiated squamous carcinoma cells have suggested that Int α6β4 has an increased turnover and also that its normally polarized cell surface distribution is lost (Witkowski et al. 2000). A reduction in the Int α6β4 content upon increasing malignancy, as well as a lack of Lm-332 deposition in Ha-Ras-induced EMT of mammary epithelial cells, has been reported (Cress et al. 1995; Maschler et al. 2005). Disappearance of Int α6β4 complex has also been detected in oral SCCs (Downer et al. 1993; Jones et al. 1993). Our results showed that in 43A cells Int α6β4 had a polarized “Swiss cheese”- or “leopard skin”-like localization in type II hemidesmosomal-like plaques colocalized with HD1/plectin and BP180 (Nievers et al. 1999). 43B cells, possibly as a result of ZEB-1 and ZEB-2 expression, lost this association and presented only small streaks of Int α6β4 and a loss of cell surface-associated plectin, which was found in colocalization with cytokeratin fibers (see also Niessen et al. 1997). SNAIL-transfected cells lacked Int α6β4 expression, indicating that suppression of Int α6β4 may be among the many downstream effects of ZEB-1 and/or ZEB-2 and SNAIL. The results suggest that in addition to desmosomes, tight and adherens junctions (De Craene et al. 2005b), also hemidesmosomes are downregulated upon EMT.
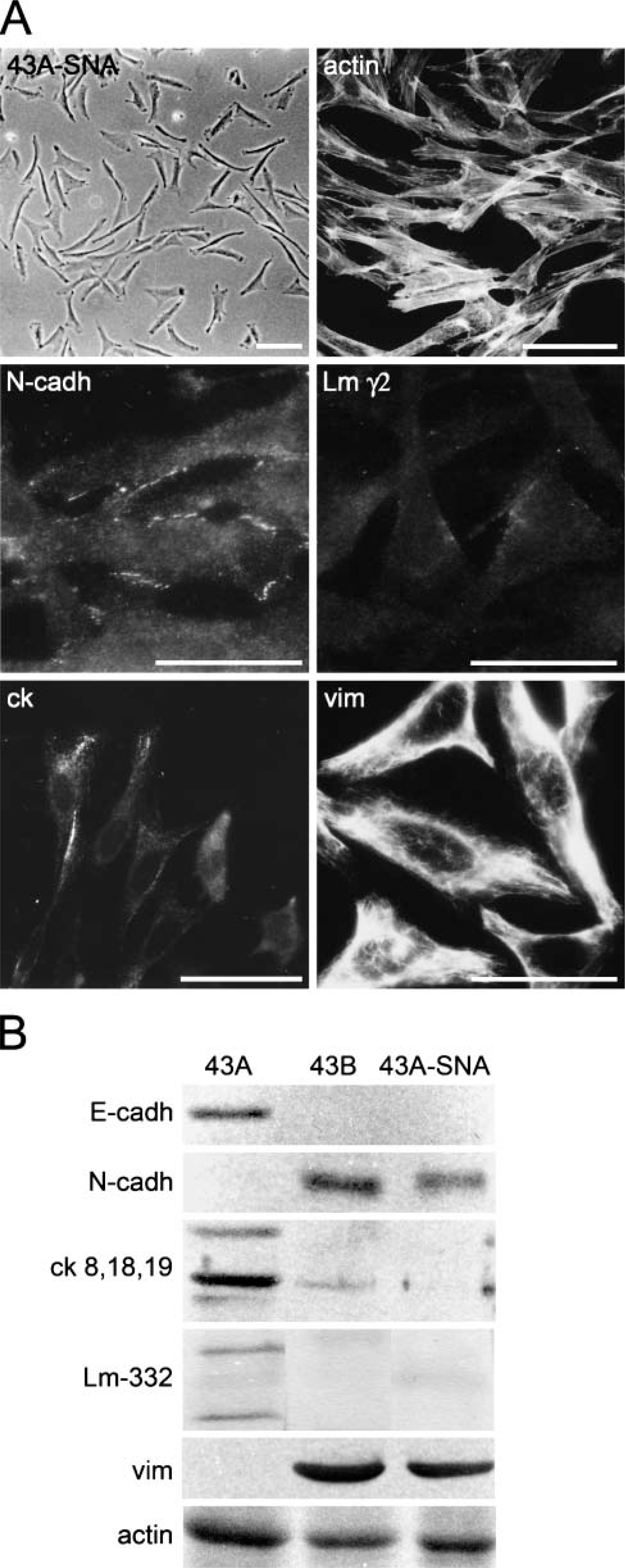
Phase-contrast microscopy, immunofluorescence, and Western blots of SNAIL-transfected 43A-SNA cells. (
Much interest has recently been devoted to the production of Lm-332 by carcinoma cells and its role in the behavior of carcinomas (Patarroyo et al. 2002; Katayama and Sekiguchi 2004; Miyazaki 2006; Ziober et al. 2006). Previous results implicate that continuous production of Lm-332 plays a role in the migration and invasion of carcinoma cells (Tani et al. 1997; for reviews, see Katayama and Sekiguchi 2004; Miyazaki 2006; Ziober et al. 2006). Other studies have suggested that especially invasive carcinoma cells contain abundant cytoplasmic γ2 chain, and that secretion of γ2 chain or β3γ2 dimer might contribute to malignancy (Koshikawa et al. 1999; Yamamoto et al. 2001). It is of note that in most of the numerous studies reporting overexpression of Lm γ2 chain, expression of α3 and β3 chains has not been addressed as critically analyzed by Ziober et al. (2006). Our results partly agree with these suggestions because we found that 43A secreted and deposited all three chains of Lm-332, whereas ZEB-1 and ZEB-2 expressing 43B cells either secreted β3γ2 dimer or contained cytoplasmic γ2 chain. These results were verified by Northern blots: 43B cells expressed a low level of γ2 mRNA and no α3 mRNA, whereas 43A cells expressed both α3 and γ2 mRNAs.
Our results suggest a role in Lm-332 expression for not only ZEB-1 and ZEB-2 but also for SNAIL because upon SNAIL transfection, expression and secretion of Lm-332 chains were totally terminated. Results with AsPC-1, BxPC-3, HPAC, and PANC-1 pancreatic carcinoma cells also suggest that endogenous SNAIL expression found in PANC-1 cells correlates with the lack of Lm-332 chain synthesis. Laminin α3A chain regulatory region contains two E-box motifs at the position of +40 (5î-CATGTG) and +87 (5î-CACCTG) (Virolle et al. 2002), the latter being identical to the sequences present in the E-cadherin promoter and the DNA-binding sites of SNAIL, ZEB-1, and ZEB-2 (Batlle et al. 2000; Grooteclaes and Frisch 2000; Comijn et al. 2001). This could imply yet another target for SNAIL, i.e., α3 chain of Lm-332. Lm α3 chain repression could therefore occur either directly by SNAIL binding to α3 E-box (43A-SNA cells) or indirectly via ZEB transcription factors (43B cells).
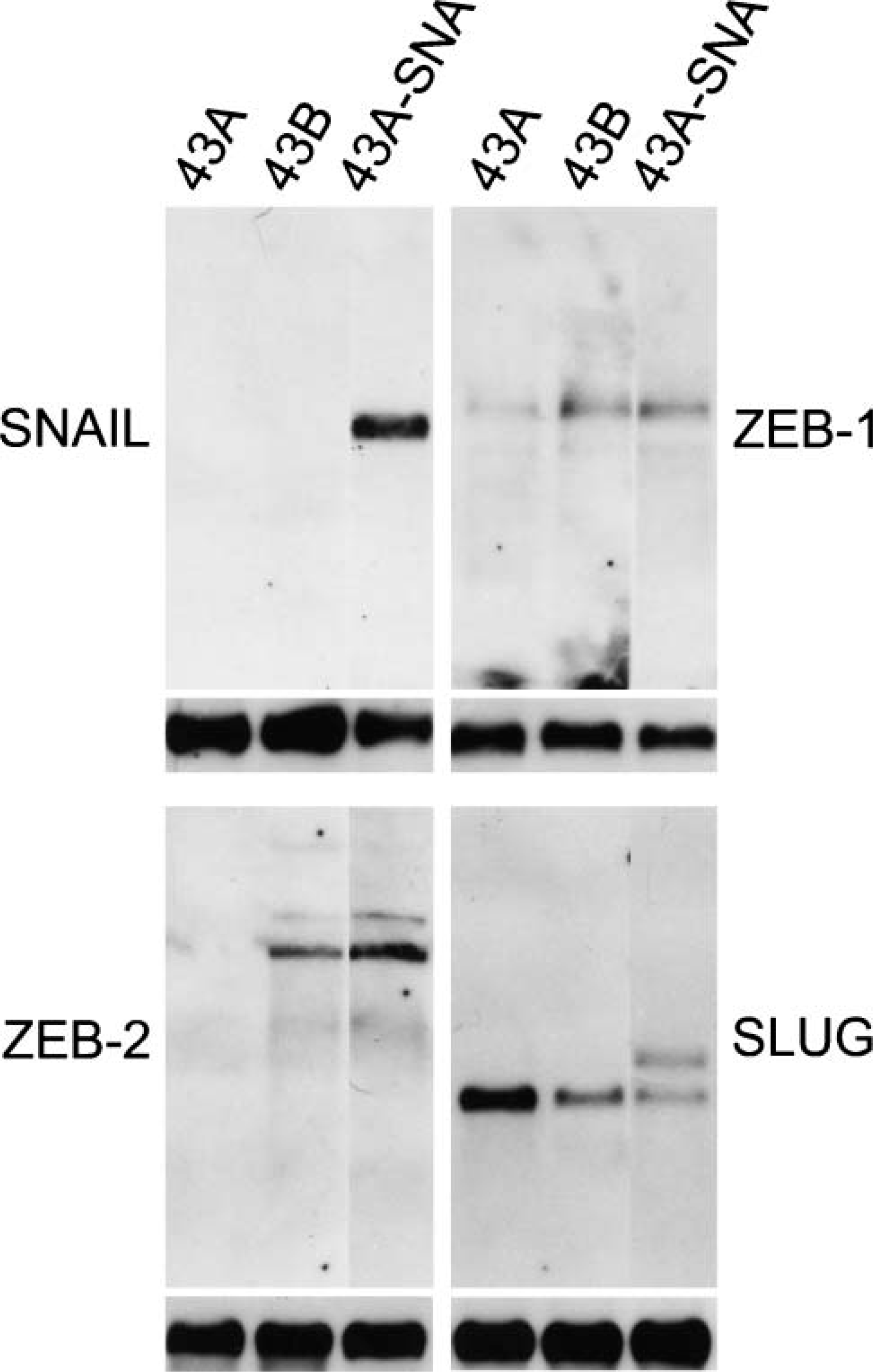
Expression of E-cadherin repressors in 43A, 43B, and SNAIL-transfected 43A-SNA cells. In Northern blots of 43A cells, a weak band for ZEB-1 and no bands for ZEB-2 were detected, whereas in 43B cells ZEB-1 and ZEB-2 were present. SLUG expression was found in every cell line, being higher in 43A cells compared with 43B and 43A-SNA cells. The probe cross-reacts with SNAIL in 43A-SNA cells (the upper band equals SNAIL in 43A-SNA cells). In 43A-SNA cells, a prominent transcript for SNAIL was detected, in addition with transcripts for ZEB-1, ZEB-2, and SLUG. GAPDH bands indicate equal mRNA loading.
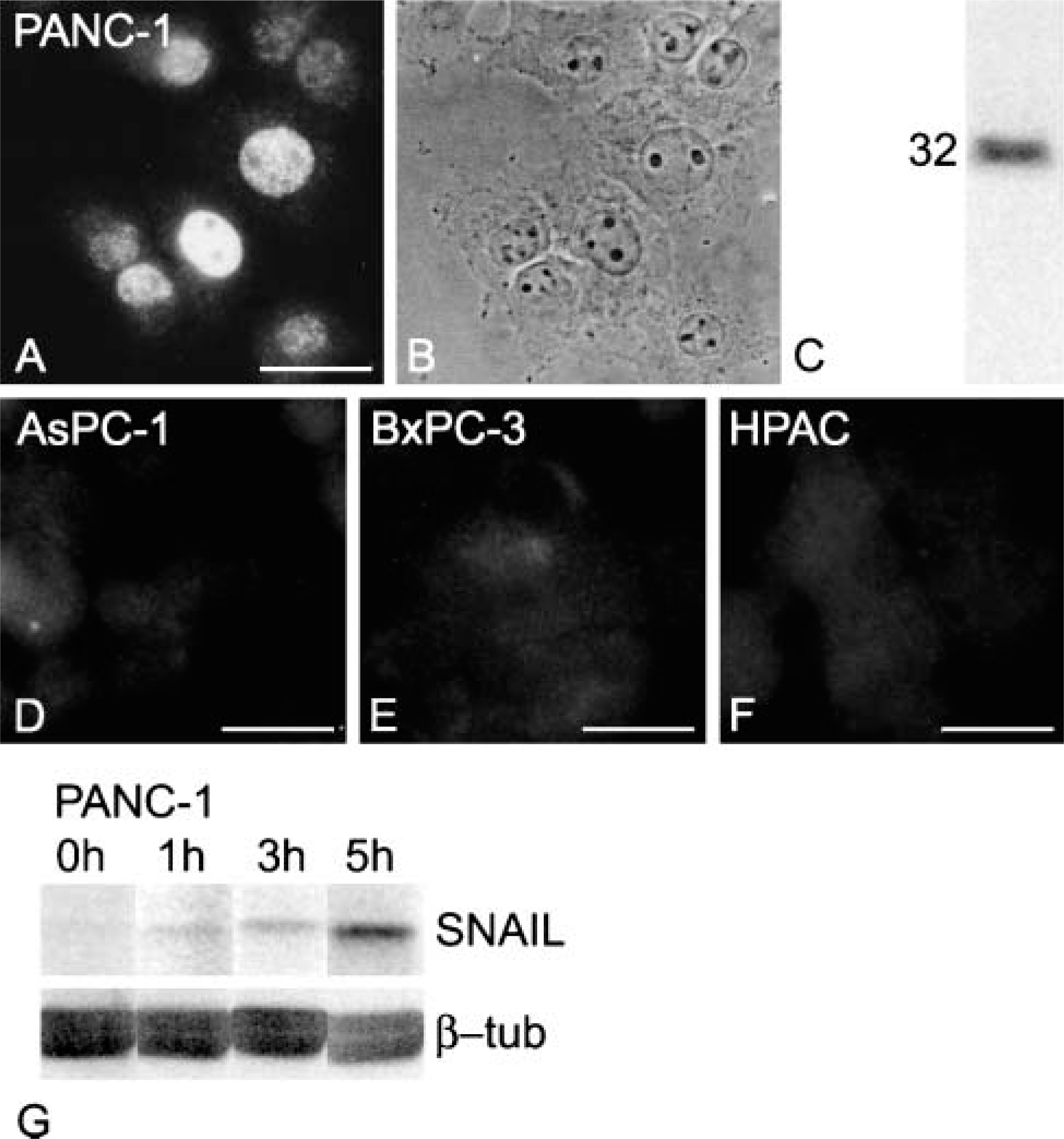
Endogenous SNAIL immunoreactivity in human pancreatic carcinoma cell lines. As detected with MAb CE2, immunoreactivity for SNAIL is found heterogeneously in the nuclei of PANC-1 pancreatic carcinoma cells (
Studies on the subcellular localization of SNAIL have suggested a cell-type-dependent localization, either nuclear or cytoplasmic (Domínguez et al. 2003; Yang et al. 2005). Recently, using a newly established antibody, localization of SNAIL was reported to be both nuclear and cytoplasmic in cells expressing endogenous SNAIL, whereas exogenous SNAIL confined only to the nucleus (Rosivatz et al. 2006). However, in immunohistochemical stainings of gastrointestinal adenocarcinomas, localization of SNAIL was limited to the nuclei in the latter study. Our results with novel MAbs CE2 and EC3 showed an exclusively nuclear localization of SNAIL in SNAIL-overexpressing interphase SCC cells. A similar, strictly nuclear localization was also detected in endogenously SNAIL-expressing PANC-1 cells showing features of EMT. Considering the short, 25-min half-life of SNAIL (Zhou et al. 2004), sensitivity of the MAbs is shown by immunostainings of endogenously SNAIL-expressing cells, although nuclear extraction or proteasome inhibitor treatment was required for Western blot detection.
Carcinoma-associated fibroblasts in tumor stroma have been suggested to derive directly from epithelial cells by EMT (Petersen et al. 2003). In line with this, we have reported expression of SNAIL in tumor-stroma interface of cervical, laryngeal, and colorectal carcinomas (Francí et al. 2006). A role for EMT has also been implicated in the pathogenesis of fibrosis of many parenchymal organs including kidney (Zeisberg and Kalluri 2004) and lung (Willis et al. 2006). Therefore, we suggest that SNAIL antibodies could be valuable tools in detecting SNAIL in carcinoma patients, thus determining cells with EMT features.
Using conditionally SNAIL- or ZEB-2-expressing colorectal carcinoma cells, SNAIL and ZEB-2 have recently been reported to extensively affect cell morphology and the epithelial cell differentiation program, including alterations in junctional complexes, Cks, and actin cytoskeleton (De Craene et al. 2005b; Vandewalle et al. 2005). Our results on human oral SCC cells and their descendants suggest a multitude of concomitant phenotypic changes: expression of E-cadherin repressors ZEB-1 and ZEB-2 together induce, for instance, loss of E-cadherin, but also emergence of N-cadherin expression and cytoplasmic accumulation of Lm γ2 chain. Expression of SNAIL exceeds the effects of those detected in endogenously ZEB-1- and ZEB-2-expressing cells, producing fibroblastoid cells with reduced cell-cell contacts, expression of N-cadherin and vimentin, and a lack of E-cadherin, Cks, hemidesmosomes, and Lm-332 chains.
Footnotes
Acknowledgements
M.T. received support from the Suomalais-Norjalainen Lääketieteen Säätiö.
We thank Dr. Tom Genetta, Dr. Antonio Postigo, and Prof. Karl Tryggvason for cDNAs and Dr. Stephen Blose, Prof. Robert E. Burgeson, Dr. Jonathan Jones, Prof. Kaoru Miyazaki, Prof. Raymond Nagle, and Dr. Katsushi Owaribe for MAbs. The excellent technical assistance of Reijo Karppinen, Hannu Kamppinen, Pipsa Kaipainen, Marja-Leena Piironen, Outi Rauanheimo, and Hanna Wennäkoski is acknowledged.
