Abstract
Hypoxia precedes neovascularization in many retinal diseases that can lead to irreversible vision loss. The transcription factor NF-κB is activated by hypoxia and regulates the expression of many genes, including angiogenic factors. The relation between the NF-κB activation and the cytokine-induced neutrophil chemoattractant (CINC), a member of the interleukin-8 (IL-8) family, was investigated by immunohistochemistry in a rat model of proliferative retinopathy presumably caused by relative hypoxia. Activated NF-κB and CINC immunoreactivity was detected in retinal glial cells in the nonperfused retina and in neovascular cells. Activated NF-κB was detected before the CINC staining, and both of these events occurred before the development of neovascularization. The intensity of both activated NF-κB and CINC staining remained increased during the development of neovascularization and then declined as neovascularization regressed. In rat retinal glial cells in vitro, dexamethasone, an inhibitor of NF-κB activation, prevented the hypoxia-induced increase in the amount of CINC mRNA. Furthermore, CINC induced neovascularization in a rat corneal pocket model. These results suggest that hypoxia-induced activation of NF-κB results in CINC production and participates in the induction of retinal neovascularization.
P
Retinal ischemia often precedes the onset of retinal neovascularization. The transcription factor NF-κB is activated by hypoxia and is believed to be central to the regulation of many inflammatory and proliferative response genes (Lenardo and Baltimore 1989; Grilli et al. 1993). Activity of NF-κB is regulated by specific inhibitors (IκBs) that maintain it in a cytoplasmic inactive form (Beg and Baldwin 1993). Several agents induce NF-κB activity, thereby promoting the rapid translocation of NF-κB into the nucleus (Baeuerle 1991), where it interacts with regulatory sites in the promoters of target genes and controls gene transcription (Grilli et al. 1993).
Recent studies have also demonstrated that NF-κB may regulate the initiation of angiogenesis in vitro (Shono et al. 1996; Stoltz et al. 1996). Shono et al. (1996) showed that NF-κB induced the production of interleukin-8 (IL-8) in endothelial cells and consequent angiogenesis in vitro. IL-8 is the most well-known CXC chemokine and contributes to many angiogenesis-dependent disorders (Endo et al. 1994; Elner et al. 1995; Singh et al. 1995). In rodents, the functions of IL-8 appear to be performed by cytokine-induced neutrophil chemoattractant (CINC) (Guex et al. 1996), whose production is also regulated by NF-κB activation (Blackwell et al. 1994; Ohtsuka et al. 1996). On the basis of amino acid sequence homology, CINC appears to be the rodent counterpart of human growth-related oncogene (GRO), a member of the IL-8 family (Watanabe et al. 1989b). CXC chemokines containing the sequence Glu-Leu-Arg (the ELR motif) are potent angiogenic factors both in vitro and in vivo (Strieter et al. 1995). CINC, IL-8, and GRO all contain this motif and promote cell migration and proliferation (Watanabe et al. 1989a; Koch et al. 1992; Driscoll et al. 1995; Nanney et al. 1995; Strieter et al. 1995). Although these findings are suggestive of pathological roles for CINC and IL-8 via NF-κB activation in neovascularization, no evidence has been shown for this mechanism in retinal neovascularization. In this study we have now investigated the roles of NF-κB and CINC in retinal neovascularization.
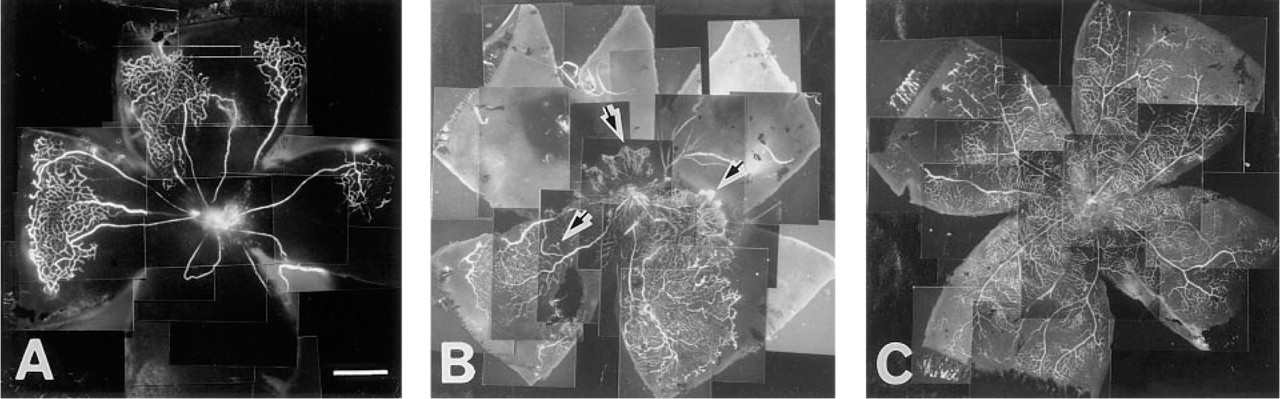
Fluorescein–dextran-perfused flat-mount retinas from animals exposed to room air or hyperoxia. (
Materials and Methods
Materials
Rabbit polyclonal anti-p65 (NF-κB) antibody was obtained from Santa Cruz Biotechnology (Santa Cruz, CA), rabbit polyclonal anti-CINC antibody was from IBL (Gunma, Japan), and rabbit polyclonal anti-glial fibrillary acidic protein (GFAP), rabbit polyclonal anti-von Willebrand factor (vWf), and mouse monoclonal anti-α-smooth muscle actin antibodies were from Dakopatts (Glostrup, Denmark). Fluorescein isothiocyanate–dextran (average molecular weight 2 X 106) was obtained from Sigma (St Louis, MO), bovine basic fibroblast growth factor (bFGF) was obtained from R&D Systems (Minneapolis, MN), CINC-1/GRO from Peptide Institute (Osaka, Japan), and ethylene vinyl acetate (EVA) from Takeda Chemical (Osaka, Japan).
Cell Culture
Primary rat retinal glial cells were isolated from postnatal Day 1 (P1)-P3 Sprague–Dawley rats as previously described (Cao et al. 1997). These cells were cultured in Dulbecco's modified Eagle's medium containing 10% FBS, 100 U/ml penicillin and 100 mg/ml streptomycin sulfate as reported.
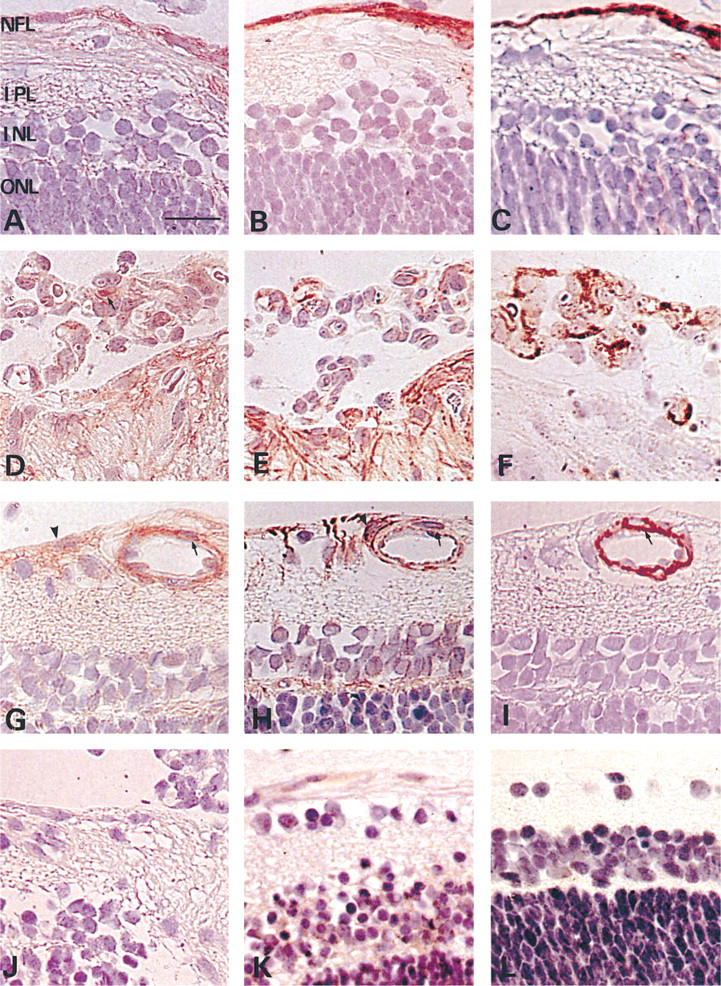
Immunohistochemistry of P22 retina after 7 days in room air subsequent to exposure to hyperoxia (
Animal Model of Proliferative Retinopathy
The experiments were reviewed and accepted by the Ethics Committee on Animal Experiments of the Faculty of Medicine, Kyushu University. Litters of P1 Brown Norway rats, together with their mothers, were exposed to 80 ± 2% oxygen for 14 days. Some rats (n = 21) were sacrificed immediately after the exposure and the remaining animals (n = 138) were transferred to room air for an additional 6 or 12 hr or for 1, 2, 3, 7, 14, 21, 28, 43, 55, 85, or 165 days before sacrifice. Rats of the same age that had been kept in room air were used as controls (n = 42).
Angiography with High Molecular Weight Fluorescein–Dextran
Rats (n = 78) were deeply anesthetized by
Immunohistochemistry for NF-κB and CINC
Rats (n = 123) were sacrificed by
Immunohistochemistry for GFAP, vWf, and α-Smooth Muscle Actin
To identify the cell types, we performed immunohistochemistry for GFAP, vWf, and α-smooth muscle actin, which are markers for glial cells, endothelial cells, and pericytes and smooth muscle cells, respectively.
Assessment of Immunohistochemistry
The incidence of preretinal neovascularization was defined as the percentage of rats showing unilateral or bilateral neovascularization. Four high-power microscopic fields (HPF) (X 320) near the optic disc (within 2 mm from the edge of the optic nerve) were examined for each retina for assessment of immunohistochemistry. Cells that stained positively for activated NF-κB and for CINC were counted by investigators who had no prior knowledge of the differences among the experimental groups. The NF-κB score was defined as the percentage of activated NF-κB-positive cells relative to the total number of cells identified in an area extending from the nerve fiber layer (NFL) to the inner plexiform layer, per HPF. Because virtually all activated NF-κB is localized in the nucleus, cells were considered positive for activated NF-κB if any nuclear staining was present. The CINC score was defined in a similar manner as the percentage of cells with positively stained nuclei or cytoplasm.
Hypoxic Conditions
Cells were incubated in Anaerocult A mini (Merck-Clevenot; Chelles, France) to produce hypoxic conditions as previously described (Ryuto et al. 1996).
Reverse Transcription PCR
Synthetic oligonucleotide primers based on the cDNA sequences of rat CINC (Huang et al. 1992) and GAPDH (Crippes et al. 1993) were prepared: CINC, 5'-ACTCCAA-CAGAGCACCATGG-3’ and 5'-TTGAGTGTGGCTATG-ACTTCG-3'; and GAPDH, 5'-CAATGTATCCGTTGTGG-ATCTGACA-3’ and 5'-GAGGCCATGTAGGCCATGAGG TCCA-3'. First-strand cDNA was synthesized from 1 μg of mRNA by incubation for 1 hr at 37C with Moloney murine leukemia virus reverse transcriptase (Gibco-BRL; Gaithersburg, MD). A portion (1 μl) of the RT reaction mixture was then subjected to PCR. After denaturation for 9 min at 95C, samples were subjected to 30 cycles of denaturation for 30 sec at 94C, primer annealing for 30 sec at 55C, and polymerization for 45 sec at 72C. Reaction products were analyzed on 2% agarose gels, and DNA was extracted from bands of the expected sizes, subcloned into the pMOS Blue T-vector (Amersham; Poole, UK), and subjected to nucleotide sequencing by the dideoxynucleotide chain termination method (Sanger et al. 1977).
Statistical Analysis
Data are presented as means ± SD, and the significance of differences was evaluated by the Kruskal-Wallis test. A p value of <0.01 was considered statistically significant.
Results
Angiography with High Molecular Weight Fluorescein–Dextran
The pattern of vascular development and neovascularization was readily apparent in retinal flat-mounts prepared from rats after fluorescein–dextran perfusion. Immediately after exposure to hyperoxia (P15), the experimental animals showed almost no perfusion of the fine branching capillaries around the optic disc, whereas the well-developed radial vessels extending from the optic disc were perfused (Figure 1A). After 14 days in room air (P29), the neovascular response, as in humans, occurred at the junction between the perfused and the nonperfused retina (Figure 1B). The vascular pattern of a normal P15 retina extended from the optic disc to the ora serrata (Figure 1C).
Distribution of NF-κB and CINC
To determine the distribution of NF-κB and CINC during the development of neovascularization, immunohistochemistry was performed. Figure 2 shows the results of a typical immunohistochemistry profile on Day 7 in room air (P22) for NF-κB (Figures 2A, 2D, and 2G) and CINC (Figures 2B, 2E, and 2H). NF-κB immunoreactivity was detected in the nuclei and cytoplasm of cells in the NFL and also surrounding the superficial vessels in an area extending from the NFL to the inner plexiform layer of the retina, being especially marked in the central retina (Figure 2A). Immunostaining for CINC was detected in the cytoplasm of those cells (Figure 2B) that were also positive for GFAP (Figure 2C). NF-κB and CINC staining was also detected in neovascular cells (Figures 2D and 2E) that stained for vWF (Figure 2F). However, some of the NF-κB-positive cells surrounding the vessels (Figure 2G) were negative for CINC (Figure 2H). Those cells stained for α-smooth muscle actin (Figure 2I). NF-κB immunoreactivity was observed at a low level in the cytoplasm of retinal pigment epithelial cells, which were negative for CINC staining (data not shown). No specific immunoreactivity was detected with rabbit nonimmune IgG as the primary antibody (Figure 2J).
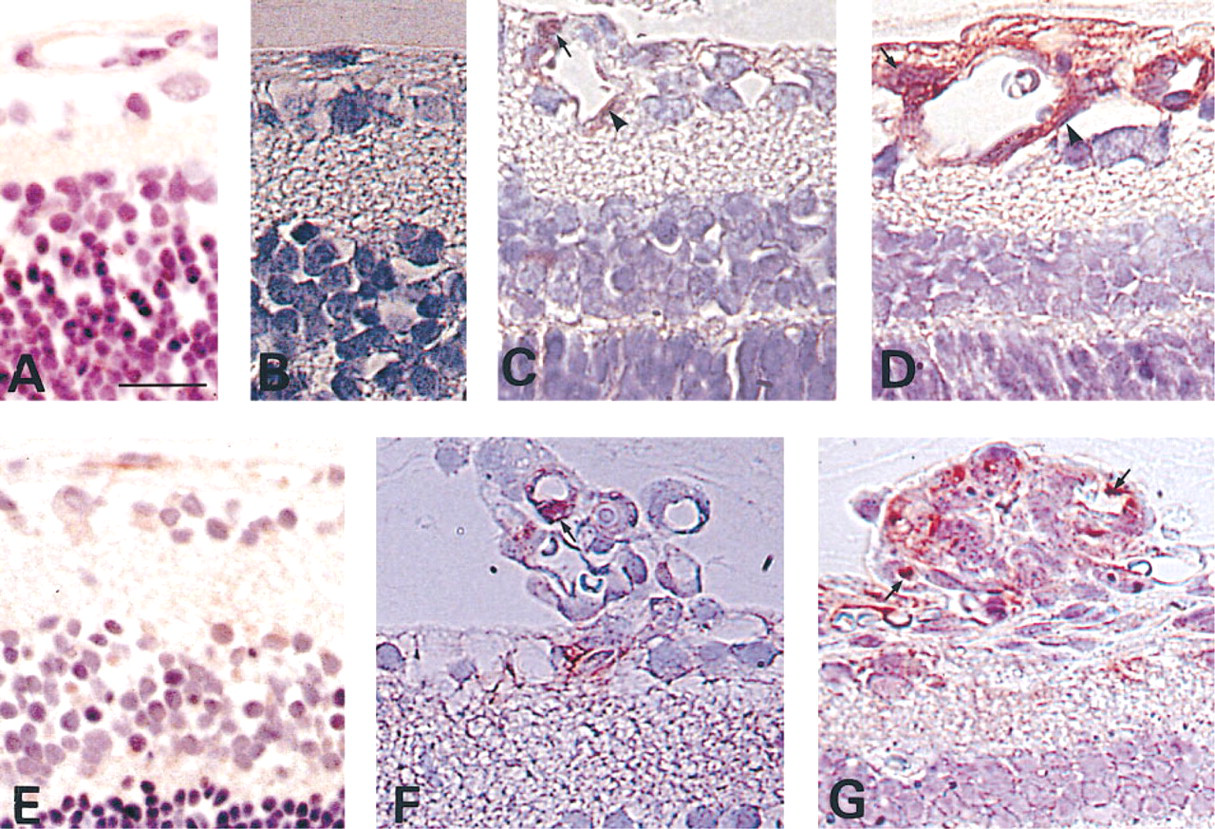
The time course of changes in the immunostaining of NF-κB and CINC in response to 14 days of hyperoxia and subsequent exposure to room air for various intervals. (
In normal controls throughout the entire observation period, NF-κB staining was weak in the cytoplasm of cells (Figure 2K) and CINC immunoreactivity was either absent or was present only in small amounts (Figure 2L).
Time Course of Immunostaining for NF-κB and CINC
Immediately after the animals had been returned to room air (P15), NF-κB staining was weak in the cytoplasm of the cells in the NFL and surrounding the vessels (Figure 3A) and CINC staining was absent (Figure 3B). After the animals had been in room air for 12 hr, intense NF-κB staining was seen in the nuclei (Figure 3C). After 24 hr in room air (P16), the intensity of NF-κB staining increased in the nuclei and the cytoplasm (Figure 3D) and CINC immunoreactivity began to be detected (Figure 3E). Immunostaining for NF-κB and CINC was also detected in the cells forming neovascular tufts during the development of neovascularization (Figures 3F and 3G). On Day 14 in room air (P29), the intensity of both NF-κB and CINC staining began to regress. By day 28 (P43) in room air, NF-κB staining was only slightly detected in the cytoplasm and CINC staining was observed in a few glial cells. At 6 months post exposure, NF-κB staining was weak and CINC immunoreactivity was either not detected or only slightly detected in the glial cells (data not shown).
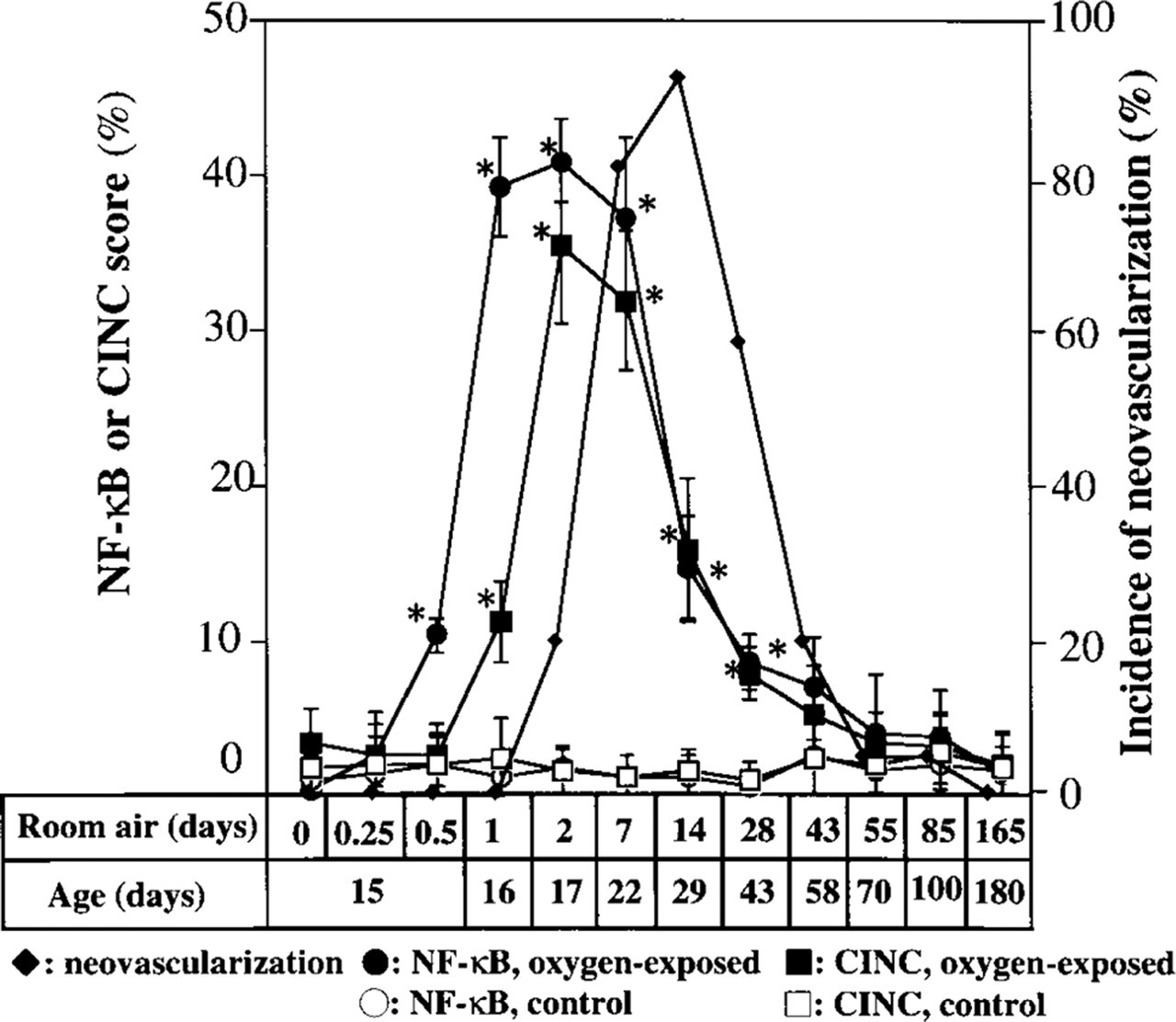
Time course of changes in activated NF-κB and CINC scores and in the incidence of retinal neovascularization both for rats exposed to hyperoxia for 14 days and then to room air for the indicated times and for control animals maintained in room air. ∗, statistically significant difference (p<0.01) compared with the value in control animals.
The activated NF-κB and CINC scores and the incidence of preretinal neovascularization are shown in Figure 4. A significant increase in the activated NF-κB score was first apparent 12 hr after the animals were returned to room air, and a significant increase in the CINC score was first detected 12 hr later. Neovascularization first became apparent after 2 days in room air. Activated NF-κB and CINC scores remained significantly increased for several weeks before returning to baseline values as the neovascularization regressed. The activated NF-κB and CINC scores of age-matched control rats remained relatively constant at low levels.
Effects of Dexamethasone on the Hypoxia-induced Increase in the Level of CINC mRNA
The relationship between NF-κB and CINC in retinal glial cells of this rat model of retinal neovascularization was investigated with rat retinal glial cells in vitro. Consistent with our observations in vivo, the CINC mRNA level increased after exposure of the retinal glial cells to hypoxia (Figure 5). Pretreatment for 3 hr with 1 μM dexamethasone, an inhibitor of NF-κB activation (Ohtsuka et al. 1996), prevented this induction.
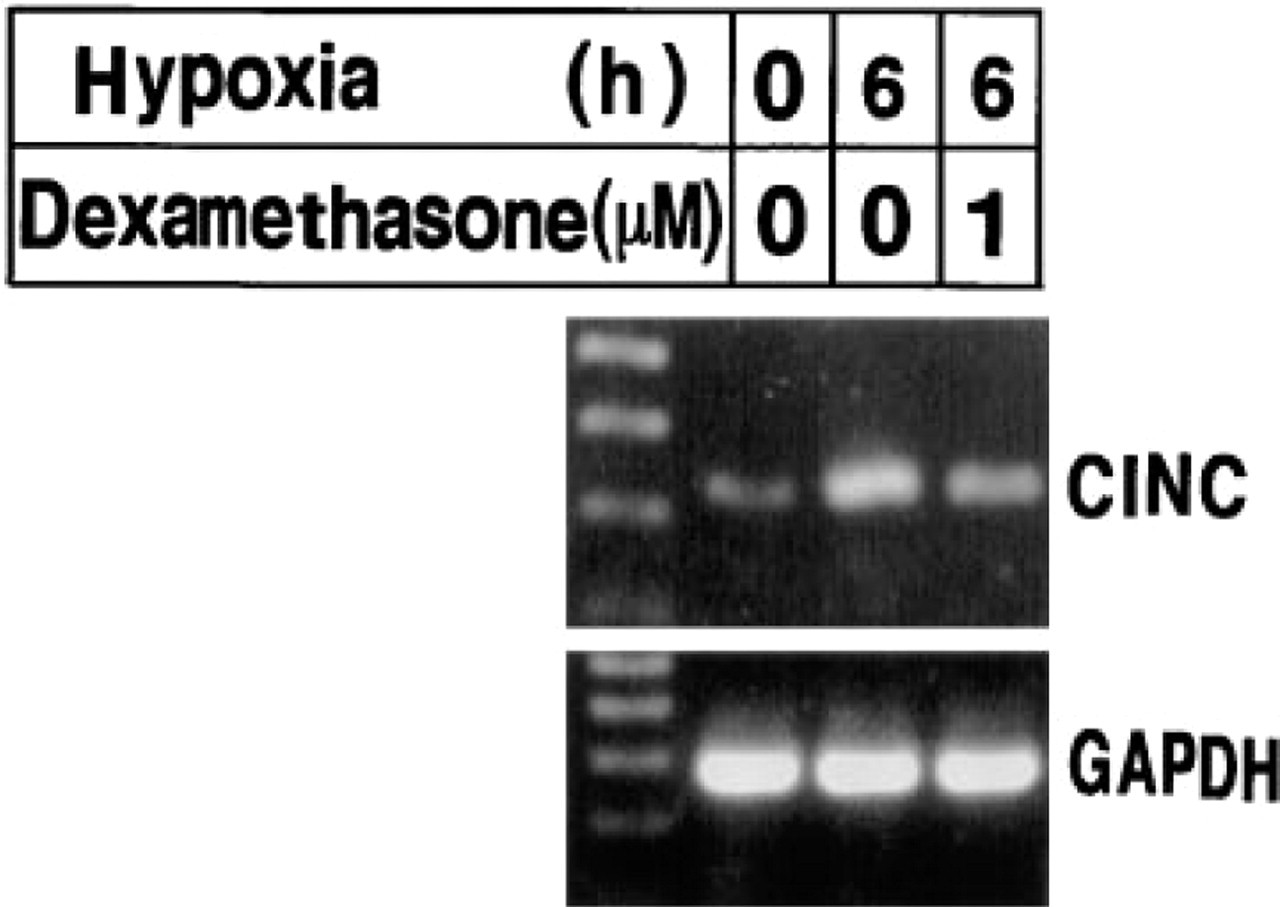
Effects of dexamethasone on the induction of CINC mRNA by hypoxia in rat retinal glial cells. Cells were incubated in DMEM containing 1% FBS for 24 hr, exposed or not to 1 μM dexamethasone for 3 hr, and then subjected or not to hypoxia for 6 hr, as indicated. Cells were lysed and mRNAs were subjected to RT-PCR as described in Materials and Methods, with primers specific for CINC (upper panel), or GAPDH (lower panel). Left lanes contain molecular size markers.
Rat Corneal Pocket Model of Neovascularization
We used the rat corneal pocket model of neovascularization to determine whether CINC can induce angiogenesis. CINC (50 ng) and bFGF (50 ng) induced positive corneal angiogenic responses in 6/6 and 7/7 corneas, respectively (Figures 6A and 6B), without evidence of significant leukocyte infiltration (assessed by light microscopy). In contrast, control buffer resulted in a positive neovascular response in 1/6 corneas treated (Figure 6C).
Discussion
We have shown that immunostaining for activated NF-κB and CINC was detected in retinal glial cells in the nonperfused retina and in neovascular cells of a rat model of proliferative retinopathy. Activated NF-κB was detected before the CINC staining, and both were detected before the development of neovascularization. The intensity of both activated NF-κB and CINC staining remained increased until neovascularization reached its maximum, and then returned to baseline values as neovascularization regressed. The time course and localization of their staining indicate that the activation of NF-κB probably induces CINC expression and that NF-κB and CINC play an important role in the development of neovascularization. We further showed that CINC could induce corneal neovascularization in vivo.
In the rat model of retinal neovascularization, exposure of the animals to hyperoxic conditions results in obliteration of the vessels of the posterior retina. Return of the animals to room air is believed to cause relative hypoxia of the nonperfused retina, resulting in quantifiable neovascular response. Although there is no direct evidence that the avascular retina is hypoxic in this model, there is evidence that this is the case in other animal models of retinal neovascularization (Pournaras et al. 1990). Fluorescein–dextran perfusion of animals exposed to hyperoxia also demonstrates complete loss of the retinal capillary bed in the central retina (Smith et al. 1994).
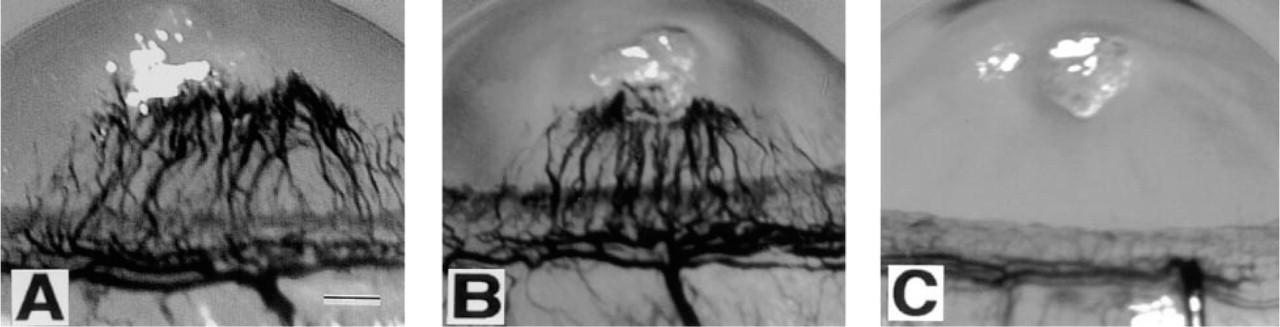
Biomicroscopic photographs of rat corneas 6 days after implantation of EVA pellets containing CINC (50 ng) (
It is therefore reasonable to assume that the non-perfused retina is hypoxic in the rat model. Hypoxia induces NF-κB activation and CINC production in vitro (Karakurum et al. 1994; Tsuruma et al. 1996). The NF-κB binding site in the promoter region of the CINC gene is indispensable for gene expression (Blackwell et al. 1994; Ohtsuka et al. 1996). Furthermore, in this study, hypoxia-induced increase of the CINC mRNA level was prevented by an inhibitor of NF-κB activation in rat retinal glial cells. Therefore, the hypoxia-induced activation of NF-κB probably promotes CINC production in retinal glial cells.
Activated NF-κB and CINC were localized to glial and endothelial cells. Glial cells in the retina also produce vascular endothelial growth factor/vascular permeability factor (VEGF/VPF), an important angiogenic factor, in response to hypoxia (Hata et al. 1995; Pierce et al. 1995). Our observations suggest that both CINC and VEGF might contribute to the neovascularization of the retina in an autocrine/paracrine manner. In contrast, we observed that pericytes and smooth muscle cells expressed NF-κB but not CINC, suggesting that activated NF-κB may induce the production of proteins other than CINC in the pericytes and smooth muscle cells of the retina (Hiscott et al. 1993; Baeuerle and Henkel 1994). Human retinal pigment epithelial cells also produce IL-8 and GRO in vitro (Wood and Richmond 1995; Kurtz et al. 1997). However, in the present study little NF-κB and no CINC immunoreactivity was detected in retinal pigment epithelial cells. The oxygen supply of the inner retina is derived solely from the retinal vessels, whereas retinal pigment epithelial cells obtain oxygen from choriocapillaries. Therefore, glial cells in the superficial layer of the retina might be more susceptible to ischemia than retinal pigment epithelial cells.
NF-κB is a pleiotropic regulator of many genes, including those encoding angiogenic factors or adhesion molecules (Grilli et al. 1993; Shono et al. 1996; Yoshida et al. 1997) and therefore may provide a target for therapeutic intervention in retinal neovascularization. In an in vitro model, NF-κB anti-sense oligonucleotides and pyrrolidine dithiocarbamate, an inhibitor of NF-κB activation, could inhibit angiogenesis (Shono et al. 1996; Stoltz et al. 1996).
In summary, we have shown that activation of NF-κB probably induces CINC production and participates in retinal neovascularization in a rat model. These observations suggest that NF-κB activation might similarly induce the expression of human homologues of CINC (IL-8 and the GRO family of polypeptides), and therefore play an important role in the pathogenesis of ischemic retinal vascular disease in humans.
Footnotes
Acknowledgements
Supported in part by the Health Sciences Research Grants Japan.
We thank M. Imamura for technical assistance.
