Abstract
In back skin sections from adolescent C57BL/6 mice, regularly distributed, perifollicular inflammatory cell clusters (PICC) were found located around the distal noncycling portion of about 2% of all hair follicles examined. The PICC and the affected hair follicles were characterized during spontaneously developed or induced hair cycle stages, using antibodies against MHC Class II,F4/80, ER-MP23, NLDC 145, CD4, CD8, γδTCR, IL-1 receptor, and ICAM-1. PICC consisted predominantly of macrophages (MAC), accompanied by a few CD4+ cells, whereas γδTCR+ and CD8+ cells were absent. During anagen and catagen, some of the PICC+ hair follicles showed variable degenerative phenomena reminiscent of scarring alopecia: thickened basement membrane, ectopic MHC II expression, MAC infiltration into the follicle epithelium, and signs of keratinocyte apoptosis. Loss of distal outer root sheath keratinocytes was detected in 10% of PICC+ hair follicles (0.2% of all hair follicles). Because PICC were located in the vicinity of the bulge region, MAC-dependent damage to follicle stem cells might eventually lead to follicle degeneration. These perifollicular MAC clusters around selected hair follicles may indicate the existence of a physiological program of MAC-dependent controlled follicle degeneration by which damaged or malfunctioning follicles are removed by programmed organ deletion (POD).
I
Here we describe a striking HF-related phenomenon that casts further doubt on exactly when cutaneous inflammatory cell infiltrates have to be considered pathological. In the course of analyzing hair cycle-dependent changes in the immune status of murine skin (Eich-müller et al. 1994; Paus et al. 1994a,c; Hofmann et al. 1996; Paus 1997) we noted dense perifollicular “clusters” of dendritic cells around the distal portion of individual HF in the unmanipulated, macroscopically uninflamed back skin of C57BL/6 mice, irrespective of the age and hair cycle stage of the mice (Eichmüller et al. 1997). Because this had not been reported previously, extensive analyses by a variety of immunohistological and histochemical stains were performed to better characterize the type of dermal cells within a cluster as well as the affected HF.
Materials and Methods
Animals and Tissue Collection
Syngeneic female C57BL/6 mice and Balb/c mice (Charles River; Sulzfeld, Germany) were housed in community cages with 12-hr light periods at the Animal Facilities of the Biomedical Research Center, Charité, Berlin, and were fed water and mouse chow ad libitum. Skin harvesting for cryosections, embedding, and sectioning were performed as previously described (Paus et al. 1994a,c). Additional skin samples were cut parallel to the epidermis to obtain serial horizontal cross-sections of HF so that multiple labeling experiments could be performed on consecutive levels of the same HF.
To obtain different spontaneously developed hair cycle stages, we took back skin of mice with all HF in telogen (6- to 8-week-old animals) or in various stages of the anagen VI/catagen transition of the hair cycle (12–15-week-old animals). In addition, one group consisted of mice with back skin in spontaneously developed catagen after anagen induction by depilation of 7-week-old mice (Paus et al. 1994a,b) and one group in anagen developed after cyclosporin A was injected IP during telogen (Paus et al. 1989). Longitudinal and horizontal sections of more than 60 mice were studied. In addition, quantitative analyses were performed on eight mice with horizontal and six mice with longitudinal sections, studying a total of approximately 10,000 HF (see below).
Immunohistology
The applied primary antibodies, source, dilution, and host species are given in Table 1. Cryosections (8–10 μm) were incubated with avidin- and biotin–blocking solution (Vector; Burlingame, CA) if the ABC method was applied, followed by another block using either protein blocking agent (Immu-notech; Krefeld, Germany) or 4% normal goat serum in TBS (Tris-buffered saline) for 20 min at room temperature (RT). The primary antibody was diluted in TBS containing 1% normal goat serum (for titers see Table 1) and applied for 1 hr (monoclonal antibodies) or overnight (polyclonal rabbit antisera). As secondary antibodies, we used biotinylated goat-anti-rat or goat-anti-hamster (Vector) diluted 1:200 in TBS containing 4% mouse normal serum for 45 min at RT, followed by an incubation with avidin–biotin-alkaline phosphatase complex (Vector) 1:100 in TBS for 30 min. After washing, a routine staining procedure for alkaline phos-phatase was used and sections were counterstained with Mayer's hematoxylin. The polyclonal rabbit antisera were detected using a rhodamine-coupled goat anti-rabbit secondary antibody applied for 1 hr at 37C, followed by washing and covering of sections with Vecta-Shield cover medium (Vector).
Double immunostaining on the same section, using anti-MHC Class II antibody and anti-NLDC145 or anti-MHC Class II antibody and anti-ICAM-1, was performed as previously described (Eichmüller et al. 1996). Apoptotic keratinocytes were visualized as described (Lindner et al. 1997), using a fluorescent TUNEL technique, a commercially available kit (ApopTag; Oncor, Gaithersburg, MD), and Hoechst 33342 (Sigma; St Louis, MO) as counterstain.
Histochemical Stains
Various histochemical protocols were applied, following the procedures described by Romeis (1989). Giemsa stains were used as an overview, for nuclei and mast cell differentiation, and to screen for infections by protozoa and mites. Bacterial infection was checked for by Gram stain and fungal infections by PAS stain. The distribution of elastic fibers throughout the connective tissue was shown by Elastica (Resorcin/ Fuchsin) stain. Finally, hematoxylin/eosin stains were used to identify possible infections by mites, to demarcate apoptotic cells, and to check whether perifollicular cell clusters could also be identified in a routine histopathological stain.
Analyses
Quantitative analyses were performed on both longitudinal and horizontal sections. Comparisons among different cell types as well as co-localization studies within the same cluster of labeled cells were done on serial cross-sections using a Zeiss microscope with a camera lucida system. All HF and the various immunohistological labelings were drawn by hand on separate transparencies, superimposed and correlated thereafter.
Statistical assessment was done using the Kruskall–Wallis test for analyses of independent groups and the Mann–Whitney U-test for comparison of independent pairs (Sachs 1992).
Antibodies: source, technique, and dilution
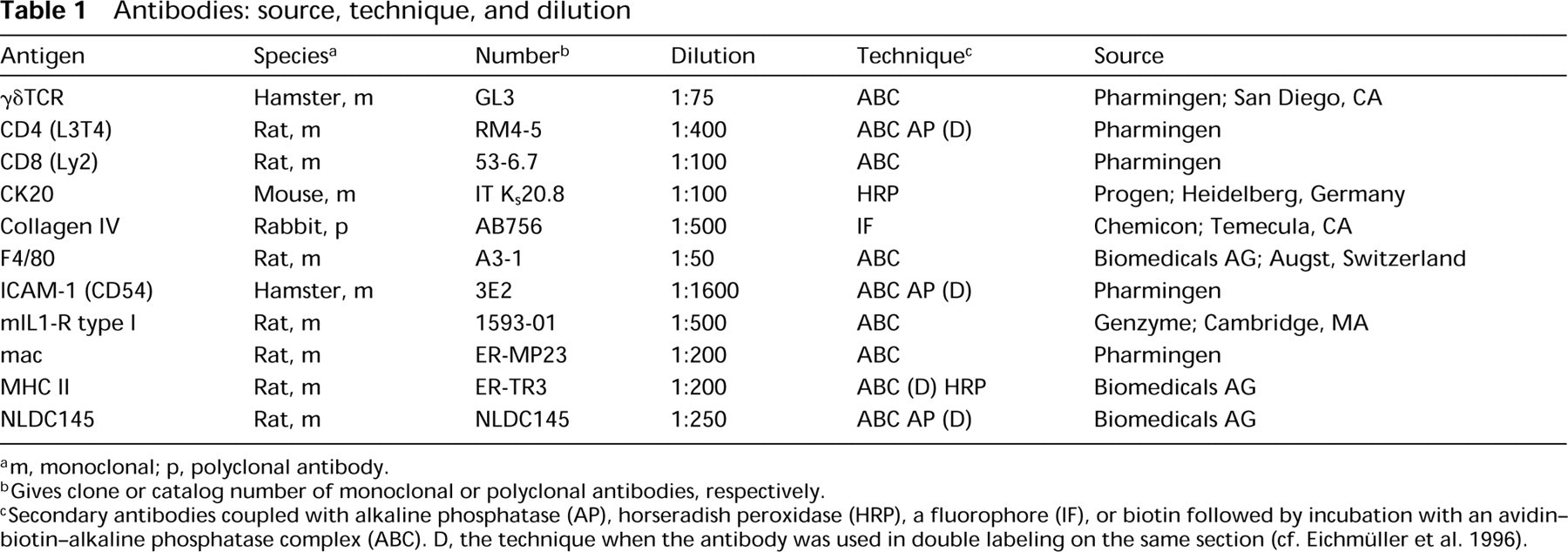
m, monoclonal; p, polyclonal antibody.
Gives clone or catalog number of monoclonal or polyclonal antibodies, respectively.
Secondary antibodies coupled with alkaline phosphatase (AP), horseradish peroxidase (HRP), a fluorophore (IF), or biotin followed by incubation with an avidin–biotin–alkaline phosphatase complex (ABC). D, the technique when the antibody was used in double labeling on the same section (cf. Eichmüller et al. 1996).
Photomicrographs were taken with a video camera and digital image analysis was performed using the computer software ISIS (Metasystems: Altlussheim, Germany).
Results
In longitudinal and horizontal sections of back skin from apparently healthy adolescent female C57BL/6 mice, we confirmed the presence of regularly spaced PICC of macrophage-like cells around individual HF. These PICC as well as the affected HF were characterized during different hair cycle stages that had either developed spontaneously or had been induced by IP cyclosporin A or by depilation. Skin sections were stained with antibodies against MHC Class II, F4/80, ER-MP23, NLDC 145, CD4, CD8, γδTCR, IL-1R, and ICAM-1. Table 2 summarizes the key results of these immunohistological analyses, which are described in detail below.
Normal Murine Skin Displays Perifollicular Inflammatory Cell Clusters
Macroscopically uninflamed, normal back skin of C57BL/6 mice with all HF in telogen that had not been manipulated in any way exhibited an increased density of MHC II-IR, dermal cells clustered around individual telogen HF, more or less evenly distributed throughout the back skin (Figure 1A). Such PICC were usually located in the vicinity of the distal HF epithelium, i.e., around the noncycling portion of the HF. PICC could also be seen around HF in the growth stage (anagen VI, Figure 1B) and in the regression stage of the hair cycle (catagen VII, Figure 1C) and were visible in both longitudinal and cross-sections of C57BL/6 back skin HF. Consecutive longitudinal sections showed that PICC enwrap the entire HF (Figures 1E and 1F). Interestingly, PICC were also detected in the skin of cyclosporin A-treated and thus immuno-suppressed animals (not shown).
Summary of immunohistological results
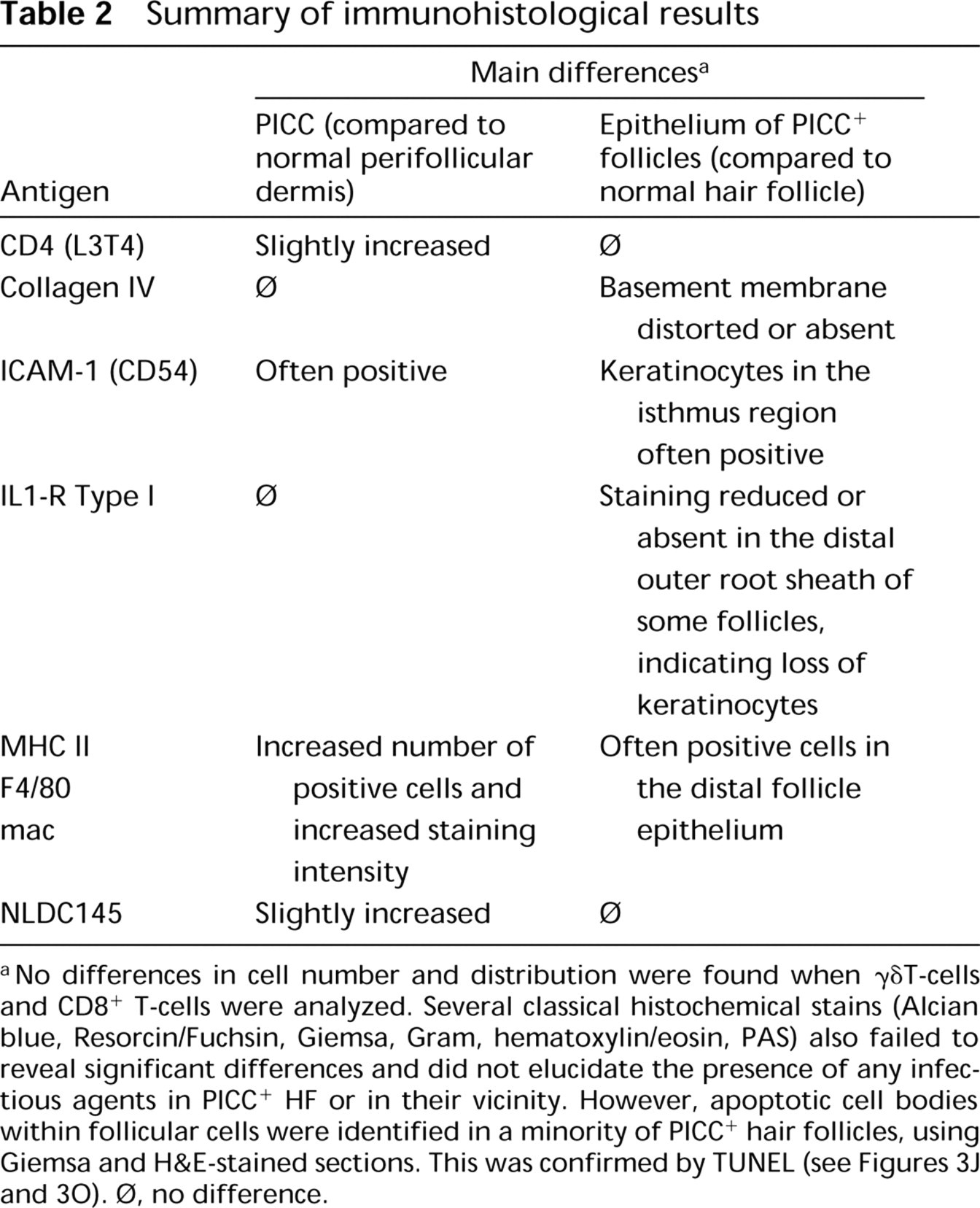
No differences in cell number and distribution were found when γδT-cells and CD8+ T-cells were analyzed. Several classical histochemical stains (Alcian blue, Resorcin/Fuchsin, Giemsa, Gram, hematoxylin/eosin, PAS) also failed to reveal significant differences and did not elucidate the presence of any infectious agents in PICC+ HF or in their vicinity. However, apoptotic cell bodies within follicular cells were identified in a minority of PICC hair follicles, using Giemsa and H&E-stained sections. This was confirmed by TUNEL (see Figures 3J and 3O). Φ, no difference.
These PICC did not appear to be a strain-specific feature of C57BL/6 mice because they were also present in Balb/c mice (Figure 1D). There were no overt signs of bacterial, fungal, or parasitic infections, either macroscopically or in any of the analyzed skin sections as checked by standard histochemical stains (PAS, Gram, Giemsa, hematoxylin/eosin; not shown).
A “cartography” of PICC distribution over the entire back skin of one untreated mouse with all HF in telogen revealed a fairly homogeneous distribution of PICC+ HF (Figure 2), with a slightly (but not significantly) lower density in the lower back region compared to the neck or central back regions (p<0.05). To further analyze the distribution of PICC as well as to perform multiple labeling studies on the same cluster of cells and on the same follicle, serial horizontal cross-sections were further investigated (e.g., Figures 1D and 1H). Counting the number of PICC in horizontal cross-sections of eight different mice, we found 111 of 6340 HF to be surrounded by PICC (1.8%).
Both Pelage and Tylotrich Hair Follicles Are Associated with PICC
Among the pelage follicles of murine back skin, a sub-population of larger, so-called tylotrich hair follicles (TF) stand out (Sundberg 1994). TF are believed to have special sensory functions, are larger, have longer hair shafts, are associated with tactile discs (Haarscheiben), and are more heavily innervated (Mann 1969; Moll et al. 1996). First, the amount of TF within normal murine back skin was estimated in sequential longitudinal sections stained with antibodies against MHC II or CK20. The latter demarcates Merkel cells which, in the back skin of C57BL/6 mice, are present only in the intraepidermal tactile discs that are invariably associated with TF (Moll et al. 1996). Analyzing 4800 HF in these longitudinal sections, about 10% of back skin follicles were identified as TF (cf. Figure 1G). Next, horizontal cross-sections were used to calculate the amount of TF, and TF were identified by their enlarged diameter, their bilobular sebaceous gland, and by their substantially thicker outer root sheath (ORS) and hair shaft (Mann 1969; Moll et al. 1996) (cf. Figure 1H). Surprisingly, when we counted 6340 cross-sections of HF, we found approximately 3% of all HF to be TF (range 2–4%). These differences in the percentage of identified TF most likely reflect the inherent methodological difficulty of unequivocally identifying a TF as such, with longitudinal sections providing a greater likelihood of identifying a TF by its intraepidermal Merkel cell disc than horizontal sections, which cannot reveal this specific TF marker (cf. Moll et al. 1996). Both TF and pelage follicles were found to be associated with PICC (see Figure 1G). A strict correlation of the follicle type (TF vs pelage) and the appearance of PICC could not be established because of the low number of detected TF in horizontal sections.
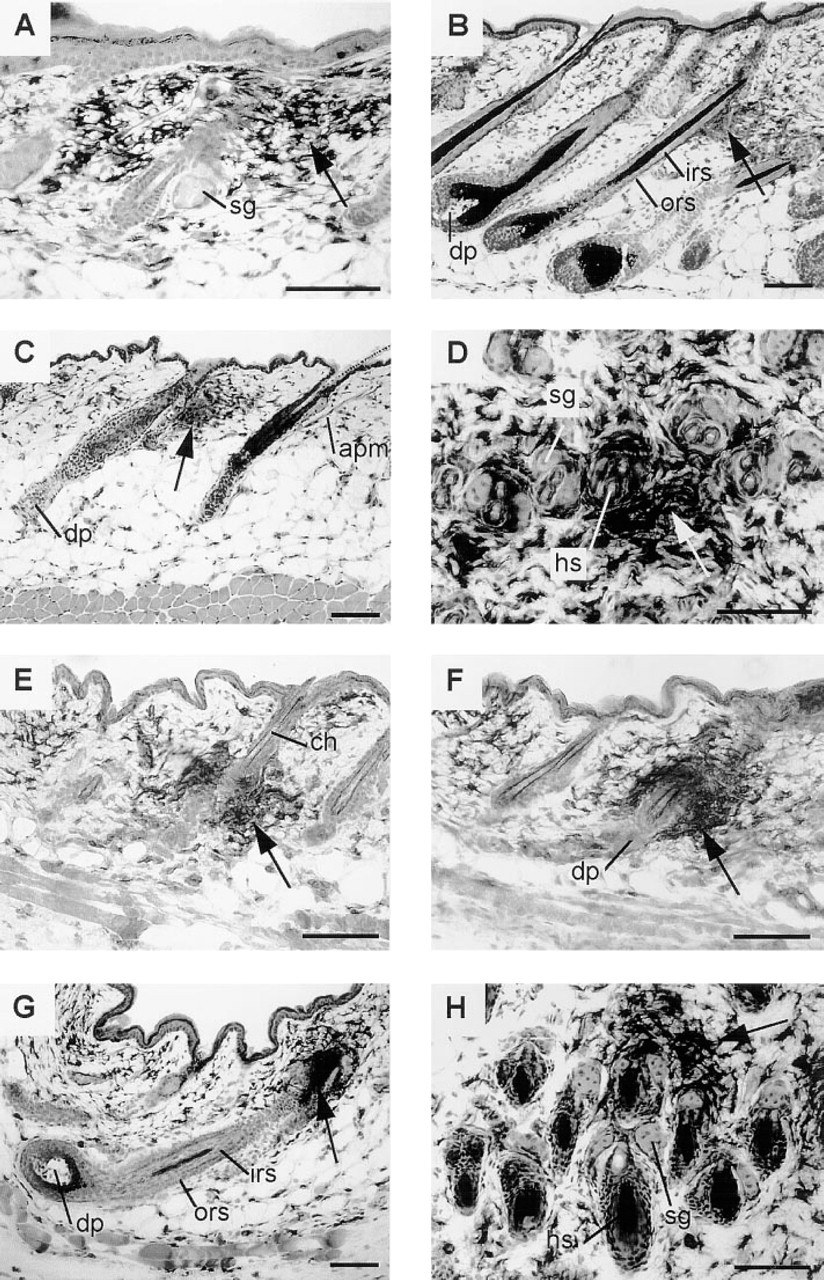
PICC are present around hair follicles, irrespectively of the hair cycle stage. (
PICC Consist Predominantly of Macrophage-like Cells
A variety of immunohistological stains on serial horizontal sections were performed to clarify the cellular composition of PICC (Figure 3). Up to eight different stains were performed in one series of consecutive sections. To identify the same HF in follow-up sections, the camera lucida system was used, and follicles and labeling results were drawn on separate transparencies, which were then superimposed (see Materials and Methods).
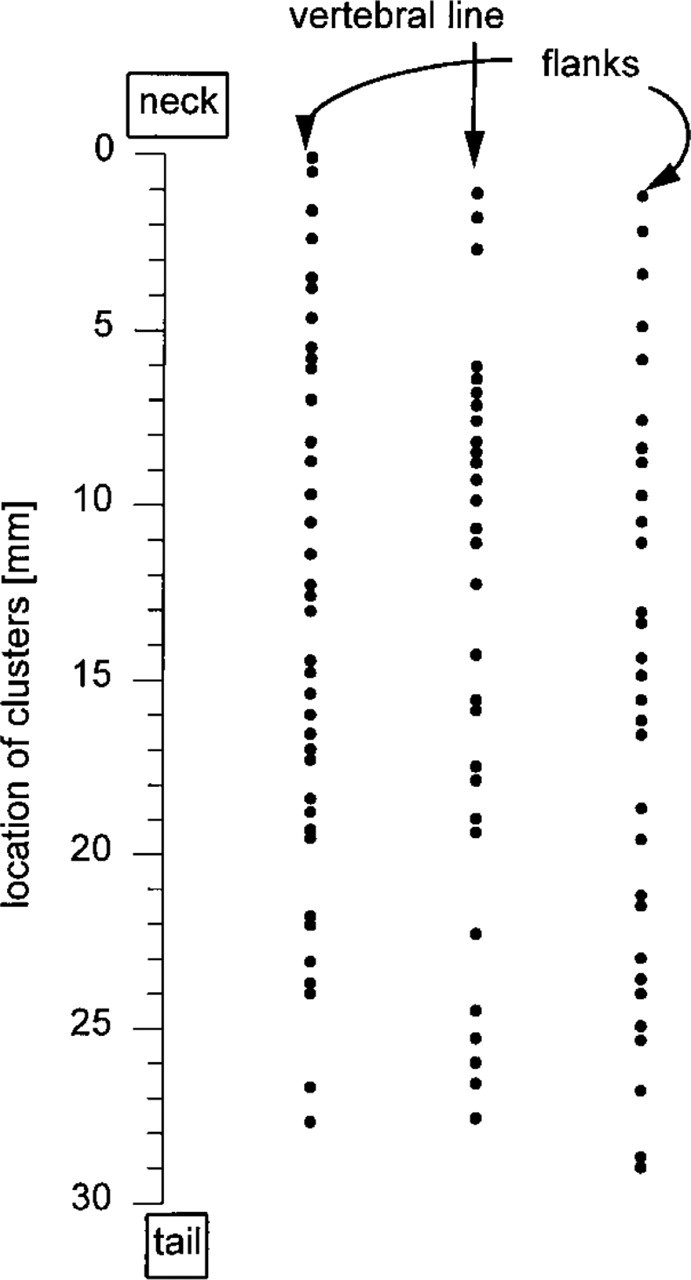
PICC are distributed quite homogeneously over the back skin. The graph shows one representative mouse. Adjacent skin specimen of the vertebral line and two parallel lines 5 mm from each side were analyzed (three blocks for each line; longitudinal sections). The exact location was measured and is displayed as distance to the caudal rim of the blocks, ending at the neck.
Most of the cells within a PICC were MHC II IR (Figures 3A, 3F, 3I, and 3K) and F4/80-IR (not shown), suggesting these cells to be macrophages (MAC) or Langerhans cells. To distinguish these two cell populations, double labeling with anti-MHC II and anti-NLDC145 antibodies was performed. Quantitative analyses of the number of cells within and outside of PICC revealed that the vast majority of cells in a PICC were not Langerhans cells (i.e., were NLDC145-negative) and that only the number of MHC II+/NLDC145−cells was significantly higher in the PICC compared to the interfollicular dermis (Figure 4).
Only very few PICC cells exhibited weak CD4-IR, but the number of CD4+ PICC cells was higher than in the surrounding dermis (Table 2) (e.g., Figure 3N compared to Figure 3K). The number of CD8-IR cells within the PICC was not increased in comparison to other skin areas (notably, the total number of CD8-IR cells in normal C57BL/6 mouse skin is normally very low). When sections were stained with an antibody against the γδT-cell receptor or against CD3, no clustered perifollicular labeled cells could be detected. Because MAC can also express CD4 and because PICC display a dendritic phenotype, PICC are most likely composed of activated MAC. No obvious alterations in the number or appearance of mast cells was found at the site of PICC when histochemical stainings were used (not shown) (see Table 2).
ICAM-1-IR was upregulated both inside the PICC and on ORS keratinocytes of PICC+ HF. Of a total of 1728 analyzed HF, of which 19 had a PICC, all of these PICC except one expressed strong ICAM-1-IR (not shown).
Double labeling of ICAM-1 and MHC II on the same cross-sections of telogen back skin of five different mice, estimation of the ICAM-1-IR staining intensity (arbitrary units) of follicular keratinocytes, and measuring of the size of the associated PICC revealed that there was a statistically significant correlation between the ICAM-1-IR intensity of the ORS keratinocytes in these PICC+ follicles and the size of the PICC (Figure 5A) (r 2 = 0.86 for a linear correlation; p<0.01). In addition, the probability of PICC to occur appeared to correlate with the follicular ICAM-1-IR intensity (Figure 5B).
Some Hair Follicles with PICC Exhibit Signs of Degeneration
Analyzing mouse back skin with all HF in late anagen VI or catagen, we noted that some follicles with a large PICC showed signs of follicle degeneration. These PICC+ follicles differed in their degree of degeneration, as evidenced by the relative integrity of the basement membrane, the presence of MHC II-IR cells within the follicle epithelium, and the loss of ORS keratinocytes. The latter express strong IL-1 receptor Type I-IR (Maurer et al. 1995), which was therefore used as a convenient immunohistological keratinocyte marker. A moderate form of degeneration (Figures 3A-3E) manifested itself by a broadened follicular basement membrane, as visualized by a collagen IV antibody (Figures 3C and 3D; compare with Figure 3E).
Other HF with a large, dense, and strongly MHC II-IR PICC also showed intrafollicular MHC II-IR cells, which may derive from an upregulation of MHC Class II on follicular keratinocytes and/or from MAC penetrating the HF (Figure 3F). The ORS keratinocytes shown in the HF of Figures 3F and 3G still express IL-1RI-IR (Figure 3G). The basement membrane is present, although it appears to be partially disrupted (Figure 3H). In these cases, the proximal–distal expansion of the PICC can span almost the entire HF (not shown).
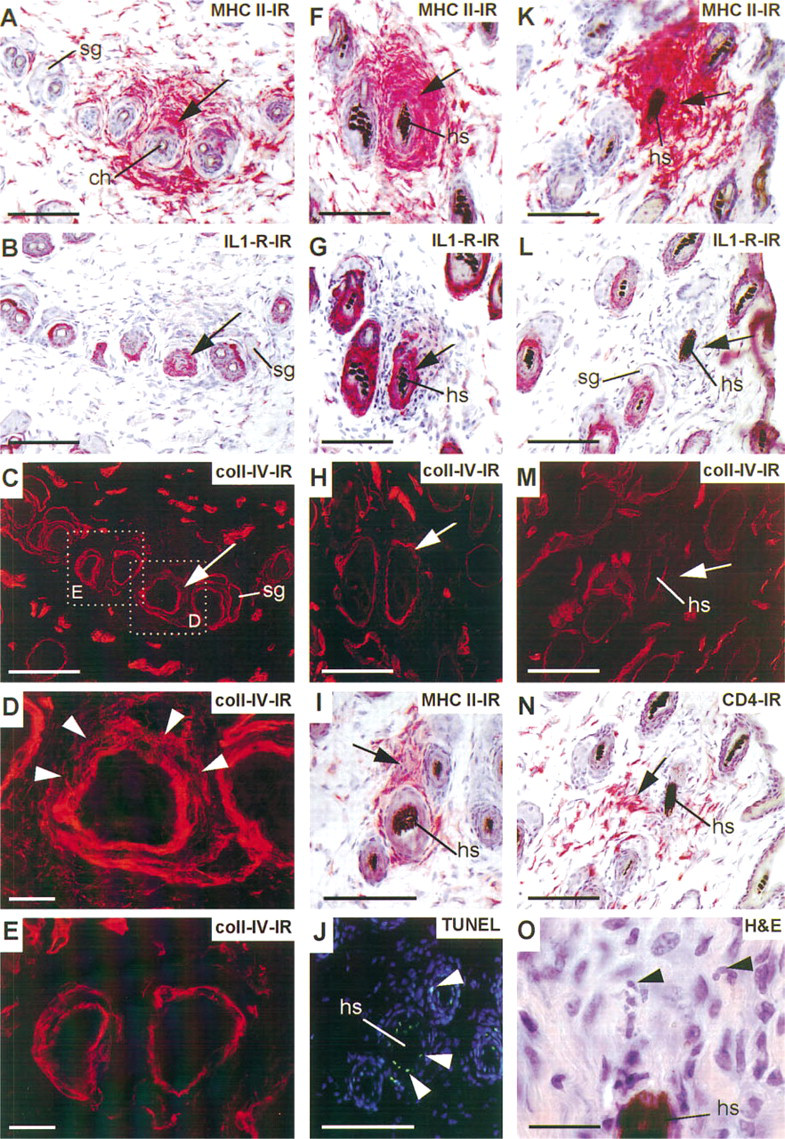
PICC and the affected hair follicles stained with various markers. Staining was performed on horizontal serial sections and longitudinal sections of untreated skin (
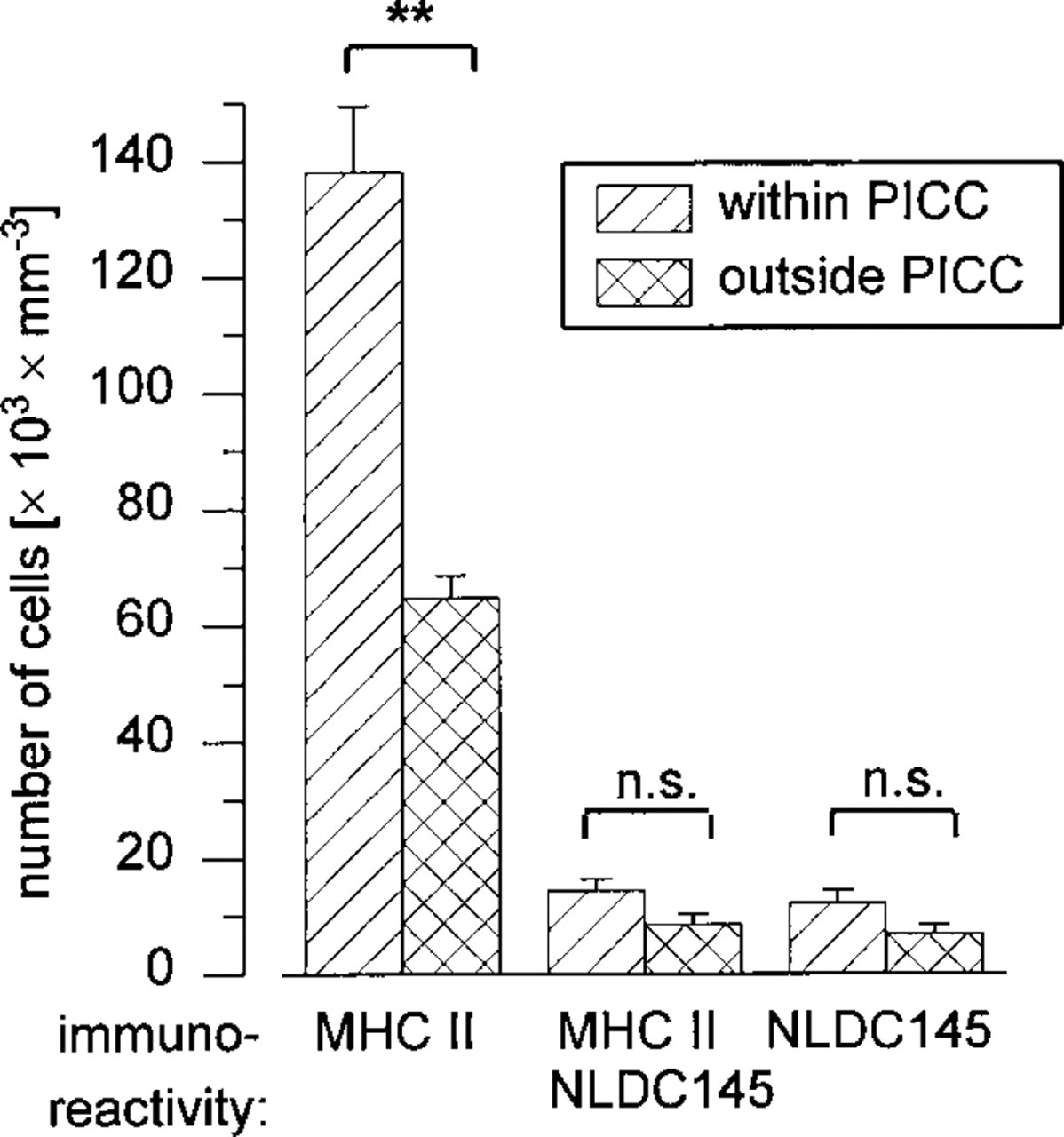
PICC consist predominantly of MHC II-IR cells. In double labeling experiments on the same sections, the number of cells per area within or outside of MHC II-IR cluster was counted separately for cells being MHC II+/NLDC145−, MHC II+/NLDC145+, and MHC II−/ NLDC145+. Bars give means of five different animals. ∗∗ p<0.01.
The clearest evidence for follicle degeneration in macroscopically completely normal–appearing anagen back skin can be seen in the follicle depicted in Figures 3K-3O. This series of cross-sections shows an HF with a large MHC II-IR PICC (Figure 3K). The follicle had lost all its ORS keratinocytes in this area, as judged by the absence of IL-1RI-IR (Figure 3L, arrow), and the basement membrane had largely disappeared (Figure 3M). A hematoxylin/eosin stain of the subsequent section revealed light microscopic signs of apoptosis (apoptotic bodies) close to the hair shaft, suggesting that the ORS keratinocytes had undergone apoptosis in this region (Figure 3O). This was confirmed in another example by in situ end-labeling, using a fluorescent TUNEL/Hoechst 33342 staining technique (Lindner et al. 1997). Figures 3I and 3J show sequential sections of PICC+ HF (Figure 3I) whose outer and inner root sheath display several TUNEL+ cells (Figure 3J).
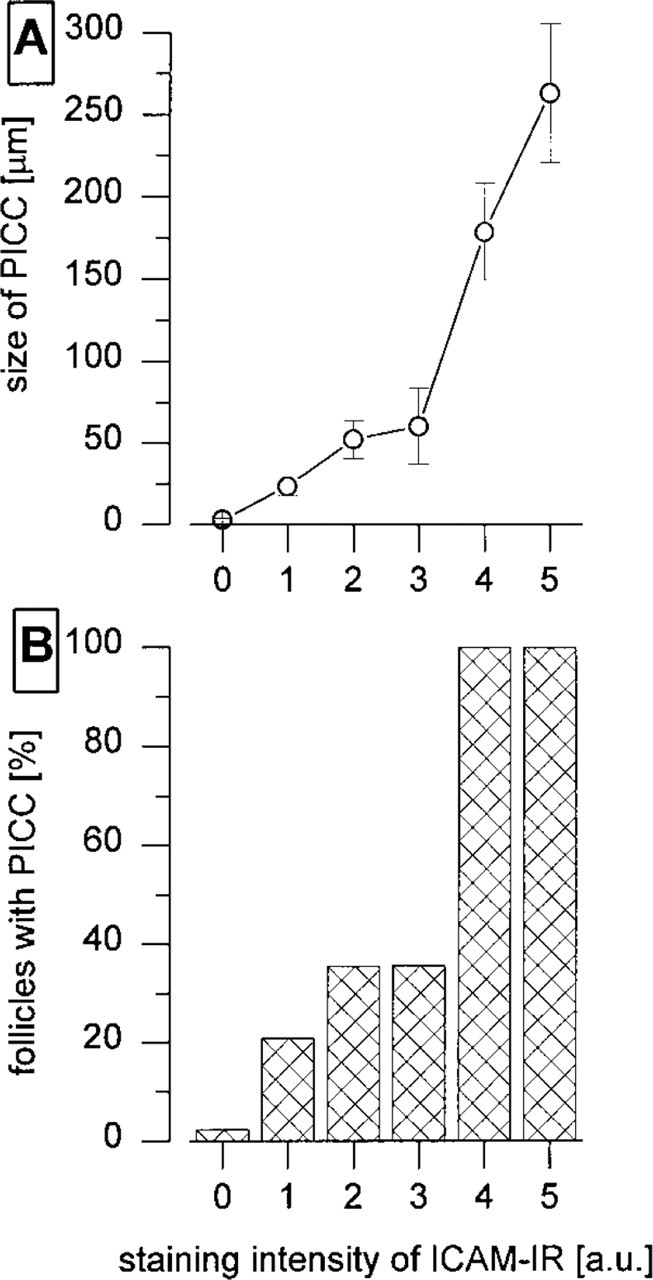
ICAM-1-IR keratinocytes correlate with PICC. This quantitation was done on untreated telogen skin using cross-sections and double labeling of MHC II and ICAM-1 on the same section. (
When eight mice of different hair cycle stages were analyzed using camera lucida-aided drawings of 6340 HF, we found this most severe type of HF degeneration in only eight HF, all of which were either in late anagen or in catagen, irrespective of whether anagen had been induced by depilation or had developed spontaneously. Approximately 10% of all PICC+ anagen and catagen HF showed signs of HF degeneration.
Discussion
Here we describe for the first time that macroscopically uninflamed, normal murine back skin contains regularly distributed PICC of largely MAC-like cells. These PICC are located around the distal noncycling portion of individual, fairly regularly spaced HF. These were present even in totally unmanipulated back skin with all HF in spontaneous telogen and in the absence of overt macroscopic or histochemical signs of skin infection, thus strongly suggesting that they are a physiological phenomenon. This did not appear to be a strain-specific curiosity or a phenomenon that depends on HF pigmentation. Two immunologically quite distinct mouse strains (C57BL/6, Balb/c), one black, the other white, both exhibit PICC+ follicles. Given the relative frequency of PICC+ follicles in normal mouse skin (around 2% of all HF) and the extensive scrutiny murine skin has been subjected to in countless immu-nohistological analyses over more than two decades (Bos 1997), it is quite surprising that this striking phenomenon has not been reported previously.
The presence, nature, and potential biological significance of PICC are interesting in a number of respects. They underscore the fact that mammalian skin can display substantial inflammatory cell infiltrates without any macroscopically visible signs of inflammation or the presence of infection, and that normal HF can display a large, dense infiltrate without suffering apparent damage (the vast majority of PICC+ follicles were morphologically indistinguishable from PICC− follicles, and signs of follicular degeneration were seen only in a minority of anagen VI and catagen follicles and not in telogen or early anagen follicles). A common problem in dermatopathological diagnosis is how to define exactly whether or not inflammatory cells located in the vicinity of an HF should be considered pathologically increased in number (Murphy 1995). Do at least some PICC in otherwise normal–appearing human skin, formerly interpreted as “pathological” (Lattand and Johnson 1975; Jaworsky et al. 1992; Hermes and Paus in press), represent only a physiological phenomenon?
PICC are always located at about the level of and in close proximity to the bulge region, the seat of follicular epithelial stem cells (Cotsarelis et al. 1990). In human skin, the occurrence of PICC at this level [as opposed to more proximally located PICC, such as the peribulbar PICC seen in alopecia areata with its reversible hair loss (Olsen 1994)], usually indicates an irreversibly damaging attack on the HF that will eventually result in permanent alopecia (cf. Murphy et al. 1991; Lavker et al. 1993; Elston and Bergfield 1994; Templeton and Solomon 1994; Hermes and Paus in press). It is most interesting, therefore, that the PICC observed in C57BL/6 and Balb/c skin in the vast majority of PICC+ follicles were not associated with any morphological signs of follicle damage, even in cases of very large and dense infiltrates (Figures 1 and 3A). Therefore, the mere occurrence of a PICC at the level of the bulge does not indicate with any certainty whether or not infiltrate-induced follicle damage is present or is bound to occur. Furthermore, because the center of the PICC is also close to the sebaceous gland, involvement of this tissue in the process of degeneration cannot be excluded.
Intriguingly, about 10% of the PICC+ anagen VI or catagen HF show signs of degeneration reminiscent of scarring or of “permanent” nonscarring alopecia (cf. Elston and Bergfield 1994; Templeton and Solomon 1994)—and this in mice with perfectly normal patterns of HF development and cycling (adolescent C57BL/6 mice). It is important to note that this follicular degeneration pattern is not a depilation-induced artifact, such as a sequela of follicle injury due to the trauma of anagen induction of depilation (cf. Argyris 1967), because PICC+ follicles with signs of ORS degeneration were also seen in spontaneously developed anagen VI HF. The few selected PICC+ HF with clear signs of permanent alopecia-like degeneration may well undergo degeneration because their stem cell-carrying ORS region has come under attack (as suggested by the large number of MHC Class II+ dendritic cells in the ORS, the upregulation of ICAM in this region, and the occurrence of ORS keratinocyte apoptosis). MAC activities, such as phagocytosis of the basement membrane of catagen hair bulbs, have long been implicated in the control of normal follicle regression (Parakkal 1969; Westgate et al. 1991; Paus 1996).
That PICC-associated follicle degeneration occurs only around individual anagen VI and catagen HF, i.e., follicles that are about to involute or are already in the process of regression, suggests that we are witnessing here an exaggerated form of the phagocytic activities MAC normally can display during HF regression (Parakkal 1969), with one key difference from normal catagen, i.e., that epithelial stem cells are eliminated. Teleologically, this segment of the hair cycle is ideally suited to permanently remove the entire follicle, provided that follicle stem cells are deleted as well, because normal follicle regression already is characterized by massive, apoptosis-mediated epithelial tissue involution in the follicle and by substantial matrix remodeling (Weedon and Strutton 1981; Paus et al. 1994d; Paus 1996; Lindner et al. 1997). In this light, it is only to be expected that the follicle degeneration detected here is not associated with “scarring” or any other disruption of skin physiology and architecture and has therefore long escaped recognition.
Naturally, the question arises as to whether this degeneration represents a physiological program of controlled follicle deconstruction or whether this kind of follicle degeneration is essentially pathological. We currently interpret PICC-associated follicle degeneration in otherwise perfectly normal murine skin as a basically physiological mechanism for removing damaged or malfunctioning follicles by programmed organ deletion (POD). Indeed, POD is a commonly employed, entirely physiological tool for tissue remodeling, which is seen, for example, in many insect and lower vertebrate species (cf. deletion of gills and tail during frog metamorphosis) as well as during embryonal and perinatal development in all mammals including humans [e.g., involution of aortic arches and omphaloenteric duct (Gilbert 1994; Müller 1997)]. However, if the degeneration of PICC+ HF is indeed a variant of physiological POD, this would be the first example of POD in the mammalian organism occurring long after birth. If POD only affects and deconstructs damaged HF, one wonders whether the deletion of such damaged tissue by activated MAC is not actually quite commonplace (cf. Vlassara et al. 1988).
Various forms of clinically perceptible, permanent alopecia [including, e.g., rare cases of androgenetic alopecia, alopecia areata, and psoriatic alopecia associated with permanent HF loss (Rook and Dawber 1991; Elston and Bergfield 1994; Templeton and Solomon 1994; Hermes and Paus in press)], may therefore represent only exaggerated, ill-controlled forms of this type of POD, some of which result in true scarring alopecia and some in a permanent loss of follicles without evidence of scarring (cf. Elston and Bergfield 1994). Therefore, it should be of profound clinical interest to dissect and identify the molecular controls of physiological follicle degeneration by POD in normal murine skin. This could provide invaluable new insights into how the progression of permanent alopecia in humans might be halted, which in most patients cannot be suppressed reliably and effectively by any current form of therapy (cf. Elston and Bergfield 1994).
A critically important question to be answered by future studies is how MAC become attracted to accumulate around individual HF. One reasonable explanation is that the follicle epithelium secretes chemoat-tractant cytokines, particularly those that it can express constitutively, such as TNF-α and IL-1αa (Bos 1997). This is supported by the observation that almost all PICC+ follicles also express “ectopic” ICAM-1-IR in the ORS at the level of the inflammatory infiltrate and that strong ICAM-1-IR can be observed in most PICC, which suggests the presence of an ICAM-1-upregulating local cytokine milieu, even though it does not reveal the source of cytokines (e.g., ORS keratinocytes vs PICC constituents). Once ICAM-1 has been upreg-ulated on the ORS, this may easily serve to attract additional inflammatory cells (cf. Zhang and Oliver 1994; Bos 1997).
Which mechanisms can be envisioned that might explain any such changes in the local cytokine milieu in and around the follicle bulge and the isthmus area? Hair follicle cycling as such, which is associated with profound alterations in the cutaneous and follicular cytokine milieu (cf. Paus 1996; Stenn et al. 1996), could be responsible. However, this appears unlikely, because of all the follicles in a single defined stage of the synchronized murine hair cycle only a small minority were PICC+ at any time point during the cycle, and no significant changes in the percentage of PICC+ follicles were detected over the hair cycle.
Alternatively, those follicles that have suffered some form of damage or malfunction might secrete cytokines that attract MAC to assist in damage repair or to initiate POD, once damage or malfunctioning has surpassed a critical threshold. Interestingly, even cyclosporin A-treated mice display PICC+ HF, suggesting that the signal for PICC accumulation is not cyclosporin A-sensitive.
In summary, our observations in murine skin suggest that the presence of even very dense perifollicular inflammatory cell infiltrates (PICC) does not necessarily indicate the presence of pathological processes. In addition, these infiltrates may serve as markers for follicles selected for programmed organ deletion (POD) and/or for HF that differ from their morphologically otherwise indistinguishable neighboring follicles in their local cytokine milieu.
Footnotes
Acknowledgements
Supported by grants from the Deutsche Forschungsge-meinschaft to RP (Pa 345/3–2, 5–1) and to IM (Mo 644/2–1).
The continued support of Prof W. Sterry is gratefully acknowledged. We thank R. Pliet, E. Hagen, and R. Böhmer for excellent technical assistance.
