Abstract
We performed a comparative investigation of the immunomorphological characteristics of lymphatic and blood microvascular endothelial cells in normal human skin, cutaneous lymphangiomas, and hemangiomas, employing a pre-embedding immunogold electron microscopic technique. We stained for cell membrane proteins that are commonly used for light microscopic characterization of blood endothelial cells. With blood microvascular endothelial cells, we observed uniform labeling of the luminal cell membranes with monoclonal antibodies (MAbs) JC70 (CD31), EN-4 (CD31), BMA120, PAL-E, and QBEND-10 (CD34), and strong staining of the vascular basal lamina for Type IV collagen under normal and pathological conditions. In contrast, lymphatic microvascular endothelial cells in normal human skin and in lymphangiomas displayed, in addition to a luminal labeling, pronounced expression of CD31 and CD34 along the abluminal cell membranes. Moreover, CD31 was preferentially detected within intercellular junctions. The expression of CD34 was mostly confined to abluminal endothelial microprocesses and was upregulated in lymphangiomas and hemangiomas. Type IV collagen partially formed the luminal lining of initial lymphatics and occasionally formed bridges over interendothelial gaps. Our findings suggest a function of transmigration protein CD31 in recruitment of dendritic cells into the lymphatic vasculature. CD34 labeling may indicate early endothelial cell sprouting. The distribution of Type IV collagen also supports its role as a signal for migration and tube formation for lymphatic endothelial cells.
Keywords
T
In contrast to our continuously increasing knowledge about the diverse functions of HDBMECs, little is known about human dermal lymphatic microvascular endothelial cells (HDLMECs). Previous morphological studies have identified the architecture of the dermal lymphatic microvascular system and have clearly defined the subcellular characteristics of HDLMECs (Ryan 1989). However, with the exception of one recent study (Erhard et al. 1996), most investigations of HDLMECs were performed by pure morphological methods. Therefore, little is known about the immunomorphological characteristics of HDLMECs in situ at the ultrastructural level.
In this study we focused our interests on the immunophenotypical and immunomorphological characteristics of HDLMECs to obtain a better understanding of the biology of this particular cellular component of the human skin. We performed a pre-embedding immunogold electron microscopic study with a panel of well-characterized EC reactive/specific monoclonal antibodies (MAbs) that are directed against cell membrane-associated proteins and Type IV collagen. After determination of the immunomorphological phenotype of the HDLMECs, we compared these findings with those on blood ECs. To assess whether the immunomorphological features of resting HDMECs in normal human skin are maintained during benign proliferation, we then investigated the ultrastructural immunomorphology of capillary hemangiomas and lymphangiomas.
Here we present the ultrastructural characteristics of resting and proliferating human dermal microvascular endothelial cells (HDMECs) in situ, that clearly allow the differentiation of these two cell populations by ultrastructural immunomorphological criteria. Because the distribution of certain surface proteins with lymphatic ECs indicates biological relevance, these findings contribute to our understanding of the function of these cells and may be helpful in clarification of the ontogeny of certain vascular skin tumors.
Materials and Methods
Tissue Specimens
Biopsies/excisions were taken under local anesthesia with 2% mepivacaine of normal human skin from 10 healthy volunteers, four patients with hemangiomas, and five with lymphangiomas. All tissue specimens underwent an identical technical procedure. Immediately after biopsy the tissue was rinsed in a precooled solution of physiological NaCl and was divided with a razor blade on a wax plate into pieces of approximately 1 mm3. From each biopsy specimen, one portion was fixed in 7.5% formaldehyde and further processed for routine histopathology. For light microscopic immunohistological studies, another part of the biopsy specimen was snap-frozen in Tissue-Tek OCT compound (Miles Scientific; Naperville, IL) using isopentane precooled in liquid nitrogen, and then stored at −70C until further use. Another part of the biopsy specimen was processed for immunoelectron microscopy. After fixation in a freshly prepared and slightly modified periodate–lysine–paraformaldehyde fixative for 4 hr at room temperature (RT) and a thorough rinse in PBS, pH 7.4, three times for 15 min at 4C, the specimens were infiltrated with 10% dimethylsulfoxide dissolved in PBS for 1 hr at 4C, snap-frozen, and stored in liquid nitrogen (McLean and Nakane 1974; Sterniczky et al. 1996).
Antibodies
We selected a set of six MAbs (Table 1) that were all known to react with the cell membrane of EC or with the basal lamina: MAbs JC70 and, reportedly, EN-4 are directed against CD31, a glycoprotein of 130 kD that is found on various hematopoietic cells such as bone marrow stem cells, CD8+ cells, monocytes, neutrophils, platelets, and EC (Parums et al. 1990; Burgio et al. 1994). This molecule is involved in homotypic and heterotypic cell adhesion and mediates the transmigration of leukocytes from the blood vessel lumen into the extravascular space (Hirschberg et al. 1980; Muller et al. 1993; Newman 1994; Liao et al. 1995; Muller 1995). QBend10 is a monoclonal reagent that recognizes CD34, a transmembrane glycoprotein of 115 kD, present on immature hematopoietic precursor cells and ECs. Its function still awaits clarification (Fina et al. 1990; Ramani et al. 1990). MAb PALE reacts with an antigen that is exclusively localized in coated pits and vesicles of blood ECs; the target protein has not yet been biochemically characterized (Schlingemann et al. 1985). MAb BMA120 detects a glycoprotein of 200 kD that is present exclusively on ECs (Alles and Bosslet 1986), and Type IV collagen is a constitutive protein of the lamina densa of basement membranes (Barsky et al. 1983).
List of first-step reagents
For light microscopic immunomorphological studies, 4-μm-thick serial cryosections were mounted on gelatin-coated slides, air-dried, and fixed in acetone for 10 min at −20C. For blocking of Fc Ig receptors, sections were preincubated in PBS, pH 7.4, supplemented with 1% bovine serum albumin (BSA) for 20–30 min at RT. For morphological orientation, the first section of each tissue block was stained with hematoxylin and eosin. For immunohistological staining the three-step avidin–biotin–immunperoxidase technique was employed using a Supersensitive Staining Kit (Bio Genex Laboratories; San Ramon, CA). Sections were incubated with the first-step antibodies (first-step antibodies used are listed in Table 1), appropriately diluted in PBS/1% BSA for 1 hr at 4C. Thereafter, the slides were thoroughly washed in PBS/0.1% BSA and consecutively reacted with an appropriately diluted biotinylated anti-mouse IgG. After three washes in PBS/0.1% BSA, the slides were incubated with peroxidase-conjugated streptavidin. Bound immunoreactants were visualized with AEC as chromogen.
Immunoelectron microscopic experiments were performed with 15–20-μm-thick cryostat sections that were prepared with a Jung CM3000 cryomicrotome (Leica; Vienna, Austria) and were immediately rinsed in PBS at 4C for 20 min. To reduce nonspecific antibody binding via Fc Ig receptors, the specimens were preincubated in PBS/1% bovine serum albumin (BSA) for 30 min at RT in 5-ml glass tubes. Afterwards, the specimens were incubated with the first-step reagents appropriately diluted in PBS/0.1% BSA for 8 hr at 4C (Table 1). After thorough rinsing in PBS/0.1% BSA, three times for 60 min at 4C, the sections were incubated with goat anti-mouse IgG (Fc) conjugated to 15-nm colloidal gold particles (Amersham International; Poole, UK), diluted in PBS/1% BSA for another 8 hr at 4C. To remove unbound immunoreagents, the tissue was washed in PBS/0.1% BSA two times for 60 min at 4C and once in 0.1% cacodylate buffer. The specimens were consecutively fixed in 2% glutaraldehyde for 1 hr at RT, rinsed in 0.1% cacodylate buffer for 1 hr at RT, postfixed in Palade's osmium, and contrasted with uranyl acetate. After dehydration of the specimens in a graded series of ethanols and infiltration with propylenoxide and Epon 812, the resin was polymerized at 60C. Thin sections were cut with a Reichert Ultracut 2000, contrasted on grids with lead citrate/uranyl acetate, and examined with a JEOL 1200 EX electron microscope.
Controls
Negative controls included omission of either the first- or the second-step reagent and substitution of the first-step reagent with an irrelevant isotype-matched antibody.
Results
Light microscopic Immunophenotype of the Dermal Microvasculature
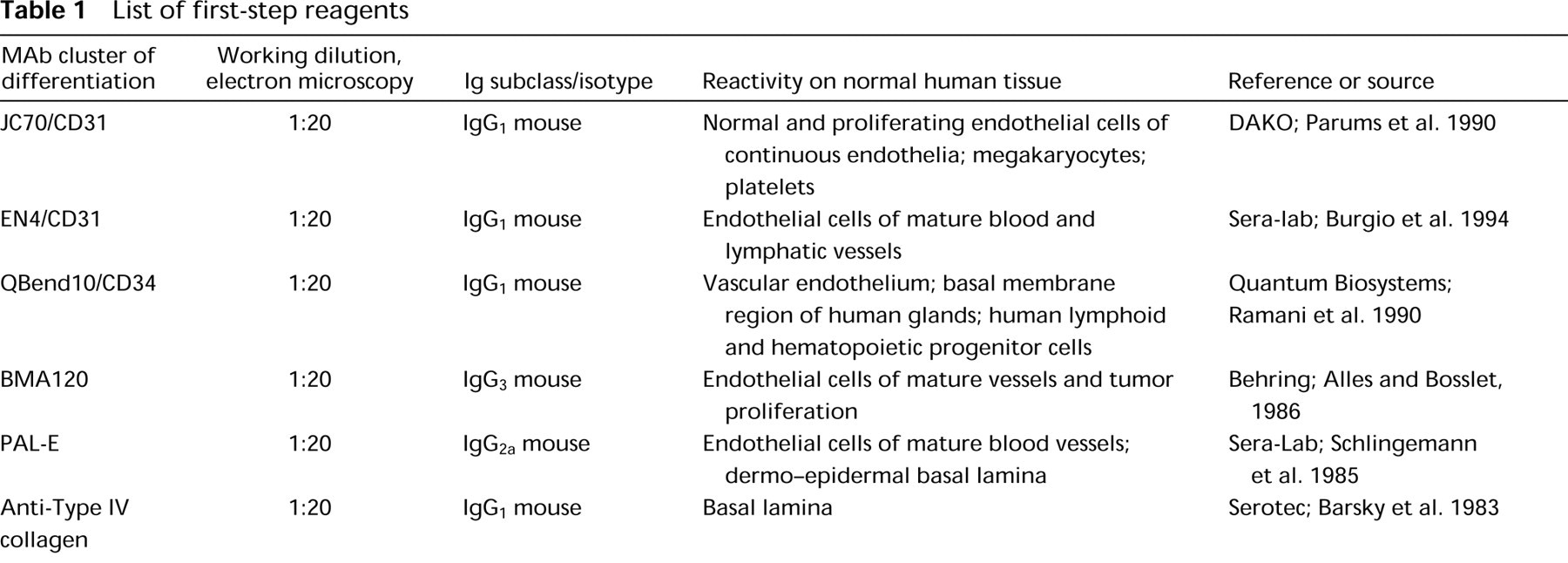
In the papillary dermis of normal human skin, ECs of blood vessels such as arterioles, capillaries, and postcapillary venules were regularly stained with all antibodies employed (Figure 1). The most intense labeling was seen with MAbs EN-4 and JC70/CD31 and, to a lesser degree, with MAbs PAL-E, QBend10/CD34, and BMA120. Staining of Type IV collagen revealed a well-developed basement membrane around these vascular structures.
Staining of serial sections of normal human skin (papillary dermis) with MAb JC70/CD31 (
In addition, we observed flattened, thin cords of cells without any discernible lumen formation. In consecutive serial sections these structures displayed reactivity with MAbs EN-4, JC-70, and BMA-120, and also labeling with MAb QBend10, but the cords failed to react with MAb PAL-E. The additional observation of a Type IV collagen-positive basal lamina around these structures most likely indicated that they represent collapsed lymphatic capillaries.
Investigating the immunophenotype of proliferating vessels of cutaneous capillary hemangiomas and lymphangiomas, we found identical immunophenotypic features as for blood and lymphatic capillaries, respectively, in normal human skin (data not shown).
Ultrastructural Morphology of Blood and Lymphatic Capillaries in Normal Human Skin
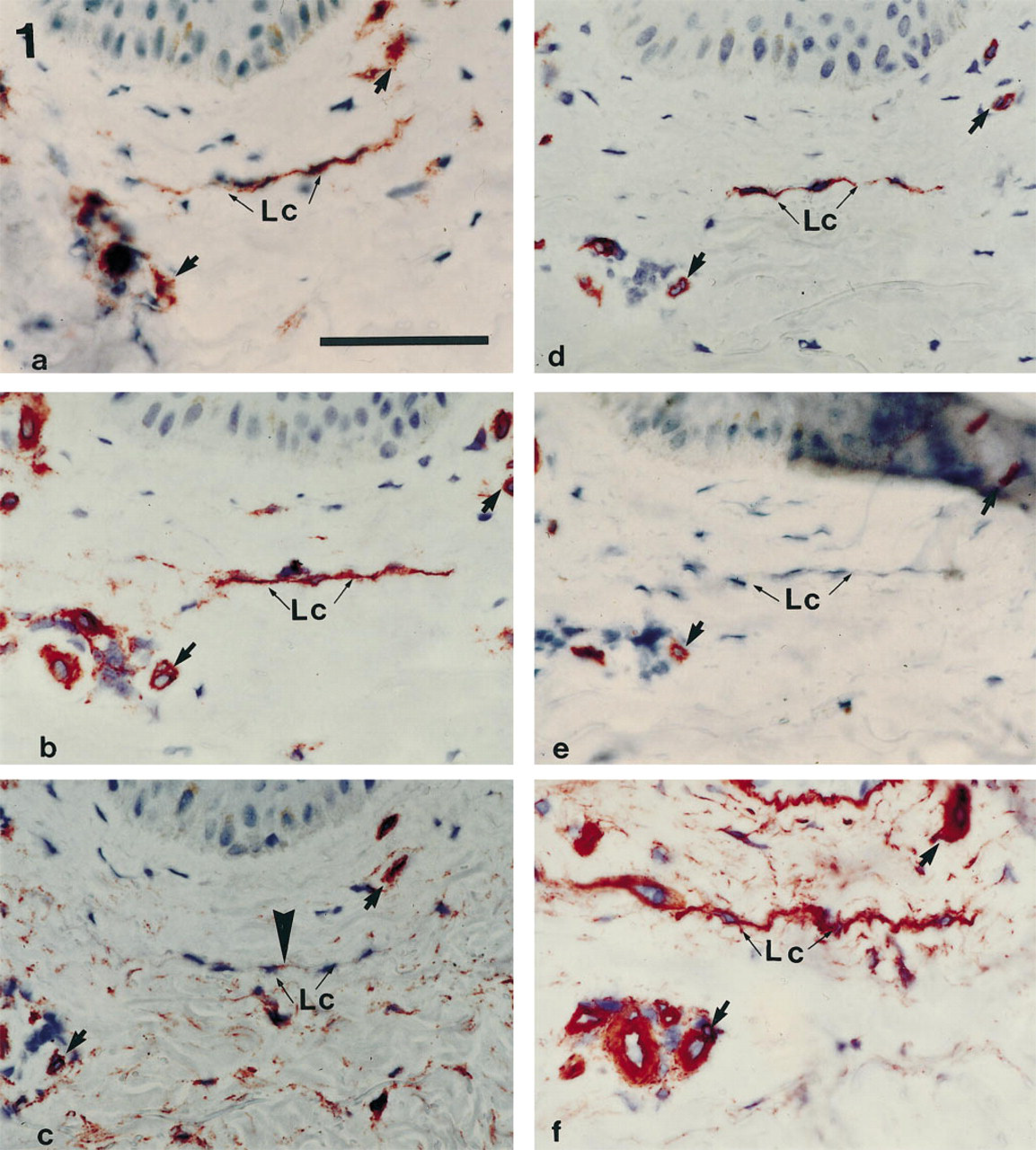
ECs of blood and lymphatic capillaries were morphologically identified in accordance with previous investigations (Thorgeierson and Robertson 1978; Daroczy 1988; Ryan 1989). The lumen of blood capillaries is formed by one or two ECs that characteristically bear Weibel–Palade bodies within their cytoplasm. The abluminal cell membrane is enveloped by a well-developed continuous basement membrane that occasionally displays reduplications of the lamina densa. Within the perivascular space one regularly observes pericytes, which are also surrounded by a basement membrane (Figure 2).
Immunoelectron microscopy of a dermal blood capillary reactive with MAb QBend10/CD34. This vessel is lined by two blunt endothelial cells (
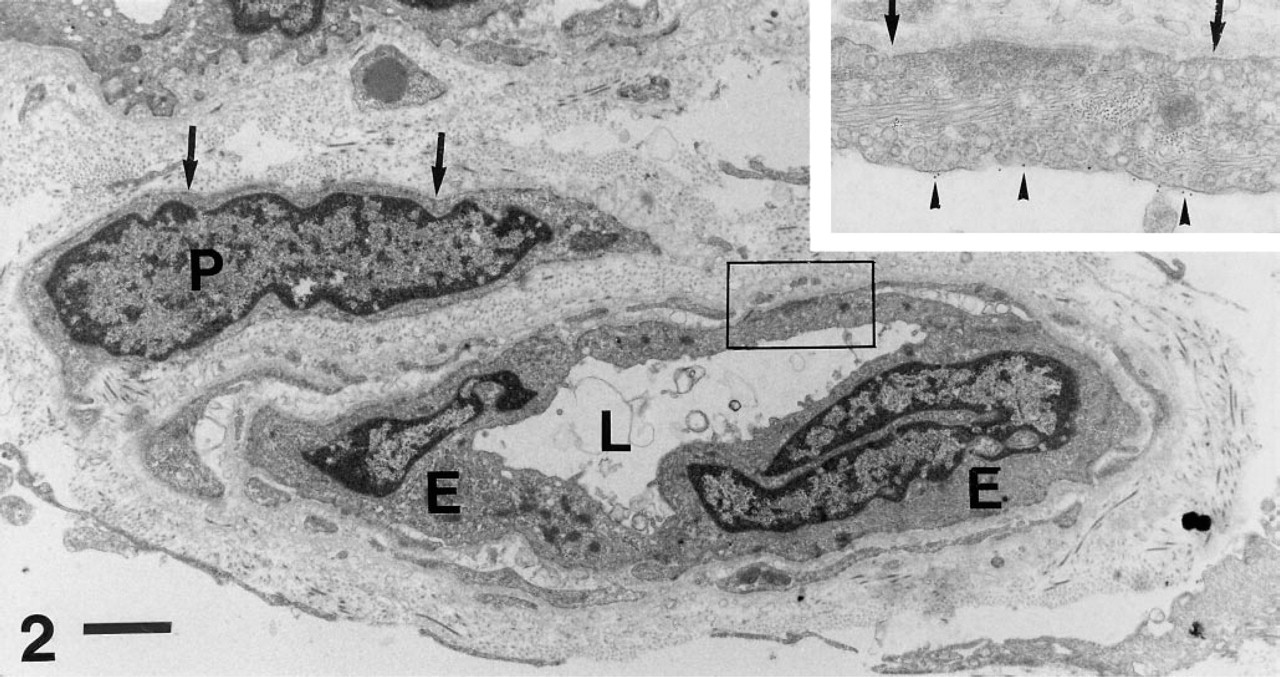
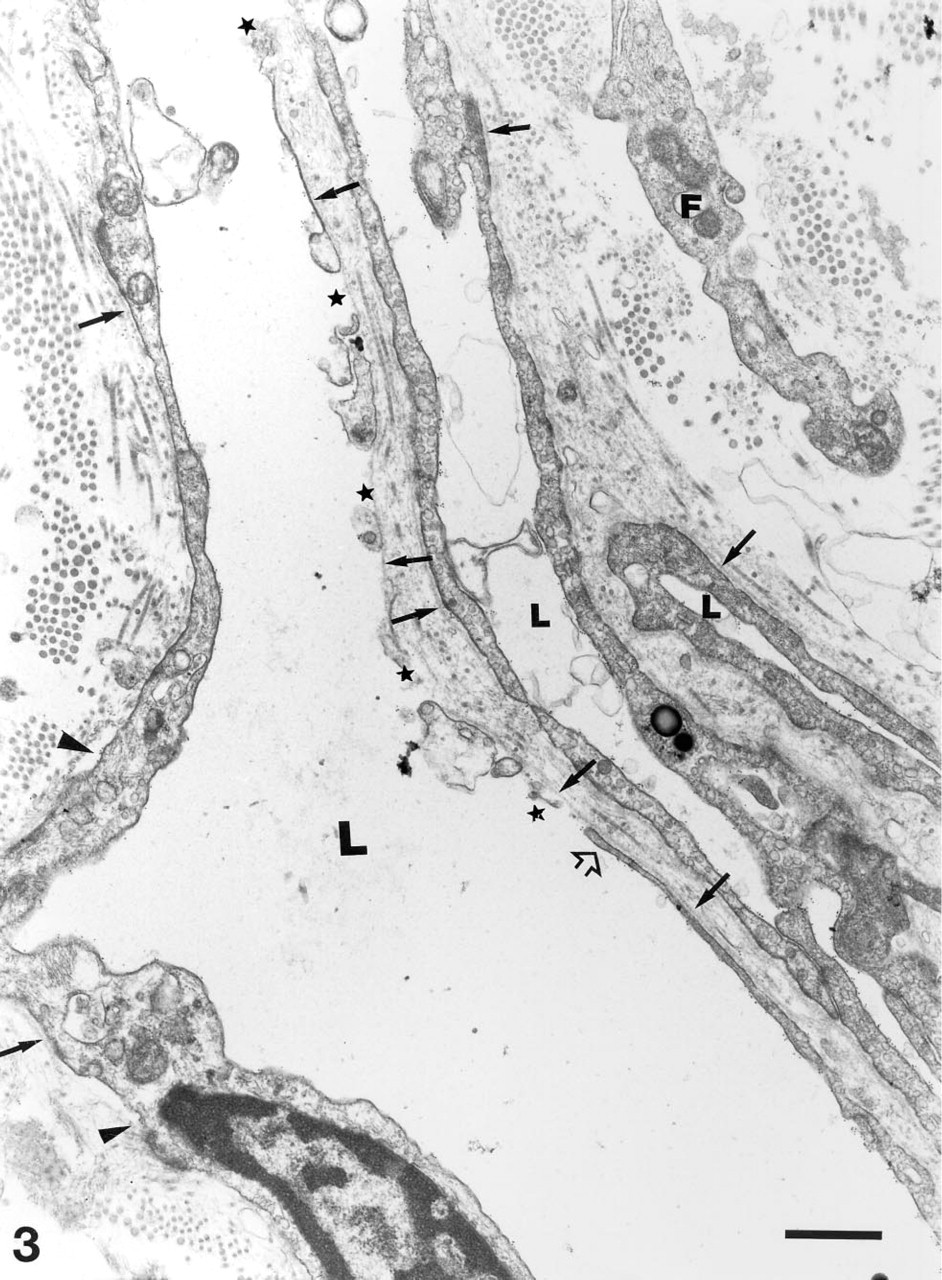
Lymphatic capillaries originate as tiny, thin open tubes that are formed by extremely attenuated, almost spindle-shaped ECs and, occasionally, are completely devoid of an EC lining, but are enveloped by lamina densa-like structures that form the vessel wall at the most distal ending of the lymphatic channels. Along such laminar structures, lymphatic EC processes align and form the capillary tube (Figure 3). The cytoplasm of lymphatic ECs lacks cell-specific organelles such as Weibel–Palade bodies. However, lipid droplets and dense bodies are frequently seen. The continuity of the endothelial lining is provided by cytoplasmic processes of lymphatic ECs that form interdigitating, overlapping, and end-to-end-type junctions (Figure 4). In addition, along these junctions the cell membranes may form characteristic intercellular adherens junctions that appear as desmosome-like structures and tight junctions. Nevertheless, large gaps are often observed between two neighboring ECs (Figure 3). Lymphatic capillaries are lined with a thin and discontinous but well-preserved basement membrane that may bridge over interendothelial gaps, thus forming the boundary between the lymphatic lumen and the interstitial connective tissue (Figures 3 and 4). Another specific morphological feature of lymphatic capillaries is the intimate association with elastic and collagen fibers and fibrils that insert either directly or via microfilaments within the abluminal cell membrane. However, lymphatic capillaries lack the presence of accompanying pericytes (Figures 3 and 4).
Immunoelectron microscopy of initial dermal lymphatic capillaries. The cleft-like lumina (
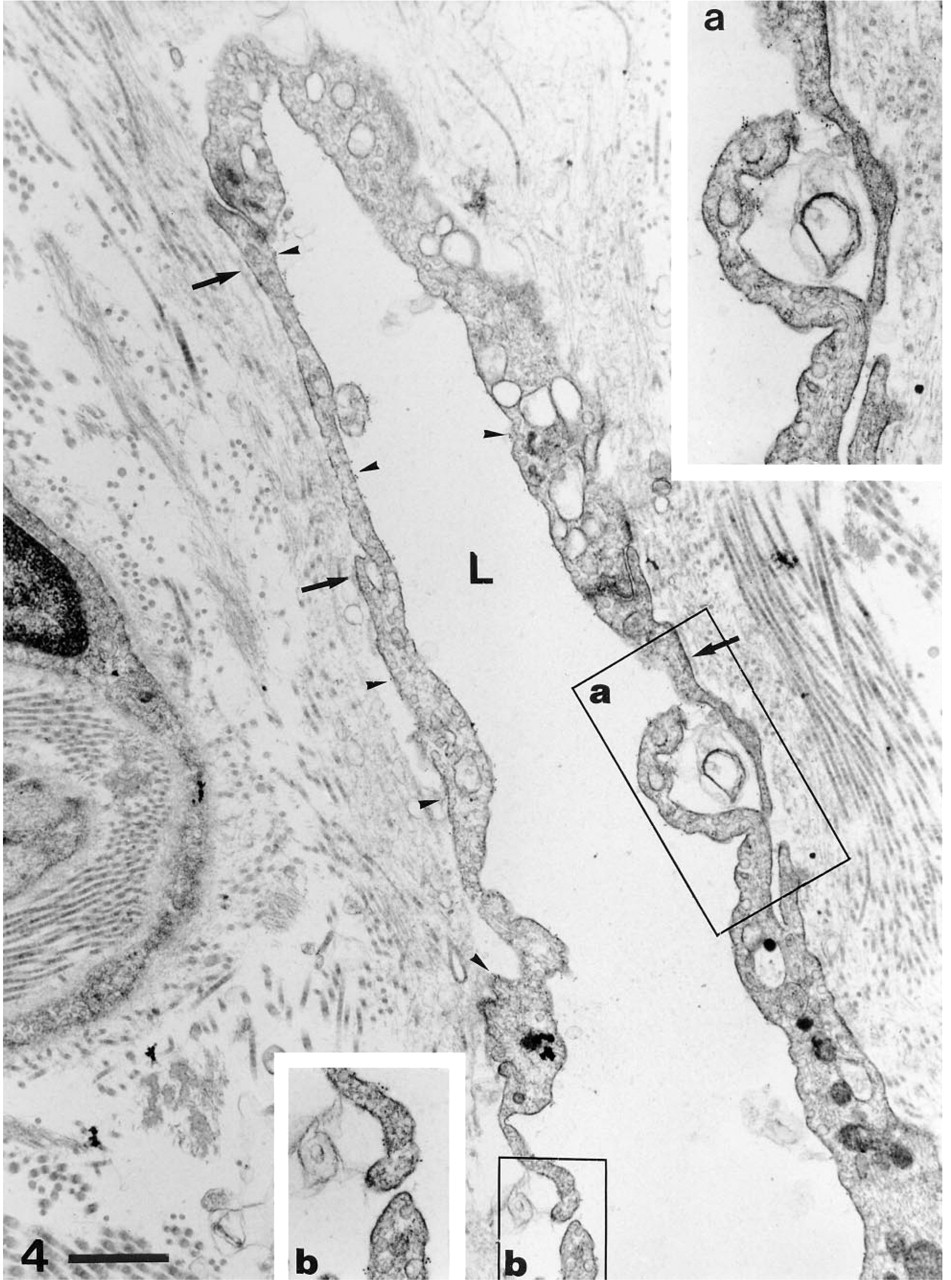
Immunoelectron microscopy of a longitudinally sectioned lymphatic capillary labeled with MAb EN-4/CD31. The narrow lumen (L) is lined by attenuated endothelial cells that form overlapping-junctions (
Immunophenotype of Blood and Lymphatic Capillaries
Morphologically characterized blood and lymphatic microvascular ECs, but no other cellular component of the dermal microvasculature of normal human skin, exhibited labeling with MAbs JC70, EN-4, QBend10, and BMA120 (Figures 1–4).
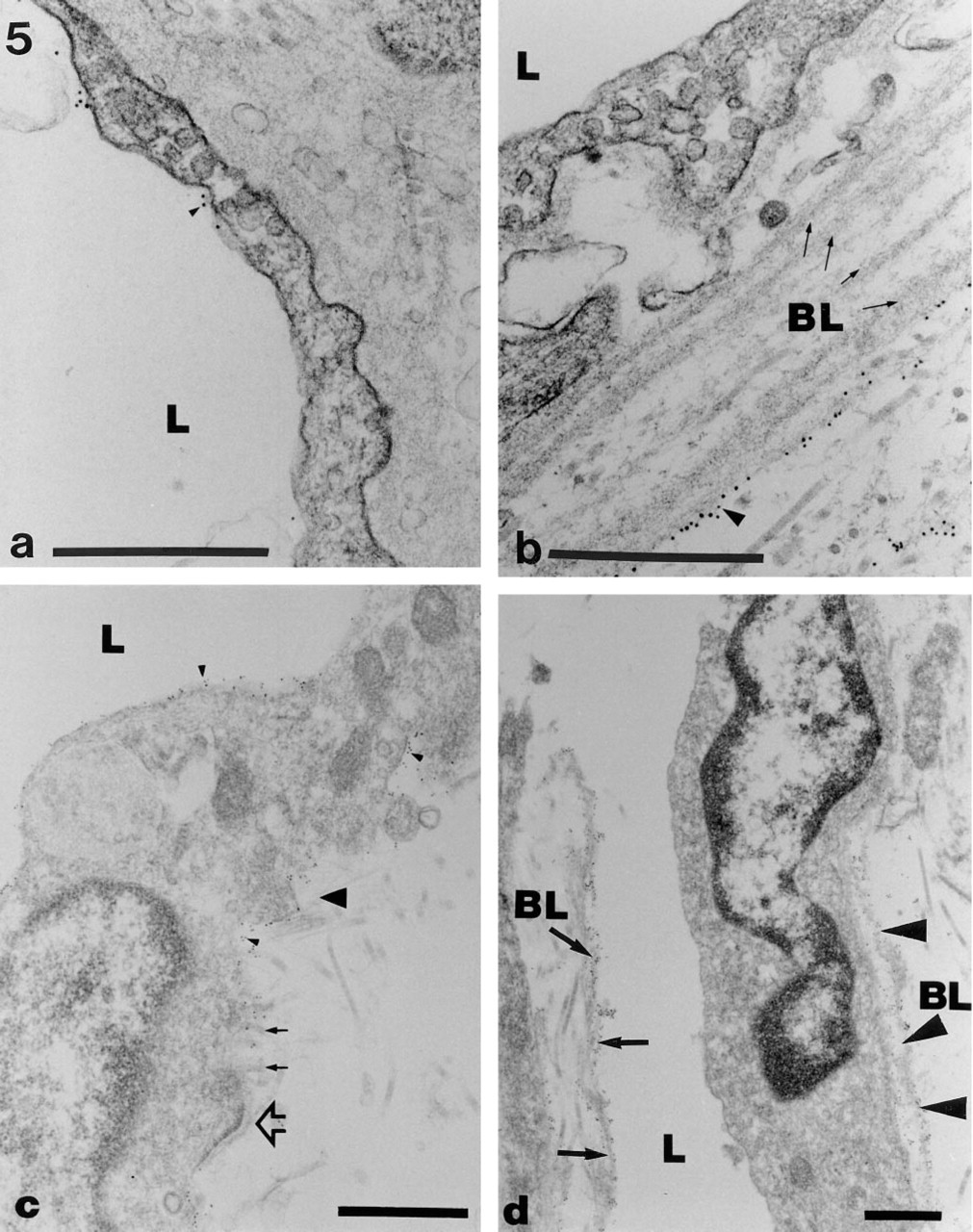
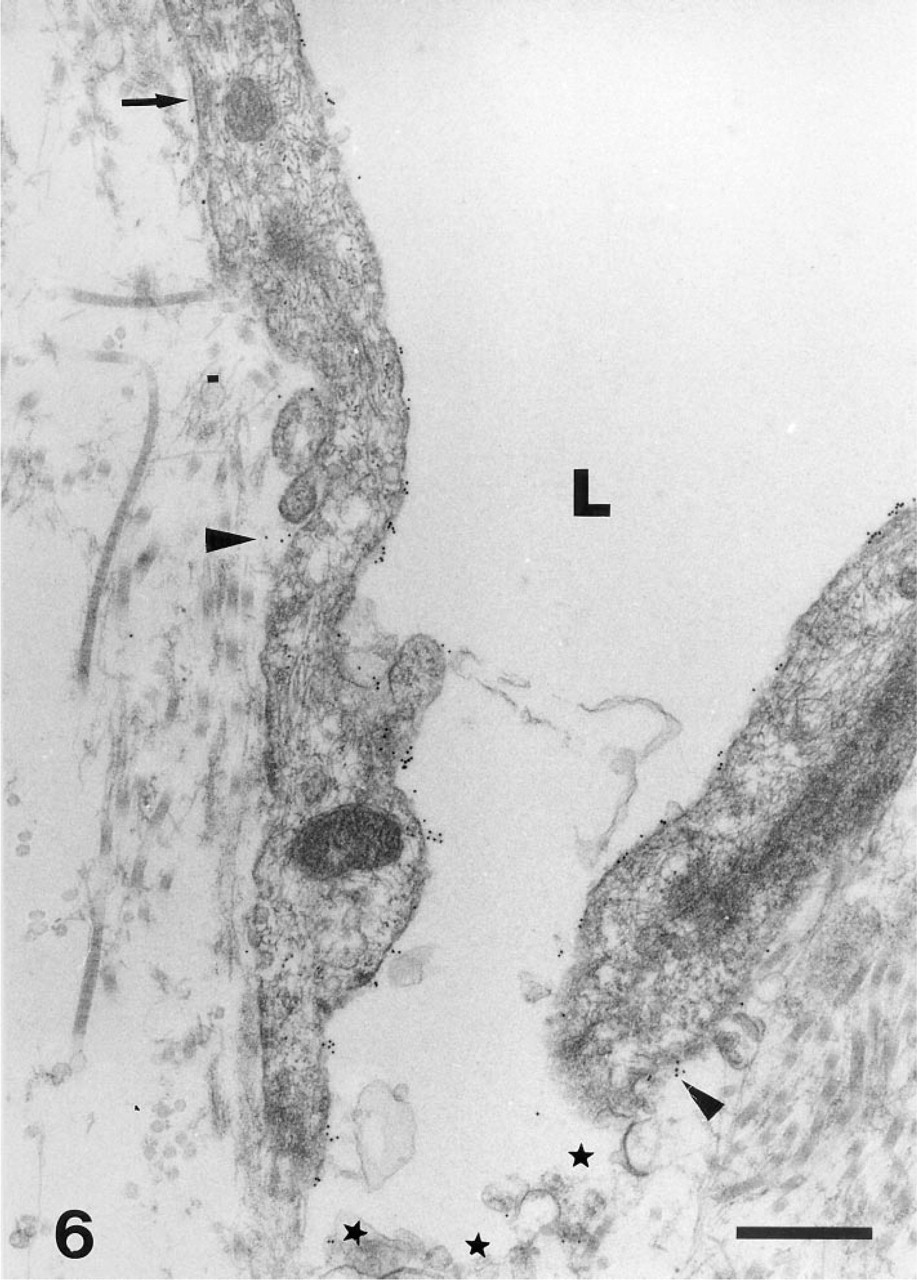
The number of colloidal gold particles indicating the binding of MAbs and thus the expression of antigens is shown in detail in Table 2. The overall labeling intensity was higher with blood capillaries than with lymphatics. Quantitative analysis of bound immunogold particles per 100 μm abluminal and luminal EC cytoplasmic membrane displayed certain characteristic immunophenotypic differences between blood and lymphatic capillaries. The most intense labeling of blood capillaries was seen with MAbs JC70 and EN 4 and was somewhat weaker with QBend10, whereas the labeling with MAbs BMA-120 and PAL-E was much weaker. Immunolabeling with PAL-E was restricted to blood microvascular ECs. PALE labeling often revealed a preferential binding to coated pits and vesicles of the luminal cell membrane, as was reported previously for cultured blood ECs (Schlingemann et al. 1985). The major differences in the immunopheno-type between blood and lymphatic microvascular endothelial cells were observed with MAbs QBend10, JC70, and EN-4, in that these reagents also stained portions of the abluminal cell membrane of lymphatic ECs (Figures 2–4). Abluminal CD34 labeling accumulated along portions of the cell membrane that were devoid of a basement membrane (Figures 2, 6). Abluminal CD31 labeling was pronounced along the cell membrane of cytoplasmic processes that formed overlapping and end-to-end-type junctions. Such junctions displayed a striking continuity of expression of CD31 from the luminal to the abluminal cell membrane (Figure 4). Labeling with MAb-BMA120 was always restricted to the luminal cell membrane of blood and lymphatic ECs and did not show a preferential binding to a defined subcellular structure.
Immunoelectron microscopic characterization of blood and lymphatic microvascular endothelial cells: gold particles per 100-μm length of luminal and abluminal cell membrane

As expected, staining for Type IV collagen demonstrated well-developed, occasionally reduplicated laminae densae around ECs and pericytes of blood capillaries. Moreover, it also confirmed the presence of a Type IV collagen-positive fragmented lamina densa with lymphatic capillaries. In addition, the laminar structures that were bridging over interendothelial gaps of lymphatic capillaries and forming the vessel walls of initial lymphatics, as shown in Figure 2, displayed a striking reactivity with anti-type IV collagen MAbs (Figure 5d).
The immunophenotype of endothelial cells in hemangiomas and lymphangiomas equals that of blood and lymphatic capillary endothelial cells, respectively, in normal human skin. CD31 expression with endothelial cells of hemangiomas is restricted to the luminal cell membrane (arrowheads,
Endothelial cells of a lymphangioma express large amounts of CD34 along the luminal cell membrane, and minute labeling is also seen along portions of the abluminal cell membrane (arrowheads). Stars indicate an interendothelial gap; arrow demonstrates portions of a basal lamina. Arrowheads indicate the lamina densa. L, lumen. Bar = 1 μm.
Immunophenotype of Hemangiomas and Lymphangiomas
To prove our findings in normal human skin, we also investigated the immunomorphological characteristics of benign microvascular tumors with blood and lymphatic differentiation, i.e., hemangiomas and lymphangiomas. As with lymphatic capillaries in normal human skin, the most striking observation was the expression of CD31 and CD34 on the abluminal cell membranes of lymphatic EC in lymphangiomas (Figures 5c and 6). Surprisingly, the most pronounced staining was seen with MAb QBend10, which indicated an increase in CD34 expression on both luminal and abluminal portions of lymphatic capillaries by a factor of 2. In addition, with hemangiomas there was a slight increase in CD34 expression (Table 2; Figures 5a and 6). In situ labeling with antibodies QBend10, JC70, EN-4, and BMA120 did not disclose a localization to any defined subcellular structure but was evenly distributed along the cell membrane. PAL-E labeling was confined to blood vessels in lymphangiomas. The lamina densa of the basement membranes of both blood and lymphatic ECs, as well as pericytes, always displayed strong expression of Type IV collagen (Figures 5b and 5d). It is of note that proliferating lymphatic capillaries, like normal lymphatic vessels, showed large interendothelial gaps. Frequently these gaps were bridged by a well-developed Type IV collagen-positive lamina densa that formed the boundary to the surrounding connective tissue (Figure 5d). As in normal skin, proliferating lymphatic ECs were intimately connected with the surrounding connective tissue by elastic and collagen fibers as well as by micro-fibrils that inserted into the abluminal cell membrane (Figure 5c).
Discussion
Light microscopic immunohistochemistry is an adequate tool to grossly characterize immunophenotypic characteristics of cellular components in vitro and in vivo and therefore plays a major role in diagnostic pathology. However, because the expression of cell surface proteins is mostly associated with functional properties, identification of the exact immunolocalization of such molecules may be helpful in the understanding of biological functions. Such investigations are limited by the resolution of light microscopic techniques and therefore require immunoelectron microscopic techniques to define the ultrastructural localization of antigens.
Immunoelectron microscopic studies of human tissue taken ex vivo are largely hampered by technical problems, because the preservation of the ultrastructural morphology by fixation with denaturing and crosslinking aldehydes strikingly reduces the antigenicity of the structures studied. In this study we employed a technique that provides excellent preservation of both subcellular morphology and antigenicity of the tissue, allowing an unequivocal assignment of the immunolabel to ECs defined as blood or lymphatic microvascular ECs by morphological criteria. Using a panel of different MAbs directed against EC and Type IV collagen on normal human skin, hemangiomas, and lymphangiomas, we found a clear heterogeneity of the immunophenotype of blood and lymphatic HD-MECs, in situ, at the ultrastructural level. The major findings of this study relate to the differential expression of CD31 and CD34 as well as Type IV collagen with lymphatic and blood microvascular ECs.
All dermal microvascular structures displayed a clear-cut labeling of the luminal cell membranes of ECs with anti-CD31 and anti-CD34 reagents as well as with MAb BMA-120, whereas PAL-E reactivity was restricted to blood microvascular ECs. The most striking observation of this study was the finding of concomitant abluminal and luminal expression of CD31 and CD34, respectively, by lymphatic microvascular endothelial cells. Abluminal expression of CD31 was never observed in blood capillaries, despite the fact that other investigators have detected CD31 on the abluminal cytoplasmic membranes of endothelial cells of cutaneous venous vessels (Erhard et al. 1996). CD31 expression was most pronounced along cytoplasmic processes of lymphatic ECs that formed overlapping and end-to-end connections. Because such sites of the cell membranes were consistently devoid of morphologically well-defined intercellular adherens junctions, i.e., desmosomes or tight junctions, these CD31-positive adhering cell processes obviously represent rather loose connections that may provide direct interaction between the vessel lumen and the extracellular space for exchange of fluid or particular substances and cellular components. CD31 was recently shown to play a crucial role in the transmigration of leukocytes from the lumen of blood microvessels into the extravascular space. The demonstration of an abluminal expression of this transmigration protein exclusively by lymphatic ECs and its preferential localization within intercellular junctions suggest that this molecule is also involved in cellular traffic in the opposite direction, e.g., recruitment of leukocytes into the lymphatic vasculature on their way into regional lymph nodes (Larsen et al. 1990; Muller et al. 1993; De Lisser et al. 1994; Newman 1994; Liao et al. 1995; Muller 1995; Lukas et al. 1996). Such phenomena occur with antigen-presenting cells and cutaneous dendritic cells in the induction phase of delayed-type hypersensitivity reactions and have been reproduced in a skin organ culture model, and with Leishmania-infected Langerhans cells that home into regional lymph nodes (Silberberg et al. 1976; Blank et al. 1993; Lukas et al. 1996; Weiss et al. 1997).
Recent investigations have clearly confined the expression of the human progenitor cell marker CD34 to blood endothelial cells (Parums et al. 1990; Ramani et al. 1990; Kuzu et al. 1992).
Most studies demonstrated CD34 along the luminal cell membrane. Occasionally however, abluminal expression of CD34 was found in vivo and in vitro (Schlingemann et al. 1990). In this study we have observed CD34 expression on lymphatic capillaries in normal skin and in lymphangiomas on both the luminal and abluminal portions of the cell membranes of ECs. This finding is new and is in contrast to previous light microscopic observations in pathological conditions such as lymphatic amyloidosis that failed to demonstrate CD34 reactivity of lymphatic vessels (Kaiserling et al. 1994). The discrepancy of these findings could be explained by the fact that CD34 expression by lymphatic capillaries is hardly discernible at the light microscopic level, which is also evident from this study. This observation, however, clearly emphasizes that immunoelectron microscopic studies are required for the ultimate immunolocalization of certain antigens. Previous observations of abluminal expression of CD34 with blood capillaries in tumor angiogenesis and during embryogenesis suggested its upregulation during sprouting and migration (Schlingemann et al. 1990). Our observations of abluminal expression of CD34 by lymphatic endothelial cell processes and also the upregulation of CD34 expression in lymphangiomas may also indicate migratory properties and suggest sites of initial EC sprouting in the early phase of lymph vessel neogenesis and proliferation (Leak 1976; Ryan et al. 1986).
In addition, angiogenesis and angioproliferation in vitro depend on the presence of the basement membrane proteins laminin and Type IV collagen, which promote the rapid alignment and differentiation of HDMECs into capillary-like tubes in vitro (Form et al. 1986; Kubota et al. 1988). Similar to these in vitro findings, we detected basement membranes in close association with CD34-positive EC processes forming the boundary of initial lymphatics as well as Type IV collagen-positive laminar structures spanning interendothelial gaps of normal lymphatic capillaries and proliferating vessels in lymphangiomas in situ. These observations suggest a role of basement membrane protein Type IV collagen as a signal for the migration, alignment, proliferation, and differentiation into mature capillaries of sprouting lymphatic microvascular ECs in vivo.
The labeling characteristics of ECs observed in normal human skin were identical to those in hemangiomas and lymphangiomas and therefore indicate a preservation of antigenic and functional properties of HDMECs in cases of benign proliferation.
Taken together, we present immunomorphological data that indicate a pronounced antigenic heterogeneity of HDMECs of blood and lymphatic origin in situ at the ultrastructural level. The characteristic immunolocalization of CD31 and CD34 by lymphatic ECs and the distribution of Type IV collagen with lymphatic microvessels allows a clear-cut immunophenotypic differentiation between these two distinct cell populations at the subcellular level and also indicates biological relevance.
Footnotes
Acknowledgements
We are indebted to Frank J. Rietveld (Department of Pathology, University Hospital, Nijmegen, The Netherlands), who helped us with the staining procedure for monoclonal antibody PAL-E.
