Abstract
The tumor suppressor maspin (
I
When added to mammary tumor cell lines, maspin inhibits their invasiveness and motility in vitro and their growth and invasiveness in vivo (Sheng et al. 1994; Zou et al. 1994; Sager et al. 1996). In addition, immunostaining studies on breast tissues derived from mammary tumor biopsy samples indicate that loss of maspin expression occurs most frequently in advanced cancers. These observations support the hypothesis that maspin functions as a tumor suppressor by blocking tumor cell migration and proliferation and is downregulated during tumor progression.
At present the molecular basis of maspin function is unknown, but its homology with the serpin superfamily suggests that it functions as a proteinase inhibitor. However, exhaustive studies utilizing recombinant protein have thus far failed to identify a proteinase inhibitory function for maspin. Furthermore, not all members of this superfamily are proteinase inhibitors, and recent structural considerations indicate that maspin function may not rely on proteinase inhibition (Hopkins and Whisstock 1994; Potempa et al. 1994; Pemberton et al. 1995). Nevertheless, the functional properties of exogenous recombinant maspin are dependent on the structural integrity of its reactive site loop (RSL) because limited proteolysis within the loop, or antibody binding to the loop, destroys the biological activity of the molecule (Sheng et al. 1994; Zou et al. 1994). Biochemical and modeling studies show that maspin has no disulfide bonds, despite the presence of eight cysteine residues, and that the RSL exists in a distorted α-helical configuration, a finding in keeping with the observed proteolytic sensitivity of this region (Fitzpatrick et al. 1996).
The maspin gene is part of a serpin locus at chromosome 18q21.3 that includes plasminogen activator inhibitor 2 (PAI-2) and the squamous cell carcinoma antigens 1 and 2 (SCCAs 1 and 2) (Schneider et al. 1995). These serpins possess uncleaved signal sequences and are considered members of a branch of the serpin family called “Ov-serpins” because of their relatedness to the well-studied secreted chicken serpin ovalbumin (Remold-O'Donnell 1993). PAI-2 is found both extracellularly in a glycosylated form and intracellularly in a nonglycosylated form, and SCCA1 is secreted by squamous cell carcinomas (Wohlwend et al. 1987; Ye et al. 1988; von Heijne et al. 1991; Kurokawa et al. 1993). Little is known about the underlying molecular mechanism(s) that allow secretion of these serpins, but the hydrophobicity of the amino terminal domains of these proteins does determine their topology. Ovalbumin is reported to have an internal signal sequence that contributes to its secretion, but there are conflicting reports about its localization. Initial studies by Lingappa et al. (1979) localized the sequence to residues 234–253, but subsequent studies by Meek et al. (1982) localized it to within the amino terminal 50–60 residues of the protein. Studies on the related serpin PAI-2 by von Heijne et al. (1991) demonstrated that its secretion was entirely dependent on the hydrophobicity of the amino terminal domain, thus supporting the previous findings for ovalbumin. However, in contrast to the strongly hydrophobic domain present in ovalbumin that results in secretion, PAI-2 has a weakly hydrophobic amino terminal domain that functions as an inefficient secretion signal, allowing it to localize both extra- and intracellularly (Belin et al. 1989). On examining the amino terminal domain in maspin we noted that its hydrophobicity is closer to that of PAI-2 than to that of ovalbumin, indicating that it might also function as an extra- and intracellular serpin, a possibility suggested in the original immunostaining studies (Zou et al. 1994).
The distribution, location, and subcellular localization of maspin in normal cells is of particular importance in view of its biological activities. Here we provide evidence that maspin, or a maspin-like protein, is present in the epithelium of many different organs, particularly in the myoepithelia of the breast, where it localizes and probably functions both intra- and extracellularly.
Materials and Methods
Northern Blotting
Northern blot analysis of purified human tissue RNAs was carried out using a maspin probe encoding the full-length maspin cDNA sequence derived from breast. Multiple-tissue Northern blots (Clontech; Palo Alto, CA) containing ∼2 μg poly(A)+ RNA from adult tissues were hybridized with the maspin cDNA probe and washed. The probe was 32P-labeled and used under standard-stringency hybridization and washing conditions (Sambrook et al. 1989). Blots were also probed with a β-actin cDNA to verify that comparable levels of poly(A) + mRNAs were present in each lane.
Antibody Purification
Biologically active recombinant maspin was purified as described previously (Pemberton et al. 1995). Polyclonal anti-maspin antiserum was raised in rabbits and IgG affinity-purified on maspin-Sepharose, followed by protein G-Sepharose chromatography.
Antibody Specificity
Cell lines from human mammary myoepithelia (HMEC; Clonetics, Palo Alto, CA) and human embryonic small intestinal epithelia (CCL-6; American Type Culture Collection, Rockville, MD) were grown according to the suppliers' instructions. The human mammary adenocarcinoma cell line MDA-MB-435 was a gift from Dr. J. Price (MD Anderson Cancer Center, Houston, TX) and was grown in MEM supplemented with 100 μM nonessential amino acids, 1 mM sodium pyruvate, 5% FBS, and 20 ml/liter MEM vitamins. Cells were grown to 80–85% confluency and lysates prepared in 150 mM NaCl, 50 mM Tris, 1% NP40 containing 10 mM EDTA, 10 μM leupeptin, 10 μM pepstatin, and 1 mM PMSF. Samples were centrifuged (5 min, 10,000 rpm; Eppendorf microfuge) and soluble and insoluble fractions retained for analysis by 12.5% SDS-PAGE and immunoblotting. Maspin was detected using a 1:10,000 dilution of the affinity-purified polyclonal anti-maspin IgG and a 1:5000 dilution of goat anti-rabbit IgG coupled to horseradish peroxidase. Immunoreactive bands were visualized by enhanced chemiluminescence (ECL; Amersham, Chicago, IL). This antibody preparation was also tested for immunoreactivity by immunoblotting with the related serpins PAI-2 (American Diagnostica; Greenwich, CT), SCCA1, ovalbumin, α1-antitrypsin, anti-thrombin III, and α1-anti-chymotrypsin (Sigma; St Louis, MO).
Immunostaining
Slides with sections of formalin-fixed, paraffin-embedded normal human tissues were purchased from Biomeda (Foster City, CA) or were a gift from Dr. N. Weidner (UCSF; San Francisco, CA). These were stained for maspin using the antigen retrieval method (Arber et al. 1993), followed by incubation with 200–500 ng/ml affinity-purified polyclonal anti-maspin IgG. The secondary antibody was biotinylated anti-rabbit IgG. Biotinylated secondary antibodies were detected with streptavidin-peroxidase and the peroxidase substrate 3,3′-diaminobenzidine. Sections were counterstained with hematoxylin. Control slides were stained using affinitypurified rabbit IgG or maspin-specific IgG preincubated with 5 μg/ml recombinant maspin.
Generation of Maspin and PAI-2 cDNA Constructs for Analysis by In vitro Transcription, Translation, and Secretion Assays
Oligonucleotide Synthesis. PCR primers were synthesized on an Applied Biosystems 394 DNA/RNA synthesizer and purified by polyacrylamide gel electrophoresis (PAGE) and chromatography on C-18 Sep-pak cartridges.
Primers for maspin and maspin S36T37S38-LLL were: MAS-1-36 (5′-AGATCTGGTACCACATGGATGCCCTGCAACTAGCA-3′), MAS-3-45 (5′-TTCTCTCCAATCT GTCTCCTGCTCCTTCTGTCACTTGCTCAAGTG-3′), MAS-4-45 (5′-CACTTGAGCAAGTGACAGAAGGAGCAGGA-GACAGATTGGAGAGAA-3′), and MK-36-36 (5′-CTTTCTCTCGAGTTAAGGAGAACAGAATTTGCCAA A-3′). Primers for maspin lacking the amino terminal 45 residues (maspin ΔNH2) PCR were: MAS-2-39, (5′-AGATCTGGTACCACCATGGCTAAAGGTGACACTGCAAAT-3′) and MK-36-36. Primers for PAI-2 were: P-1-35 (5′-GGGAAAACTAGTCCATGGAGGATCTTTGTGTGGCA-3′) and P-2-33 (5′-GGGAAAGTCGACTTAGGGTGAGCAAAATCTGCC-3′).
Maspin and PAI-2 cDNA Constructs. Maspin, maspin ΔNH2, and PAI-2 cDNAs containing the complete coding regions of maspin and PAI-2 were generated by PCR using Vent polymerase (New England Biolabs; Beverly, MA). For maspin cDNA, 1 μM each of MAS-1-36 and MK-36-36 PCR primers and 10 μg/ml of pMas32 DNA template (Zou et al. 1994) were used. For PAI-2 cDNA, 1 μM each of P-1-35 and P-2-33 PCR primers and 10 μg/ml PAI-2-4 DNA template (Webb et al. 1987) were used. The maspin ΔNH2 cDNA was generated as described for maspin cDNA, except that PCR primer MAS-2-39 was used in place of MAS-1-36. Thirty cycles of PCR were performed in a Perkin-Elmer DNA Thermal Cycler, with each cycle consisting of a 1-min denaturation step at 94C, a 2-min annealing step at 55C, and a 3-min extension step at 72C. An additional 7-min extension step was included after the last cycle.
Maspin STS-LLL cDNA was generated in two steps by overlapping PCR, using mismatched primers to generate the mutations (Daugherty et al. 1991). Two separate PCRs were performed as above for maspin using (a) MAS-1-36 and MAS-4-45 PCR primers to amplify the mutant amino terminus and (b) MAS-3-45 and MK-36-36 PCR primers to amplify the remaining cDNA sequence. The full-length cDNA construct incorporating these mutations was generated by fusion of the two pieces using overlapping PCR and primers MAS-1-36 and MK-36-36.
The final products were processed as described previously (Kiefer et al. 1992), digested with Acc65 I and Xho I, gel-purified, ligated into Acc65 I/Xho I-digested pcDNA3 (Invitrogen; Carlsbad, CA) and introduced into E. coli strain HB101. All bacterial and DNA manipulations were performed using standard procedures (Sambrook et al. 1989). To confirm the sequence of the PCR products, DNA sequencing of plasmid DNA was performed by the dideoxy chain termination method using the T7 Sequenase 7-deaza-dGTP sequencing kit (Amersham).
Cell-free Transcription, Translation, and Translocation Assays
Synthetic constructs were transcribed and translated in vitro using T7 RNA polymerase in a coupled transcription, rabbit reticulocyte lysate translation system according to the supplier's instructions (Promega; Madison, WI). Reactions were carried out in triplicate in the presence of canine pancreatic microsomal membranes in a volume of 25 μl to 50 μl and supplemented with [35S]-methionine. To determine if the translated proteins had been secreted into the microsomes, protease protection assays were performed as described previously (von Heijne et al. 1991). Proteinase K (final concentration 1 mg/ml) was added to one set of reaction mixes and proteinase K and detergent (0.1% SDS) were added to another. Samples were incubated on ice for 1.5 hr and proteinase K was inhibited with PMSF. Samples were analyzed by SDS-PAGE and autoradiography.
Human Mammary Myoepithelial Cell-based Protease Protection Assay
Human mammary myoepithelial cells (HMECs) were grown to 80–95% confluence in 6-well plates. Triplicate sets of cells were treated as follows: (a) lysed in situ with RIPA buffer for 30–40 min (25 mM Tris-HCl, pH 7.5, 150 mM NaCl, 1% NP-40, 0.1% SDS); (b) trypsin released with a solution of 0.05% trypsin, 0.53 mM EDTA, pelleted, and residual trypsin neutralized with trypsin neutralizing solution (Clonetics); or (c) lysed in situ as described in a and then treated with 0.05% trypsin, 0.53 mM EDTA for the same amount of time required to release the cells in b. Trypsin was neutralized as above. Cells and lysates were retained for maspin quantitation and analysis by SDS-PAGE and immunoblotting as described above.
Immunostaining of Human Mammary Myoepithelial Cells in Culture
HMECs were cultured and grown as described above on microscope slides. Two staining procedures were used to distinguish between secreted and total maspin. (a) Cells were preincubated with anti-maspin antibody for 1 hr, then washed with PBS, before fixing and staining, or (b) cells were fixed, permeabilized, then incubated with anti-maspin antibody before staining.
Cells were fixed with 1% glutaraldehyde and stained with 50–200 ng/ml affinity-purified polyclonal anti-maspin antibody.
Subcellular Fractionation
Subcellular fractionation of HMECs was performed as described previously for the RS11846 B-cell lymphoma line, with slight modification (Krajewski et al. 1993). Cells were washed twice with PBS, mechanically detached with a cell scraper, and resuspended in 0.5–1.0 ml of MES buffer (17 mM morpholinopropanesulfonic acid, pH 7.4, 2.5 mM EDTA, 250 mM sucrose) containing protease inhibitors (1 mm PMSF, 2 μg/ml pepstatin, and 1 μg/ml leupeptin). The suspension was homogenized with 60 strokes of a Dounce homogenizer and the samples were transferred to Eppendorf tubes and spun at 500 x g for 10 min at 4C to pellet the nuclear fraction. The supernatant was re-centrifuged at 10,000 x g for 15 min at 4C to obtain the heavy membrane fraction (mitochondria, lysosomes, peroxisomes) and then at 150,000 x g at 4C for 60 min to obtain the light membrane fraction (plasma membrane, microsomes, endoplasmic reticulum) and cytosolic fractions (supernatant). Each fraction was subjected to two cycles of centrifugation to remove cross contamination, then reconstituted to the same volume in SDS-PAGE sample buffer, and equivalent volumes of each were analyzed for maspin content by SDS-PAGE and immunoblotting.
Results
Maspin mRNA Is Expressed in Multiple Organs
Figure 1A shows the expression, distribution and size of maspin mRNAs in a number of human organs. Maspin mRNA is expressed in the placenta, testis, prostate, colon, small intestine, and thymus. In these organs the mRNA is approximately 3.0
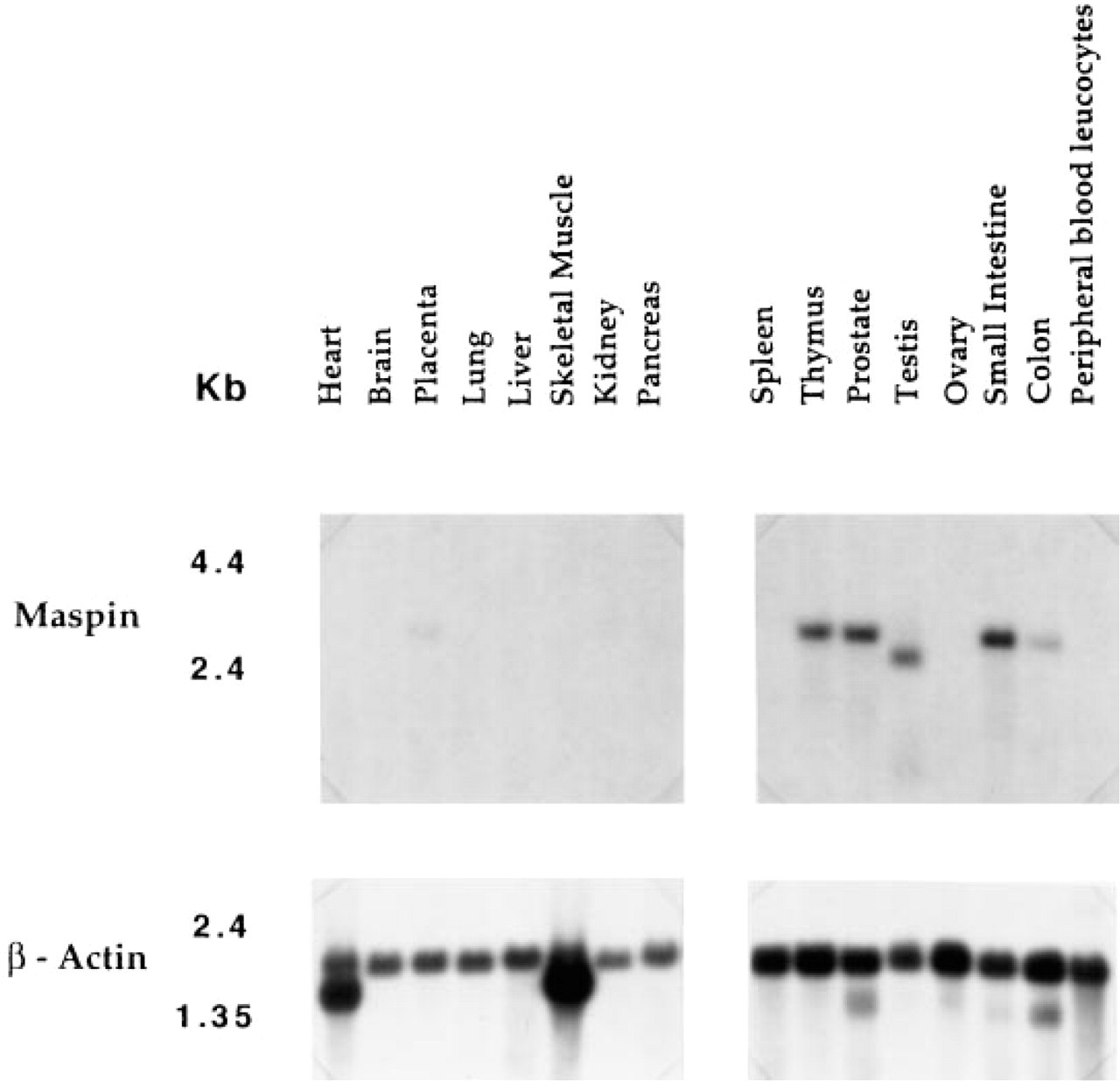
Tissue distribution of maspin mRNAs by Northern blotting.
Specificity of Anti-maspin Antibodies on Immunoblotting
The purified polyclonal IgG anti-maspin antibody preparation reacted with purified recombinant maspin and maspin produced by normal human mammary epithelial cells (Figure 2). Maspin was not detected in the mammary epithelial tumor cell line MDA-MB-435 but was present in the embryonic small intestinal cell line CCL-6. In each case, only one band of 42 kD was detected, consistent with the size of recombinant maspin and the previously reported size of HMEC maspin. Under these conditions, this antibody preparation did not react with any of the related serpins that were tested, further demonstrating the monospecificity of this preparation.
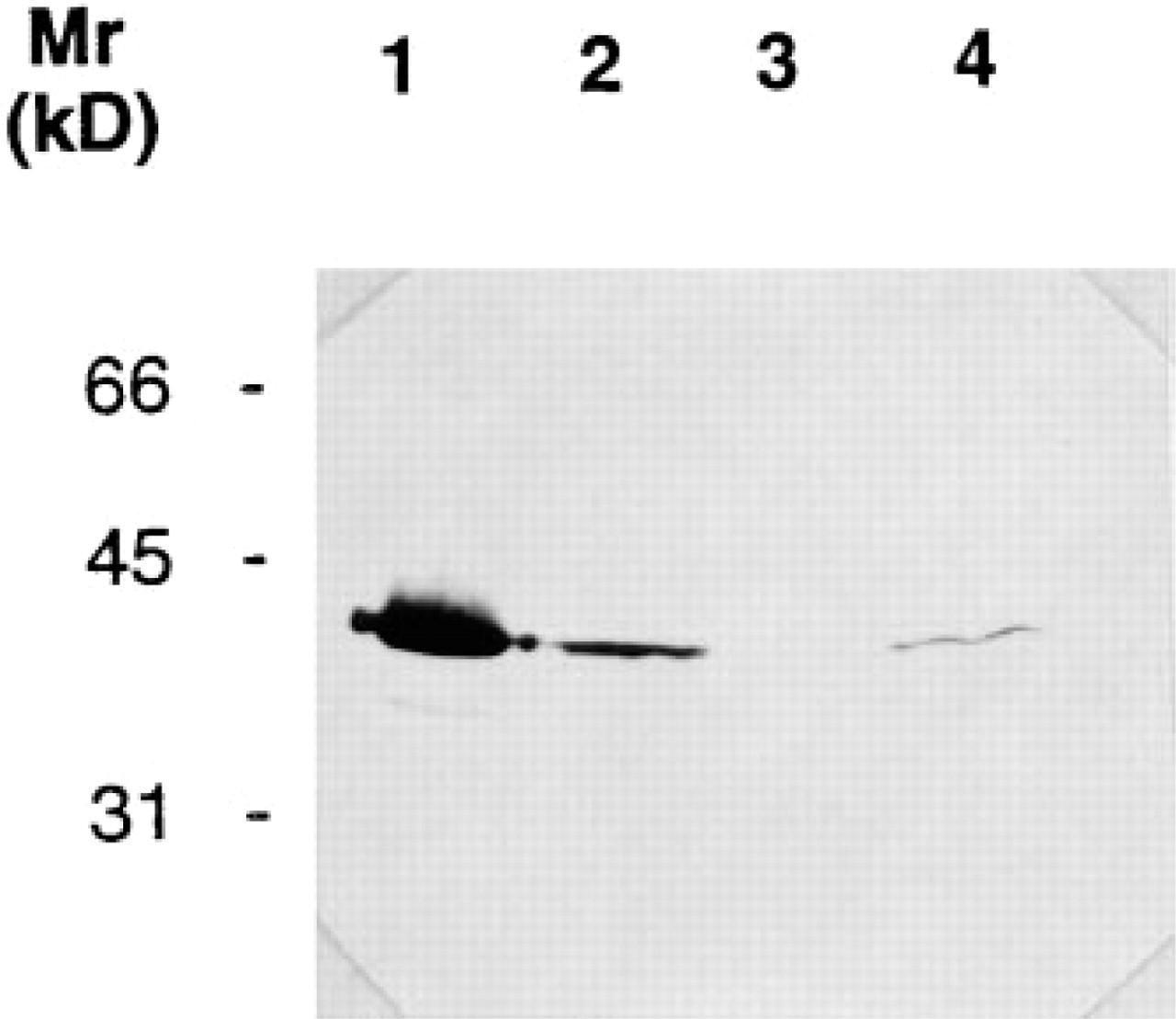
Immunoreactivity of affinity-purified anti-maspin IgG with mammary and intestinal cell line lysates by immunoblotting. Lane 1, purified recombinant maspin; Lane 2, soluble human mammary epithelial cell lysate; Lane 3, soluble MDA-MB-435 tumor cell lysate; Lane 4, soluble CCL-6 embryonic small intestinal cell lysate.
Tissue Immunolocalization of Maspin
Figure 3 shows representative results of immunostaining studies on multiple unrelated tissue samples performed at different times over a 2–3-month period using affinity-purified polyclonal anti-maspin IgG. For each example, Panel 1 represents the staining observed with an affinity-purified nonspecific control rabbit IgG at low magnification. Panels 2 and 3 represent staining with maspin-specific IgG at both low and high magnifications. As described previously, maspin was routinely detected in breast (Figure 3A), where it was found almost exclusively in the myoepithelial cells lining the duct (Zou et al. 1994). Breast luminal cells stained much less intensely for maspin. The structure of prostate ducts is similar to that of the breast, but they do not possess a defined myoepithelial layer. Nevertheless, the equivalent basal cell layer stained as intensely for maspin as the breast myoepithelium (Figure 3B). Like the breast, prostate luminal epithelial cells stained much less intensely for maspin.
The simple columnar epithelia of the gastrointestinal tract also stained for maspin, ranging from intense staining in the mucosal epithelium of the small intestine (Figure 3D) to much weaker staining in the mucosal epithelium and intestinal glands of the colon (Figure 3E) and mucosal epithelium of the stomach (Figure 3C). In the stomach, we also observed stronger maspin staining in epithelial cells of the basal crypts, whereas motile epithelial cells further up the microvilli did not stain as strongly.
In the placenta, maspin localized to cytotrophoblasts associated with the chorionic villi (Figure 3H). In the testis, maspin was present in the complex germinal epithelium of the seminiferous tubule and myoid cells (Figure 3G). Maspin was also present in the stratum functionale of the uterus (Figure 3F), stratified squamous epithelium of the tonsil (in this example it is heavily infiltrated with lymphocytes) (Figure 3I), intralobular duct epithelia of the pancreas (Figure 3J), kidney tubule epithelia (Figure 3K), and skin (Figure 3L), where it was abundant in the stratum granulosum, spinosum, and basale (but not in clear cells).
Little or no staining of any tissue section was observed with the affinity-purified control rabbit polyclonal IgG preparation, and all staining was abolished by preincubation of the antibody with 5 μg/ml recombinant maspin. We did not observe any staining for maspin in the brain, pituitary, adrenal, heart, liver, spleen, or thyroid.
Evidence That Maspin Is an Intracellular and Extracellular Serpin
Several lines of evidence indicate that maspin is predominantly an intracellular serpin that is also present extracellularly.
In vitro transcription, translation, and secretion studies demonstrate that small amounts of maspin do get translocated into the Golgi, but the bulk of it remains cytoplasmic. Figure 4 shows the results of this study. As previously reported by Von Heijne et al. (1991) we found (a) that PAI-2 translocation into microsomes is relatively inefficient, resulting in less than 5% of PAI-2 being protected from proteinase K digestion (PAI-2; Figure 4, Lane 2, arrow) and (b) that degradation of untranslocated PAI-2 by proteinase K in the absence of detergent results in the generation of a proteolytically resistant “core” of PAI-2 (Figure 4, Lane 2, +) that cannot be further degraded in the presence of detergent (Figure 4, Lane 3). Like PAI-2, maspin was also poorly translocated into microsomes, but the amount translocated was marginally more (maspin; Figure 4, Lane 2). Furthermore, and as also reported for PAI-2, translocated maspin transcripts appear to be of several slightly different sizes, possibly reflecting post-translational modifications such as glycosylation. However, unlike PAI-2, maspin was completely degraded by proteinase K in the presence of detergent (maspin; Figure 4, Lane 3). As predicted, maspin lacking the amino terminal 45 residues was not translocated at all (maspin ΔNH2; Figure 4, Lane 2), whereas maspin possessing an amino terminus of increased hydrophobicity was very efficiently translocated, as evidenced by its increased resistance to proteolysis by proteinase K (maspin STS-LLL; Figure 4, Lane 2). However, we noted a slight discrepancy in molecular weight between maspin STS-LLL translated in the presence of microsomes (Figure 4, Lane 1) and the product treated with protease in the absence of detergent (Figure 4, Lane 2). Nevertheless, in the presence of detergent this construct was totally degraded (Figure 4, Lane 3).
To further address the issue of maspin secretion, we examined the proteolytic sensitivity and immunoreactivity of maspin produced by a normal HMEC line. Figures 5 and Figures 6 show the results of these studies. Under the culture conditions employed, this cell line synthesized approximately 0.5–1.0 nM of maspin/106 cells (Figure 5A), of which none was detectable in cell culture medium by immunoblotting. Releasing the cells with trypsin resulted in partial degradation of some maspin, but the bulk of it was resistant to proteolysis (Figure 5B). Maspin present in cells lysed with detergent was completely degraded by the addition of proteinase (Figure 5C).
Pretreatment of the HMEC line with the polyclonal anti-maspin IgG antibody preparation before fixing and staining showed that maspin is found at the cell surface, where it localizes in a reticular pattern (Figure 6B). In permeabilized fixed cells, maspin staining is much stronger and is also present throughout the cytoplasm (Figure 6C). This cell line did not stain with an affinity-purified nonspecific rabbit IgG preparation (Figure 6A). These results were observed for all cells grown on the microscopic slide and were repeated on several different occasions to verify consistency.
Fractionation of human mammary epithelial cells demonstrated that maspin is present predominantly (<95%) as a soluble protein in the cytoplasm of these cells and that small amounts may also be found associated with heavy, light, and nuclear membrane fractions (Figure 7).
Discussion
The results from Northern blot, tissue immunostaining, and immunoblot analyses clearly show that maspin is expressed in a variety of organs. Maspin mRNA is seen at low levels in the colon and placenta and at higher levels in the prostate, small intestine, testis, and thymus. Tissue immunostaining studies show that maspin, or a maspin-like protein, is associated with myoepithelial cells of the breast, basal cells of the prostate, mucosal and glandular epithelia of the gastrointestinal tract, endometrium of the uterus, germinal epithelium and myoblasts of the testis, cytotrophoblasts of the placenta, squamous epithelium of the tonsil, glandular epithelia of the pancreas, tubule epithelia of the kidney, and stratified epithelial layers of the skin. It has been shown previously that breast myoepithelial cells in culture synthesize maspin (Zou et al. 1994), and we have demonstrated here that small intestinal cells in culture synthesize a protein with an identical Mr and immunoreactivity to maspin. In some organs we detected both maspin mRNA by Northern blotting and maspin, or a maspin-like protein, by immunostaining, whereas in others we could detect only a maspin-like protein. This is probably due to several factors. First, the commercial source of tissue used to prepare mRNA cannot be controlled for age or site of sampling. Second, the relative sensitivities of the assays probably differ. For example, Northern blot analysis did not detect maspin mRNA in kidney, whereas immunohistochemical procedures did. We therefore propose to use more sensitive DNA/RNA-based detection systems, such as RT-PCR and in situ hybridization, to identify and quantitate the relative amounts of maspin produced by each of these specific epithelial cell types in both normal and cancerous states, because the presence of maspin in other tissues suggests that its downregulation may be involved in carcinomas other than breast.
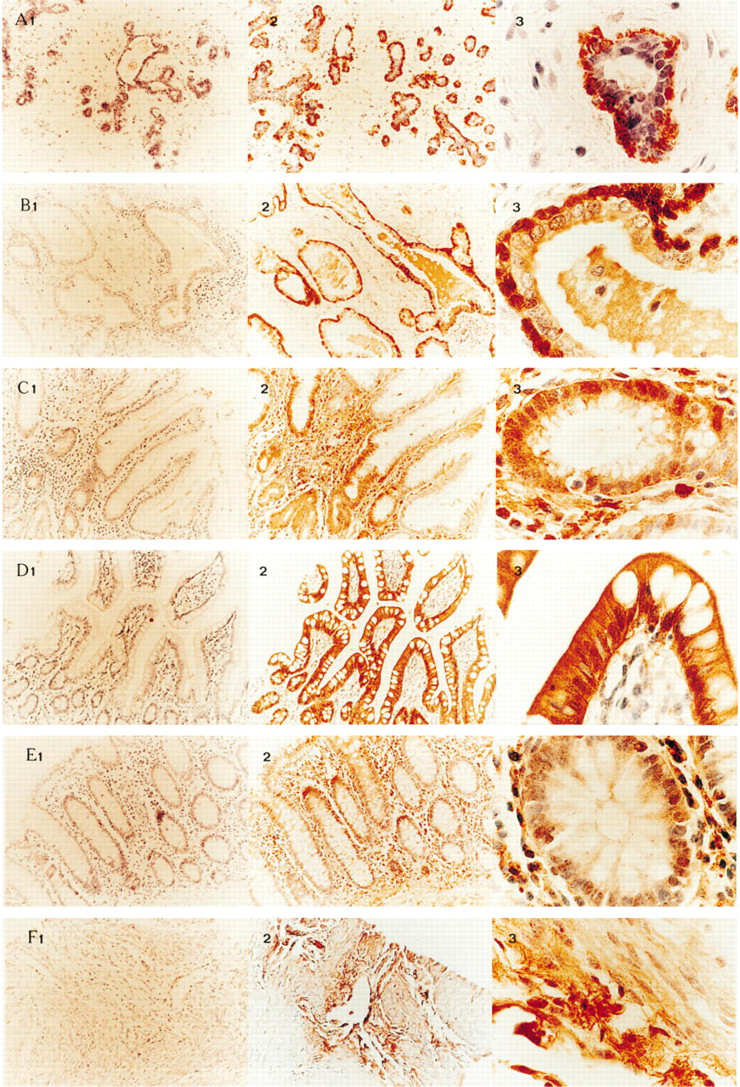
Normal tissue immunostaining for maspin.
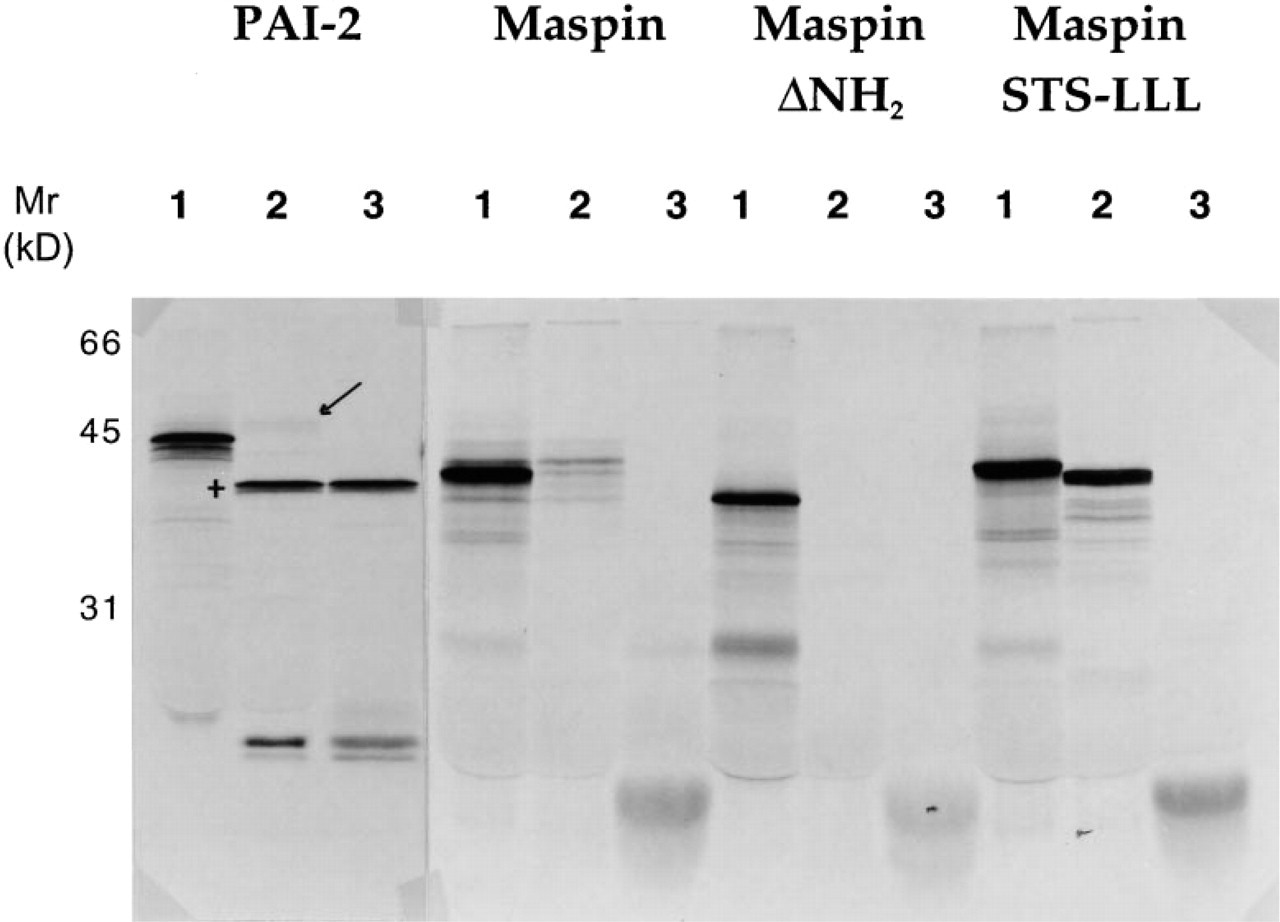
Maspin-translated In vitro partitions between the cytosol and microsomal membranes. Maspin and PAI-2 translation products were analyzed by autoradiography as decribed in Materials and Methods. For each construct, three assays were performed.
These histological results also suggest that levels of maspin, or a maspin-like protein, can differ significantly within the same tissue type. The best example of this was seen in the stomach, in which epithelial cells at the base of the crypts stained with anti-maspin antibodies whereas migratory cells further up the villi did not stain as strongly. Other examples of this are the breast and prostate, in which such staining clearly distinguished myoepithelial and basal cell populations from adjacent luminal epithelial cell populations. These observations suggest that maspin downregulation may be a normal event in cell differentiation, allowing epithelial cells to proliferate and migrate in response to an appropriate stimulus. In the case of breast epithelium, prolactin or estrogen could fulfill this role. Such a process in itself could contribute to the maintenance of normal organ architecture but, when coupled with aberrant proteinase expression, extracellular matrix degradation, and tumor cell locomotion, could lead to invasive metastatic cancer.
Several independent pieces of evidence show that maspin is an intracellular protein that also localizes extracellularly in breast myoepithelia.
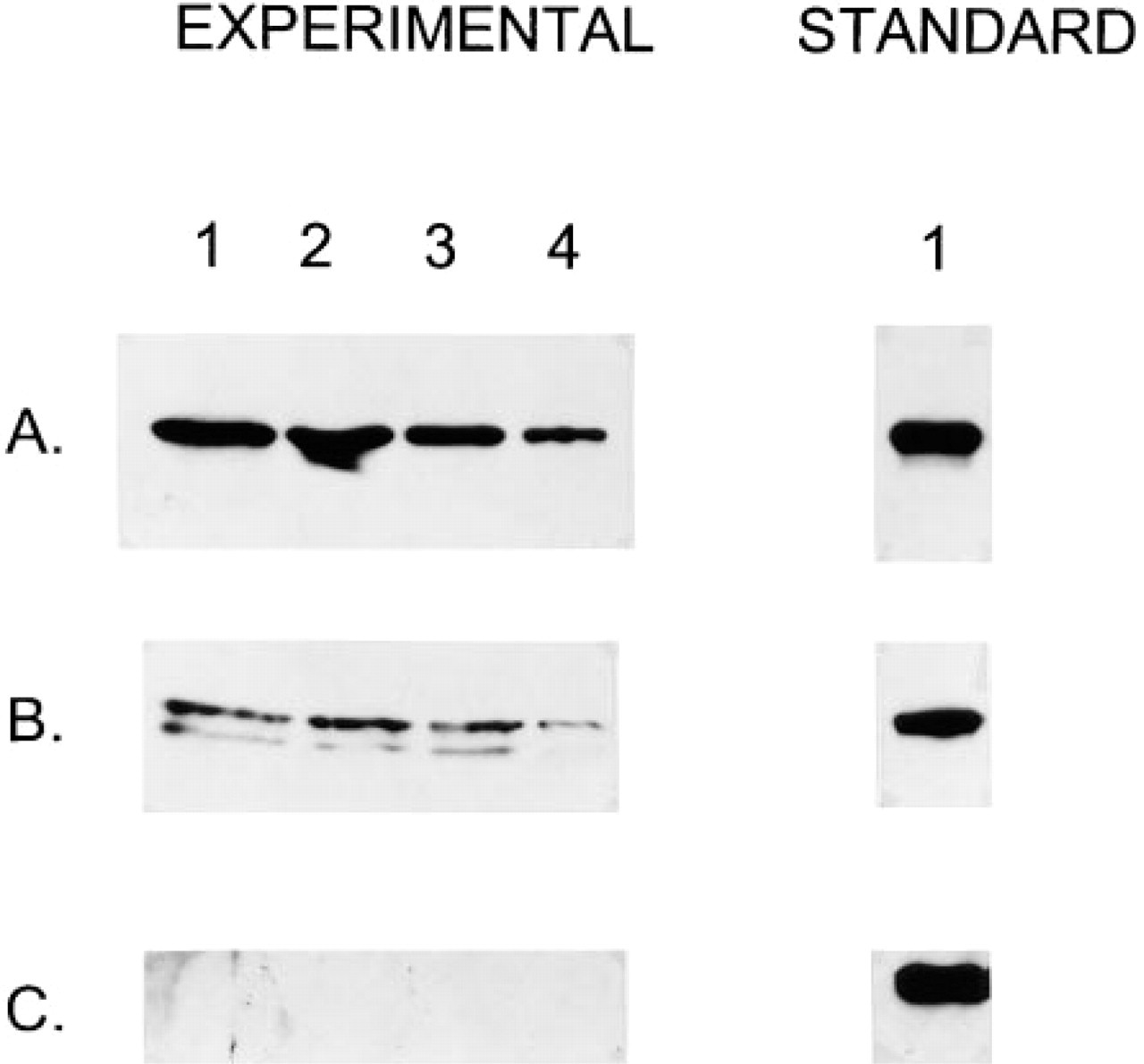
Quantitation and proteolytic sensitivity of maspin synthesized by cultured human mammary myoepithelial cells. Maspin was detected and quantitated by immunoblotting.
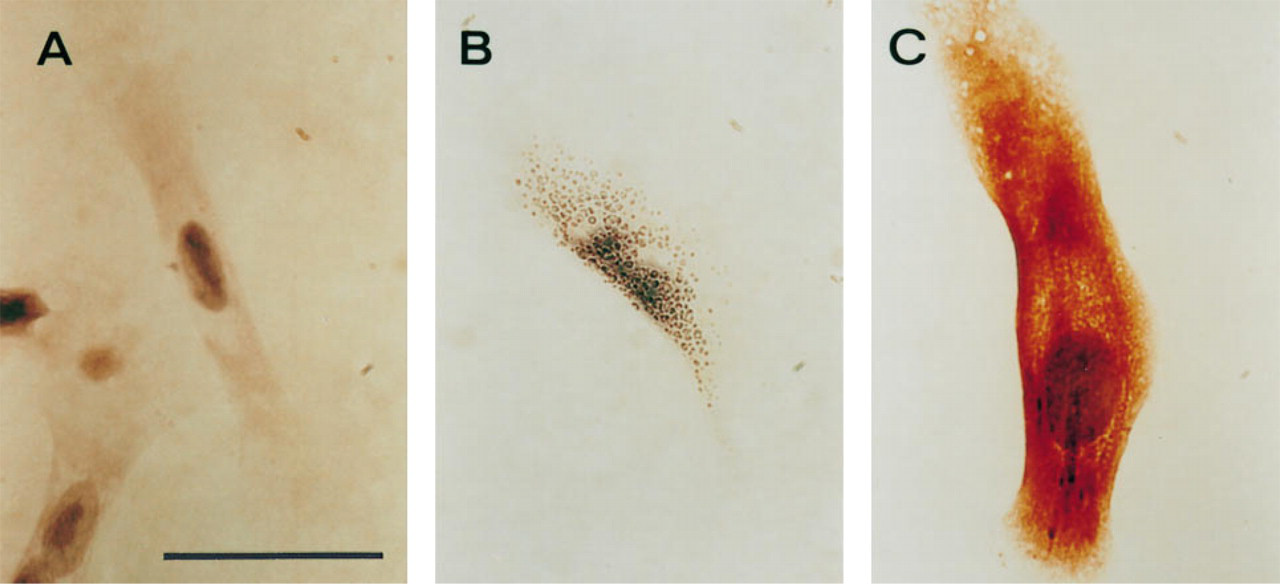
Immunostaining of human mammary myoepithelial cells in culture.
In vitro transcription, translation, and secretion assays clearly show that small amounts of maspin are translocated into the endoplasmic reticulum and that this process is dependent on the hydrophobicity of an uncleaved signal encoded within the amino terminus of the protein. Nevertheless, the bulk of maspin remains soluble within the cytoplasm. In these studies we noted a slight difference in molecular weight between maspin STS-LLL translated in the presence of microsomes and this product treated with protease, suggesting that this construct is still susceptible to very limited proteolysis. We have previously noted that the RSL of maspin is extremely sensitive to limited proteolysis, and we propose that the action of endogenous microsomal proteases may account for the difference observed here.
Further evidence supporting the dual localization of maspin was provided by protease protection assays, immunostaining, and fractionation studies performed on cultured mammary myoepithelial cells. These results demonstrate that mammary myoepithelial cells in culture possess a small amount of extracellular maspin that is accessible to limited proteolysis and antibody ligation, but that the majority of maspin is sequestered intracellularly in a soluble form. Although the role of extracellular and/or vesicle-associated maspin in tumor supression is not well defined, the results presented here suggest that the RSL of maspin at the cell surface is accessible to proteolysis. Previous results have also shown that the RSL is accessible to binding by RSL-specific antibodies. Furthermore, the addition of nanomolar amounts of exogenous recombinant maspin to several metastatic tumor cell lines blocks their invasiveness and motility and this activity is reliant on the integrity of the RSL (Sheng et al. 1994; Zou et al. 1994). These findings suggest that maspin at the surface of human mammary myoepithelial cells in a breast duct could conceivably function in a paracrine fashion to control the invasiveness and motility of adjacent luminal cells.
This is the first direct demonstration that maspin is a soluble intracellular protein, and this finding suggests that maspin regulation of cell motility, proliferation, and invasiveness may be, in part, mediated through an intracellular function. We have recently noted that the RSL of maspin is homologous to a sequence in MAP1B (Pemberton 1997), a microtubule-associated protein highly expressed and localized at the leading edge of lamellipodia in outgrowing motile neurites. We therefore propose that the physiological tumor-suppressing functions of maspin may also be mediated, in part, through direct interaction with intracellular motor protein(s).
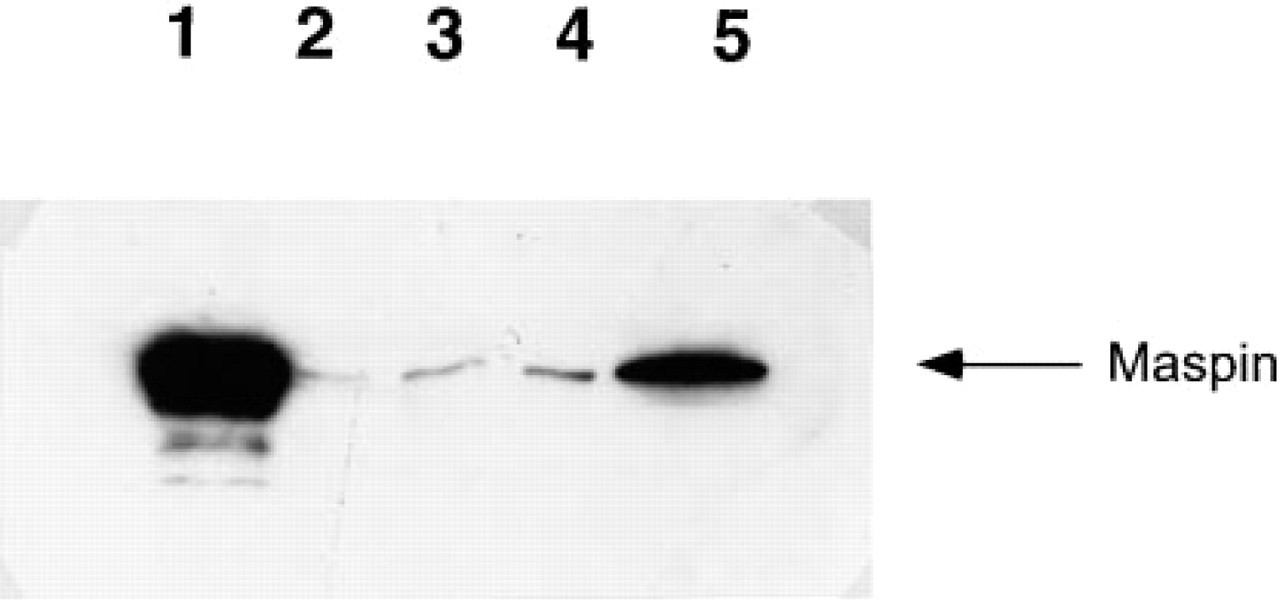
Detection of maspin in human mammary myoepithelial subcellular fractions by immunoblotting. Lane 1, 200 ng recombinant maspin; Lane 2, nuclear fraction; Lane 3, heavy membrane fraction; Lane 4, light membrane fraction; Lane 5, cytosol.
In conclusion, we have shown that maspin, or maspin-like immunoreactivity, is expressed in a number of organs in which it is associated almost exclusively with epithelia. This suggests that, in addition to the breast, maspin may regulate the development of a number of other carcinomas. In vitro assays indicate that maspin is predominantly a soluble intracellular serpin that translocates into secretory vesicles and is also present at the cell surface. These findings suggest that the biological function(s) of soluble intracellular maspin is probably different from those of vesicle-associated or extracellular maspin, and that the elucidation of their relative contributions and mechanisms of action is of paramount importance in understanding the role of maspin in suppressing tumor growth and metastasis.
Footnotes
Acknowledgements
We thank Ian Bathurst and Vaclav Prochazka for their invaluable technical assistance and advice.
