Abstract
In this immunocytochemical study, we used light and electron microscopic observations in combination with morphometry to analyze the processing of pro-opiomelanocortin (POMC) in melanotrope cells of the intermediate pituitary of Xenopus laevis adapted to either a white or a black background. An antiserum was raised against a synthetic peptide including the cleavage site between ACTH and β-lipotropic hormone in Xenopus. Western blotting revealed that this antiserum recognizes only a 38-kD protein, the POMC prohormone, from extracts of Xenopus neurointermediate pituitary. Light immunocytochemistry showed differential immunostaining for anti-POMC compared to anti-α-MSH. Anti-POMC was predominantly found in the perinuclear region, whereas anti-α-MSH yielded staining throughout the cytoplasm. Immunogold double labeling revealed that electron-dense secretory granules (DGs) show high immunoreactivity for anti-POMC and low immunoreactivity for anti-α-MSH. Electron-lucent granules (LGs) are immunoreactive to anti-α-MSH only. Moderately electron-dense granules (MGs) revealed intermediate reactivity compared to DGs and LGs. Background light intensity has significant effects on the morphology and the immunoreactivity of the secretory granules. Black-adapted animals have 4.5 times as many DGs and MGs as white-adapted animals. In addition, the MGs in black animals show 42% more anti-α-MSH immunogold than the MGs in white animals. Together, these findings indicate that the three granule types represent subsequent stages in granule maturation. Adaptation to a black background stimulates the formation of young immature granules, while at the same time the processing rate during granule maturation increases.
T
The present study aimed to confirm this hypothesis by investigating whether different granule types represent different stages in granule maturation, whether processing of POMC in the secretory granules is a gradual process and occurs in parallel with the transition of DGs via MGs to LGs, and whether POMC processing is under the influence of background light intensity. For this purpose, a POMC antiserum was raised in this study against a synthetic peptide (ST-62) corresponding to the cleavage site between adrenocorticotropic hormone (ACTH) and β-lipotropic hormone (β-LPH) of Xenopus POMC. In rat melanotropes, the homologous sequence contains the first proteolytic cleavage site of murine POMC (Tooze et al. 1987; Tanaka et al. 1991; Tanaka and Kurosumi 1992). The antiserum was characterized using light microscopy and immunoblotting. Subsequently, the processing of POMC to α-MSH was investigated with quantitative immunoelectron microscopy, using the POMC antiserum and an antiserum to α-MSH.
Materials and Methods
Production and Characterization of POMC Antiserum
Peptide synthesis was carried out according to a protocol described previously (see King et al. 1990; Tanaka et al. 1992). In short, a peptide (ST-62) including the cleavage site between ACTH and β-LPH of Xenopus POMC (Glu-Leu-Arg-Arg-Glu-Leu-Ser-Leu-Glu-Cys; for amino acid sequence of Xenopus POMC, see Martens et al. 1985) was synthesized using an automated peptide synthesizer (Model 431A; Applied Biosystems, Foster City, CA). After purification of the peptide by reverse-phase HPLC, an antibody was raised in a guinea pig. The specificity of guinea pig anti-POMC was examined by preabsorption tests with the corresponding ST-62 peptide or with synthetic α-MSH (N-acetyl-α-MSH-NH2; Sigma, St Louis, MO) at concentrations of 1–100 mg/ml for 16 hr at 4C, before their immunocytochemical use. The specificity of the rabbit anti-α-MSH serum used in this study has been shown previously by noncompetitive binding and preabsorption tests, which showed that the antiserum specifically recognizes α-MSH (Tanaka and Kurosumi 1986).
Western Blotting
The specificity of the POMC antiserum was also tested in a Western blot. Neurointermediate lobes of black-adapted animals were homogenized in 100 μl sample buffer consisting of 62.5 mM Tris-HCl (pH 6.8), 12.5% glycerol, 1.25% SDS, 0.0125% bromophenol blue, and 2.5% β-mercaptoethanol. Samples and markers (
Animals
Adult Xenopus laevis were obtained from laboratory stock and fed trout pellets (Trouvit; Trouw, Putten, The Netherlands) once a week. The animals were kept under constant illumination and water temperature was 22C. Before the experiments, animals had been adapted to either a white or a black background for 3 weeks. The melanophore dispersion state was consistently 1.0 ± 0.1 for white adaptation and 4.9 ± 0.1 for black adaptation, according to the index of Hogben and Slome (1931).
Light Immunocytochemistry
After decapitation and dissection, brains with pituitary glands were fixed for 2 days in Bouin-Hollande, dehydrated, and embedded in paraffin. Serial sagittal sections (5 μm) were mounted on gelatin-coated glass slides. Sections were deparaffinized and rinsed in distilled water and PBS (0.01 M sodium phosphate buffer, 0.14 M NaCl, 0.1% sodium azide, pH 7.5). Then the sections were treated with 1% bovine serum albumin (BSA) in PBS for 2 hr, guinea pig anti-POMC (1:10,000) or rabbit anti-α-MSH (1:5,000) for 16 hr, donkey anti-guinea pig or goat anti-rabbit antiserum (1:100; Nordic) for 2 hr, and finally with guinea pig and rabbit PAP complex (1:100; Nordic) for 90 min. After washing in PBS, sections were treated with 10 mg 3,3′-diaminobenzidine tetrahydrochloride (DAB; Sigma), 0.25% nickel ammonium sulfate and 0.005% H2O2 in 100 ml 0.05 M Tris-HCl buffer (pH 7.6) to reveal peroxidase activity. Then sections were dehydrated and mounted.
Freeze-substitution and Immunoelectron Microscopy
Animals were decapitated and freshly dissected neurointermediate lobes were fixed in 1% glutaraldehyde in 0.05 M sodium phosphate buffer (pH 7.4) for 16 hr. Then the lobes were treated with 0.2% borohydride and 0.4% glycine in the buffer for 15 min, and cryoprotected by immersion in a graded series of glycerol (10%-20%-30% in the buffer, 30 min per concentration). Lobes were rapidly frozen in liquid propane (−180C) and transferred to a precooled chamber (−90C) in a quick-freezing apparatus (Reichert-Jung; Vienna, Austria). Then they were freeze-substituted by methanol containing 0.5% uranyl acetate and warmed at a rate of 4C per hour to −45C. Embedding in Lowicryl HM20 resin (BioRad) was carried out in three steps with an increasing ratio of resin to methanol. Polymerization of Lowicryl was performed by
Control Experiments
To test the specificity of the immunoreaction, incubation of sections in preabsorbed POMC and α-MSH antisera and immunostaining without use of a primary antiserum was executed. To examine crossreactivity of the immunogold products, the mixture of immunogold was used in single labeling experiments with both anti-POMC and anti-α-MSH. With anti-POMC, only goat anti-guinea pig immunogold (10-nm gold particles) was present, and with anti-α-MSH only goat anti-rabbit immunogold (15-nm gold particles) was found, indicating that crossreactivity between the species is absent.
Quantitative Evaluation and Statistics
For quantitative evaluation of the double labeling with anti-POMC and anti-α-MSH, three white- and four black-adapted animals were studied. Per animal, secretory granules were counted in at least 15 melanotrope cells and expressed as numbers of DGs, MGs and LGs per 100 μm2 cytoplasmic area. For each secretory granule, the numbers of 10-nm (POMC immunoreactivity) and 15-nm (α-MSH-immunoreactivity) gold particles were counted on photomicrographs (final magnification x 40,000), and expressed as numbers of gold particles per granule (Gp/G). A random selection procedure (animals, cells, granules) was maintained throughout the experiment. Data were analyzed with one-way analysis of variance (Bliss 1967) (α = 5%) followed by Duncan's multiple range test (Steel and Torrie 1960). The analysis was preceded by tests for joint assessment of normality (Shapiro and Wilk 1965) and homogeneity of variance (Bartlett's test; see Bliss 1967).
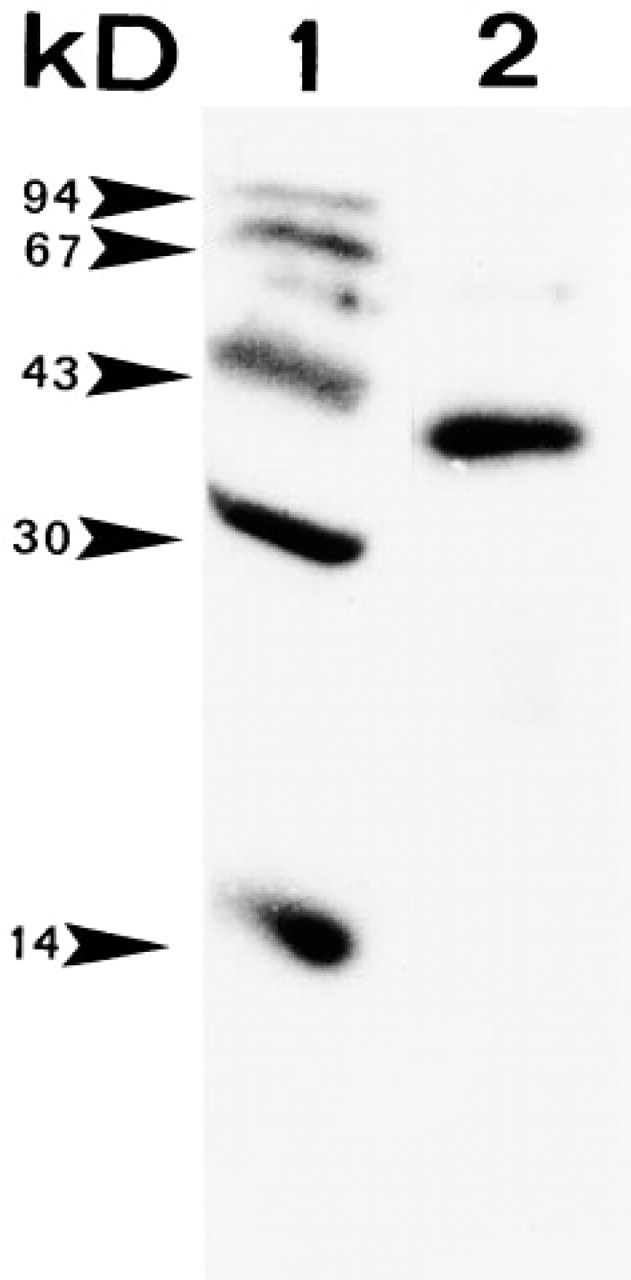
Western blot analysis obtained with the anti-ST-62 anti-serum (Lane 2). Anti-ST-62 recognizes one band (38 kD) corresponding to glycosylated POMC. Lane 1 shows molecular weight markers.
Results
Anti-ST-62 Recognizes POMC
To test the specificity of the ST-62 antiserum for Xenopus POMC, immunoblotting was carried out with neurointermediate lobes of black-adapted Xenopus. The antiserum appeared to recognize a protein with a molecular weight of 38 kD, corresponding to the POMC precursor. No other bands were detected with the ST-62 antiserum, which will be referred to below as anti-POMC (Figure 1).
Differential Immunostaining with Anti-POMC and Anti-α-MSH at the Light Microscopic Level
The intermediate lobe of the pituitary of Xenopus consists of over 95% melanotrope cells, which can readily be identified on the basis of their characteristic round shape. Immunocytochemistry with anti-POMC showed intense staining of the cytoplasm, particularly in the perinuclear region of the melanotropes (Figures 2A and 2B, arrows). With anti-α-MSH, strong staining throughout the cytoplasm of all melanotrope cells was observed in both black- and white-adapted animals (Figure 2C). Preabsorption of the antiserum with ST-62 completely abolished staining, whereas preabsorption with anti-α-MSH did not affect staining (data not shown).
Immunogold Labeling of the Secretory Granules with Anti-POMC and Anti-α-MSH
Freeze-substitution fixation yielded adequate ultra-structural preservation of the neurointermediate lobe, revealing melanotrope cells, folliculostellate cells, and axons with varicosities and synaptic terminals. Melanotropes of black-adapted animals showed a highly developed rough endoplasmic reticulum (RER) and an extensive Golgi apparatus (Figures 3B and 3C), in contrast to white-adapted animals, in which these cell organelles were almost absent (Figure 3A). With respect to secretory granules, DGs, MGs, and LGs occurred in both black- and white-adapted animals. In black-adapted animals, significantly more DGs and MGs per cytoplasmic area were present than in white-adapted animals (10.7 ± 1.39 DGs and 14.4 ± 2.79 MGs in black-adapted vs 2.4 ± 0.77 DGs and 3.8 ± 0.61 MGs in white-adapted animals; p>0.05) (Figure 5). In white-adapted animals, the granule population consisted almost exclusively (i.e., more than 90%) of LGs. Significantly more LGs were found in white-adapted animals than in black-adapted animals (340.0 ± 29.26 LGs in white-adapted vs 108.6 ± 27.60 LGs in black-adapted animals; p>0.05) (Figure 5).
Immunoelectron microscopy of freeze-substituted neurointermediate lobes with anti-POMC and anti-α-MSH showed specific immunostaining in both black-adapted and white-adapted animals. Strong double labeling was observed for secretory granules, whereas background labeling over cytoplasm, mitochondria, cell nuclei, and extracellular spaces with either of the antisera was barely present (Figures 3 and Figures 4).
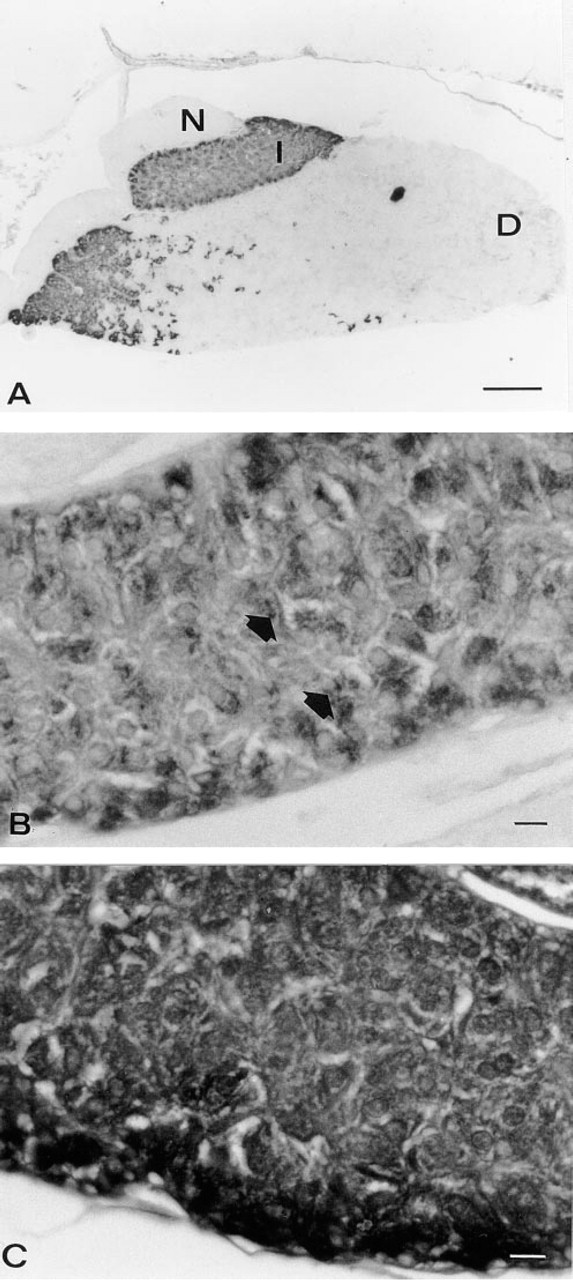
Light micrographs of the pituitary of a black-adapted Xenopus immunostained using the PAP method for POMC (
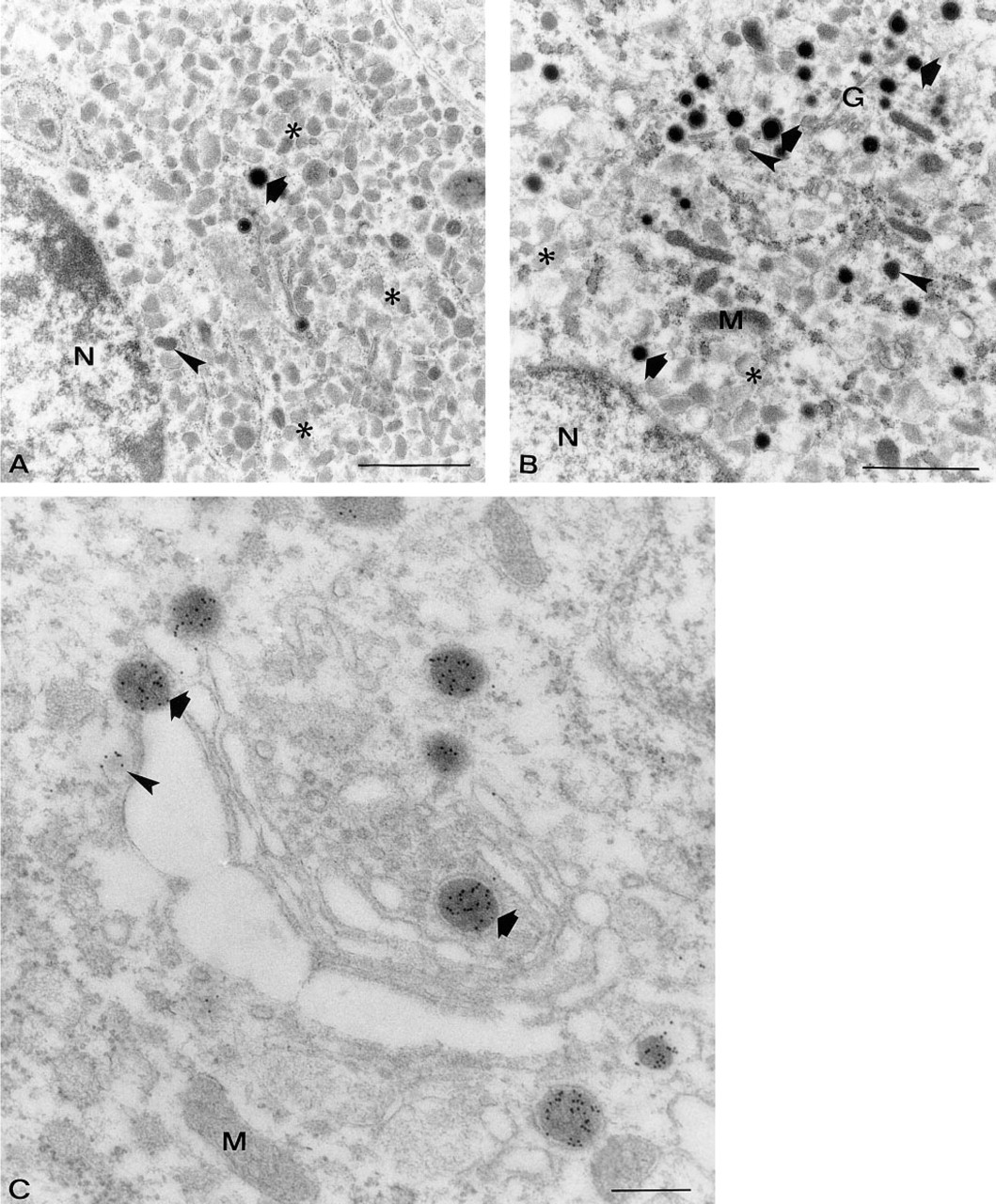
Electron micrographs of pars intermedia fixed by freeze-substitution, showing melanotrope cells of a white-adapted (
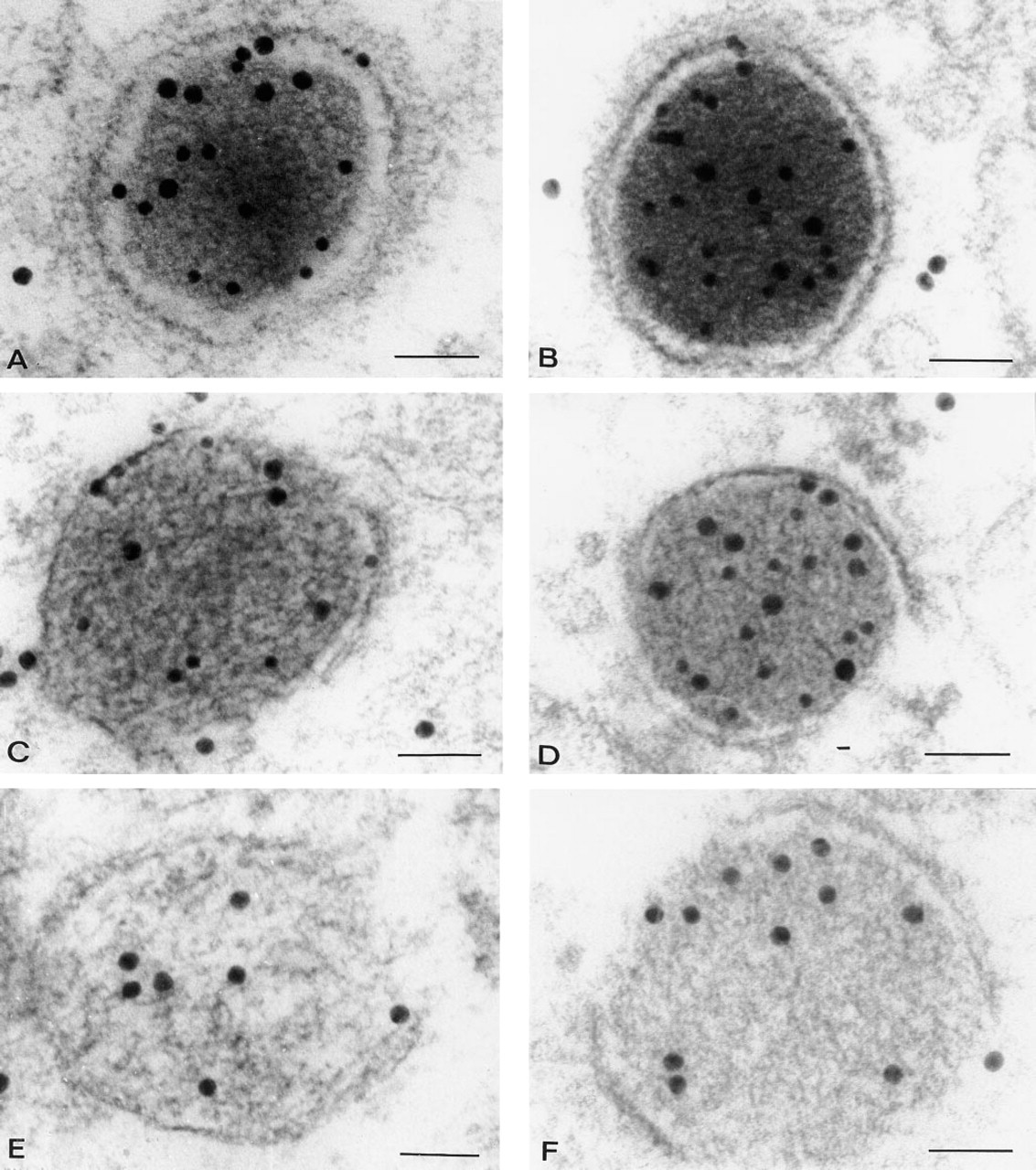
Double labeled DGs, MGs, and LGs in a melanotrope cell of a white-adapted (
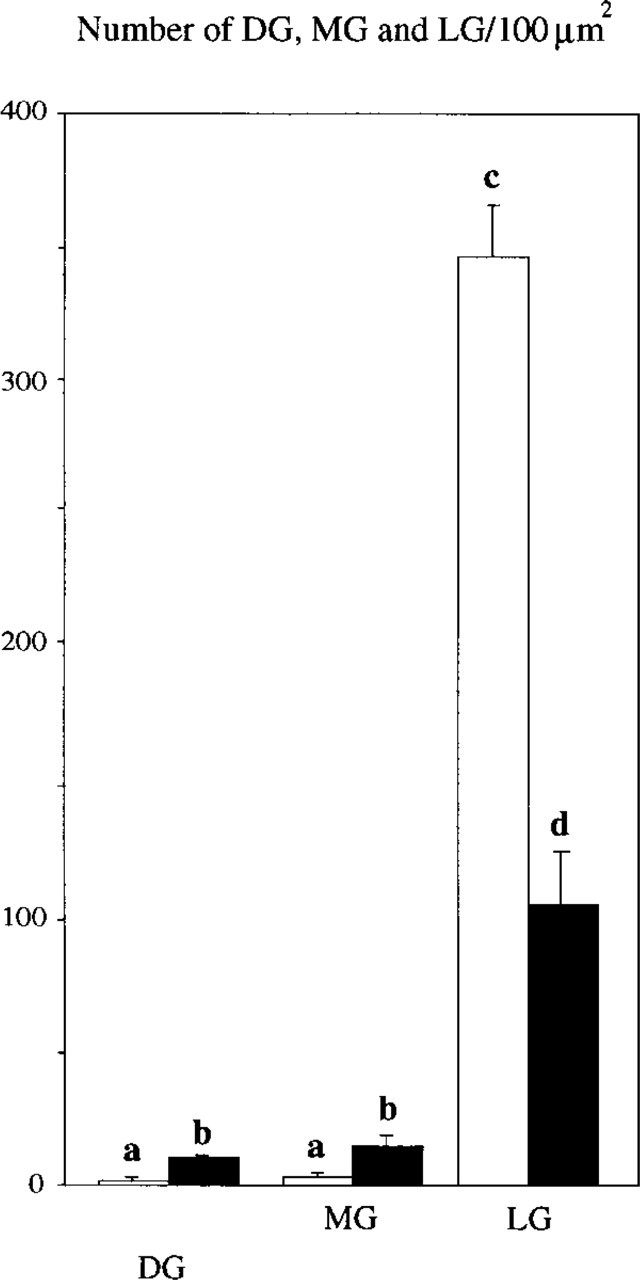
Mean numbers of DGs, MGs, and LGs in melanotrope cells of white-adapted (open bars) and black-adapted (closed bars) animals per 100 μm2 cytoplasmic area and SEM. Different superscripts on top of columns indicate that groups differ significantly (p>0.05).
For anti-POMC, the RER was negative but some immunogold labeling occurred in dilated cisternae of the Golgi apparatus (Figure 3C, arrowheads). With respect to granule types, a differential staining pattern with anti-POMC was found (Figures 3C and 4A–4F). In both black-adapted and white-adapted animals, virtually all DGs (i.e., more than 98%) showed high labeling intensities, 7.1 ± 0.60 Gp/G in black-adapted and 6.6 ± 1.70 Gp/G in white-adapted animals (Figures 4A, 4B, and 6A). DGs that were located within or close to the Golgi apparatus were strongly immunopositive (Figure 4B). Between 10 and 20% of these DGs were seen budding off from the trans-Golgi network (Figure 3C). Around 80% of the MG showed immunolabeling with anti-POMC, but the labeling intensity was about one third lower than in the DGs (4.1 ± 0.66 Gp/G in black, 3.9 ± 0.20 Gp/G in white animals) (Figures 4C, 4D, and 6A). In both black-adapted and white-adapted animals, the LGs were almost devoid of immunogold labeling. Only about one in four granules was labeled and usually revealed only a single gold particle (0.3 ± 0.06 Gp/G in black-adapted, 0.3 ± 0.05 GP/G in white-adapted animals) (Figure 6A). Preabsorption of the antiserum with the ST-62 peptide and omission of the first antiserum completely abolished the immunostaining (data not shown).
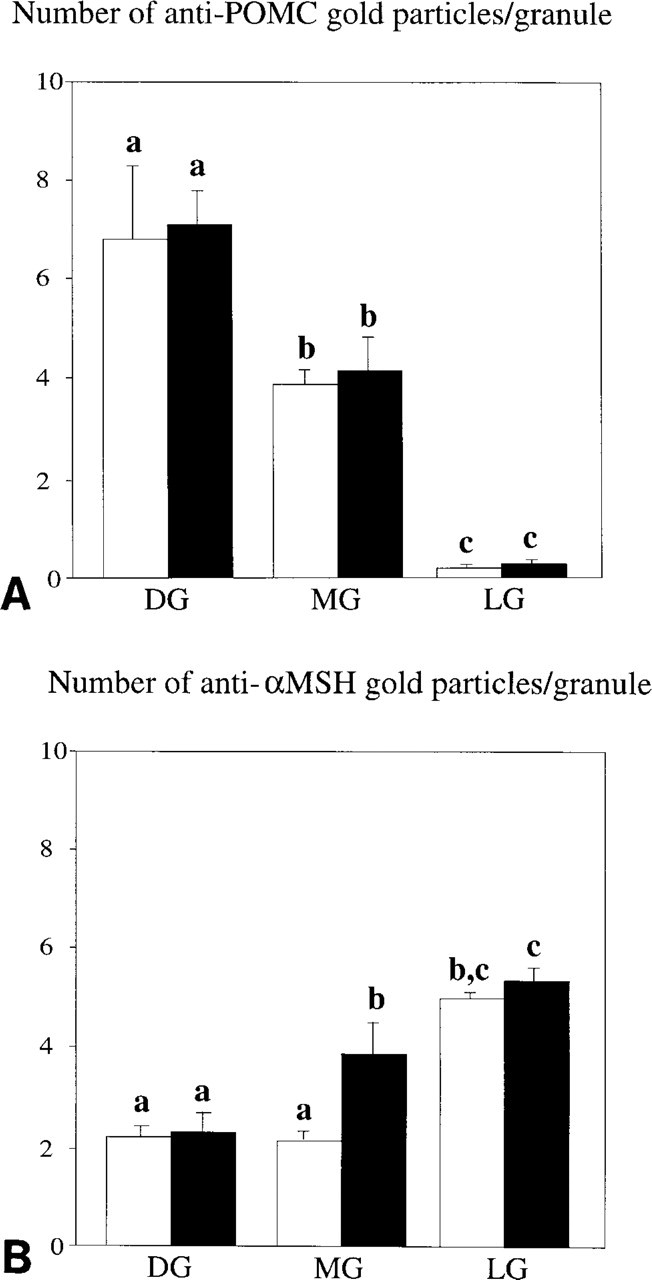
Labeling intensities for anti-POMC (
For anti-α-MSH, immunogold labeling was completely restricted to secretory granules. Virtually all granules were labeled (i.e., more than 98%), both in black-adapted and white-adapted animals (Figures 4A–4F). The granule types obviously showed differential labeling, with a staining pattern that was completely opposite to that obtained with anti-POMC. Weakest labeling was found over DGs (2.4 ± 0.30 Gp/G in black-adapted, 2.3 ± 0.17 Gp/G in white-adapted animals) (Figure 6B), especially DGs in contact with or close to the Golgi apparatus. Background light intensity had a clear effect on the MG. Whereas in white-adapted animals MGs did not differ in immunogold labeling compared to DGs (2.2 ± 0.21 Gp/G), in black-adapted animals MGs showed much stronger reactivity, which was 42% above the labeling of DGs (3.9 ± 0.61 Gp/G; p>0.05) (Figure 6A). Both in black-adapted and in white-adapted animals, LGs were much more strongly labeled than MGs (+40% and +48%, respectively) (Figure 6).
Discussion
Specificity of the Antisera
The antisera used in this study were carefully tested by immunoblotting and preabsorption tests. For the anti-α-MSH serum, the high specificity for α-MSH has been shown previously (Tanaka and Kurosumi 1986). The anti-POMC serum was tested in preabsorption tests. They showed a total loss of immunoreactivity after addition of the ST-62 peptide but not after preabsorption with an excess of synthetic α-MSH. Moreover, Western blot showed that only one protein band of 38 kD reacts with the antiserum. This molecular weight corresponds to that of the precursor POMC in Xenopus (Martens et al. 1980,1985). These results demonstrate that the anti-POMC serum specifically recognizes the precursor protein POMC.
First Cleavage Site of POMC in Xenopus
The immunoblot data reveal that the anti-POMC serum used in this study recognizes only one protein, POMC itself. This means that the sequence (Glu-Leu-Arg-Arg-Glu-Leu-Ser-Leu-Glu-Cys) to which the antiserum had been raised contains the first proteolytic cleavage site of Xenopus POMC. Earlier studies showed that this sequence includes the first proteolytic site of rat POMC as well (Tooze et al. 1987; Tanaka and Kurosumi 1992).
Light Immunocytochemistry Reveals Different Locations of POMC and α-MSH
The light immunocytochemical data show a differential staining pattern with anti-POMC compared to that obtained with anti-α-MSH. Whereas POMC occurs particularly in the perinuclear region of the melanotropes, α-MSH is found throughout the cytoplasm of the melanotrope cell. This observation indicates that POMC is contained within the perinuclear RER, the Golgi apparatus, and/or in immature secretory granules lying near the Golgi apparatus, whereas mature α-MSH appears to be present in secretory granules dispersed throughout the cell. The ultrastructural double labeling experiment confirms this idea (see below).
Different Granule Types Represent Different Stages in Granule Maturation
The ultrastructural observations show that the RER in the melanotrope cells of Xenopus laevis does not stain with either of the two antisera. Because it is highly unlikely that the RER would not contain POMC, the reason for the absence of anti-POMC immunoreactivity may be that the POMC molecules are packed very tightly together so that they cannot be reached by the POMC antibodies. On the other hand, it may be that POMC is too low in concentration to be detected or that the POMC molecule has another conformation that makes it unrecognizable for the antibodies. Immunoreactivity to anti-POMC is present within Golgi cisternae, suggesting that the Golgi apparatus contains POMC in a less tightly packed form and that this POMC is not yet processed to α-MSH. As the ultra-structural study also shows, the Golgi forms small granules with an electron-dense content, previously defined as electron-dense granules (DGs; Roubos and Berghs 1993). The immunoelectron microscopic study reveals that DGs are strongly positive with the anti-POMC serum, especially those still attached to the Golgi apparatus, indicating that the DGs contain a high amount of POMC. In the DGs, relatively few POMC molecules are processed to α-MSH, because the DGs poorly react with the anti-α-MSH serum and hence appear to contain only a small amount of α-MSH. Therefore, we conclude that these granules represent the first stage of the melanotrope secretory granule. In contrast to DGs, LGs are never seen associated with the Golgi apparatus. As to their contents, the situation is opposite to that of the DGs. Their immunoreactivity to anti-POMC and anti-α-MSH indicates that LGs contain a very low amount of POMC but a high amount of α-MSH. Apparently, in this granule stage almost all POMC has been processed to form α-MSH. Therefore, they appear to represent a late step in secretory granule maturation. MGs show an intermediate picture, both in morphology and in immunoreactivity. The size of these granules, their degree of electron density, and their immunolabeling with anti-POMC and anti-α-MSH are all intermediate compared to the DGs and LGs. Therefore, the MGs appear to represent an intermediate, transient step in granule maturation, in between the DGs and LGs, in which POMC is only partially processed to α-MSH (and probably to other POMC endproducts). Apparently, POMC is gradually cleaved in the secretory granules, which simultaneously change their morphological characteristics. The first cleavage of POMC may start within the trans-Golgi network, whereas the last cleavage activity of POMC may take place within mature LGs, which eventually release their fully processed endproducts by exocytosis. Interesting in this respect would be the immunolocalization of endoproteases PC1 and PC2 in the secretory granule types (Braks et al. 1992). Recently, a co-localization of PC1 with POMC and with POMC peptides was demonstrated in secretory granules of AtT-20 cells, confirming the hypothesis of gradual POMC processing during granule maturation (Tanaka et al. 1997).
Effects of Background Adaptation on Granule Maturation and POMC Processing
It is well known that adaptation to a black background has a stimulatory effect on the biosynthesis of POMC (Martens et al. 1980,1982; Ayoubi et al. 1992). The present study shows that this process is reflected at the morphological level by the fact that DGs, and especially the DGs associated with the Golgi apparatus, are much more numerous in melanotrope cells of black-adapted animals than in melanotropes of white-adapted Xenopus. In addition, the MGs are affected by background light, as is clear from the greater number of these granules in black-adapted animals, indicating a higher turnover of DGs to MGs. Whereas the amounts of POMC and α-MSH per DG do not appear to be related to the state of background adaptation, for MGs, the situation is different. The immunoreactivity of MG to α-MSH is higher under black than under white background conditions, suggesting that the rate of processing of POMC to α-MSH in the MGs is increased in black-adapted animals. As to the LGs, melanotrope cells of animals adapted to a white background clearly contain more of these granules than cells of black-adapted toads. Because under white background conditions melanotropes do not or barely release α-MSH (Jenks et al. 1993), it must be concluded that these cells have accumulated LGs, containing fully processed peptides, during their 3-week adaptation stay on a white background. Data show that LGs of black- and white-adapted animals do not differ in immunoreactivity to α-MSH. Most likely, the secretory granules in animals on a white background have had time enough (i.e., 3 weeks) to process all POMC to α-MSH, catching up in this way with the more rapid production of α-MSH under black background conditions. After a longer period on a white background, Xenopus appears to be fully ready for a fast adaptive response to a black background stimulus, because its melanotrope cells are crowded with LGs that contain fully processed peptides.
In conclusion, the present study shows that POMC processing to POMC endproducts, including α-MSH in secretory granules of the melanotrope cell of Xenopus laevis, is a gradual process, which occurs in parallel with the morphological transition of DGs via MGs to LGs. The DGs are the immature granules and the LGs are the mature granules, where the MGs represent an intermediate stage between DGs and LGs. Adaptation to a black background stimulates both the formation of immature granules and the intragranular processing of POMC, which is concomitant with the transition between the granule types.
Footnotes
Acknowledgements
Supported by grants from the EU HCM (ERBCHRXCT-920017) and the NWO/MW-INSERM exchange program.
We thank Huub Geurts and Tony Coenen for technical assistance, and Ron Engels for animal care.
