Abstract
We examined the distribution of copper-zinc superoxide dismutase (CuZn-SOD) in adult rat central nervous system by light and electron microscopic immunocytochemistry, using an affinity-purified polyclonal antibody. The enzyme appeared to be exclusively localized in neurons. No immunoreactivity was seen in non-neuronal cells. The staining intensity was variable, depending on the brain region and, within the same region, on the neuron type. Highly immunoreactive elements included cortical neurons evenly distributed in the different layers, hippocampal interneurons, neurons of the reticular thalamic nucleus, and Golgi, stellate, and basket cells of the cerebellar cortex. Other neurons, i.e., pyramidal cells of the neocortex and hippocampus, Purkinje and granule cells of the cerebellar cortex, and the majority of thalamic neurons, showed much weaker staining. In the spinal cord, intense CuZnSOD immunoreactivity was present in many neurons, including motor neurons. Pre-embedding immunoelectron microscopy of the neocortex, hippocampus, reticular thalamic nucleus, and cerebellar cortex showed cytosolic and nucleoplasmic labeling. Moreover, single membrane-limited immunoreactive organelles identified as peroxisomes were often found, even in neurons that appeared weakly stained at the light microscopic level. In double immunogold labeling experiments, particulate CuZn-SOD immunoreactivity co-localized with catalase, a marker enzyme for peroxisomes, thus demonstrating that in neural tissue CuZnSOD is also present in peroxisomes.
Keywords
R
Both enzymatic and nonenzymatic systems are involved in the removal of ROS. Superoxide dismutases (SODs) are a family of metalloenzymes that catalyze the dismutation of O2-· to H2O2, which, in turn, is reduced to water by catalase or peroxidases. Mammalian cells possess three forms of SODs with distinct localizations, i.e., extracellular SOD (EcSOD), manganese SOD (MnSOD), and copper-zinc SOD (CuZnSOD) (Fridovich 1989).
MnSOD is located in the mitochondria, and CuZn-SOD is predominantly found in the cytosol and nucleoplasm (Slot et al. 1986). Recent biochemical and morphological studies have shown that CuZnSOD is also present in peroxisomes of rat hepatocytes and fibroblasts (Dhaunsi et al. 1992; Wanders and Denis 1992) and of human fibroblasts and hepatoma cells (Keller et al. 1991). Peroxisomes are cytoplasmic organelles containing many H2 O2-producing oxidases and catalase, and are therefore primarily involved in oxygen metabolism. The presence of CuZnSOD in the peroxisomes, however, is still controversial (Crapo et al. 1992; Kobayashi et al. 1993) and to date has not been demonstrated in the nervous system.
The importance of CuZnSOD in the CNS is emphasized by the finding that this enzyme exerts a protective action against brain injury and neuronal death both in vitro and in vivo (Imaizumi et al. 1990; Matsumiya et al. 1991; Tagaya et al. 1992; Uyama et al. 1992; He et al. 1993; Greenlund et al. 1995). Moreover, transgenic mice overexpressing the human CuZnSOD gene were found to be more resistant than their nontransgenic normal littermates to oxidative stress, including ischemia-reperfusion injury (Chan 1994). Conversely, loss of endogenous CuZnSOD anticipates the neuronal pathological changes due to focal or global cerebral ischemia in rat (Liu et al. 1993, 1994).
CuZnSOD has also been involved in other neurodegenerative diseases, such as Parkinson's disease (Saggu et al. 1989; Marttila et al. 1990; Nishiyama et al. 1995), in which an imbalance between free radical production and their scavengers has been demonstrated (Yoshikawa 1993).
Interest in elucidating the precise role of CuZnSOD in neural tissue has furtherly increased in recent years, because point mutations in the gene encoding this enzyme have been detected in a proportion of families with familial amyotrophic lateral sclerosis (FALS) (Deng et al. 1993; Rosen et al. 1993; Gurney et al. 1994).
Despite the importance of CuZnSOD in neural tissue, light and electron microscopic studies on the localization of this enzyme are few and are focused on restricted brain areas (Thaete et al. 1986; Liu et al. 1994; Pardo et al. 1995).
In this article we describe the distribution of CuZn-SOD in the rat CNS by use of a recently developed immunocytochemical procedure that allows great signal amplification (Adams 1992; Moreno et al. 1995). We also performed immunoelectron microscopy in selected brain areas, i.e., the neocortex, the hippocampus, the reticular thalamic nucleus, and the cerebellar cortex, with the aim of investigating the intracellular distribution of the enzyme, with particular regard to the peroxisomes.
A preliminary report of our results has been published (Moreno et al. 1994).
Materials and Methods
Antibodies
Affinity-purified rabbit polyclonal anti-CuZnSOD antibody was a generous gift of Dr. M. Giorgi, University of L'Aquila, Italy (Rossi et al. 1994). As antigen, CuZnSOD purified from rat liver according to Yano (1990) (a generous gift of Prof. G. Rotilio, University “Tor Vergata,” Rome, Italy) was used.
Sheep polyclonal anti-catalase antibody was a generous gift from Prof. D. Fahimi (University of Heidelberg, Germany).
Immunoblotting
The specificity of the affinity-purified anti-CuZnSOD was tested by immunoblotting. This was performed as follows. Rat liver CuZnSOD, goat erythrocyte CuZnSOD (a generous gift from Prof. L. Calabrese, University of Rome 3, Italy), rat brain, and rat liver homogenates, suspended in 60 mM Tris-HCl buffer, pH 6.8, containing 2% sodium dodecyl sulfate, 5% β-mercaptoethanol, 10% glycerol, and 0.001% bromophenol blue, were heated to 92C for 15 min to ensure that all the native proteins were denatured to the monomeric form. Thirty ng of the purified proteins, 60 μg and 100 μg of liver and brain proteins, respectively, were loaded onto a 15% polyacrylamide gel. SDS-PAGE electrophoresis was performed according to Laemmli (1970). Western blotting was performed according to Towbin et al. (1979). Immunostaining was obtained by incubating the nitrocellulose sheets with the primary antibody (1:5000) and then with goat anti-rabbit IgG conjugated with alkaline phosphatase (1:2000) (Sigma Chemical; St Louis, MO). The immunocomplexes were revealed according to Kincaid (1988).
Controls were performed by omission of the primary antibody. Because of the scarcity of available antigen, we were unable to perform controls with the primary antibody previously adsorbed with excess antigen.
Animals
Twenty adult Wistar rats of either sex, weighing about 250 g and kept on a standard diet and water ad libitum, were deeply anesthetized with pentobarbital (35 mg/kg). Animals were housed and handled according to approved guidelines.
Light Microscopic Immunocytochemistry
Animals were perfused through the aorta at room temperature (RT) with Ringer's variant without calcium, pH 7.3, followed by 4% freshly depolymerized paraformaldehyde with or without 0.05% glutaraldehyde in 0.12 M phosphate buffer, pH 7.3. One hr after perfusion the brains were dissected and cut coronally or sagittally on a Vibratome at 25 μm. Sections were collected in 0.12 M phosphate buffer, pH 7.4, and then transferred to PBS containing 2% nonfat dry milk and 0.05% Triton X-100 for 1 hr. Sections were then incubated with anti-CuZnSOD diluted 1:6000–1:12,000 in PBS containing 1% nonfat dry milk and 0.02% Triton X-100 overnight at 4C. After rinsing with buffer, sections were processed applying an amplification procedure of the avidin-biotin method (Adams 1992), with the modifications previously described (Moreno et al. 1995). Controls performed by omission of the primary antibody were negative.
Biotinylated secondary antibody, normal goat serum (NGS), and avidin-biotin–horseradish peroxidase (HRP) (standard ABC kit) were purchased from Vector (Burlingame, CA), NHS-biotin from Pierce Chemical (Rockford, IL), and tyramine and 3,3′-diaminobenzidine (DAB) from Sigma.
Pre-embedding Immunoelectron Microscopy
Specimens were treated for immunocytochemistry as above. After the immunoreaction, sections were rinsed with buffer and then postfixed in OsO4 for 1 hr, dehydrated, and flat-embedded in Epon. Selected areas were remounted on Epon blanks and sectioned on a Reichert Ultracut ultramicrotome. Ultrathin sections were briefly contrasted with uranyl acetate and photographed in a Philips 400 T electron microscope.
Post-embedding Double Immunogold Labeling
Vibratome coronal sections 100 μm thick were collected in 0.12 M phosphate buffer, pH 7.4, and then dehydrated in graded alcohols, progressively lowering the temperature (from 4C to −20C). Sections were infiltrated with Unicryl (British BioCell; Cardiff, UK), according to Scala et al. (1992), at −20C for 48 hr, and then at RT overnight. Polymerization was carried out at −10C under UV light.
Other 100 μm Vibratome sections were dehydrated and embedded in London Resin White (LR White) at 4C.
Ultrathin sections were sequentially incubated with (a) NGS diluted 1:10 in 10 mM PBS containing 1% bovine serum albumin (BSA) (Medium A) for 15 min, (b) rabbit anti-CuZnSOD diluted 1:40 in Medium A, overnight, (c) goat anti-rabbit IgG conjugated to 10-nm colloidal gold (British BioCell), diluted 1:30 in Medium A containing 0.01% gelatin for 1 hr, (d) sheep anti-catalase diluted 1:10 in Medium A, overnight, and (e) donkey anti-sheep IgG conjugated to 5-nm colloidal gold (British BioCell), diluted 1:30 in Medium A containing 0.01% gelatin for 1 hr. All incubations were carried out at RT, (except for b and d, at 4C). Grids were briefly contrasted with uranyl acetate and photographed in a Philips 400 T electron microscope.
Results
Immunoblotting
Figure 1 shows the immunoblotting obtained by using the affinity-purified antibody produced against rat liver CuZnSOD. The antibody recognizes both the rat liver antigen used for immunization and goat erythrocyte CuZnSOD. In rat liver and brain homogenates, the antibody recognizes only one band at the MW corresponding to the CuZnSOD monomer (17 kD). Controls are negative.
Light Microscopic Immunocytochemistry
[Note: For all Figures the terminology follows that of Paxinos and Watson (1986).] Immunoreactivity to CuZnSOD was seen exclusively in neurons. No staining was detectable in glial or endothelial cells. The intensity of immunoreaction was variable, depending on the area and on the neuronal type considered.
In the main olfactory bulb, appreciable staining was observed in periglomerular cells in the glomerular layer, in a few neurons located in the external plexiform layer, in mitral cells, and in many elements of the granule cell layer (Figure 2A).
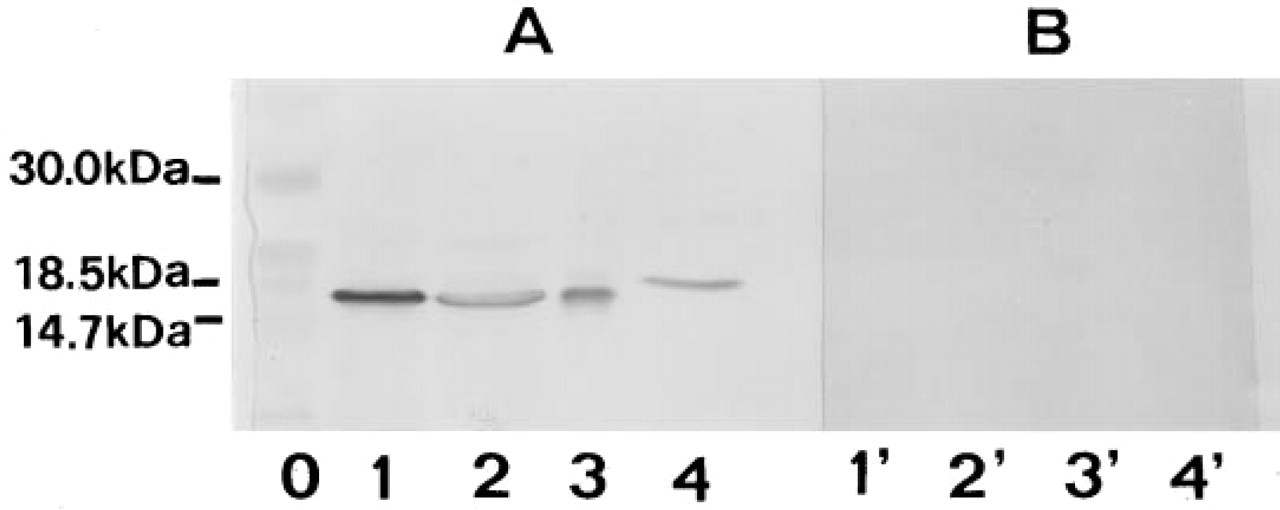
Immunoblotting with the affinity-purified anti-CuZnSOD polyclonal antibody. (
The piriform cortex appeared extremely rich in CuZnSOD-positive neurons. Because of their number, size, and location in the three layers and in the subjacent endopiriform nucleus, most of the positive cells were interpreted as GABAergic interneurons (not shown).
In the neocortex, especially in the frontal area, many highly immunoreactive neurons were seen in all the layers (Figure 2B). On the basis of their shape and orientation, most of these cells were identified as GABAergic interneurons (Mugnaini and Oertel 1985). Pyramidal cells showed weaker immunoreactivity.
In the telencephalon, immunostaining was detected in many cells of the caudate putamen, including spiny neurons and presumably cholinergic interneurons (Figure 2C), in the ventral pallidum, in the islands of Calleja (Figure 2D), and in some neurons of the septal nuclei (Figure 2E).
In the hippocampal formation, the immunoreaction product was concentrated in interneurons. These cells were evenly distributed in all layers of Ammon's horn, the dentate gyrus, and the subiculum (Figures 2F–2H). Hippocampal pyramidal cells and granule cells were weakly positive.
In the diencephalon, the most remarkable degree of staining was seen in the reticular nucleus of the thalamus (Figure 3A). The anterodorsal thalamic nucleus and lateral and medial geniculate nuclei also showed distinct labeling, whereas other ventral and dorsal thalamic nuclei displayed little immunoreactivity.
In the mesencephalon, intense immunoreactivity was found in neurons of all the layers of superior colliculus (Figure 3B). A moderate positivity was also detected in the central gray substance and in the inferior colliculus (Figure 3C). The substantia nigra displayed intense immunoreactivity in both pars reticulata and pars compacta. Staining was more evident in the neuronal cytoplasm than in the nucleus (Figure 3D). The magnocellular part of the red nucleus also showed some positivity, the staining being confined to the neuronal cytoplasm (Figure 3E). Many other nuclei of the mesencephalon, such as dorsal and ventral nuclei of the lateral lemniscus, raphe nuclei, and interpeduncular nucleus, appeared rather rich in CuZnSOD (Figure 3F).
In the hindbrain, almost all the centers appeared distinctly positive for CuZnSOD. The nuclei that exhibited the highest level of immunoreactivity included the pontine nuclei, nucleus abducens, supragenual nucleus, gigantocellular reticular nucleus, ventral and dorsal cochlear nuclei, spinal trigeminal nucleus, principal trigeminal sensory nucleus, nucleus of the trapezoid body, facial nucleus, and lateral vestibular nucleus (Figure 3G–3J).
In the cerebellum, small neurons of deep cerebellar nuclei were moderately positive (Figure 3K). In the cerebellar cortex, remarkable immunostaining was seen in Golgi cells of the granular layer and in basket and stellate cells of the molecular layer, whereas granule and Purkinje neurons were negative (Figure 3L).
In the spinal cord, neurons of all the Rexed's laminae, comprising motor neurons and interneurons, displayed intense immunostaining (Figures 4A and 4B). In many areas, the positive neurons showed immunoreaction product in both their cytoplasm and nucleus. However, neurons of certain areas, such as mitral cell layer, substantia nigra, red nucleus, abducens nucleus, and reticular gigantocellular nucleus, deep cerebellar nuclei primarily showed cytoplasmic immunoreactivity (Figures 2A, 3D, 3E, 3G, 3H, and 3K). On the contrary, other neurons, such as Golgi cells of the cerebellar cortex, primarily displayed nuclear positivity (Figure 3L). In all the neurons, the nucleolus appeared negative (e.g., inset in Figure 3L).
Pre-embedding Electron Microscopic Immunocytochemistry
In all the brain regions examined at the ultrastructural level, CuZnSOD immunoreactivity was seen exclusively in neurons. The immunoreaction product was rather homogeneously diffused in the cytosol and in the nucleoplasm of the positive neurons. Densely stained cytoplasmic bodies, bounded by a single membrane and measuring 0.2–0.3 μm in diameter, were often observed. Endoplasmic reticulum, Golgi apparatus, vacuoles, mitochondria, and nucleolus were always unstained.
In the cerebral neocortex the immunoreaction product appeared concentrated in heterogeneous nonpyramidal neurons, ranging from 7 to 18 μm in mean diameter (Figure 4C).
In the hippocampus, intense positivity was seen in small interneurons (not shown). Weakly immunoreactive pyramidal cells were occasionally observed. In these cases, the nucleus showed a relatively stronger labeling than the cytoplasm. These cells sometimes contained positively labeled organelles that resembled peroxisomes (Figure 4D).
In the thalamic reticular nucleus (Figures 4E, 4F, 5A, and 5B), the majority of the neurons showed intense immunoreactivity. Glial cells, as in other brain regions, were devoid of staining (Figures 5A and 5B). A number of axons and dendrites, probably belonging to the positive neurons, were immunoreactive. Other neurites appeared immunonegative, irrespective of the presence or absence of a myelin sheath.
In the cerebellar cortex, stellate and basket cells of the molecular layer were highly immunoreactive (Figure 5C). Parallel fibers originating from granule cells were negative, whereas other axons and dendrites were specifically stained (not shown). Purkinje cells showed weak positivity for CuZnSOD. In fact, only a few of these neurons were recognized as immunoreactive, and even in these cases the immunoreaction product was non-homogeneously distributed, being more evident in the nucleoplasm. However, clearly specific and intense immunostaining was rather frequently observed in single membrane-limited bodies measuring 0.2–0.4 μm in diameter, whereas other organelles were negative (Figure 5D). Golgi cells of the granular layer showed cytoplasmic and nuclear staining (Figure 5E) and also immunoreactive organelles (Figure 5E, inset). Granule cells showed very weak but specific CuZnSOD immunoreactivity in their nuclei, apparently only in correspondence with euchromatin (Figure 5E).
Postembedding Double Immunogold Labeling
The comparison between LR White- and Unicryl-embedded sections revealed that the neural tissue morphology was not satisfactorily preserved by the former resin (not shown), whereas the latter yielded better ultrastructural preservation. Unicryl was therefore chosen for postembedding experiments.
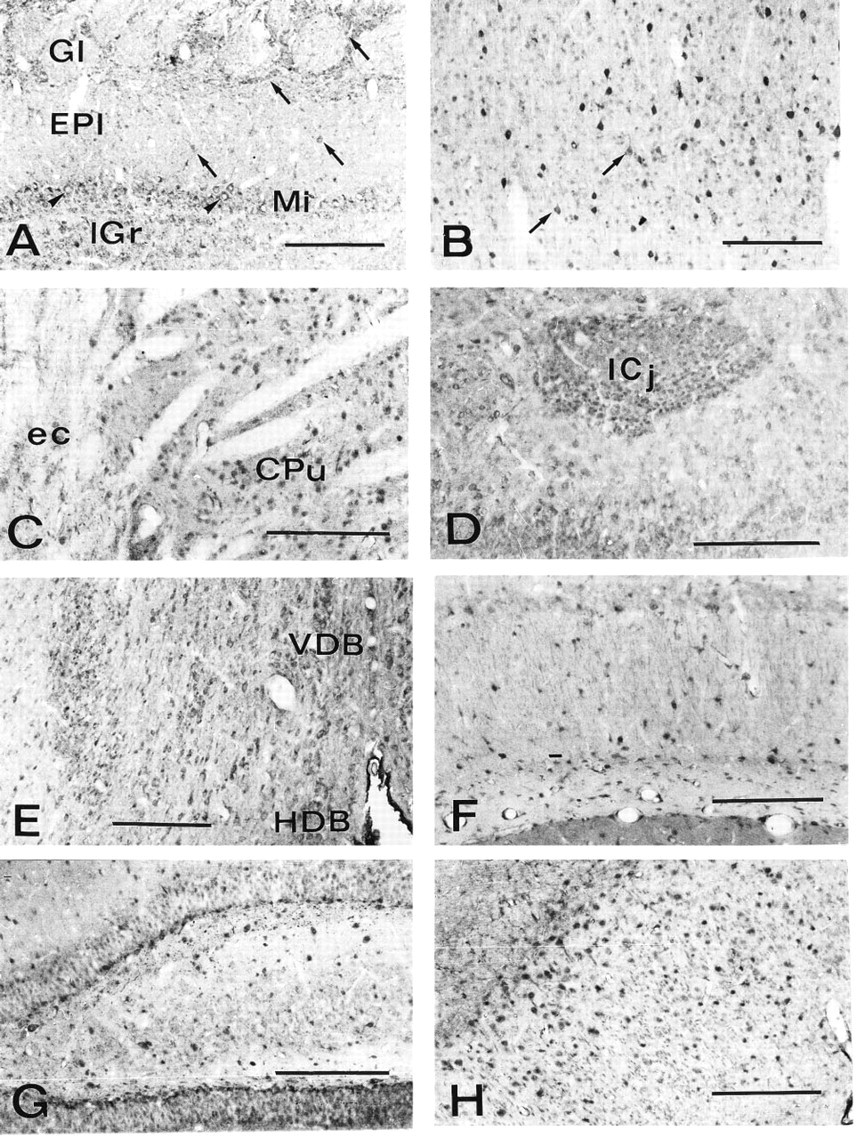
Coronal sections of rat brain processed for CuZnSOD immunocytochemistry. (
For double immunogold labeling, the cerebellar cortex was selected because of the presence of some cell types showing peroxisome-like positivity for CuZnSOD and known to contain catalase (Moreno et al. 1995), i.e., Golgi and Purkinje cells. In these cells, organelles measuring 0.15–0.35 μm in diameter showed intense immunogold labeling for CuZnSOD, revealed by 10-nm gold particles (Figure 5F). Anti-catalase antibody (revealed by 5-nm gold) co-localized with anti-CuZnSOD in the same organelles, thus confirming their peroxisomal nature (Figure 5F). Examination of 15 double labeled organelles belonging to either Golgi or Purkinje cells gave a mean content of 5.2 gold particles of 5 nm (SD 2.17) and 26 gold particles of 10 nm (SD 4.97).
Discussion
In the present investigation we have obtained a complete map of the distribution of CuZnSOD in the rat CNS by means of an extremely sensitive immunocytochemical procedure based on the avidin-biotin system (Adams 1992). This study also describes for the first time the ultrastructural localization of CuZnSOD in neural cells and demonstrates that the enzyme is also present within the peroxisomes.
Regional Distribution of CuZnSOD
Our light and electron microscopic observations demonstrate that CuZnSOD is an exclusively neuronal enzyme, the immunostaining being absent from any non-neuronal element, including glial and endothelial cells. This result is in agreement with other reports dealing either with expression of CuZnSOD mRNA in the human hippocampus and mesencephalon (Ceballos et al. 1989; Zhang et al. 1993) or with immunolocalization of the protein in the gerbil hippocampus (Matsuyama et al. 1993). Furthermore, both CuZnSOD mRNA and protein were detected only in neuronal elements in the brains of transgenic mice carrying the human CuZnSOD gene (Ceballos-Picot et al. 1991). In this respect, it is worth mentioning that immunologically active CuZnSOD was detected in reactive glial cells in gerbil hippocampus after ischemic insult (Kato et al. 1995).
Although specific immunoreactivity is detectable in most, if not all, neurons, different degrees of staining are clearly distinguished in the various brain regions. High staining levels are found in all subdivisions of the CNS and particularly in the cerebral cortex, the hippocampus, the thalamus, the superior colliculus, the substantia nigra, the cerebellar cortex, and the spinal cord. However, within these areas, not all the neurons show similar immunoreactivity.
On the basis of biochemical data, a non-homogeneous distribution of CuZnSOD in various areas of rat brain has been suggested previously (Ciriolo et al. 1991; Carrillo et al. 1992). Our data are in overall agreement with these reports because appreciable immunoreactivity was detected in the caudate nucleus, the cerebral cortex, hippocampus, pons, and medulla oblongata. Some discrepancies between the biochemical and morphological results can be easily explained, keeping in mind that the former reflect the mean values of CuZnSOD biochemical activity of different cell types within the same area, which may include highly positive cells together with negative cells.
The morphological studies available on the distribution of CuZnSOD in neural tissue also point to a differential expression of this enzyme. Immunohistochemical studies dealing with CuZnSOD localization recently carried out in specific CNS regions (Liu et al. 1994; Pardo et al. 1995) are in agreement with our results, because many positive neurons were observed in the neocortex, striatum, and spinal cord. Moreover, intense CuZnSOD immunoreactivity has been described in pyramidal cells and interneurons of the hippocampal regions CA1 and CA3, whereas dentate granule cells showed only moderate staining (Liu et al. 1993; Kato et al. 1995).
Our light and electron microscopic observations are in partial agreement with these studies, because we demonstrate that in the hippocampal formation interneurons are strongly immunostained for CuZnSOD, whereas pyramidal cells and granule cells show weaker immunoreactivity.
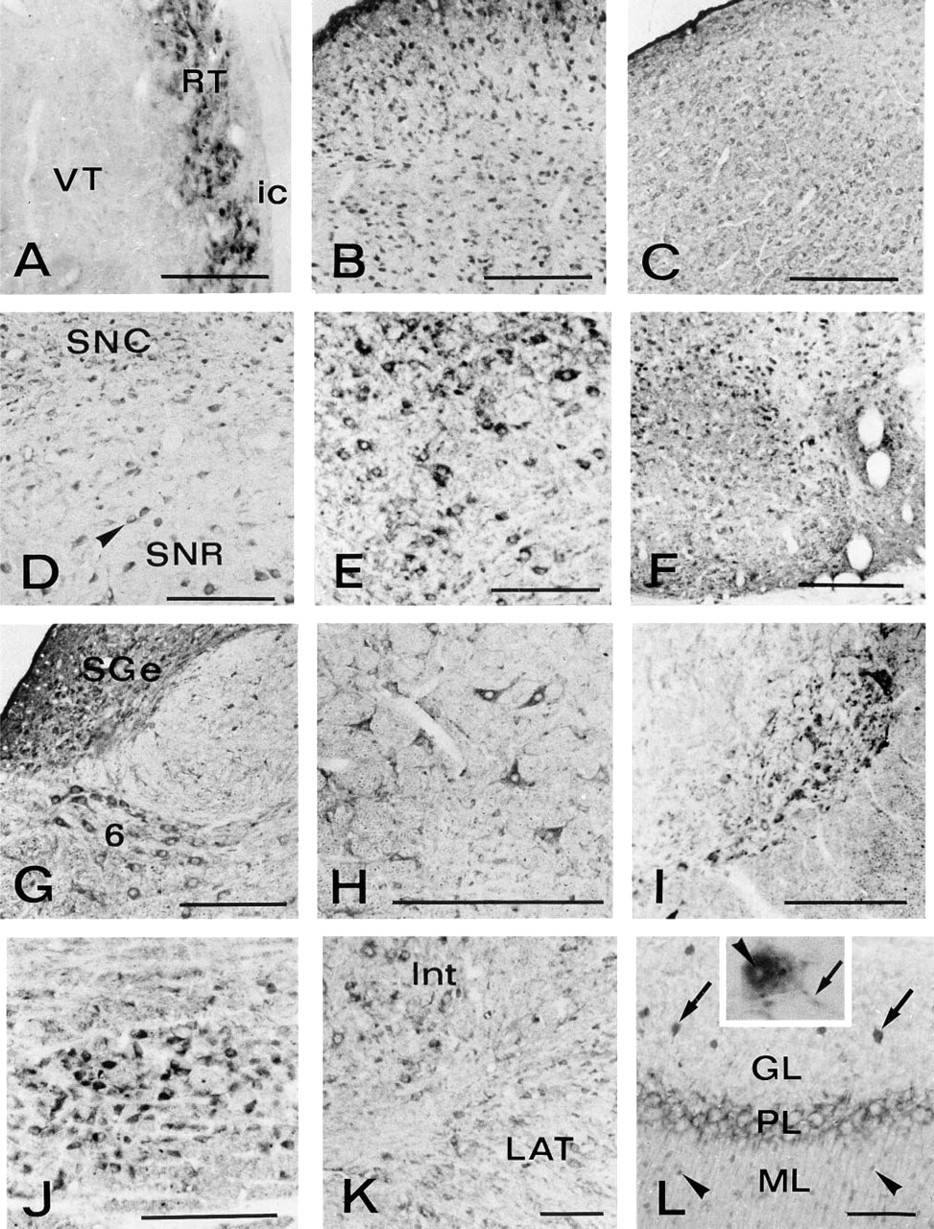
CuZnSOD immunocytochemistry in rat brain. (
In human mesencephalon, in situ hybridization revealed preferential expression of the CuZnSOD mRNA in dopaminergic neurons, particularly in the melanized ones, thus suggesting a role for the enzyme in the removal of superoxide anion generated by catecholamine metabolism (Zhang et al. 1993). Consistently, in our specimens many aminergic areas are identified as immunoreactive (substantia nigra, central gray substance, locus ceruleus). However many other strongly positive neurons use different neurotransmitters. In fact, interneurons of the piriform cortex, neurons of the reticular thalamic nucleus, principal neurons in the corpus striatum, neurons of the dorsal nucleus of lateral lemniscus, and Golgi, basket, and stellate cells of the cerebellar cortex are GABAergic. Furthermore, the cholinergic motor neurons of the cranial and spinal nuclei are also positive.
The factors involved in the differential expression of CuZnSOD in the various neuronal populations therefore remain obscure, and cannot solely depend on the specific neurotransmitter used by the neuron, at least as far as it concerns the classic mediators.
Functional Considerations
The fact that CuZnSOD is expressed exclusively in neurons may suggest that these cells require a specific protective mechanism against the superoxide anion, although a role of CuZnSOD through its metabolite H2O2 or through a still unknown function, possibly linked to the presence of copper and zinc in its molecule, cannot be ruled out.
Therefore, the issue concerning the functional implications of CuZnSOD abundance in certain neuronal subtypes is still to be addressed. Although in some instances the presence of CuZnSOD appears to favor a greater protection against ROS-mediated neuronal damage (Chan 1994; Medina et al. 1996), in other cases high levels of CuZnSOD may even be responsible for neuronal injury, as probably occurs in Down's syndrome and in some strains of transgenic mice that overexpress CuZnSOD (Brooksbank and Balazs 1984; Amstad et al. 1994).
Correspondingly, some of the strongly positive neurons described in our specimens can be identified as oxidative stress resistant neurons (i.e., GABAergic interneurons of hippocampus and cerebral cortex) (Schlander et al. 1988), whereas others clearly belong to subtypes typically vulnerable to a variety of injuries most probably involving ROS (i.e., motor neurons of spinal cord, affected by ALS; neurons of the substantia nigra, affected by Parkinson's disease; and also principal neurons of the striatum, affected by Huntington's disease).
This apparent contradiction is probably due to the dual aspect of the function of SOD, one as a free radical scavenger (as the principal O2-· metabolizing enzyme) and the other as a ROS generator (as an H2O2-producing enzyme).
Concerning the H2O2 produced by cytosolic CuZn-SOD, one could envision that this molecule, leaked out of the neurons, is captured and eliminated by astrocytes, which have been reported to contain glutathione peroxidase (Damier et al. 1993) and to protect neurons against H2O2 toxicity (Raps et al. 1989; Desagher et al. 1996).
CuZnSOD as a Peroxisomal Enzyme
The comparison between the distribution of CuZnSOD in the rat CNS and that of catalase (McKenna et al. 1976; Moreno et al. 1995) allows further considerations. In many instances we have found an overlap of CuZnSOD and catalase immunostaining, e.g., in Golgi cells of the cerebellar cortex, interneurons of the hippocampus, reticular thalamic nucleus, substantia nigra pars reticulata, interpeduncular nucleus, motor neurons of cranial and spinal nuclei, and neurons of septal nuclei (Moreno et al. 1995).
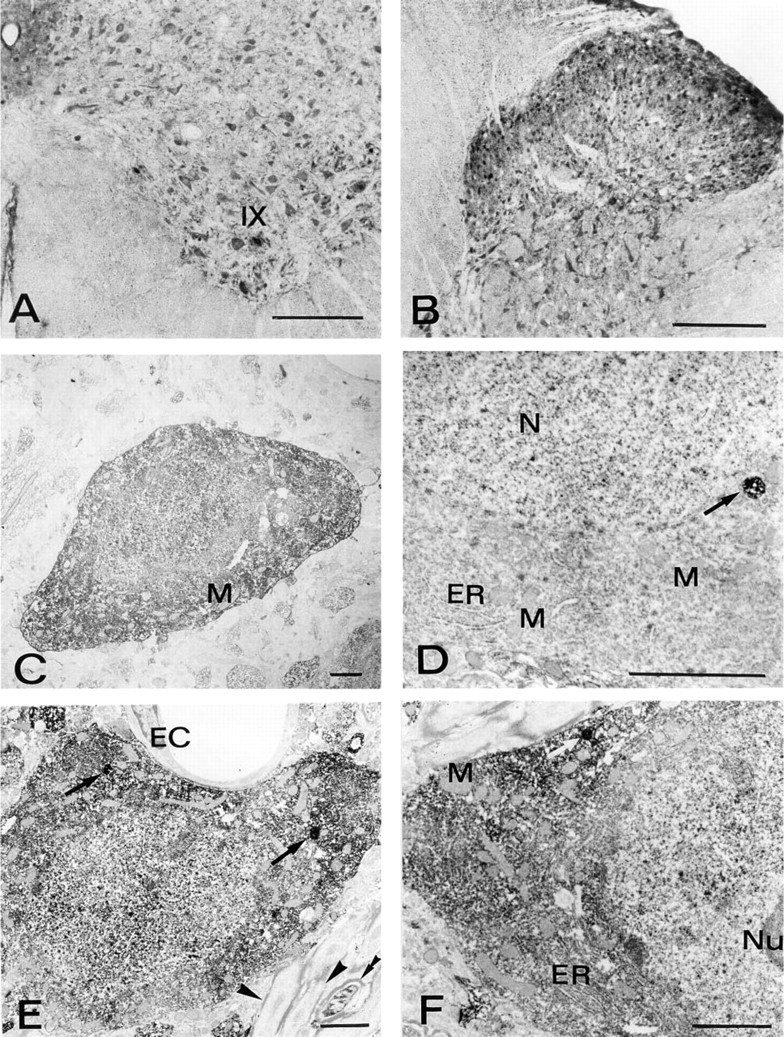
CuZnSOD immunocytochemistry in rat brain. (
In general, catalase is predominantly localized to peroxisomes, whereas CuZnSOD is mainly a cytosolic enzyme. However, ultrastructural examination of many different areas of the brain gives strong indication of a particulate CuZnSOD as well. We first interpreted these vescicles as peroxisomes on the basis of their size and morphology; even the dishomogeneous immunostaining observed within these organelles is in keeping with their peroxisomal nature, because a subcompartmentalization of peroxisomal enzymes has been largely demonstrated (for review see Van Den Munckhof 1996). Postembedding double immunogold labeling confirmed that the CuZnSOD-positive particles are true peroxisomes. The low catalase/SOD rate, as evaluated by morphometric analysis in double immunolabeled specimens, is not in contrast to this conclusion. Even the absence of catalase from SOD-positive organelles would not contradict their peroxisomal nature, because catalase-negative peroxisomes have been recently recognized (Van Roermund et al. 1995; Willcke et al. 1995), thus confirming the high degree of heterogeneity of these particles (Cimini et al. 1993) and the involvement of other systems in removal of H2O2 (Singh et al. 1994).
The positivity for CuZnSOD in the peroxisomes does not always correlate with a similar degree of immunoreactivity in the cytoplasm or nucleus. In fact, neurons apparently negative or weakly stained at the light microscopic level, such as pyramidal cells of the neocortex and hippocampus and Purkinje neurons of the cerebellar cortex, are often found to contain intensely labeled vacuoles.
Therefore, the compartimentalization of CuZnSOD within peroxisomes strongly suggests a specific function of this enzyme in the removal of intraorganellar by generated O2-·, independent of the situation in the cytosolic and nuclear compartments.
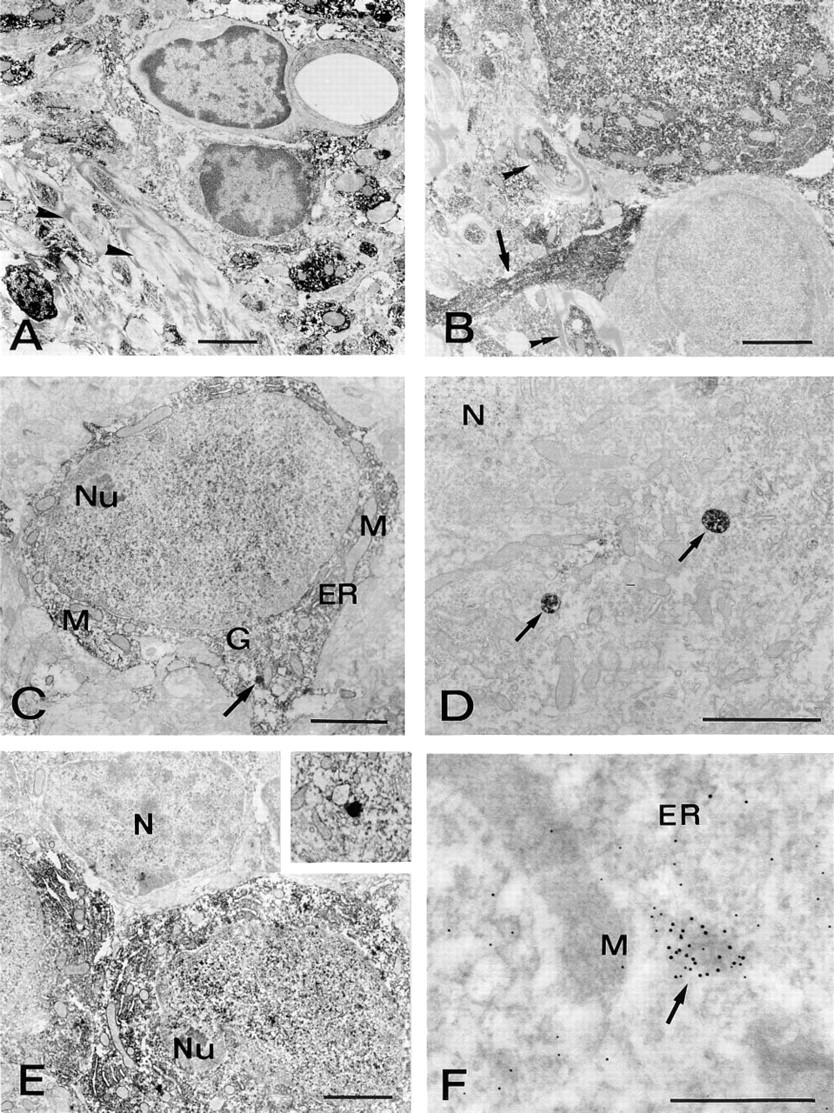
CuZn immunoelectron microscopy. (
Footnotes
Acknowledgements
Supported by MURST 40% grant, CNR grant no. 95.02196.CT04, and in part by the European Concerted Action “Peroxisomal Leukodystrophy” BMH4 CT 96–1621.
We wish to thank Dr E. Mugnaini for critical reading of the manuscript and for helpful discussion, Dr S. Stefanini for immunogold analysis, and Dr A. Cimini for immunoblotting. We gratefully acknowledge Dr L. Cristiano, Dr B. Pruiti, and Dr P. Aimola for skillful technical assistance.
