Abstract
Menkes disease is an X-linked disorder of copper metabolism. Excess amounts of copper in the kidney of Macular mice, a model for this disease, were found as copper–metallothionein (Cu–MT) from kidney of the mice. Histochemical studies of Cu–MT based on its autofluorescent emission properties showed that the protein was predominant in the proximal convoluted tubule (PCT) cells of the cortex. PCT cells are known to be the primary site of the nephrotoxicity caused by heavy metals. MT mRNA was also observed in the cortex, indicating that the protein was biosynthesized in this region. On the basis of these results, we suggest that biosynthesis and degradation of Cu–MT occur repeatedly in the PCT cells of the cortex. We also compared the histochemical localization of Cu–MT in Macular mice and Long–Evans cinnamon rats, a model for Wilson's disease. The significance of this comparison is discussed.
M
Recently, the gene encoding a P-type, cation-transporting ATPase was shown to be responsible for the decrease in hepatic copper content in Menkes patients (Chelly et al. 1993; Mercer et al. 1993; Vulpe et al. 1993). However, the developmental mechanisms of the abnormal copper accumulation in the kidneys of Menkes patients and mouse models have not been sufficiently investigated.
Copper is an essential trace element that requires a delicate cellular balance between necessary and toxic concentrations. Metallothionein (MT), a low molecular weight heavy metal binding protein, plays an important role in copper homeostasis and detoxification (Kojima and Kägi 1978; Kägi and Kojima 1987). Because it is generally accepted that surplus copper in the eukaryotic cells is bound to MT, excessive amounts of copper in the renal and intestinal cells of patients with Menkes disease are believed to exist in the form of Cu–MT. Kelly and Palmiter (1996) suggested that MT was essential to protect the embryonic placenta of Menkes disease against copper toxicity.
Hepatic copper accumulation is observed in some other disorders, such as Wilson's disease (Scheinberg and Steinlieb 1984). Nakayama et al. (1996) and Sakurai et al. (1992a,b, 1993) reported that large amounts of Cu–MT are present in the liver of Long–Evans cinnamon (LEC) rats (Li et al. 1991a,b; Okayasu et al. 1992), a model for Wilson's disease.
To gain insights into the developmental mechanism of the abnormal copper metabolism, we investigated the histochemical distribution of Cu–MT in the kidney of Macular mice by a method based on the auto-fluorescent emission properties of thiolate clusters in the protein (Nakayama et al. 1996; Okabe et al. 1996). Macular mice, discovered by Nishimura (1984), are a model for Menkes disease. Before the histochemical study, we identified a copper binding protein purified from the kidney of Macular mice as Cu–MT. The elucidation of its distribution in the kidney would greatly benefit understanding of the system for copper transport, storage, reabsorption, and excretion in Menkes disease. We also compared the renal localization of Cu–MT in Macular mice with that previously described by us in LEC rats (Okabe et al. 1996). The physiological importance of the difference between the macular mouse and the LEC rat is discussed.
Materials and Methods
Materials
Original breeding pairs of the Macular mouse strain were kindly supplied by Dr. M. Nishimura. The C3H male mice used as a control, weighing 20–25 g, were bought from Japan SLC (Hamamatsu, Japan). Both strains of mice were fed MF-4 standard pellets ad libitum (Oriental Yeast; Tokyo, Japan), containing 1.5 mg of copper/100 g, and water, and were housed under conditions of constant temperature (22 ± 1C) and humidity (50 ± 10%). Sephadex G-75, DEAE-Sephadex A-25, Octyl-Sepharose CL-4B, and ProRPC HR5/2 were obtained from Pharmacia LKB Biotechnology (Uppsala, Sweden). Other chemicals were reagent grade. Nitroblue tetrazolium (NBT) and 5-bromo-4-chloro-3-indoryl phosphate (BCIP) were obtained from Sigma (St Louis, MO). Zeta probe membrane and nylon membrane were from Bio-Rad (Richmond, CA) and Pall Ultrafine Filtration (Glen Cove, NY), respectively. The DIG DNA labeling and detection kit was bought from Boehringer Mannheim (Mannheim, Germany).
Copper Therapy of Macular Mice
Copper was administered to the mice by sc injections in the mid-dorsal region of 10 μl of copper chloride solution (10 μg of copper/g bw in 0.9% NaCl) on the morning of postnatal Days 7 and 10. Injections were made with a 10-μl Terumo microsyringe MT-10 (Tokyo, Japan).
Purification of Copper Binding Protein from Kidneys of Macular Mice
Copper binding proteins from 7.5 g of the kidneys were purified by a simple method that we previously described (Suzuki–Kurasaki et al. 1996). Briefly, 7.5 g of kidney from Macular mice was homogenized (1:1, w/v) in 50 mM Tris-HCl buffer, pH 8.6, and centrifuged at 9000 x g for 30 min. The fat-free supernatant was subsequently recentrifuged at 100,000 x g for 1 hr. The cytosol was applied to a column (2.5 × 100 cm) of Sephadex G-75 and the fractions were analyzed for metal concentrations with a Hitachi frame atomic absorption spectrophotometer model 180–80 (Hitachi; Tokyo, Japan) and absorbance at 280 nm with a Beckman spectrophotometer model DU-65 (Fullerton, CA).
Further purification of the low molecular weight copper-containing fractions was carried out on a column of DEAE-Sephadex A-25 (2.1 × 4 cm) with a linear gradient. The main copper-containing peak was immediately subjected to a column (1.1 × 3 cm) of Octyl-Sepharose CL-4B. The pass-through fractions were gathered.
Finally, analytical and preparative chromatography was carried out on a reverse-phase FPLC column, ProRPC HR5/2, attached to the fast protein liquid chromatography (FPLC) equipment from Pharmacia LKB Biotechnology. Fractions were collected and measured for absorbance at 220 nm with a Beckman spectrophotometer model DU-50 and for copper content with a Hitachi flameless atomic absorption spectrophotometer model 170–70. The isolated protein was lyophilized. All steps were performed at 4C.
Amino Acid Analysis
The proteins were oxidized with performic acid at 4C for 16 hr and dried with a Yamato rotary evaporator model RE-40 (Tokyo, Japan) at 40C. Amino acid analysis after hydrolysis at 110C with 6 N HCl for 24 hr was performed on a Hitachi amino acid analyzer model 305.
Histochemical Observation of Luminescence from Cu–MT
All procedures were carried out according to the methods of Okabe et al. (1993), with some modifications. Zeta probe membranes were used to observe the overall kidney and glass slides were used for the microscopic observations of the fluorescent signal of Cu–MT (section thickness: membrane blotting 20 μm; glass slides 5μm). Membrane blotting was processed according to the method of Okabe et al. (1993). Briefly, tissues removed from animals perfused transcardially were frozen and cut with a cryostat microtome. The sections were mounted on dry Zeta probe membranes in a cryostat box and thawed at 25C. The membranes were incubated on filter paper pre-wetted with a 40 mM Tris-HCl buffer, pH 8.0, for 10 min at 4C. After the transfer, the tissues were removed by a high-pressure jet spray. Tissue-mounted glass slides were immersed in 70% ethanol.
The fluorescent emissions of Cu–MT were visualized and photographed by using an Olympus BX-50–FLA (Tokyo, Japan) biological microscope system with U-MWU filter cube (DM-400 dichroic mirror, 330–385-nm excitation filter, 530-nm barrier filter) (Tokyo, Japan) with a 100-W mercury lamp as a
Northern Hybridization
To examine the genomic expression of Cu–MT in the kidney, the distribution of mRNA encoding MT was observed according to the method of Sato et al. (1996). A cDNA fragment of 450 base pairs, including a full length of the rat MT-I coding region, was fractionated by polyacrylamide gel electrophoresis and used as a probe. The coding region of rat MT-I was labeled with DIG-11-dUTP by a random-primed DNA labeling method (Sambrook et al. 1989). Renal sections (20 μm) were blotted onto the nylon membranes by the method of Okabe et al. (1993). The nylon membrane blotted with a renal section was hybridized by the method of Sambrook et al. (1989), and the signals were detected according to the instruction manual for the DIG DNA labeling and detection kit. All molecular biological techniques were performed by the methods of Sambrook et al. (1989).
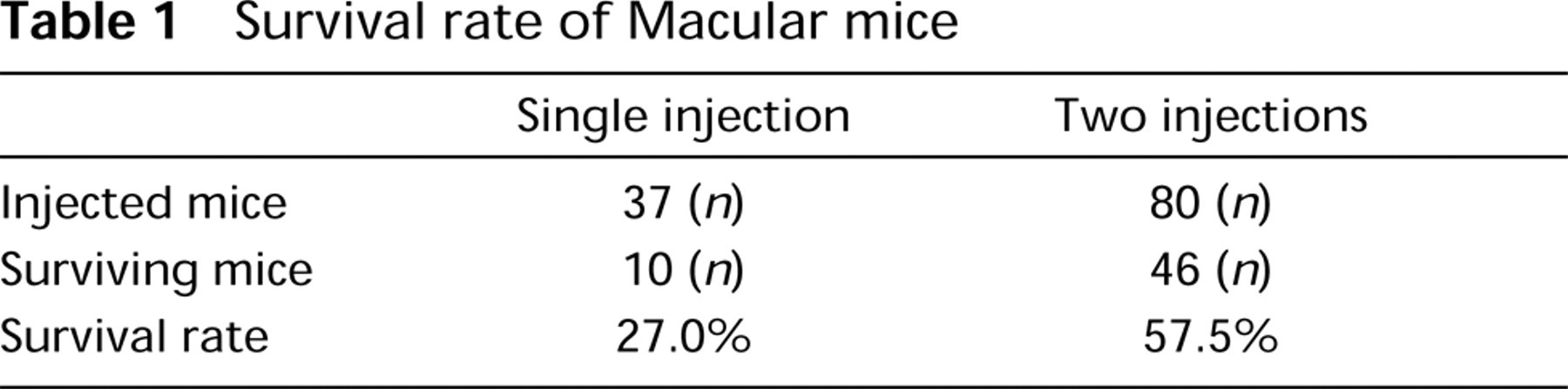
Survival rate of Macular mice
Histochemical Study with Acid Phosphatase
The histochemical distribution of acid phosphatase was investigated by the method of Gossrau et al. (1978), using BCIP as a substrate. The incubation with 0.5 mM BCIP, 0.5 mM NBT, and 45 mM dimethylformamide in 40 mM CH3COOH and 100 mM CH3COONa was carried out at 25C for 60 min.
Results
Treatment of Macular Mice
Because some investigators have reported that a single dose of copper at 7th days after birth permitted the mutant mice to survive, we injected copper (10μg of copper/kg bw) into the mice at Days 7 and 10 after birth and compared their survival rates (Table 1). We found that mutant mice injected twice with copper had higher longterm survival rates than mice that received a single injection. The significant difference of the survival rates between two groups of mice receiving one or two injections was observed (p>0.005) by χ2 study. Other studies used single injections (Mann et al. 1979b; Nagara et al. 1981; Chelly et al. 1993) and two injections (Suzuki and Nagara 1981; Kodama et al. 1993).
Purification of Low Molecular Weight Copper Binding Protein from Kidneys of Macular Mice
Figures 1A and 1B show representative elution patterns of Sephadex G-75 chromatography of the kidney extracts from Macular mice and control mice, respectively. No peak corresponding to the low molecular weight protein (Peak A) in the chromatogram of Macular mice can be detected in the chromatogram of the control mice. The Kd value of Peak A is 0.76, which agreed well with that of MT from the liver of rats reported by Winge and Miklossy (1982). Peak A was directly applied to a column of DEAE-Sephadex A-25 (Figure 1C). Copper binding proteins in the major peak from anion-exchange chromatography were found in the pass-through fraction of Octyl-Sepharose chromatography.
The final purification step of the protein was carried out by FPLC on a reverse-phase ProRPC column (Figure 1D). One copper-containing peak was eluted at a retention time of 8.5 min, the same as the retention time of Cu–MT from the livers of copper-injected rats (Suzuki-Kurasaki et al. 1996).
Amino Acid Composition
Table 2 lists the amino acid composition of the copper binding protein obtained by FPLC. Typically for mammalian MTs, the protein showed a high cysteine content and completely lacked aromatic amino acids and histidine. The composition agreed with that deduced from the cDNA sequence of MT (Huang et al. 1977). The copper binding protein in the Macular mouse kidney was identified as Cu–MT.
Overall Localization of Cu–MT
We examined the overall histochemical localization of Cu–MT in the kidney of Macular mice by direct autofluorescent observation (Okabe et al. 1993; Nakayama et al. 1996) based on fluorescent emissions yielded by Cu+-thiolate clusters in Cu–MT (Beltramini et al. 1987; Stillman et al. 1989). Fluorescent signals were observed in the cortex of the kidney (Figure 2A). The kidney of the control mouse did not exhibit fluorescent signals (Figure 2B). We have proved that the fluorescence is essentially attributable to Cu–MT (Okabe et al. 1993; Nakayama et al. 1996), although the specificity of the histochemical detection of copper or MT by other several analytical methods has been conflicting (Elmes et al. 1987; Williams et al. 1989; Schmid et al. 1993).
Expression of MT mRNA
The expression of MT mRNA was investigated to determine whether Cu–MT in the cortex was biosynthe-sized in the kidney or transported from other tissues (Figures 2C and 2D). A high level of mRNA was expressed throughout the renal cortex. The expression of MT mRNA was not detected in the renal medulla and pelvis of the mutant mice. The kidneys of the control mice did not exhibit expression of MT mRNA. Both MT mRNA and its transcripts were detected in the renal cortex of Macular mice, indicating that biosynthesis of MT occurred in this region.
Microscopic Observation of Fluorescence from Cu–MT
To determine the detailed localization of Cu–MT in the kidney, microscopic observation was carried out. In the Macular mice, yellow fluorescence was observed exclusively in the proximal convoluted tubules (PCT) of the cortex (Figure 2E). The renal proximal tubules are the most sensitive sites in kidneys exposed to large doses of heavy metals (Dorian et al. 1992). No fluorescence was observed in the glomeruli, Bowman's capsules, and the distal tubules (data not shown). Fluorescent emissions were not observed in the renal cortex (Figure 2F), medulla, and pelvis (data not shown) of the control mouse, even by microscopic observation.
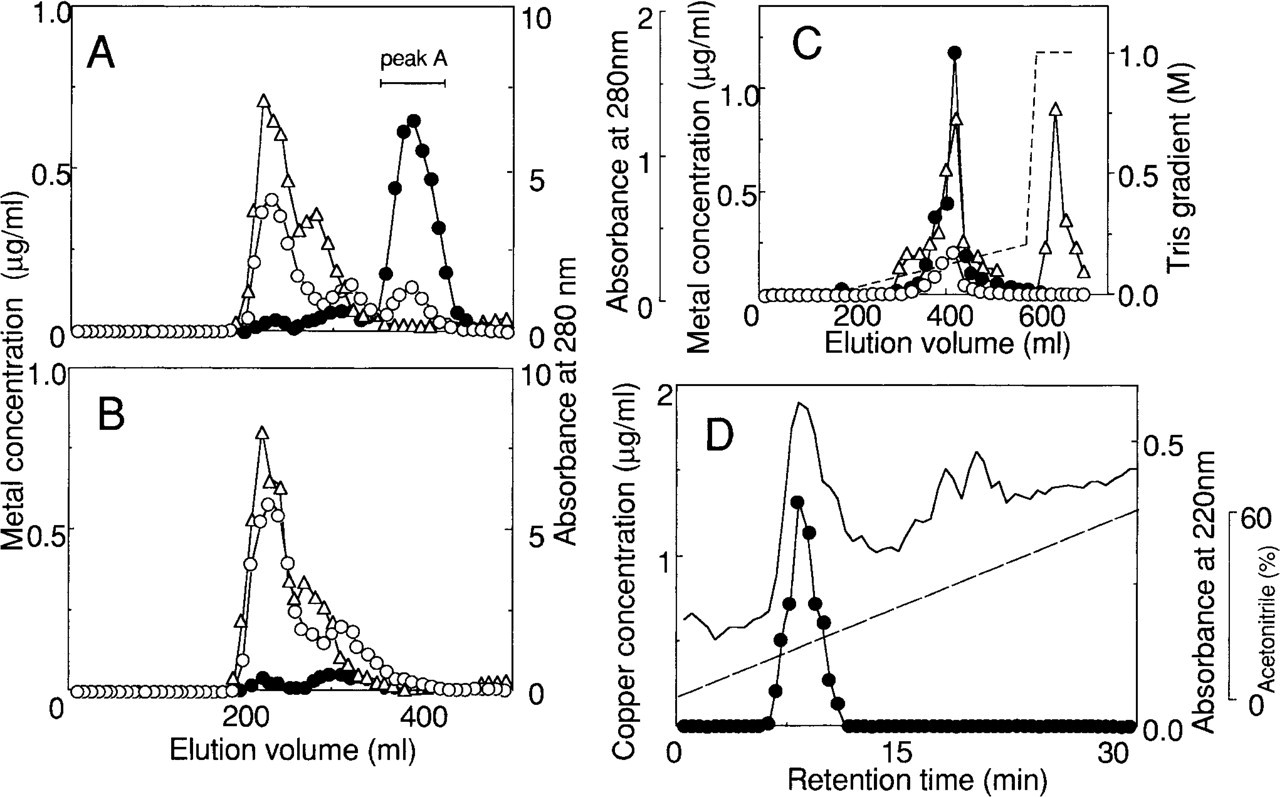
Sephadex G-75 chromatography of the renal cytosol of Macular mice
To clarify the subcellular distribution of Cu–MT in the PCT cells, the mechanism of the renal damage caused by copper was investigated. Strong emission signals of spherical granules (Figures 2G and 2H) indicated that Cu–MT was concentrated in certain organelles, such as the lysosomes. Emissions from Cu–MT appeared also in the cytoplasm (Figure 2G) and in some nuclei of the PCT cells (Figure 2H). It has been reported that MT bound heavy metals present in nuclei (Sakurai et al. 1993; Tohyama et al. 1993; Cherian 1994; Suzuki et al. 1994).
In the quenching test of the blot, the fluorescent emission disappeared after soaking in 1 mM HgCl2 (data not shown) as we described previously (Okabe et al. 1996; Nakayama et al. 1996). Therefore, the specificity of the fluorescent detection of Cu–MT was verified.
Localization of Acid Phosphatase
To confirm that Cu–MT exists mainly in the lysosomes, we investigated the histochemical localization of acid phosphatase in the PCT cells. This enzyme is recognized as a histochemical marker for lysosomes (Gossrau 1978). Yellow signals of Cu–MT (white arrows in Figure 2J) showed acid phosphatase activity (black arrows in Figure 2I) in the same section of the kidney, suggesting that Cu–MT is highly concentrated in the lysosomes.
Microscopic Observation of Fluorescence from Cu–MT in LEC Rat Kidney
LEC rats are a model for Wilson's disease. Copper and Cu–MT were abnormally accumulated in LEC kidneys. To compare the localization of the fluorescence from Cu–MT in Macular mouse kidney with that in LEC rat kidney, microscopic observation of the fluorescence from Cu–MT was carried out. Orange autofluorescence from Cu–MT was detected in the outer stripe of outer medulla and yellow signals from Cu–MT were observed in the PCT cells of the renal cortex of LEC rat (Figure 2K) as previously described (Okabe et al. 1996).
Amino acid composition of Cu-MT from the Macular mouse kidney compared with the composition deduced from the cDNA sequence of mouse MT-I (Schmid et al. 1993) a
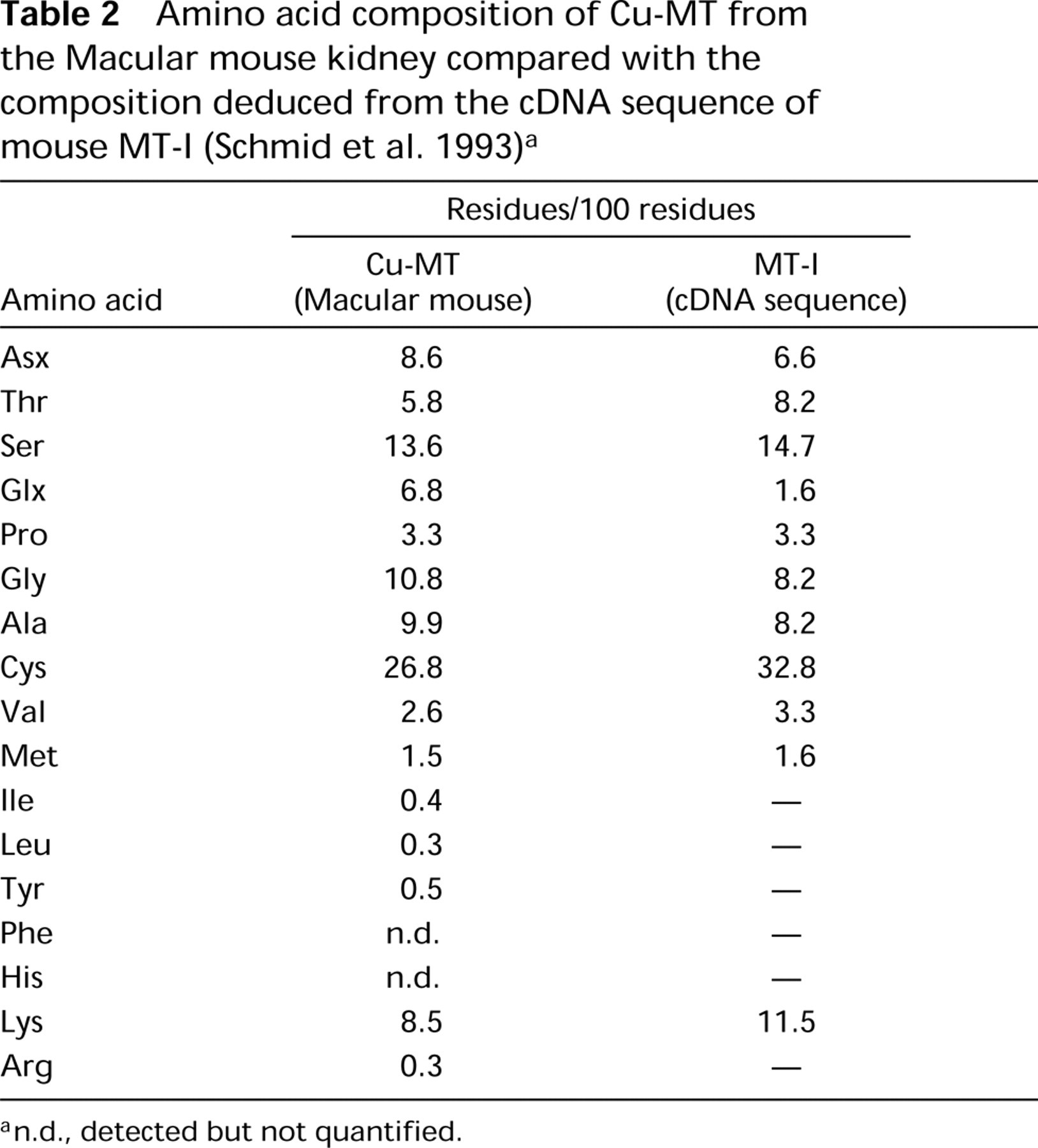
a n.d., detected but not quantified.
Discussion
Menkes disease is a disorder of copper metabolism (Menkes 1962). Accumulation of abnormal amounts of copper in the kidney and intestinal wall of patients and in mouse models of disease is a significant feature. This study showed that excess amounts of copper were present as Cu–MT in the kidney of Macular mice. The properties of Cu–MT autofluorescence enabled us to observe the histochemical localization of the protein directly, as previously described (Nakayama et al. 1996; Okabe et al. 1996). We verified that the fluorescent emission obtained was from Cu–MT. In the kidney of Macular mice, Cu–MT was found in the lysosomes, cytoplasm, and nuclei of the PCT cells in the cortex. Because both Cu–MT and MT mRNA were observed in the cortex, these data suggest that Cu–MT in the cortex is biosynthesized in this region.
There have been few reports on the direct identification of Cu–MT from the tissues of Menkes patients or from model animals for Menkes disease. Many investigators have believed that the low molecular weight protein associated with Menkes disease is Cu–MT because of its features, e. g., the metal (Chan et al. 1978; Onishi et al. 1981) and cysteine contents (Prins et al. 1981; Crane and Hunt 1983). Our study confirmed the view of earlier studies that Cu–MT is present in Menkes patients and in animal models for Menkes disease. We successfully purified a copper binding protein from the model mice by a method established previously (Suzuki–Kurasaki et al. 1996) and identified it as Cu–MT.
Microscopic studies of the detailed localization of Cu–MT showed that it is dominant in the PCT cells of the cortex in Macular mice (Figure 2E). It has been reported that after copper administration Cu–MT accumulates mainly in the renal PCT cells (Williams et al. 1989; Evering et al. 1990; Schmid et al. 1993). The physiological significance of this result is noteworthy, because Foulkes et al. (1978) reported that Cd–MT bound to the brush border of the PCT cells after glomerular filtration. PCT cells are considered to be the primary site of the nephrotoxicity caused by heavy metals, such as cadmium (Foulkes 1978; Onishi et al. 1981).
The subcellular distribution of Cu–MT in the PCT cells of Macular mice showed a large concentration in lysosomes (Figure 2E). This result coincides with the evidence of Evering et al. (1990) that Cu–MT is incorporated into the lysosomes in the copper-loaded rat kidney. Both the cytosol and nuclei of the PCT cells of Macular mice showed emissions from Cu–MT (Figures 2G and 2H). Because Cu–MT diffuses in the cytoplasm, the emission intensity in the cytoplasm could be relatively sparse. We confirmed that the cytoplasm contained Cu–MT because we purified Cu–MT from the cytoplasm of the kidneys of Macular mice. The localization of Cu–MT in Macular mouse kidney is similar to that observed in the copper-loaded rat kidney (Evering et al. 1990). Kodama et al. (1993) demonstrated that copper ions in the PCT cells of Macular mice were found in the organelle-free cytoplasm. The distribution of copper ions in the PCT cells of Macular mice did not correspond to the distribution of Cu–MT, which is consistent with the finding of another study that there is some difference in the histochemical behavior of free copper ions and the copper of Cu–MT (Schmid et al. 1993).
Some studies (Squibb et al. 1979; Cain and Holt 1983; Dorian et al. 1992) found that cadmium ions were released from Cd–MT intracellularly by the lysosomal degradation. Therefore, it is believed that Cu–MT in the PCT cells of Macular mice is involved in lysosomal degradation. The significance of the histochemical localization of Cu–MT in Macular mice is not completely clear, but our study on the mRNA expression extends the notion that the biosynthesis and degradation of Cu–MT occur repeatedly by lysosomal degradation. Both Cu–MT and MT mRNA were found in the renal cortex of the Macular mouse, suggesting that Cu–MT is continually synthesized in this region. That is, free copper ions are released from Cu–MT by lysosomal degradation in the PCT cells and are reabsorbed by these cells, especially in the S1 and S2 segments. The released copper ions induce MT mRNA to newly synthesize the protein. However, the newly synthesized Cu–MT was also degraded in lysosomes in the same region. Trace amounts of yellow fluorescence from Cu–MT were observed in the S3 segment of the proximal straight tubules (PSTs), because copper ions are scarcely reabsorbed in the S3 segment owing to the axial heterogeneity of the reabsorption system in the proximal tubules. Our observations suggest that Cu–MT and/or released copper ions were accumulated in the PCT cells. The consideration is supported by the theory of a lysosomal system for Cd–MT described by Nordberg and Nordberg (1987).
Whereas orange fluorescent emissions were observed in the S3 segment in the kidney of LEC rats (Figure 2K), yellow fluorescence appeared in the PCT cells of Macular mice. Emissions of a model complex of Cu–MT changed from orange to yellow according to a decrease in the molar ratio of copper to thiol (Elmes et al. 1987). We believe that the shortwave yellow signals from the protein indicate the oxidation of Cu–MT and that the release of copper ions from the protein results in a decrease in the ratio of copper to thiol. Yellow fluorescence suggests that the protein is in the degradation process.
LEC rats are an inbred strain. These animals exhibit an abnormal accumulation of copper in tissues and die because of the hepatotoxicity induced by copper accumulation; (Li et al. 1991a,b; Okayasu et al. 1992). It is interesting to compare the histochemical distribution of Cu–MT in Macular mice with that in LEC rats. As shown in Figure 2K, orange autofluorescence from Cu–MT was observed in the outer stripe of the outer renal medulla and yellow signals were detected in the PCT cells of the renal cortex of the LEC rat. The change in emission color was assumed to be caused by the partial release of copper ions, suggesting oxidation of Cu–MT in lysosomes in the LEC renal cortex. A high level of MT mRNA was expressed in the outer stripe of the outer renal medulla but not in the cortex (Okabe et al. 1996; and data not shown), unlike in the Macular mouse. These results suggest that the Cu–MT in the outer stripe of the outer renal medulla of the LEC rat is biosynthesized in this region (Okabe et al. 1996). The Cu–MT observed in the renal cortex of the LEC rat has been believed to be transported from other organs by a certain mechanism, to be taken up by the PCT cells after glomerular filtration, and to be degraded in lysosome in the region (Okabe et al. 1996). In our study, the expression of mRNA in the renal cortex of the Macular mouse suggests that the Cu–MT in the cortex is biosynthe-sized and that copper, but not Cu–MT, is transported by a certain carrier, such as albumin in the bloodstream, to the kidney. Copper ions released from the Cu–MT in the renal cortex of the LEC rat were believed to induce Cu–MT in the outer stripe of the outer renal medulla (Okabe et al. 1996). However, in Macular mice Cu–MT was not found in the outer stripe of the outer renal medulla, suggesting that the copper ions released from the Cu–MT in the cortex did not induce Cu–MT in the outer stripe of the outer renal medulla. These observations supported the consideration that a lysosomal cycle releasing copper ions from Cu–MT repeatedly contributes to the synthesis of Cu–MT in the cortex.
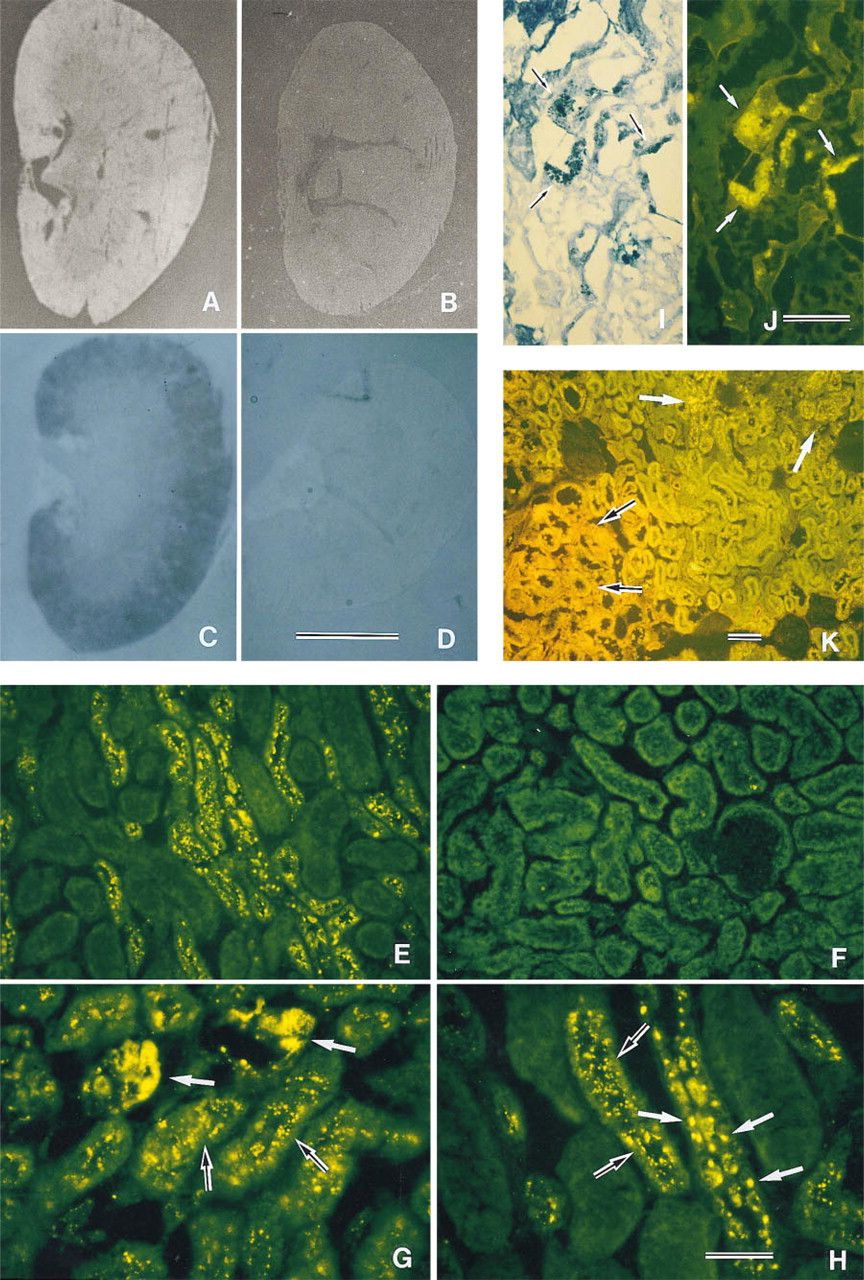
Histochemical distribution of Cu–MT. Renal sections from Macular mice
The apparent discrepancy in copper-transporting systems between Menkes disease and Wilson's disease may be due to the candidate genes for these conditions. Two types of P-type cation-transporting ATPases encoded by an x chromosome-linked gene have been reported to be concerned with copper metabolism: one is a copper-efflux pump (ATP7A) (Camakaris et al. 1995), which is a candidate gene for Menkes disease (Chelly et al. 1993; Mercer et al. 1993; Vulpe et al. 1993), and the other (ATP7B) is an enzyme suggested to be responsible for Wilson's disease (Bull et al. 1993; Petrukhin et al. 1993). These two distinct P-type cation-transporting ATPases showed a 60% homology of their amino acid sequences. In normal organisms, it is believed that copper ions are reabsorbed into PCT cells and that the reabsorbed copper ions are then released into the veins. In the kidney of Macular mice, it was postulated that the efflux function of the ATP7A was subject to a hereditary form of damage and that the copper was accumulated in the PCT cells in the renal cortex. Then, the copper ions bound to Cu–MT repeatedly contributed to the synthesis of Cu–MT in the cortex. Recently, ATP7A was reported to be present in the Golgi apparatus (Petris et al. 1996; Yamaguchi et al. 1996) and was continuously recycled between the Golgi and the plasma membrane (Petris et al. 1996). The lysosomal recycling of Cu–MT in the renal cortex of the Macular mouse might be caused by a dysfunction of ATP7A in the Golgi apparatus.
We have identified Cu–MT in the kidney of Macular mice, and we suggest that Cu–MT is biosynthe-sized in the renal cortex. Localization of Cu–MT in the kidney of these mice has been also confirmed. These findings, including the lysosomal cycle postulated here, give some clues to future studies on the mechanism of the abnormal copper accumulation in Menkes disease.
Footnotes
Acknowledgements
We are grateful to Dr M. Nishimura, Institute for Experimental Animals, Hamamatsu University of Medicine, Hamamatsu, Japan, who provided us with a breeding pair of Macular mice, and to Prof Yutaka Kojima, Graduate School of Environmental Earth Science, Hokkaido University, for suggestions and discussion during this study.
