Abstract
Differences between benign prostatic hyperplasia (BPH) and normal prostate tissue at the level of mRNA expression provide an opportunity to identify candidate genes for this disease. A cDNA subtraction procedure was used to isolate differentially expressed genes in BPH. The subtraction was done by solution hybridization of BPH cDNA against excess normal prostate cDNA. We identified known, EST, and novel genes by sequence and database analysis of the subtracted cDNAs. Several of these cDNAs were used as probes in Northern blotting analysis to confirm over-expression of their corresponding mRNAs in BPH tissues. One highly upregulated sequence of interest shared identity with a known mRNA encoding human NELL2, a protein containing epidermal growth factor-like domains. NELL2 was not previously reported to be expressed in prostate and may code for a novel prostatic growth factor. In situ hybridization analysis of hyperplastic prostate specimens demonstrated that NELL2 mRNA expression is predominantly localized in basal cells of the epithelium. Disease-related changes in the levels of NELL2 may contribute to alterations in epithelial–stromal homeostasis in BPH. (
B
Prostate tissue was obtained after written consent from each patient and was approved by the local ethics committee. mRNA isolated from BPH and normal prostate mRNA was subjected to a subtractive and kinetic enrichment procedure (Hubank and Schatz 1994). For in situ hybridization (ISH), digoxigenin-labeled probes were hybridized to transurethral resectioned prostate (TURP) specimens and detected using the tyramide signal amplification method and DAB staining (NEN Life Sciences; Boston, MA).
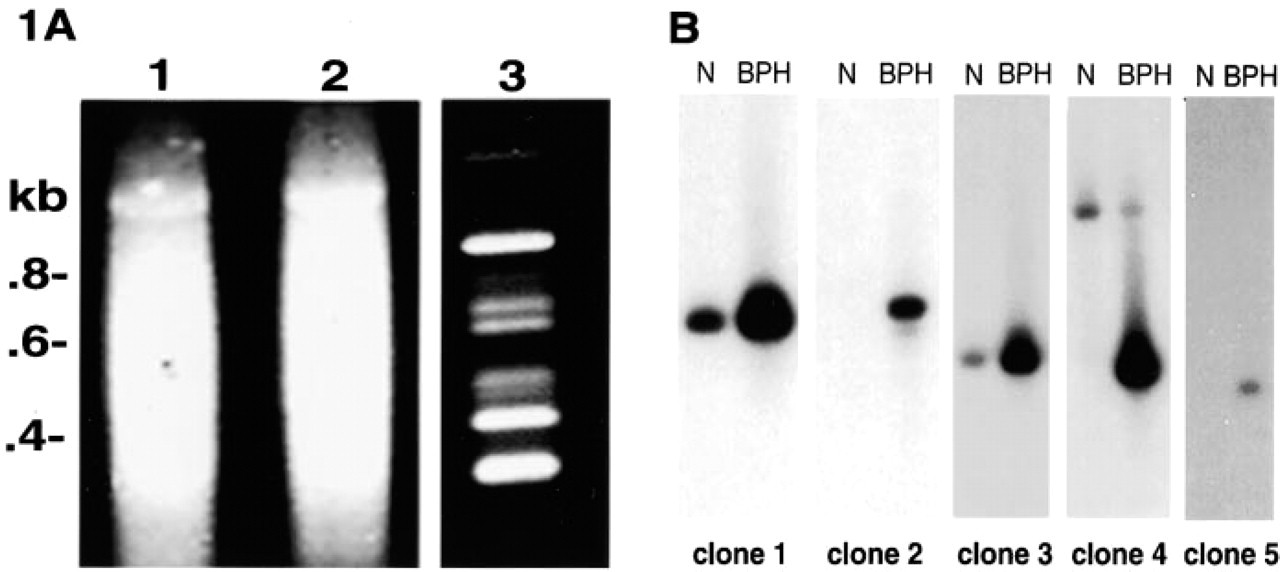
Before subtraction, cDNA populations from normal and BPH tissue were indistinguishable (Figure 1A, Lanes 1 and 2, respectively). By an iterative process of subtractive and kinetic enrichment, cDNA fragments that were upregulated in BPH were selected (Figure 1A, Lane 3). Sequence and database analysis of these fragments identified at least 15 known, EST, and novel genes. Five clones were hybridized to Northern blots to verify expression patterns. Figure 1B shows the detection of highly upregulated mRNA in BPH (clones 1 and 3) in addition to mRNA specific for BPH (clones 2, 4, and 5) compared to normal prostate.
Sequences representative of clones 1 and 3 were further analyzed and revealed identity to lactoferrin and NELL2, respectively. Lactoferrin is an iron-binding glycoprotein expressed in inflamed prostate (Reese et al. 1992) and may play a role in providing protection against tumor development (Damiens et al. 1998). Lactoferrin is expressed in the secretory component of the epithelium in TURP specimens (Figure 2A), and its upregulation in BPH may be a response to neutralize the enhanced proliferative activity of this disease. NELL2 is a neuronal protein containing EGF-like domains with similarities to thrombospondin, and has not previously been identified in prostate. Kuroda et al. (1999) recently showed abundant expression of NELL2 mRNA in colorectal adenocarcinoma SW480 cells, suggesting that NELL2 participates in the growth and differentiation of cancer cell lines. NELL2 mRNA is predominantly expressed in the epithelial basal cell layer in TURP specimens (Figure 2B). EGF is also abundantly expressed in the basal cells of BPH and may overstimulate epithelial cell proliferation (De Miguel et al. 1999). Altered expression of other growth factors has been implicated in the disruption of molecular mechanisms that regulate cell death and proliferation in the aging and diseased prostate (Gomella and Godwin 1997). We have identified NELL2 as a putative growth factor regulator of prostate epithelial cells. When upregulated, this protein may act synergistically with other growth factors contributing to alterations in epithelial–stromal homeostasis in BPH. Analysis of prostate specimens from an expanded patient population is in progress.
(
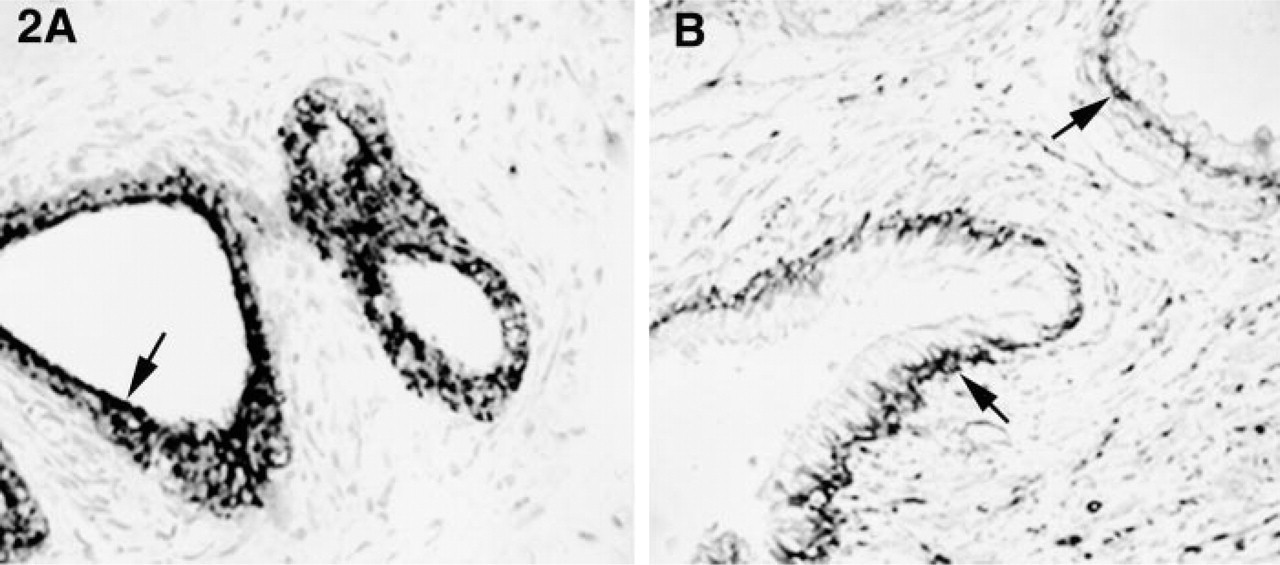
ISH of TURP specimens with antisense probes for lactoferrin (
