Abstract
Signal amplification in immunohistochemistry via binding of biotinylated tyramine to proteins near the site of peroxidase-labeled antibodies is a promising new technique, but studies investigating a wide range of markers are lacking. The tyramine amplification technique (TAT) was investigated on 85 antibodies using a simple and fast protocol, and TAT results were compared to those obtained with conventional immunohistochemistry. Using TAT, most of the markers could be 5- to 50-fold further diluted and still showed identical staining results compared with standard stainings (maximal 500-fold). However, the variable reactivity of the different markers with TAT underlines the need for individual testing of every antibody to determine the optimal dilution. Some antibodies against cell adhesion molecules could be demonstrated for the first time in archival, formalin-fixed tissue sections. TAT, if carefully evaluated, offers a revolutionary improvement for modern immunostaining, either to increase sensitivity or primary antibody dilutions (cost reduction). From a methodological point of view, immunohistochemistry has not reached its limits by far and TAT is an important progressive step in this developmental process.
I
Today, individual reports about the tyramine amplification technique (TAT) indicate that this is a further important development in IHC, promising to improve the sensitivity and extend the possible applications of IHC to new limits.
This amplification technique is based on the characteristic ability of tyramine to become “sticky” (chemically) after oxidation/radicalization (Gross and Sizer 1959). The respective epitope of interest is marked with peroxidase enzymes via specific antibodies, and the labeled tissue is then incubated with biotinylated tyramine and H2O2. The peroxidase enzyme catalyzes a reaction resulting in “radicalized” tyramine, which then binds covalently to nearby tissue molecules. Finally, the biotin on the bound tyramine serves as a tracer molecule that can easily be visualized by standard ABC techniques (avidin–biotin–enzyme complex).
Investigators began taking advantage of this tyramine effect for their procedures a few years ago, but the potential of this amplification technique is far from being fully recognized. Initially used for enhancing ELISA and Western blotting, it has now been adapted for immunohistochemistry and in situ hybridization (Bobrow et al. 1989, 1991, 1992; Adams 1992; Kerstens et al. 1995; Merz et al. 1995; de Haas et al. 1996). However, no reliable data are currently available for a broad range of commonly used antibodies although this technique may be interesting for many laboratories: because of increased sensitivity, antibody dilutions (cost reduction), and the number of antibodies applicable on paraffin sections.
Alphabetical list of antibodies tested with tyramine amplification technique (TAT) a
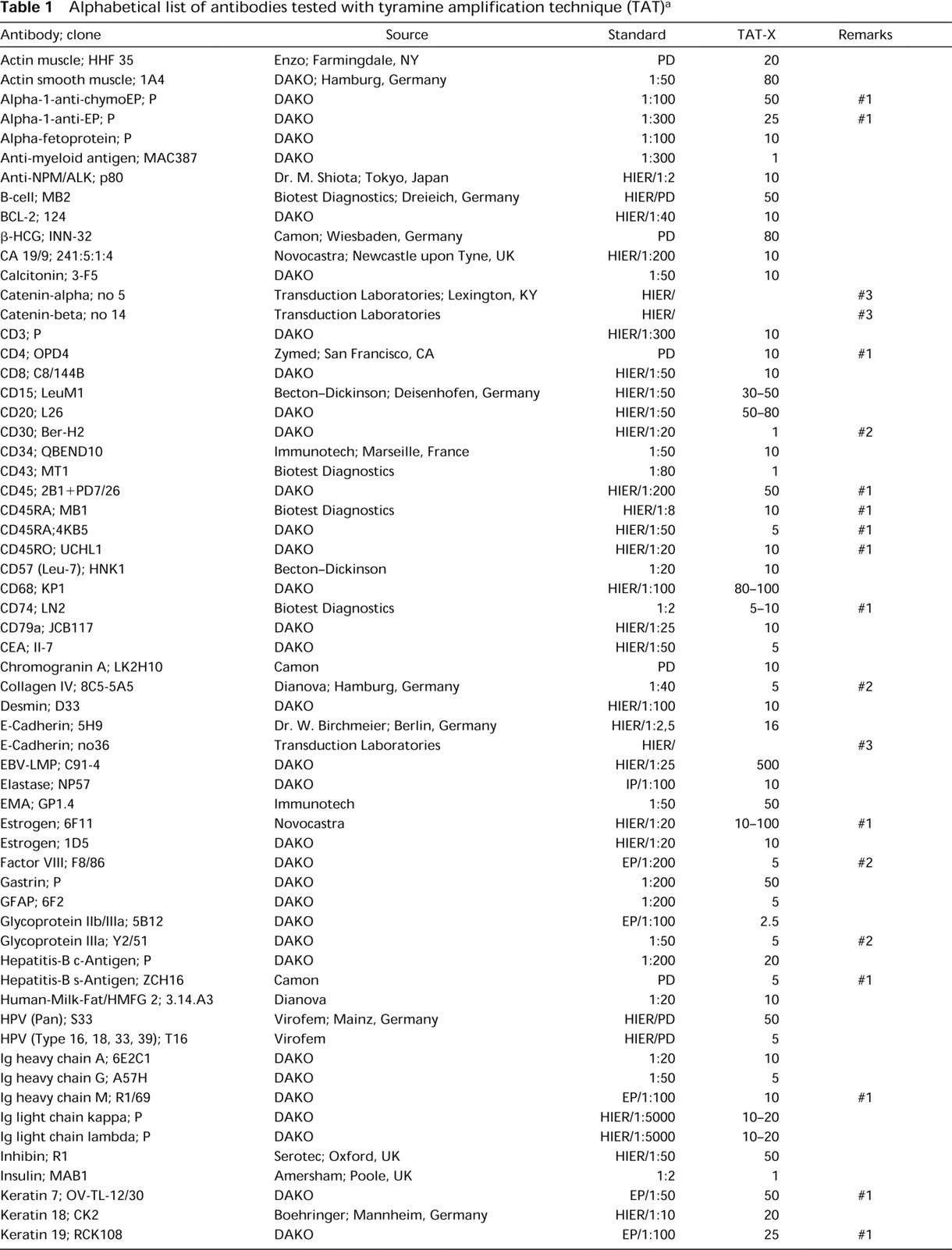
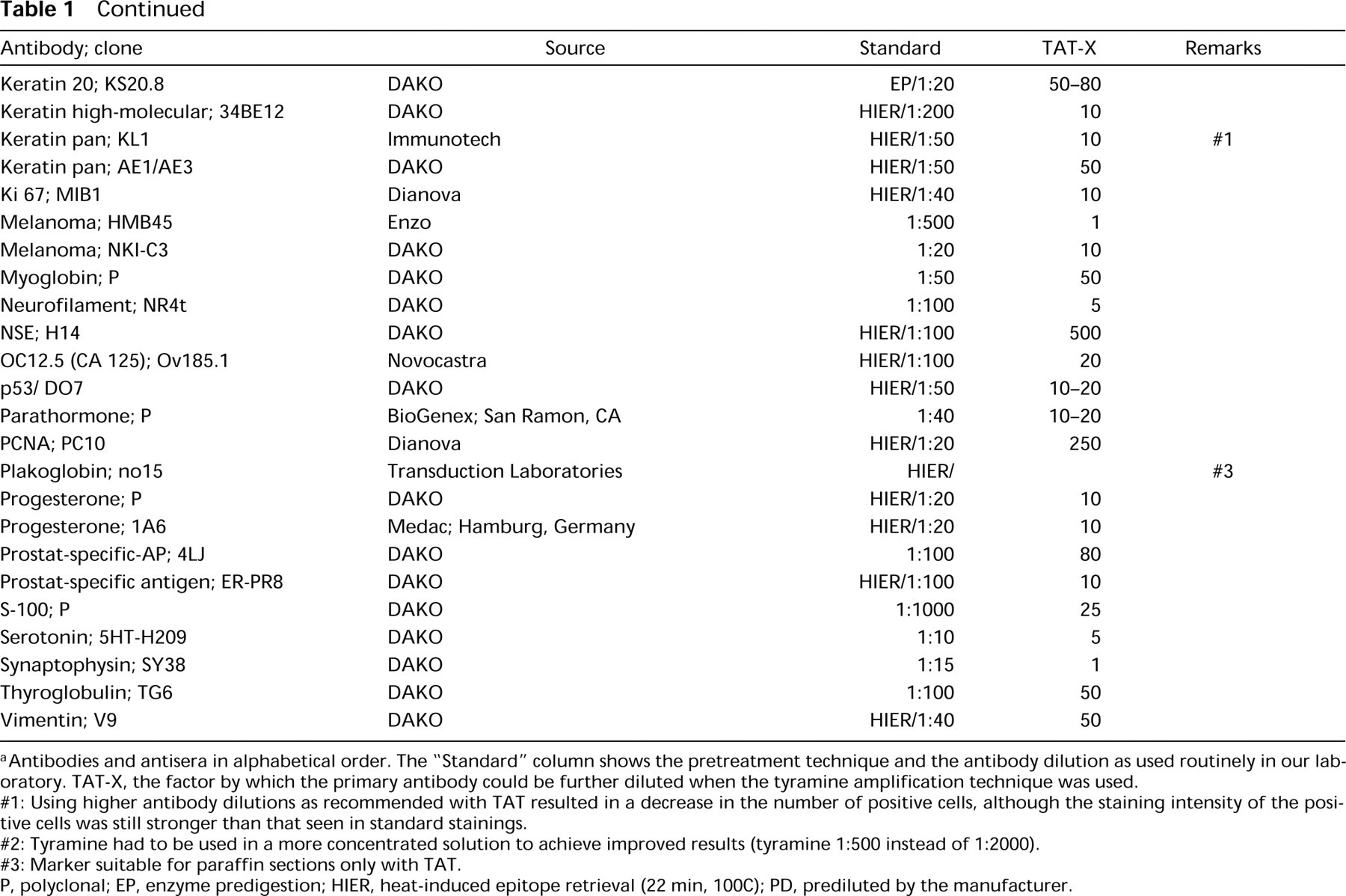
aAntibodies and antisera in alphabetical order. The “Standard” column shows the pretreatment technique and the antibody dilution as used routinely in our laboratory. TAT-X, the factor by which the primary antibody could be further diluted when the tyramine amplification technique was used.
#1: Using higher antibody dilutions as recommended with TAT resulted in a decrease in the number of positive cells, although the staining intensity of the positive cells was still stronger than that seen in standard stainings.
#2: Tyramine had to be used in a more concentrated solution to achieve improved results (tyramine 1:500 instead of 1:2000).
#3: Marker suitable for paraffin sections only with TAT.
P, polyclonal; EP, enzyme predigestion; HIER, heat-induced epitope retrieval (22 min, 100C); PD, prediluted by the manufacturer.
Therefore, the aim of this study was to evaluate the effects of the new TAT with a panel of monoclonal and polyclonal antibodies that are commonly used by many pathologists. The question to answer was whether the additional steps in the staining protocol and the additional costs, either for purchasing ready-to-use kits for tyramine signal amplification or for biotinylating tyramine by oneself, truly offer an improvement for the daily routine. Because almost identical procedures for signal amplification using tyramine have been named differently by different authors and commerical suppliers [CARD, TSA, CSA, Immuno-Max: (Bobrow et al. 1989); DuPont, Dordrecht, Netherlands; DAKO, Hamburg, Germany; (Merz et al. 1995)], we prefer the neutral abbreviation “TAT.”
Materials and Methods
Biotinylation of tyramine was done according to the protocol of Adams (1992). In brief, tyramine hydrochloride (6.2 mg; Sigma, Deisenhofen, Germany, T-2879) was diluted in 50 mM borate buffer (8 ml, pH 8.0), containing sulfo-NHS-LC-Biotin (20 mg; Pierce, Rockford, IL, 21335), and biotinylation was performed overnight under continuous stirring. The solution was filtered (0.45 syringe filter), and 5-μl aliquots were stored (−70C) until use.
Routinely fixed paraffin-embedded archival tissues were cut, mounted on poly-
Slides were immunostained either without pretreatment, with enzyme digestion, or with HIER as indicated in Table 1. Epitope retrieval was performed by boiling the slides in monohydrated citrate buffer (pH 6.0, 0.01 M) in a microwave oven (100C, 22 min).
For comparison of detection methods, the respective preteatment was simultaneously done for both the standard slides and the slides to be stained by TAT, and all stainings for a given marker were simultaneously performed the same day using identical solutions of primary antibodies (standard dilution), detection system, and substrate (new fuchsin). Therefore, any variations caused by factors other than the use or omission of TAT were excluded.
Stainings with TAT were done using at least five different antibody dilutions: standard (normal dilution used without TAT) and a 5 x, 10 x, 50 x, 100 x higher dilution. If the results were not conclusive, further dilutions were tested.
For stainings (all incubations with 100 μl per slide at 37C), tissue sections were blocked with 3% H2O2 (10 min), drained, blocked against endogenous biotin activity with TNB 0.5% (DuPont; Wilmington, DE) (15 min) and drained. After incubation with primary antibody (60 min), biotinylated secondary antibody (DAKO, Hamburg, Germany; monoclonal E0354, polyclonal E0432; 30 min) and avidin-horseradish peroxidase (DAKO P0397; 30 min), the tyramine solution, 1:2000 in 0.3% H2O2/TBS (5 min) was applied. Finally, incubations with avidin–biotin–anti-AP complex (DAKO K391; 30 min) and substrate (new fuchsin; 30 min) were performed, followed by brief hemalum counterstaining. Slides were washed in TBS after each incubation step.
Staining intensity, number of specific cells marked, staining pattern (nuclear, cytoplasmic, membranous), and background signal (unspecific binding in surrounding tissue) were noted. The highest possible dilution of primary antibody using TAT showing results identical to those of the standard staining was evaluated. For some of the markers, it is more realistic to give a range rather than a single “best dilution.” This applies either when the difference in staining results between two dilutions tested was too small to be distinguished reliably or when in different cases two different dilutions showed optimal results. To achieve reliable results, at least four routinely fixed cases were tested with each marker (average <20 cases/marker).
Results
A total of 85 markers, either monoclonal (n = 73) or polyclonal (n = 12) were included in this methodological study. For the majority of markers, IHC using TAT showed identical staining results but allowed at least a fivefold higher dilution of primary antibodies (74/85; 87%). The factor by which antibodies could be further diluted varied between 2.5-fold (GP IIb/IIIa) and 500-fold (NSE, EBV-LMP) (Table 1).
The staining signal was almost always considerably enhanced by TAT. TAT produced either a strong signal or showed no signal at all; no different gradations in intensity could be observed. This is in contrast to conventional IHC, in which an increase in primary antibody dilution often results in a slow decrease in signal intensity.
No unexpected new positivity or implausible staining signals were observed in this study. If the dilution of particular primary antibody exceeded a certain level (with TAT), the number of cells positive for the respective antibody decreased (Table 1, remark #1). In those cases, the staining pattern changed so that previously positive cells became negative while the majority of the other cells were still strongly positive. One group of antibodies (collagen IV, GPIIIa, factor VIII, and CD30; Table 1, remark #2) showed peculiar staining results with our TAT protocol described in Materials and Methods. Either partial or total false-negative staining results were observed. When collagen IV was tested in renal tissue, strong signals in the basement membranes of the capillaries were detected, whereas the basement membrane of the Bowman's capsule and of the tubuli showed no positivity. This was in contrast to results seen with standard stainings. GPIIIa and factor VIII, when tested on bone marrow biopsies, exhibited a strong signal in the endothelium of small vessels, but no positivity was seen in the megakaryocytes. For CD30 (BerH2), no signal was observed in several cases of Hodgkin's disease, whereas strong signals were found in the diagnostic giant cells of the same case if stainings were performed conventionally.
For these markers, all stainings were repeated twice, showing identical results. Therefore, a thorough checkerboard testing using higher and lower tyramine dilutions (between 1 : 100 and 1 : 10,000; standard 1 : 2000) and antibody dilutions (between 0.1- and 1000-fold) was performed (data not shown).
The best results were achieved only when tyramine was used in a more concentrated dilution (1 : 500), then allowing a fivefold increase in antibody dilution. Using this modified concentration of tyramine, a typical staining pattern was observed with no false-negativity.
Those antibodies previously described as being unsuitable in paraffin sections are marked in Table 1 (remark #3).
Discussion
Table 1 gives the most complete overview available to date of the beneficial effects of TAT on commonly applied antibodies in routine pathology. Most markers could be diluted between 5- and 50-fold higher than before, results that are in good accordance with previous reports on single markers (Bobrow et al. 1989). In our protocol, visualization was performed with alkaline phosphatase. The enzymatic activity of the peroxidase enzymes already bound on the secondary antibody was not used for substrate conversion and color development. Therefore, the signals obtained in this study are achieved solely via tyramine detection. Furthermore, the results were compared to standard stainings using identical pretreatment (e.g., enzymatic predigestion or HIER). Our enhancement factors evaluated on routine archival tissues are lower than those of a recent study that investigated only a few antibodies (Merz et al. 1995). However, this published report allows no clear-cut distinction between enhancement effects resulting from epitope retrieval and those due to tyramine amplification.
The great differences in enhancement between the different markers seen here are difficult to explain. The composition of the surrounding tissue may affect the deposition of activated tyramine because this substance is known to react with electron-rich moieties on the tissue surface (i.e., tyrosine, tryptophan) (Bobrow et al. 1989). Our protocol and antibody dilutions are intended as a starting point for other investigators. Because of differences in tissue processing, fixation, and staining systems, including the use of commerically available biotinylated tyramine, results may differ from those of this study. Therefore, individual modifications of the protocol may be beneficial for others to achieve optimal results. However, in our hands the protocol described here offered the best compromise between simplicity, reliability, and beneficial increase in sensitivity (or antibody dilution). For even more increased sensitivity, additional modifications in the TAT protocol may be useful, e.g., repetition of tyramine incubation and/or prolongation of the tyramine incubation period through the use of highly viscous diluents (Merz et al. 1995; van Gijlswijk et al. 1996).
At rather high antibody dilutions, some of the markers showed a paradoxical effect when TAT was used. Although most cells still displayed a stronger staining signal compared to standard stainings, a proportion of cells became totally negative, i.e., the number of positive cells decreased in a given tissue. This observation may be important for markers in which percentages of positivity are decisive for the evaluation, e.g., proliferation markers. Antibodies showing this phenomenon are marked in Table 1, and dilutions are recommended where this effect was not observed. Some antibodies were enhanced only when a more concentrated tyramine solution was used, as indicated in Table 1 (1:500 instead of 1:2000). Although the underlying reason remains obscure, this methodological modification allowed a further increase in the signal amplification rate. Therefore, the concentration of tyramine may serve as an additional mechanism by which the sensitivity can be further increased, as mentioned above. Detailed studies are needed to determine if all these additional refinements in technique are truly useful in general or if they are too complicated or time-consuming for routine purposes. Our protocol and recommended dilutions (Table 1) showed reliable results, and the technique was fast and easy to perform. TAT is an important progressive step in the developmental process of IHC, a technique that has not reached its limits thus far. The next milestone awaits us.
