Abstract
Θ-Toxin (perfringolysin O), a cholesterol-binding toxin, was partially proteolyzed and biotinylated (BCΘ) to eliminate hemolyzing activity and was used as a cytochemical probe. In fixed cells, binding of BCΘ was intense in the plasma membrane, especially at the base of apical microvilli and in lateral processes. The labeling was abolished by pretreatment with filipin, digitonin, or tomatin. When living cultured cells were treated with BCΘ and then with either fluorescein–avidin D or colloidal gold–streptavidin, the labeling in fine dots was distributed on the cell surface without local concentration as long as cells were kept on ice. When the temperature was raised to 37C after treatment, the probe formed discrete large patches and became sequestered to caveolae. Binding of BCΘ alone without the secondary reagents did not cause redistribution even at 37C. Because the plasma membrane maintains integrity even after binding of BCΘ, the probe can be used not only for cytochemical labeling of fixed cells but for pursuing the behavior of crosslinked cholesterol molecules in living cells. By use of this new probe, the present study revealed that crosslinked cholesterol in the plasma membrane is sequestered to caveolae.
C
Relative enrichment of cholesterol in caveolae compared to non-caveolar portions of the plasma membrane has been believed to bring about the above results. Two lines of studies have been cited to support the assumption. First, a morphological study showed that a ring of filipin–sterol complexes is preferentially formed at the orifices of caveolae (Simionescu et al. 1983). Second, biochemical studies revealed that a detergent-insoluble complex obtained by sucrose density-gradient centrifugation is enriched with caveolin (Sargiacomo et al. 1993) and contains a high proportion of cholesterol (Brown and Rose 1992). However, for the filipin cytochemistry, a later study could not show a preferential formation of filipin–sterol complexes in caveolae (Simons and Severs 1986). Furthermore, it has been questioned whether the detergent-insoluble complex represents purified caveolae (Fra et al. 1994; Gorodinsky and Harris 1995; Schnitzer et al. 1995; for review see Kurzchalia et al. 1995).
Recently, we showed by immunocytochemistry that antibodies to glycolipids and sphingomyelin bind to the cell surface without local concentration and are sequestered to caveolae when they are crosslinked with secondary antibodies (Fujimoto 1996). Glycolipids and sphingomyelin were reported to be highly concentrated in the detergent-insoluble complex (Brown and Rose 1992). These results suggest that detergent treatment and crosslinking by antibodies may exert similar effects on the lipid molecules and make them accumulate to caveolae. It would therefore be reasonable to assume that cholesterol, another lipid molecule enriched in the detergent-insoluble complex, is also sequestered to caveolae when crosslinked with specific ligands.
Several probes have been used for cytochemical labeling of cholesterol (Elias et al. 1979), but most of them extract cholesterol from the membrane and therefore are disruptive to the membrane structure. In the present study we employed a nicked and biotinylated derivative of bacterial cytolysin, Θ-toxin of Clostridium perfringens (the derivative is called as BCΘ hereafter). As described previously for a nicked and methylated Θ-toxin (Ohno–Iwashita et al. 1990), BCΘ retains specific binding activity to cholesterol but lacks hemolyzing activity (unpublished observations). By using this reagent as a probe, we could analyze the effect of cholesterol crosslinking without damaging the cellular integrity.
A portion of this work was presented previously in preliminary form (Fujimoto et al. 1996).
Materials and Methods
Reagents
Recombinant Θ-toxin was prepared from the periplasm of E. coli strain BL21(DE3) harboring pNSP10, a plasmid containing the Θ-toxin gene (Sekino–Suzuki et al., submitted for publication). The toxin was purified and nicked with subtilisin Carlsberg as reported previously (Ohno–Iwashita et al. 1986). The nicked toxin was biotinylated with sulfosuccin-imidyl-6-(biotinamido)hexanoate (Vector Labs; Burlingame, CA) (unpublished observations). Briefly, the nicked toxin was mixed with an equal amount of the bio-tinylating reagent in 50 mM borate buffer, pH 8.6, and was incubated for 2 hr at 25C with stirring. The reaction mixture was applied to a Sephadex G-25 column equilibrated with PBS and the protein fractions were added with 1 mg/ml bovine serum albumin and stored at 4C. Binding specificity of a nicked Θ-toxin for cholesterol has been reported previously (Ohno–Iwashita et al. 1988). Rabbit anti-caveolin antibody (Transduction Laboratories; Cincinnati, OH), fluorescein–avidin D (Vector), colloidal gold–streptavidin (British BioCell; Cardiff, UK), and rhodamine-conjugated goat anti-mouse IgG+IgM and rhodamine-conjugated goat anti-rabbit IgG antibodies (Jackson ImmunoResearch Labs; West Grove, PA) were obtained from commercial suppliers.
Cells
Pam 212, a mouse transformed keratinocyte cell line (Yuspa et al. 1980), and 3Y1, a rat fibroblast cell line (obtained from Japanese Cancer Research Resources Bank, Tokyo) were grown on coverslips in Dulbecco's modified Eagle's medium plus 10% fetal bovine serum.
Labeling of Fixed Specimens
Cultured cells were rinsed and incubated in a serum-free medium for at least 2 hr before experiments to minimize the possibility that cholesterol in the bovine serum would affect the results. Mouse erythrocytes were used after extensive washing with PBS. The cells were fixed with 0.1% glutaraldehyde for 10 min. In some experiments, more rigorous fixation (1% glutaraldehyde for 1 hr) was applied, but the result was not changed. For fluorescence microscopy, autofluorescence was quenched with 1 mg/ml sodium borohydride. The samples were treated with 1% bovine serum albumin (fatty acid-free grade; Seikagaku Kogyo, Tokyo, Japan) for 30 min, incubated with 10–30 μg/ml BCΘ for 30 min, and then with either fluorescein–avidin D (20–50 μg/ml) or colloidal gold–streptavidin (diluted to 1:30) for 30 min. For controls, the samples were pretreated with either 300 μM filipin, 160 μM digitonin, or 150 μM tomatin (Sigma Chemical; St Louis, MO) for 30 min, and the BCΘ solution was also added with 1:10 concentration of the above reagents. All the procedures were done at room temperature.
Treatment of Living Cells
Pam 212 cells were cooled on ice for 15 min, and were treated with 10–30 μg/ml BCΘ and then with either fluorescein-avidin D or colloidal gold–streptavidin, both for 60 min on ice. After rinsing, some samples were immediately fixed with 3% formaldehyde for more than 30 min, and others were incubated for 5–30 min at 37C before fixation.
To exclude the possibility that the final fixation caused redistribution of bound BCΘ, a group of labeled cells was observed without fixation. The result was equivalent to the cells labeled and fixed by the above method. Therefore, the distributional change of BCΘ observed in the present experiment was not an artifact induced by fixation.
In an experiment, living Pam 212 cells were labeled with BCΘ on ice as above, and their apical plasma membranes were isolated on Alcian blue-coated coverslips (Rutter et al. 1985). After brief fixation and pretreatment, rabbit anti-caveolin antibody and then a mixture of fluorescein–avidin D and rhodamine-conjugated goat anti-rabbit IgG antibody were applied. All procedures were done on ice.
Microscopic Observation
For fluorescence microscopy, samples were observed and photographed with a Zeiss Axiophot microscope. For thin-section electron microscopy, the colloidal gold-labeled samples were further fixed with 2% glutaraldehyde, postosmicated with 1% osmium tetroxide, stained en bloc with 0.5% aqueous uranyl acetate, dehydrated, and embedded in Spurr's resin. For freeze-fracture electron microscopy, glutaraldehyde-fixed samples were soaked in 25% glycerin, inverted on a mixture of polyvinyl alcohol and glycerin (Pauli et al. 1977), and frozen in a liquid nitrogen slush. They were freeze-fractured at −110C in a Balzers BAF401 apparatus (Balzers High Vacuum; Balzers, Liechtenstein). The platinum/carbon replicas were observed after extensive rinses in water without digestion (Pinto da Silva and Kan 1984).
Results
Labeling of Fixed Cells with BCΘ
When fixed cells were incubated with BCΘ, and then with fluorescein–avidin D, labeling occurred on the entire cell surface. Figure 1A shows the labeling of 3Y1 cells, a rat fibroblast cell line. Previous results have shown that a nicked Θ-toxin binds specifically to membrane cholesterol in living cells (Ohno–Iwashita et al. 1988). To examine if the binding is specific even after the aldehyde fixation, control experiments were performed. First, incubation of fixed cells with fluorescein–avidin D alone without the BCΘ treatment did not cause labeling (Figure 1B). Second, the specimens were pretreated with reagents specific for cholesterol and then incubated with BCΘ; filipin, digitonin, and tomatin were applied at the concentration used previously for cytochemical labeling (Elias et al. 1979). In mouse erythrocytes (not shown) as well as in 3Y1 cells (nFigures 1C–1E), all of the three reagents were effective in inhibiting the binding of BCΘ. These results showed that the labeling with BCΘ is specific for cholesterol.
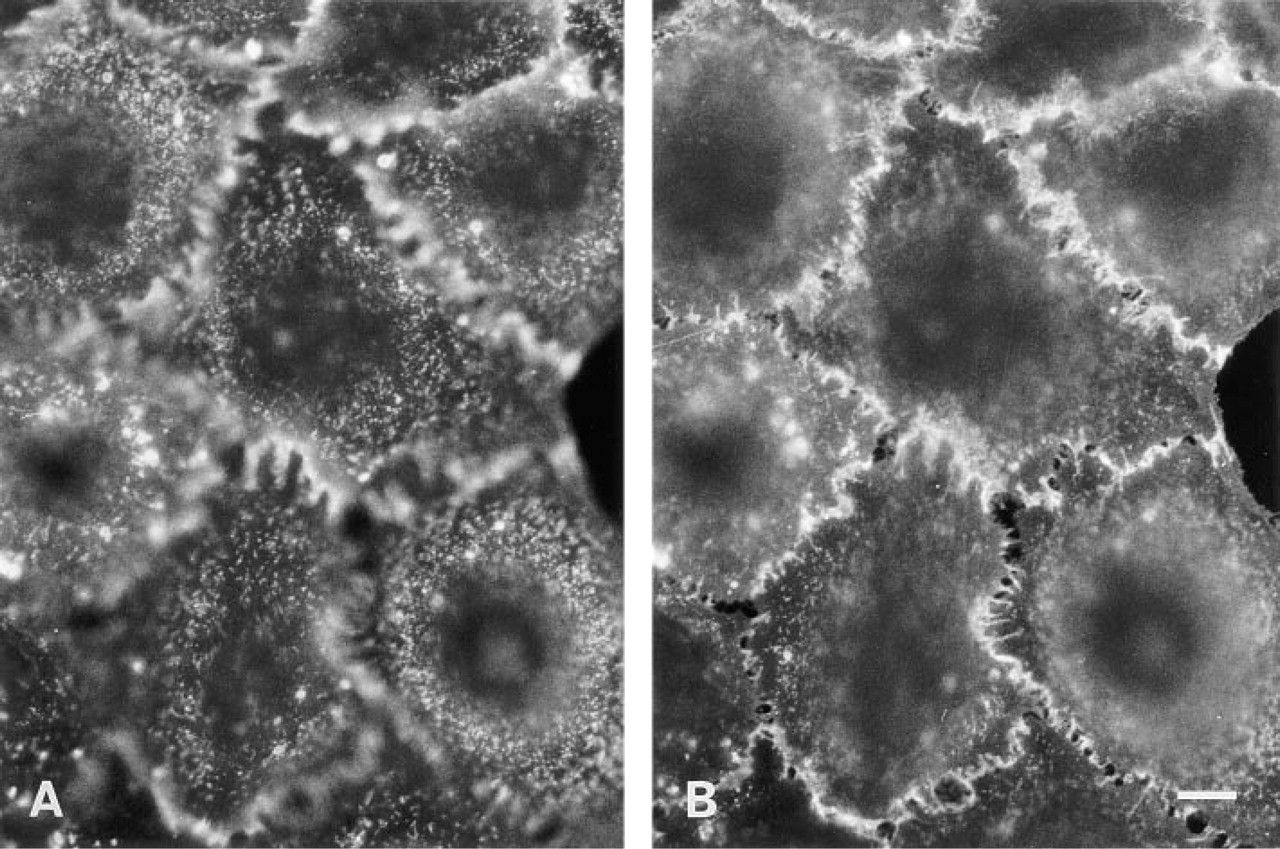
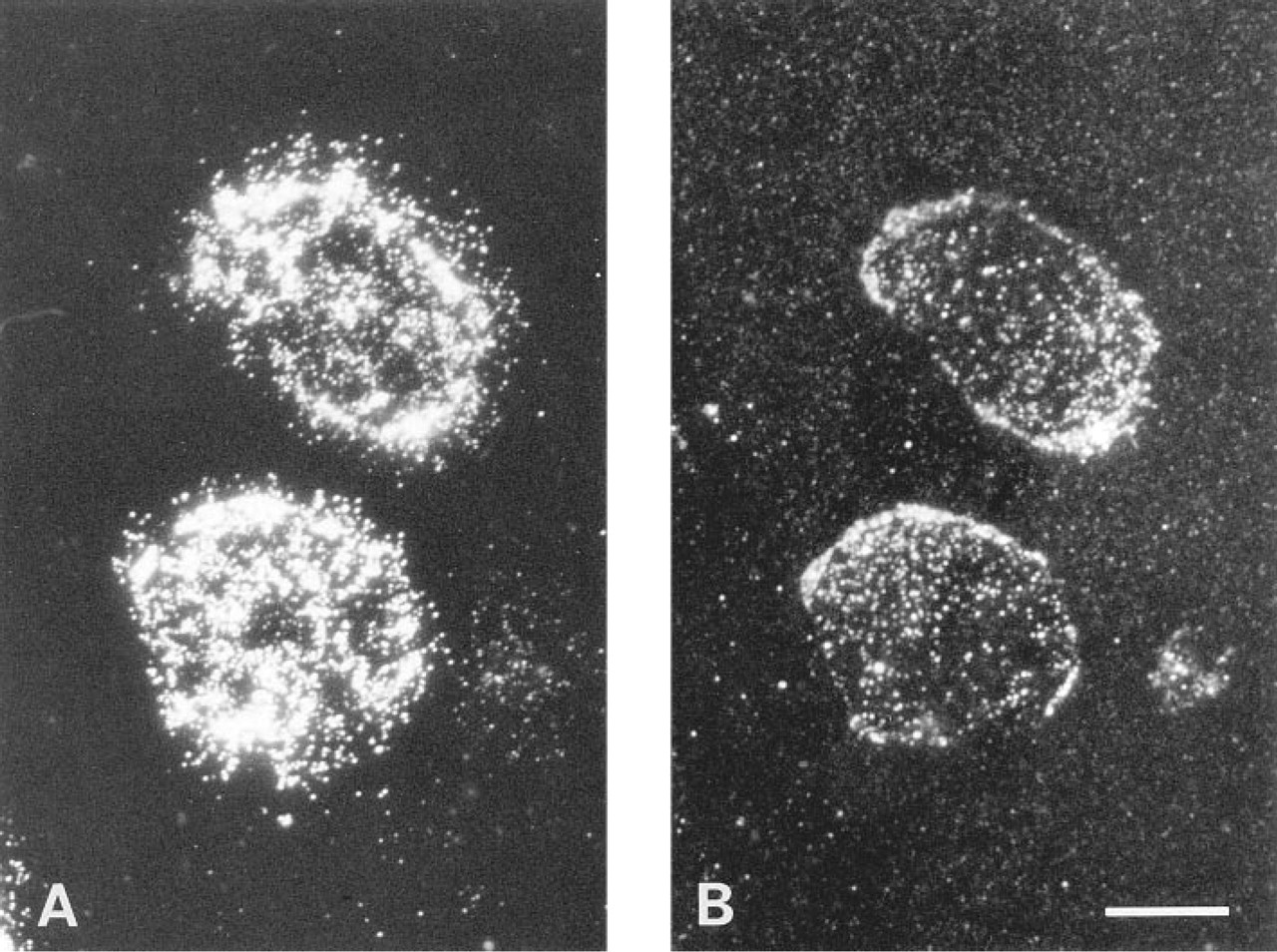
In Pam 212 cells, in addition to general labeling, intense fluorescence was observed as speckles on the apical surface; lateral processes also showed strong labeling (Figure 2). Next, fixed Pam 212 cells were treated with BCΘ and then with colloidal gold–streptavidin and were observed by label-fracture electron microscopy (Pinto da Silva and Kan 1984). By this method, a two-dimensional distribution of the labeling on the apical cell surface could be observed. Most gold particles were seen in small clusters on the E face; notably, they were preferentially distributed around the base of microvilli, which appeared as dents in the freeze-replica (Figure 3A). Because the replica was not digested with bleach, some microvilli remained intact and were observed as long dark rods. Caveolae were seen as small bumps, but gold particles were not concentrated in or around them (Figure 3B). Identification of these structures as caveolae was done previously by decoration with anti-caveolin antibody (Fujimoto 1996).
Treatment of Living Cells with BCΘ
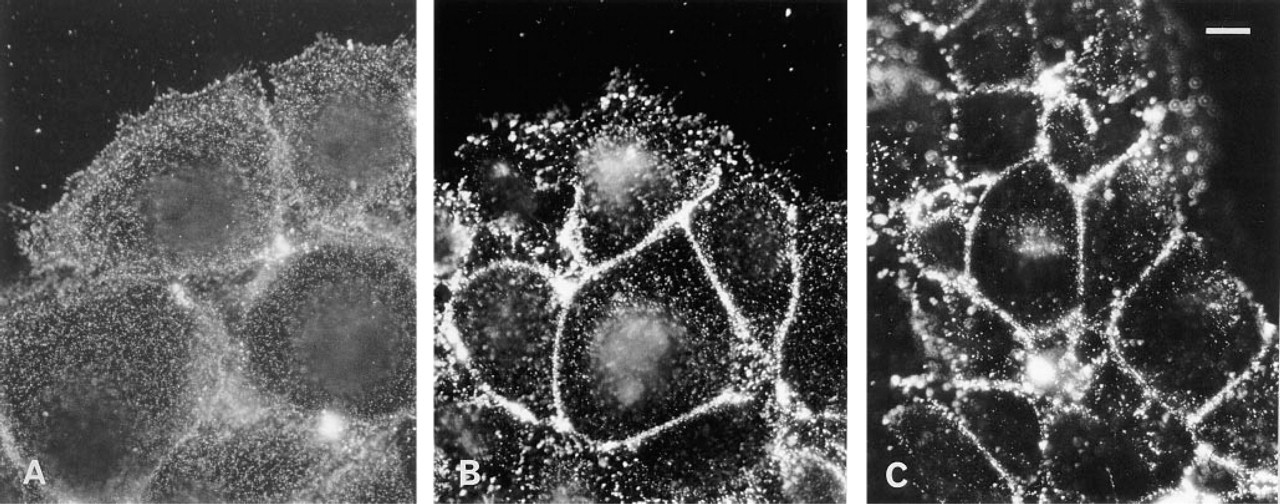
Antibodies to GPI-anchored proteins, glycolipids, and sphingmyelin bound to the entire surface of Pam cells, but became concentrated to caveolae when further treated with secondary antibodies and incubated at 37C (Fujimoto 1996). To examine if cholesterol behaves in a similar manner when crosslinked, living Pam cells were treated with BCΘ and then with fluorescein–avidin D. When the cells were treated with BCΘ on ice and fixed immediately, the labeling was seen as fine dots distributed on the cell surface without local concentration (Figure 4A). In contrast, when the temperature was raised to 37C after treatment, the labeling became concentrated at the cell periphery; the concentration was obvious at 10 min and increased further at 30 min (Figures 4B and 4C).
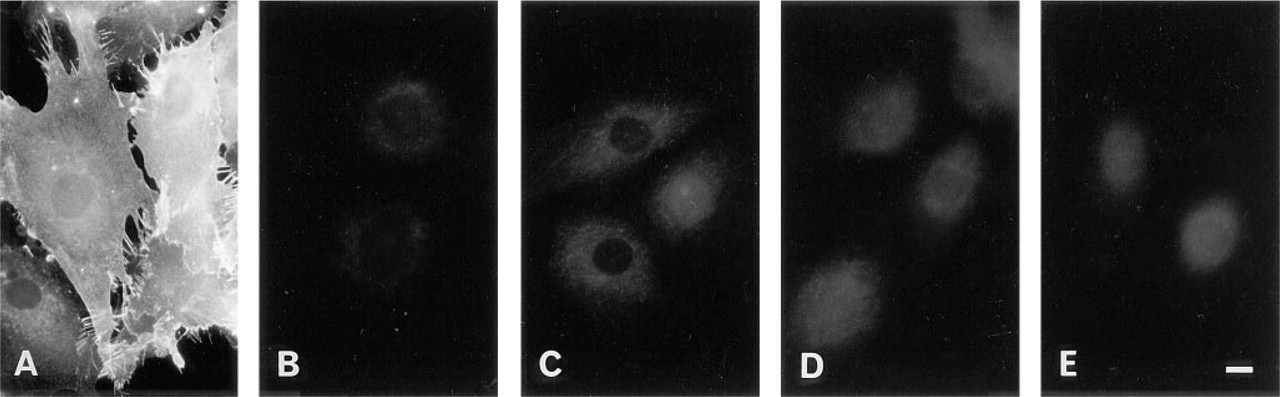
Control experiments to examine the specificity of the BCΘ labeling. Fixed rat 3Y1 fibroblasts were incubated with various combination of reagents.
Fixed mouse Pam 212 cells were incubated with BCΘ and fluorescein–avidin D. Fluorescence micrographs of the same cells at two different focal planes.
Because caveolae are accumulated in the vicinity of intercellular contact in Pam 212 cells (Fujimoto et al. 1995), the dots observed by the BCΘ labeling on ice (Figure 4A) are not likely to match caveolae. To confirm this, the apical plasma membranes of the cells treated with BCΘ alone on ice were isolated on adherent coverslips and immunolabeled for caveolin. Caveolin was localized as dots, more concentrated in some areas compared to other areas of the isolated membrane (Figure 5B). In the same membranes, binding of BCΘ, which was visualized by fluorescein–avidin D after fixation, appeared in speckles, and their distribution did not coincide with caveolin (Figure 5A).
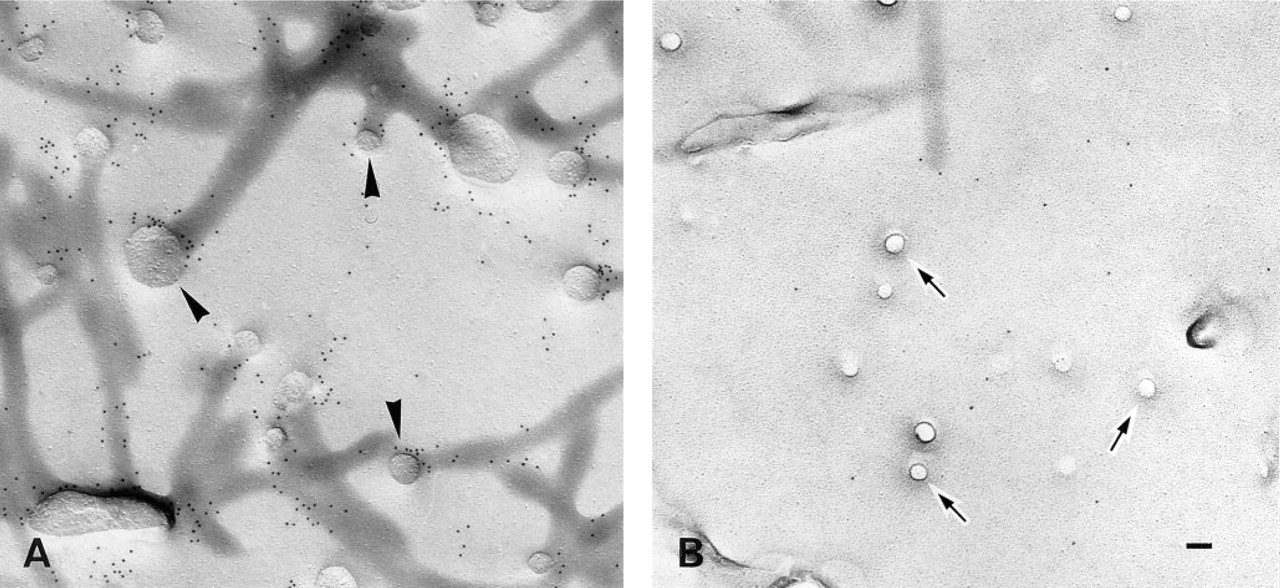
Next, to observe the fine distribution of crosslinked BCΘ, the specimen incubated for 30 min at 37C was subjected to label-fracture electron microscopy. Compared to the labeling in fixed cells (Figure 3B), gold particles were observed more concentrated in the caveola-rich area (Figures 6A and 6B). They were seen on the bumps themselves and on the flat membrane area close to the bumps. The same specimen was processed for thin-section electron microscopy. Most gold particles were seen in caveolae (Figure 6C).
Freeze-fracture replicas of labeled cells observed without digestion. Fixed mouse Pam 212 cells were incubated with BCΘ and then with colloidal gold–streptavidin. The E face of the apical surface.
Living Pam 212 cells were treated with BCΘ on ice, fixed, and then incubated with fluorescein–avidin D
The apical plasma membrane of living Pam cells was treated with BCΘ alone, isolated on adherent coverslips, and fixed. Fluorescein–avidin D was applied to visualize the distribution of BCΘ, and rabbit anti-caveolin and rhodamin-conjugated donkey anti-rabbit antibodies were used to detect caveolin.
Discussion
BCΘ as a Cytochemical Probe for Cholesterol
Many probes have been used to visualize the two-dimensional distribution of 3β-OH sterols (mainly cholesterol) in biomembranes. They are classified into three categories: polyene antibiotics (Andrews and Cohen 1979; Elias et al. 1979), saponins (Elias et al. 1978), and a combination of enzymes (Jones and Miyai 1981; Pelletier and Vitale 1994). Filipin, which belongs to the first category, has been used most frequently, but it has several drawbacks as a cytochemical probe. First, because it binds and extracts membrane cholesterol, the affected membrane loses its integrity (Bolard 1986); living cells treated with the probes are permeabilized. Second, what can be observed by electron microscopy is not the probes themselves but rather the deformation caused by the filipin–cholesterol complex. Therefore, false-negative results could occur if a membrane region is resistant to deformation (Severs et al. 1981; Severs and Simons 1983; Clark et al. 1987; Severs 1995). Third, although the probes are small molecules, they penetrate poorly in tissue specimens (Fujimoto and Ogawa 1983). These limitations should be considered when results obtained with filipin are interpreted.
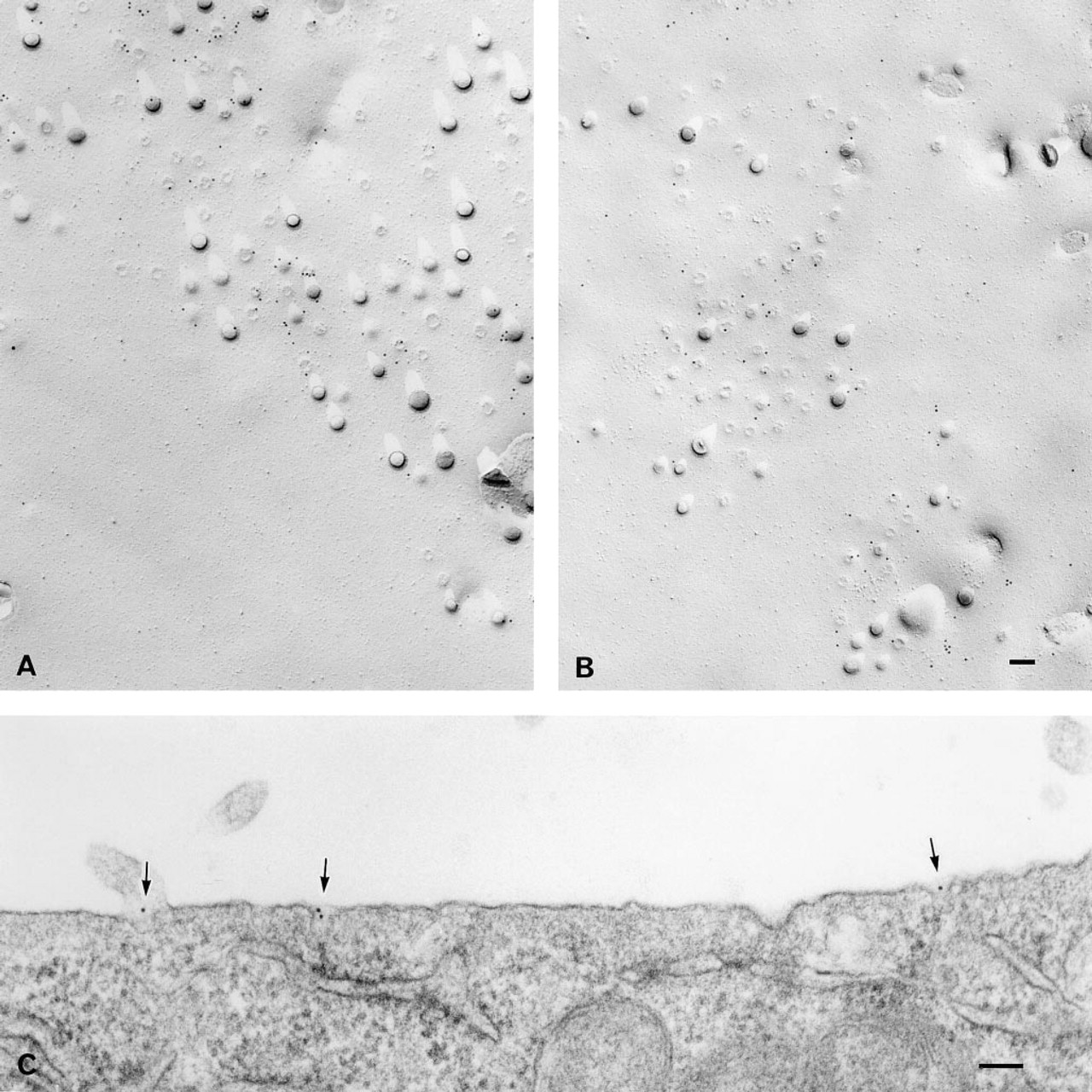
Living Pam 212 cells were treated on ice with BCΘ and colloidal gold-streptavidin and were incubated at 37C for 30 min.
In the present study, we utilized a nicked and biotinylated bacterial toxin, BCΘ, which retains specific cholesterol-binding activity but lacks hemolyzing effect (unpublished observations). When used as a probe to living cells, BCΘ appeared less damaging to the membrane than filipin or digitonin. A dye exclusion test showed that more than 95% of lymphocytes excluded trypan blue after incubation with 30 μg/ml BCΘ for 30 min at 37C; even when they were further treated with avidin D and incubated for 30 min at 37C, more than 80% of the cells excluded the dye (Fujimoto et al., unpublished observations). Because BCΘ is a protein of 53 kD, it cannot penetrate the plasma membrane and its binding is limited to the cell surface. Therefore, BCΘ may not be useful as a general cytochemical probe, but this property is an advantage when applied to living cells. Another difference between BCΘ and filipin or digitonin is that localization of BCΘ itself can be visualized by streptavidin conjugated to colloidal gold. Because the detection does not require membrane deformation, the rigidity of the membrane should not affect the labeling.
On the other hand, although the binding of BCΘ to cholesterol was shown to be specific, several problems remain to be solved. First, stoichiometry of the binding has not been determined. Therefore, whether the binding of BCΘ alone crosslinks multiple cholesterol molecules is not known. Second, it has been shown that nicked Θ-toxin binds to cholesterol with two different affinities (Ohno–Iwashita et al. 1988). This implies that cholesterol in a membrane may be composed of heterogeneous populations; local concentration of cholesterol and/or adjacent membrane molecules may change its existence mode. BCΘ may detect some populations of cholesterol more efficiently than others (Ohno–Iwashita et al. 1991). Third, because of the size of BCΘ, its binding to cholesterol is very probably affected by steric hindrance. Fourth, it is not clear whether BCΘ can recognize cholesterol bound to membrane proteins. This factor is important in interpreting the results concerning caveolae because caveolin is a cholesterol-binding protein (Murata et al. 1995) and highly enriched in caveolae. Because of these factors, especially the last two, all cholesterol molecules in a membrane may not be approached and bound by BCΘ with an equal probability.
In the present study, when cells were first fixed and then labeled, intense labeling around the base of microvilli was seen. On the other hand, such concentration was not observed in cells treated with the same combination of the reagents on ice and then fixed. Fixation may affect the state of plasmalemmal cholesterol molecules. For example, reactivity to cholesterol oxidase dramatically increased after fixation (Lange et al. 1984). The underlying mechanism is not clear, but because the aldehyde fixative is unlikely to modify cholesterol itself, membrane proteins may be primarily affected, and then influence surrounding cholesterol molecules.
Caveolar Sequestration of Crosslinked Cholesterol in Living Cells
The relationship between cholesterol and caveolae has been examined in many studies, as cited in the Introduction (Rothberg et al. 1990,1992; Schnitzer et al. 1994; Smart et al. 1994). More recently, caveolin was shown to be a cholesterol-binding protein (Murata et al. 1995). These results strongly indicate that cholesterol is important in maintaining the structure and function of caveolae.
An early cytochemical result showed that the filipin–cholesterol complex is formed densely at the orifices of caveolae (Simionescu et al. 1983). However, the preferential formation of the complex around caveolae was observed only after a short exposure to filipin; after a prolonged incubation, the complex was seen with equal density in other membrane areas (Simionescu et al. 1983). Moreover, the result was not reproducible in a careful re-examination (Simons and Severs 1986). Because the formation of the filipin–cholesterol complex can be affected by several factors in addition to the cholesterol content (Severs et al. 1981; Severs and Simons 1983; McGookey et al. 1983; Clark et al. 1987; Severs 1995), enrichment of cholesterol in caveolae or at their orifices could not be concluded from the result of filipin cytochemistry.
On the other hand, by biochemical analysis, a relatively high content of cholesterol was recovered in the detergent-insoluble complex (Brown and Rose 1992), in which caveolin is markedly concentrated (Sargiacomo et al. 1993). The complex has been often treated as isolated caveolae, but a similar complex was obtained from cell types that do not possess caveolae (Fra et al. 1994; Gorodinsky and Harris 1995). Moreover, other molecules enriched in the detergent-insoluble complex, e.g., GPI-anchored proteins, glycolipids, and sphingomyelin, were not concentrated in caveolae when examined by immunocytochemistry (Mayor et al. 1994; Schnitzer et al. 1995; Fujimoto 1996). These results indicate that the detergent-insoluble complex very likely contains non-caveolar as well as caveolar membranes (Kurzchalia et al. 1995; Schnitzer et al. 1995).
In the present study, binding of BCΘ did not occur preferentially in the vicinity of caveolae in fixed and in unfixed specimens incubated on ice. Only after crosslinking with avidin D or streptavidin and incubation at 37C was bound BCΘ sequestered to the caveola-rich region. Because of the problems discussed above, whether cholesterol is enriched in or around caveolae in native cells cannot be concluded from the BCΘ experiment. However, the pattern of redistribution after crosslinking was the same as for GPI-anchored proteins (Mayor et al. 1994; Fujimoto 1996), glycolipids, and sphingomyelin (Fujimoto 1996). This result is consistent with the assumption that at least some molecules concentrated in the detergent-insoluble complex are sequestered with caveolin artificially.
Summing up, the present study introduced a new cytochemical probe for membrane cholesterol. It can be applied to living cells with minimal damage to cellular integrity. This probe bound to the plasma membrane of living cells without marked concentration, but was sequestered to the caveola-rich region when crosslinked with the secondary reagent. The result showed that not only GPI-anchored proteins and sphingolipids (Mayor et al. 1994; Fujimoto 1996) but also cholesterol became concentrated to caveolae after crosslinking.
Footnotes
Acknowledgements
Supported by Grants-in-Aid for Scientific Research (B) (no. 08457001) and for Exploratory Research (no. 08878121) of the Ministry of Education, Science, Sports, and Culture of the Japanese Government and by a research grant from the Ciba–Geigy Foundation (Japan) for the Promotion of Science.
We are grateful to Ms Fujie Miyata and Ms Yukiko Takahashi for their excellent technical and secretarial assistance.
