Abstract
This study focused on the intracellular signal transduction system and microtubule-associated proteins (MAPs), such as MAP-2 and Tau protein. The modulation of these proteins and their correlation with ultrastructural changes were investigated in rat pituitary prolactin (PRL) cells. Adult female Wistar rats were treated with estrogen and bromocriptine and their pituitary glands were removed for analysis of the expression of tubulin, MAP-2, Tau protein, protein kinase C (PKC), and calcium calmodulin (CaM) kinase. Western blot analysis showed that estrogen increased and bromocriptine decreased the expression of PKCα, β1, β2, CaM kinase α, β, MAP-2, and Tau protein. MAP-2 and Tau protein, which are cytosolic proteins, being translated on free ribosomes, were associated with the membrane of whirling rough endoplasmic reticulum (RER) in estrogen-treated cells and dissociated with vesiculated RER induced by bromocriptine. These results suggested that the modulation of MAP-2 and Tau protein may reflect changes of PKC and CaM kinase, and that the quantitative changes and intracellular modulation of MAPs induced by estrogen and bromocriptine, i.e., estrogen-induced association and bromocriptine-induced dissociation of MAP-2 and Tau protein with membrane of RER, may reflect the dynamics of microtubules and are associated with structural changes in the RER and changes in the synthesis and intracellular transport of PRL.
P
Materials and Methods
Tissue Preparation
Thirty-two adult female Wistar-Imamichi rats were injected IM every 4 weeks with 5 mg estradiol dipropionate (E2 depot: Ovahormon Depot; Teikoku Zoki, Tokyo, Japan), and were sacrificed 7 weeks after the first injection. Half of the female rats given estrogen for 7 weeks were also injected SC with 1 mg bromocriptine (Sandoz Pharmaceutical, Basel, Switzerland) for 4 days before sacrifice. For the control study, 16 other untreated, age-matched female Wistar-Imamichi rats were sacrificed. The pituitary tissues were removed, fixed overnight in 4% paraformaldehyde dissolved in 0.01 M PBS, pH 7.4, and served for histochemical analyses. For light microscopic immunohistochemical staining, the tissues were embedded in paraffin blocks using routine techniques. For pre-embedding electron microscopic immunohistochemistry, the tissues were immersed in graded concentrations of sucrose in PBS (10% for 1 hr, 15% for 2 hr, and 20% for 4 hr), then embedded in OCT compound (Tissue-Tek; Miles Laboratories, Elkhart, IN). For the Western blotting assays, pituitary glands were homogenized and stored at 4C. For Northern blot hybridization for MAP-2 mRNA, the total RNA was isolated from the pituitary glands. A venous blood sample was taken from each rat just before sacrifice and the serum PRL level was determined by radioimmunoassay. As described in a previous report (Matsuno et al. 1995), the serum PRL levels in untreated control female rats, those given estrogen alone for 7 weeks, and those with estrogen plus bromocriptine administration ranged from 489.2 to 1306.3 (mean 1147.5), from 956.3 to 7937.6 (mean 3193.2), and from 178.6 to 1038.0 (mean 438.8) ng/ml, respectively. There was a statistically significant difference among these groups (p<0.05), and thus the effects of estrogen and bromocriptine were confirmed by evaluating serum PRL levels.
Light Microscopic Immunohistochemical Double Staining of PRL and MAP-2 or Tau Protein
To reveal the correlation between PRL and MAP-2 or Tau protein expression in a single cell, light microscopic immunohistochemical double staining of these proteins was carried out. Four-μm-thick sections of routinely processed paraffin blocks were mounted on 3-aminopropylmethoxysilane-coated slides, deparaffinized in xylene, and rehydrated through graded ethanol to water. The endogenous peroxidase activity was blocked with 0.3% H2O2 in methanol for 30 min. After the pretreatment with normal sheep sera, the sections were immunostained with the commercially available antibodies, anti-MAP-2 antibody [mouse monoclonal antibody, 1:50 diluted in bovine serum albumin (BSA)-PBS; Leinco Technologies, Manchester, UK] and anti-Tau antibody (mouse monoclonal antibody, 1:50 diluted in BSA-PBS; Chemicon International, Temecula, CA) at room temperature for 1 hr. Horseradish peroxidase (HRP)-linked antibody against mouse immunoglobulin (IgG) (sheep, 1:50 diluted in BSA-PBS; Amersham International, Poole, UK) was applied as the secondary antibody for 30 min. The immunohistochemical localization of MAP-2 and Tau protein was developed with freshly prepared 3,3′-diaminobendizine tetrahydrochloride (DAB) dissolved in 0.05 M Tris-HCl, pH 7.6, and 0.017% H2O2 for 7 min. After the slides were washed in 0.1 M glycine-HCl, pH 2.2, anti-PRL antibody [rabbit polyclonal antibody, 1:100 diluted in BSA-PBS; National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK), Bethesda, MD] was applied on the same section. Alkaline phosphatase-linked anti-rabbit IgG (goat, 1:50 diluted in BSA-PBS; Dako, Carpinteria, CA) was applied as the secondary antibody, and positive reactions for anti-PRL antibody were developed with 5-bromo-4-chloro-3-indolyl phosphate and nitroblue tetrazolium. Nuclear staining was carried out with methyl green. Normal rat brain tissue was used for the positive control study of immunohistochemical staining of MAP-2 and Tau protein, and for the negative control study normal murine and rabbit sera were substituted for the primary antibodies.
Pre-embedding Electron Microscopic Immunohistochemistry of MAP-2 and Tau Protein
Six-μm-thick frozen sections mounted on 3-aminopropyl-methoxysilane-coated slides were used. The endogenous peroxidase activity was blocked with 0.3% H2O2 in 0.01 M cold PBS for 30 min. After pretreatment with normal goat sera, they were immunostained with the above mentioned commercially available antibodies against MAP-2 and Tau protein at 4C overnight. Biotinylated antibody against mouse IgG (goat, 1:400 diluted in BSA-PBS; Dako) was applied as the secondary antibody for 30 min. Immunoreactions were followed by streptavidin and biotin complex (ABC) labeled with HRP using the Vectastain ABC kit (Vector Laboratories; Burlingame, CA) for 30 min. After being dipped in the freshly prepared DAB dissolved in 0.05 M Tris-HCl, pH 7.6, the sections were developed with 0.017% H2O2 in DAB solution for 7 min. After osmification and dehydration with ethanol, tissue sections were embedded in Quetol-812, which was polymerized at 60C for 2 days. Then ultrathin sections were made and inspected using electron microscopy (JEOL 1200VT).
Western Blotting Analyses for PKCα, β1, β2, γ, CaM Kinase α, β, Tubulin α, β, MAP-2, and Tau Protein
Fifteen pituitary glands obtained from five untreated control female rats, five rats given estrogen alone for 7 weeks, and five rats with estrogen plus bromocriptine treatment were homogenized in ice-cold 50 mM Tris-HCl, pH 7.5, containing 2 mM ethylene glycol tetraacetic acid (EGTA), 1 mM dithiothreitol (DTT), and 0.001% leupeptin. Each tissue homogenate was centrifuged at 100,000 × g at 4C for 1 hr and the supernatants were used for electrophoresis. The protein concentration of each sample was measured and the amount of each sample applied to polyacrylamide gel was adjusted for equal loading. After electrophoresis, each sample was transferred to nitrocellulose membrane. For Western blotting of PKC, anti-PKCα, β1, β2, γ antibodies (rabbit, polyclonal, 1:200 diluted; Santa Cruz Biotechnology, Santa Cruz, CA) and subsequently HRP-labeled anti-rabbit IgG (donkey, 1:500 diluted in Tween 20-PBS; Amersham International) were applied to each lane as the primary and secondary antibodies, respectively. For Western blotting of CaM kinases, MAP-2, and Tau proteins, antibodies against CaM kinase α, β (mouse monoclonal, 1:500 diluted; Gibco, Gaithersburg, MD), tubulin α (mouse monoclonal, 1:300 diluted; Sigma Immunochemicals, St Louis, MO), tubulin β (mouse monoclonal, 1:150 diluted; Sigma), MAP-2 (1:100 diluted), and Tau (1:200 diluted), and subsequently HRP-labeled anti-mouse IgG (sheep, 1:300 diluted in Tween 20-PBS; Amersham International) were applied to each lane as the primary and secondary antibodies, respectively. Immunoreactivity for each protein was visualized by freshly prepared DAB solution with 0.02% H2O2. Western blotting assays were examined for every five samples from each group.
Northern Blot Hybridization for MAP-2 mRNA
Oligonucleotide Probe Used for EM-ISH of MAP-2 mRNA. The sequence of the anti-sense oligonucleotide probe for MAP-2 mRNA was 5′-dGCT GTG GTT TTC CGCTTA ACA CAG GAG AGA TGA GTT GGT C. The probe was synthesized with a DNA synthesizer (Applied Biosystems model 392; Foster City, CA).
Northern Blot Hybridization. The total RNA was isolated from 15 pituitary glands of five untreated control female rats, five rats given estrogen alone for 7 weeks, and five rats with estrogen plus bromocriptine treatment, as described by Chirgwin et al. (1979). Thirteen μg total RNA was applied to each lane, separated by electrophoresis using formaldehyde-1% agarose gel, and transferred to a nitrocellulose membrane by the capillary elution method. Hybridization with 32P-labeled oligonucleotide probe for rat MAP-2 mRNA and 32P-labeled cDNA probe for β-actin mRNA, which was used to check for equal loading, was carried out at 42C, overnight. After hybridization the membrane was washed with SSC, 0.1% sodium dodecyl sulfate (SDS), and was exposed to Kodak RP film for a week. The hybridization signal densities of MAP-2 and β-actin mRNAs were analyzed densitometrically, and the MAP-2 mRNA: β-actin mRNA ratio was determined. Normal rat brain tissue was used for the positive control study. The mean and standard deviation (SD) of MAP-2 mRNA: β-actin mRNA ratio of each group was calculated and examined by Student's t-test.
Evaluation of In Vitro MAP-2 Phosphorylation by Estrogen and Bromocriptine in Female Rat Pituitary Cells
Anterior pituitary glands of female Wistar rats were separated from neurointermediate lobes, minced, dispersed and incubated in 0.25% trypsin-EDTA at 37C for 1 hr, followed by centrifugation at 1000 rpm for 5 min. The aliquots were washed briefly with 0.01 M PBS (Ca- and Mg-free) and viability was examined with Trypan blue. The cells were washed briefly with culture medium: Dulbecco's modified Eagle's medium (DMEM) H-21 with 10% calf serum, 100 U/ml penicillin-streptomycin, 25 μg/ml fungizone, 100 μg/ml gentamycin, and 0.1 μM ascorbic acid. Cells (5.8 × 106) were plated in 35-mm collagen-coated dishes in 2 ml of the above-mentioned culture medium, and incubated at 37C, in 95% air/5% CO2 overnight. They were labeled for 2.5–3 hr with 0.3 mCi carrier-free [32P]-Pi (ICN Pharmaceuticals, Irvine, CA). Drug treatment was performed as follows: Group 1, 17β-estradiol (Sigma) 1 × 10−7 M in 100% ethanol (six dishes); Group 2, 17β-estradiol 1 × 10−7 M and bromocriptine 1 μM in 100% ethanol (six dishes); Group 3, vehicle (10% ethanol-1 mM HCl-DMEM) (six dishes). The concentration of these drugs followed those described in previous reports (Carter et al. 1993; Song et al. 1989). Treatment of cells with the drugs was terminated by quickly rinsing with ice-cold PBS (Ca- and Mg-free) and extracting in 10 mM NaPi, pH 7.2, 0.15 M NaCl, 1% Nonidet P-40, 0.1% SDS, 1 mM EGTA, 50 mM NaF at 37C, in 95% air/5% CO2 for 10 min. An aliquot of the soluble extract was immunoprecipitated with a monoclonal antibody against rat MAP-2 protein and resolved by 7.5% SDS gel electrophoresis. The MAP-2 band was localized by autoradiography and analyzed densitometrically. The mean and SD of MAP-2 phosphorylation of each group were calculated and examined by Student's t-test.
Results
Light Microscopic Immunohistochemical Double Staining of PRL, MAP-2, Tau Protein
MAP-2. Localization of MAP-2 in PRL cells was detected in pituitary glands from rats without estrogen treatment (Figure 1A). MAP-2- and PRL-positive cells were increased after prolonged exposure to estrogen (Figure 1B), and were decreased after bromocriptine treatment (Figure 1C).
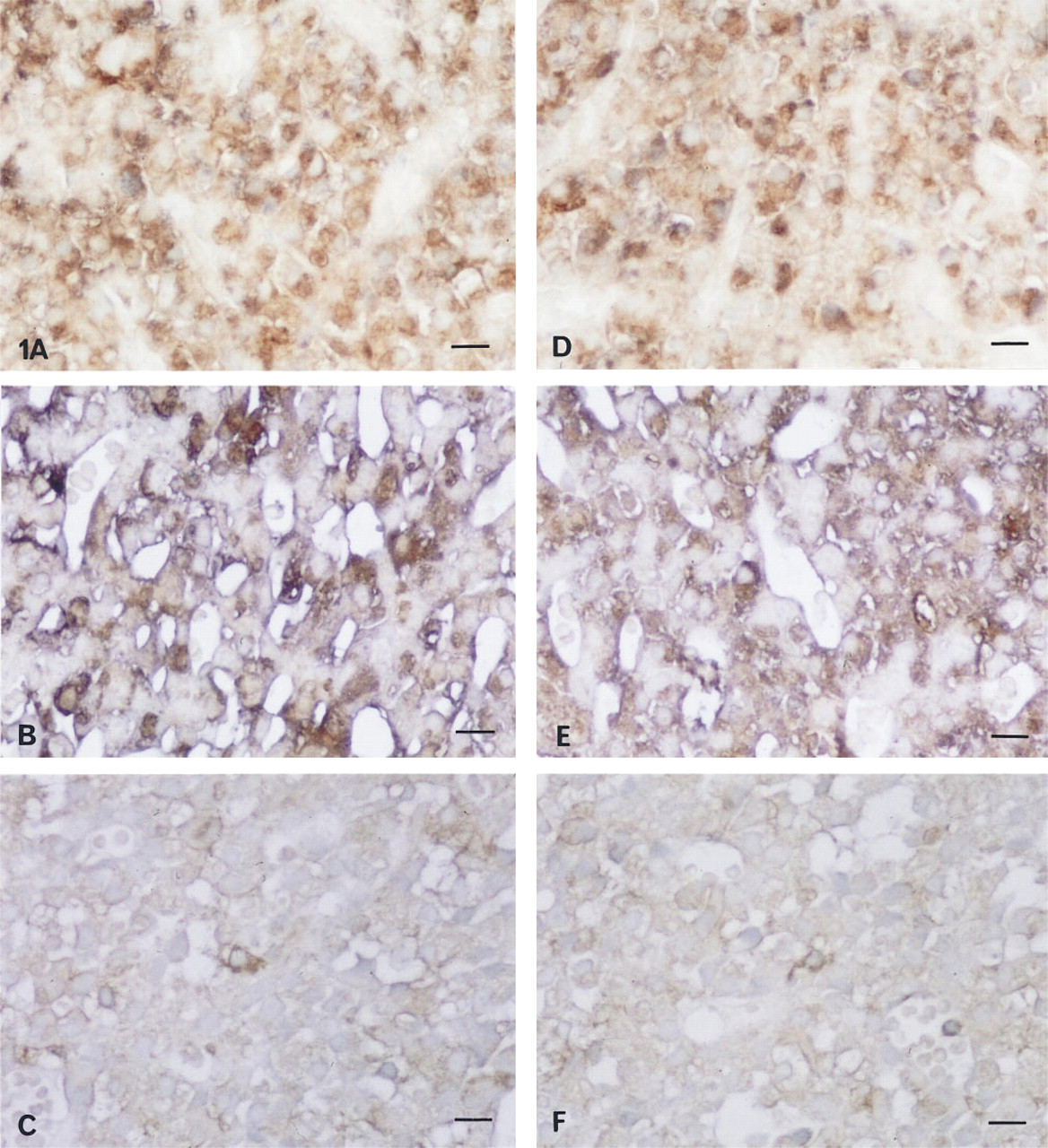
Light microscopic immunohistochemical double staining of PRL, MAP-2, and Tau protein. Localization of MAP-2 (brown) in PRL (purple) cells was detected in pituitary glands from rats without estrogen treatment
Tau Protein. Localization of Tau protein in PRL cells was detected in pituitary glands from rats without estrogen treatment (Figure 1D). Tau protein- and PRL-positive cells were increased after prolonged exposure to estrogen (Figure 1E) and were decreased after bromocriptine treatment (Figure 1F).
Pre-embedding Electron Microscopic Immunohistochemistry of MAP-2 and Tau Protein
MAP-2. MAP-2 was immunostained in the cytosol of cells from rats without estrogen treatment (Figure 2A) and was not observed on the RER (Figure 2B). MAP-2 was immunostained in the cytosol of cells from rats with prolonged exposure to estrogen (Figure 2C) and was also positively immunostained on the whirling RER (Figure 2D). When rats were treated with estrogen plus bromocriptine, MAP-2 was negative on the vesiculated RER (Figure 2E). The cells were identified as PRL cells on the basis of their secretory granules (approximately 100–250 nm in diameter), as described by Osamura et al. (1982), and of characteristic morphological changes in the RER induced by these drugs.
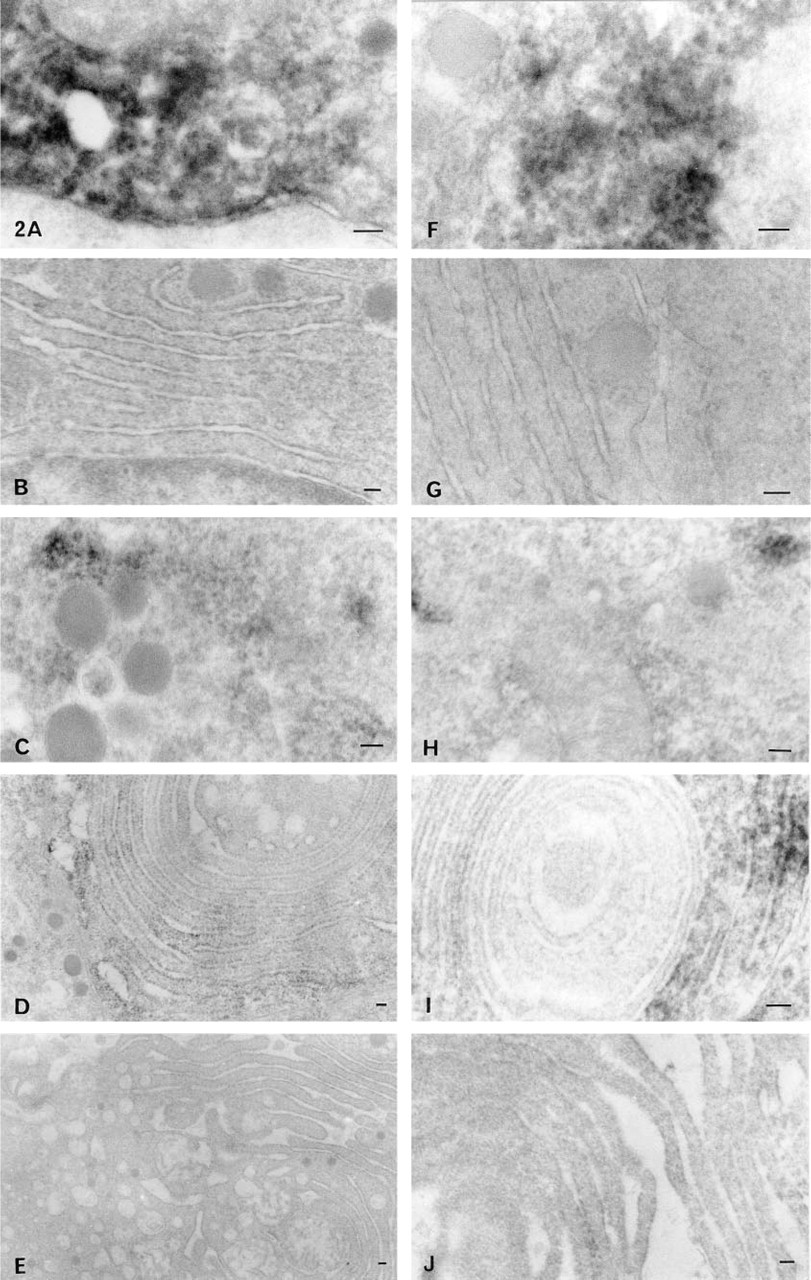
Pre-embedding electron microscopic immunohistochemistry of MAP-2 and Tau protein. MAP-2 was immunostained in the cytosol of cells from rats without estrogen treatment
Tau Protein. Tau protein was detected in the cytosol of cells from rats without estrogen treatment (Figure 2F) and was not observed on the RER (Figure 2G). Tau protein was immunostained in the cytosol of cells from rats with prolonged exposure to estrogen (Figure 2H) and was also positively immunostained on the whirling RER (Figure 2I). After treatment with estrogen plus bromocriptine, Tau protein was negative on the vesiculated RER (Figure 2J).
Western Blotting Analyses for PKC α, β1, β2, γ, CaM Kinase α, β, Tubulin α, β, MAP-2, and Tau Protein
PKCα, β1, β2, γ. Estrogen increased the amounts of PKCα (80 kD), β1 (80 kD), and β2 (80 kD) in pituitary cells, and they were all decreased by subsequent bromocriptine treatment (Figure 3). PKCγ was not expressed in pituitary cells.
CaM Kinase. Estrogen increased the amounts of CaM kinase α (54 kD) and β (60 kD) in pituitary cells, and both were decreased by subsequent bromocriptine treatment (Figure 3).
Tubulin α, β. Tubulin α (55 kD) and β (55 kD) were detected before treatment. No significant changes in the amounts of both tubulin subunits were evoked by estrogen and bromocriptine (Figure 3).
MAP-2. Estrogen increased the amount of MAP-2 (300 kD) in pituitary cells, and it was decreased by subsequent bromocriptine treatment (Figure 3).
Tau Protein. Estrogen increased the amount of Tau protein (50 kD) in pituitary cells, and it was decreased by subsequent bromocriptine treatment (Figure 3).
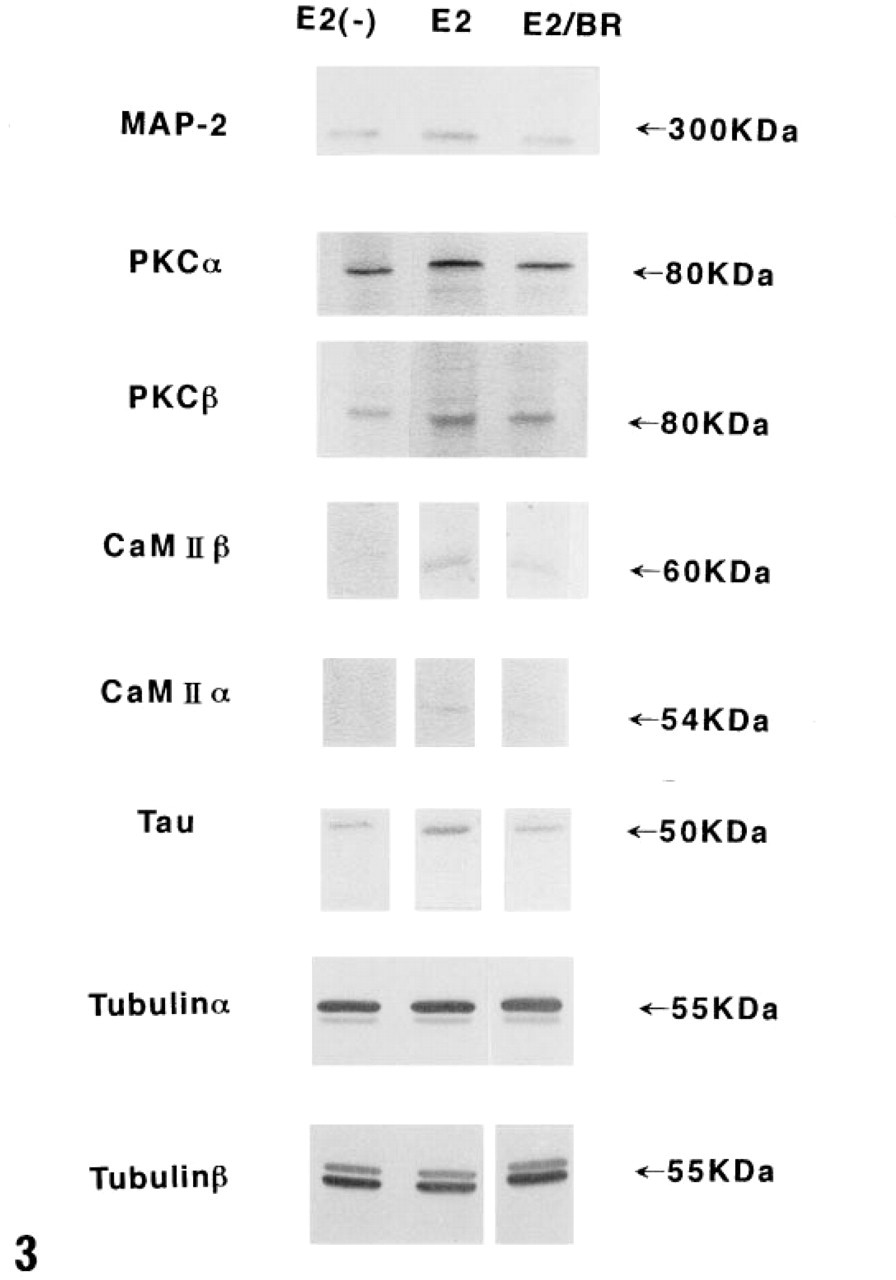
Western blotting analyses for PKCα, β1, β2, γ, CaM kinase α, β, tubulin α, β, MAP-2, and Tau protein. Estrogen increased the amounts of PKC α (80 kD), β1 (80 kD), and β2 (80 kD) in pituitary cells, and all were decreased by subsequent bromocriptine treatment. PKCγ was not expressed in pituitary cells. Estrogen increased the amounts of CaM kinase α (54 kD) and β (60 kD) in pituitary cells, and both were decreased by subsequent bromocriptine treatment. Tubulin α (55 kD) and β (55 kD) were detected before treatment. No significant changes in the amounts of both tubulin subunits were evoked by estrogen and bromocriptine. Estrogen increased the amount of MAP-2 (300 kD) in pituitary cells and was decreased by subsequent bromocriptine treatment. Estrogen increased the amount of Tau protein (50 kD) in pituitary cells, and it was decreased by subsequent bromocriptine treatment. E2(-), untreated control female rats; E2, female rats given estrogen alone for 7 weeks; E2/BR, female rats given bromocriptine after treatment with estrogen for 7 weeks.
The results observed in these Western blotting assays were common to the five samples of untreated control female rats, those given estrogen alone for 7 weeks, and those with estrogen plus bromocriptine treatment.
Northern Blot Hybridization for MAP-2 mRNA
Using a 32P-labeled oligonucleotide probe for rat MAP-2 mRNA, a 9.0-KB transcript was detected on the nitrocellulose membranes (Figure 4). The MAP-2 and β-actin mRNA hybridization signal density ratios of pituitary cells from untreated control female rats, rats with prolonged estrogen treatment, and rats treated with estrogen plus bromocriptine were evaluated by densitometry, and the mean values obtained were 1, 2.454 (SD 0.146), and 1.862 (SD 0.175), respectively (Figure 4). The MAP-2 and β-actin mRNA hybridization signal density ratio of normal rat brain tissue was 0.729. There was a statistically significant difference in the signal densities of MAP-2 mRNA from untreated control female rats, those given estrogen alone for 7 weeks, and those with estrogen plus bromocriptine treatment (p<0.001).
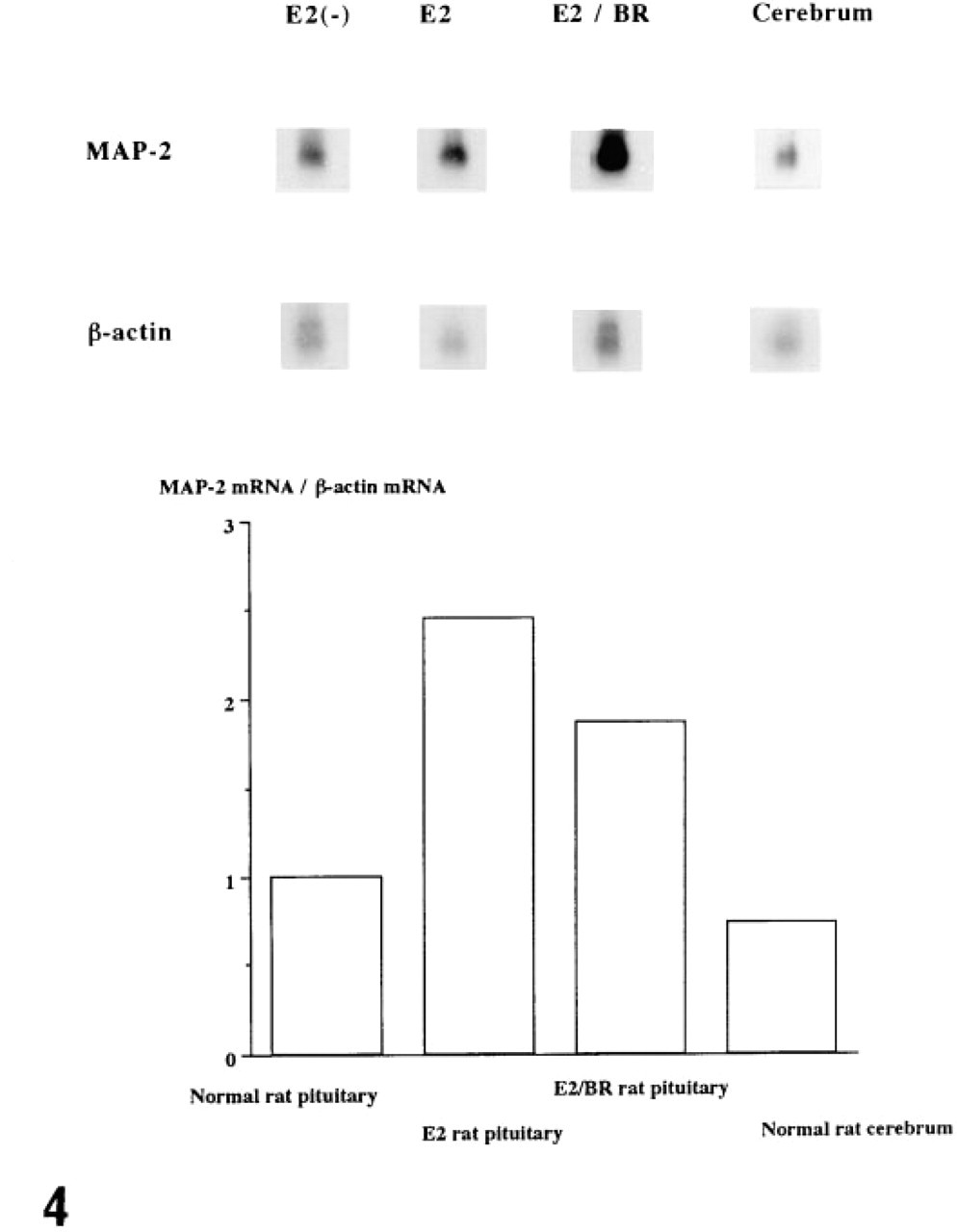
Northern blot hybridization for MAP-2 mRNA. By use of a 32P-labeled oligonucleotide probe for rat MAP-2 mRNA, a 9.0-KB transcript was detected on the nitrocellulose membranes. The MAP-2 and β-actin mRNA hybridization signal density ratios of pituitary cells from untreated control female rats, rats with prolonged estrogen treatment, and rats treated with estrogen plus bromocriptine, were evaluated by densitometry, and the values obtained were 1, 2.454, and 1.862, respectively. The MAP-2 and β-actin mRNA hybridization signal density ratio of normal rat brain tissue was 0.729. E2, female rats given estrogen alone for 7 weeks; E2/BR, female rats given bromocriptine after treatment with estrogen for 7 weeks.
In Vitro Phosphorylation of MAP-2
When the mean extent of phosphorylation of MAP-2 in control cells was defined as 1.000, the corresponding degree of phosphorylation in cells treated with 17β-estradiol and cells treated with 17β-estradiol plus bromocriptine was 1.283 and 0.913, respectively. Statistical analysis showed that there was no significant difference among these three groups (p > 0.05).
Discussion
The present experiments showed by Western blotting analyses that estrogen and bromocriptine have a significant effect on intracellular signal transduction systems, including PKC and CaM kinases. Estrogen increased and bromocriptine decreased the amounts of PKCα, β1, and β2. These findings agree with the results of our previous study, which demonstrated the respective elevation and reduction of total PKC activity after estrogen and bromocriptine administration (Matsuno et al. 1995). Estrogen-induced activation of PKC has already been suggested by Maeda and Lloyd (1993), and the reducing effect of dopamine agonists on PKC activity shown in the present study agrees with the results of Chuang et al. (1993). We also examined another important intracellular signal transduction system, CaM kinase α, β, and found that both subunits of CaM kinase were increased by estrogen and decreased by bromocriptine. Using cultured cells transfected with D2 receptors, Senogles (1994) demonstrated that dopamine influences intracellular signal transduction, and his finding also agrees with our results showing that bromocriptine has a reducing effect on PKC and CaM kinase. On the basis of these observations, it is considered that estrogen and bromocriptine respectively activate and reduce intracellular signal transduction.
Another target of the present experiment was to clarify the major factors related to the changes in cellular ultrastructure and to cell proliferation. MAP-2 and Tau protein are heat-stable, microtubule-associated proteins and both promote microtubule assembly. Our Western blot analyses of MAP-2 and Tau protein showed that estrogen increased and bromocriptine decreased both of these proteins. The estrogen-induced increase and the bromocriptine-induced decrease of MAP-2 were also demonstrated at the mRNA level by our Northern blot hybridization study. Electron microscopic observation showed that MAP-2, which is one of the cytosolic proteins, and was immunostained in the cytosol of untreated cells, was observed both in the cytosol and on the whirling RER after prolonged exposure to estrogen. Electron microscopy also revealed that MAP-2 was not immunostained on the vesiculated RER induced by bromocriptine. In addition, the same modulations of Tau protein induced by estrogen and bromocriptine were revealed by electron microscopy. These electron microscopic findings suggest the estrogen-induced association and bromocriptine-induced dissociation of MAP-2 and Tau protein with the membrane of RER. The microtubule network has an important role in the distribution and movement of intracellular organelles, such as the RER (Lee and Chen 1988; Terasaki et al. 1986), Golgi apparatus (Van De Moortele et al. 1993; Rogalski and Singer 1984; Pavelka and Ellinger 1983), and endosomes and lysosomes (Bomsel et al. 1990; Matteoni and Kreis 1987). Van De Moortele et al. (1993) studied the morphological effect of nocodazole, which inhibits microtubule assembly, on the Golgi apparatus, and found that it induced dilatation of the apparatus. Brefeldin A, a fungal metabolite, is known to inhibit protein transport from the ER to the Golgi apparatus, resulting in reorganization of the Golgi apparatus with vesiculation of the Golgi cisternae and their eventual disappearance (Nishikawa and Sasaki 1995; Lippincott-Schwarz et al. 1987, 1990; Fujiwara et al. 1988). Lippincott-Schwartz et al. (1990) have referred to the role of a microtubule-dependent pathway to the Golgi apparatus. These morphological changes in the Golgi apparatus appear to be similar and comparable to the alterations of the RER observed in our bromocriptine-treated pituitary cells. Van De Moortele et al. (1991) examined a thyroliberin-induced rapid and transient reorganization of cytoskeletal proteins such as microtubules, intermediate filaments, and microfilaments in GH3B6 cells, suggesting a correlation with the reorganization of microtubules and the remodeling of Golgi membranes. In our study, the quantitative changes and intracellular modulation of MAPs induced by estrogen and bromocriptine, i.e., estrogen-induced association and bromocriptine-induced dissociation of MAP-2 and Tau protein with the membrane of RER, are considered to reflect the dynamics of microtubules and to be associated with structural changes in the RER and changes in the synthesis and intracellular transport of PRL.
The microtubules are considered to play an important role in the secretion of PRL (Martinez de la Escalera and Weiner 1992). Martinez de la Escalera and Weiner (1992) studied MAP phosphorylation in lactotrophs, induced by thyrotropin-releasing hormone and dopamine and stated that dopamine withdrawal promoted the phosphorylation of various MAPs, including MAP-2, and increased PRL secretion. On the basis of their results, dopamine was suggested to suppress the phosphorylation of MAP-2 and to inhibit PRL secretion. MAPs can be phosphorylated by various protein kinases, including cAMP-dependent kinase, multifunctional CaM kinase, PKC, insulin-stimulated MAP kinase, and tyrosine kinase (Jefferson and Schulman 1991). Phosphorylation of MAP-2 was shown to influence the polymerization state of microtubules and the ability of tubulin to interact with the membrane (Hoshi et al. 1988). Our in vitro study of the phosphorylation of MAP-2 induced by estrogen and bromocriptine did not demonstrate statistically significant changes. Although in an in vivo model of estrogen- and/or bromocriptine-treated rat pituitary cells phosphorylation of MAP-2 is difficult to determine, it can be postulated that these drugs might affect the in vivo phosphorylation of MAP-2, which resulted in the above mentioned characteristic structural changes of the RER. The microtubule network also has an important role in cell mitosis. Taxol, which promotes the assembly of microtubules, is known to inhibit mitosis and recently has been suggested as a promising anti-cancer drug (Crossin and Carney 1981; Schiff et al. 1979). On the basis of our findings, we speculate that quantitative changes of MAP-2 and Tau protein may be involved in cell proliferation induced by estrogen and cell involution induced by bromocriptine. The present study appears to be the first report showing that the modulation of cytoskeleton-related proteins, such as MAP-2 and Tau protein, induced by estrogen and bromocriptine, may be related to cell proliferation, to ultrastructural changes in the RER, and to the synthesis and transport of PRL.
Footnotes
Acknowledgements
This study was partly supported by Grants for Scientific Research (nos. 07771082 and 06404051) from the Ministry of Education, Science and Culture of Japan.
We thank Reiko Matsuura, Johbu Itoh, and Shigeru Munakata for technical and photographical assistance.
