Abstract
The presence of neuronal nitric oxide synthase (nNOS) in two populations of pituitary cells, gonadotrophs (LH) and folliculostellate (FS) cells, suggests that pituitary nitric oxide (NO) is involved in the control of hormone secretion. We have used single and double immunostaining and quantitative procedures to investigate possible gender-related differences in the nNOS expression pattern in the anterior pituitary lobe and its possible alterations in different endocrine situations. Our results reveal a sexual dimorphism in the pattern of nNOS expression. In males, nNOS is mainly found in FS cells, whereas only a few LH cells express nNOS. Conversely, in females, nNOS is mainly found in LH cells. After gonadectomy, paralleling an increase in LH cell size and serum luteinizing hormone (LH) levels, there is nNOS upregulation in LH cells and nNOS downregulation in FS cells. After testoterone replacement, LH cells become nNOS-immunonegative again. In lactating rats, LH cells overexpress nNOS, but LH cell size and serum LH levels are low. This suggests that, depending on its cellular source, pituitary NO can exert either an inhibitory or a stimulatory effect on hormone secretion. When released from FS cells, NO exerts a paracrine inhibitory effect, and when released from gonadotrophs it exerts an autocrine or paracrine stimulatory effect on LH or prolactin secretion, respectively.
N
With immunohistochemical and in situ hybridization techniques, it has been possible to map the distribution of NOS in various tissues, including the endocrine system. In the posterior lobe of the pituitary gland, nNOS has been demonstrated in axon terminals that come from magnocellular hypothalamic neurosecretory neurons (Bredt et al 1990; Vanhatalo and Soinila 1995). In the anterior lobe of the pituitary gland, nNOS is expressed in two cell populations, gonadotrophs (LH cells) and folliculostellate (FS) cells (Ceccatelli et al. 1993; Wang et al. 1997). FC cells do not produce hormones but share certain characteristics with mononuclear phagocytic cells (Vankelecom et al. 1992). In addition, iNOS has been induced by interferon-7 in FS cells in anterior pituitary cultures (Vankelecom et al. 1997).
Functional studies suggest that NO may be involved in the secretion of different pituitary hormones (Kato 1992; Brunetti et al. 1993; Ceccatelli et al. 1993; Duvilanski et al. 1995; Rivier 1995; Pinilla et al. 1998). The fact that certain endocrine manipulations, such as gonadectomy, produce NOS upregulation in pituitary cells (Ceccatelli et al. 1993; Wang et al. 1997) also suggests a role for NO in the control of anterior pituitary functions, particularly in the secretion of luteinizing hormone (LH). Both autocrine and paracrine interactions have been proposed as mechanisms involved in these effects on pituitary secretion (Ceccatelli et al. 1993; Ceccatelli 1997; Vankelecom et al. 1997; Wang et al. 1997). At present, however, the precise role of pituitary NO in the control of anterior pituitary secretion is unknown. Stimulatory (Pinilla et al. 1998), inhibitory (Ceccatelli et al. 1993) and no direct effects (Bonavera et al. 1993) of NO on LH secretion have been reported.
With the aim of contributing to a better understanding of the functional meaning of NO in the pituitary gland, and particularly in LH secretion, we have investigated possible gender differences in the pattern of nNOS activity under normal conditions, and the effect of different endocrine manipulations on its expression, by using immunocytochemical and double immunofluorescence techniques. In addition, we used quantitative analysis of the number and size of LH cells (Kovacs and Horvath 1975; Wynick et al. 1990; Catt and Dufau 1991) and serum LH levels as functional parameters of endocrine activity.
Materials and Methods
Animals and Tissue Preparation
Male and female Sprague–Dawley rats (IFFA-Credo; Lyon, France) weighing 260–280 g were studied. The following endocrine models were used: male rats castrated 15 days beforehand (n = 5), castrated with testosterone replacement for 3 days (1.7 mg/d; n = 4), and intact male rats (n = 5); and ovariectomized (n = 5), lactating female (15 days after delivery; n = 4), and female rats in the diestrous stage of the estrous cycle (n = 5). Surgical interventions were performed under aseptic conditions, and all animals used in the research described here were acquired and cared for in accordance with the guidelines published in the NIH Guide for the Care and Use of Laboratory Animals and the principles presented in the “Guidelines for the Use of Animals in Neuroscience Research” by the Society for Neuroscience.
Rats were heavily anesthetized with chloral hydrate and transcardially perfused with 150 ml of heparinized 0.9% saline and 300 ml of 4% paraformaldehyde in 0.1 M phosphate buffer, pH 7.4 (PB). Before perfusion, blood samples of 1 ml were obtained from anesthetized animals for subsequent determination of serum LH levels, using a rat luteinizing hormone EIA system kit supplied by Amersham (Poole, UK). Pituitary glands were removed and stored in the same fixative at 4C for 2 hr. After cryoprotection in 30% sucrose in PB overnight at 4C, 20-μm-thick coronal sections were cut with a freezing microtome. Sections were collected in three parallel series, one for single NOS immunostaining, another for either LH (as a marker of gonadotrophs), prolactin (as a marker of lactotrophs) or S-100 (as a marker of FS cells) single immunocytochemistry, using 3,3-diaminobenzidine (DAB) as chromogen, and the third one for either NOS/LH or LH/prolactin double immunofluorescence, or single NOS, prolactin, or S-100 immunofluorescence. Bearing in mind that fixative conditions are critical in the histochemical and immunocytochemical detection of NOS-expressing cells (Matsumoto et al. 1993; González-Hernández et al. 1996), to ensure the validity of comparison, fixative parameters, including volume and concentration of paraformaldehyde and postfixation time, were carefully observed. In addition, sections were mounted on gelatinized slides, so that those from the experimental groups (castrated, castrated with testosterone replacement, ovariectomized, and lactating females) were on the same slide as, and close to, those from the corresponding material from the control groups (intact males and females in the diestrous stage), and all immunoreactions were developed simultaneously by using the same concentration of antibodies and chemical reactives.
Immunocytochemistry
For single immunolabeling, sections were immersed for 20 min in 3% H2O2 to inactivate endogenous peroxidase and incubated for 60 min at room temperature (RT) in the preincubation solution (PIS): 4% normal horse serum (NHS; Vector Laboratories, Burlingame, CA) or 4% normal goat serum (NGS; Vector Laboratories) in 0.1 M PBS, pH 7.4, containing 0.1% Triton X-100 (TX-100; Sigma, St. Louis, MO). Thereafter, they were incubated overnight in a gently shaking humid chamber in PIS containing one of the following primary antibodies. For nNOS, we used a rabbit anti-nNOS polyclonal antibody (1:2000; Zymed Laboratories, San Francisco, CA); for LH, a mouse anti-human β3LH monoclonal antibody (1:3000; Serotec, Oxford, UK); for prolactin, a rabbit anti-rat prolactin polyclonal antibody (1:3000; a gift from Dr. A.F. Parlow, UCLA Medical Center, Torrance, CA); and for S-100, a rabbit anti-S-100 polyclonal antibody (1:800; Sigma). After several rinses, sections were incubated for 2 hr in biotinylated horse anti-mouse antiserum (1:200; Vector) or biotinylated goat anti-rabbit antiserum (1:200; Vector) and 1:200 NHS or 1:200 NGS in PBS. Immunoreactions were visible after incubation for 1 hr at RT in ExtrAvidin-peroxidase (1:5000; Sigma) in PBS, and after 10 min in 0.005% DAB and 0.001% H2O2 in cacodylate buffer. After several rinses in PBS, the slides were dehydrated, cleared in xylene, and coverslipped with DPX (BDH Chemicals; Poole, UK).
For immunofluorescence, sections were immersed for 60 min in PIS and overnight in PIS containing one of the primary antibodies used in immunocytochemistry at double concentration. For double immunofluorescent staining, we followed the same procedure, but the incubation contained a mixture of two of the primary antibodies. The immunofluorescent labeling was visible after incubation for 3 hr in 1:100 fluorescein isothiocyanate-conjugated goat anti-mouse IgG (Sigma) and 1:150 lissamine–rhodamine-conjugated goat anti-rabbit IgG (Jackson ImmunoResearch; West Grove, PA) in PBS containing 1:200 NGS (Vector). After several rinses, sections were air-dried, coverslipped with Vectashield (Vector), and examined under epifluorescence microscopy using appropriate filters. For each type of immunocytochemistry and immunofluorescence, control experiments were performed by omission of the primary antibodies, resulting in negative staining.
Quantitative analysis
Three quantitative parameters were evaluated: the size and number of single- and double-labeled endocrine cells, which provide a semiquantitative approach to their secretory activity and the degree of NOS upregulation in different endocrine situations, and the LH plasma levels, a biochemical parameter usually used as an index of gonadotroph activity.
Single (NOS or LH)- and double (NOS and LH)- stained cell profiles were counted in four rats per group to obtain a semiquantitative estimate of the proportion of gonadotrophs showing NOS activity in each experimental group. Counts were performed in five randomly selected sections per rat, dividing each section into square fields of 300 μm × 250 μm at ×400. Measurements of the maximal diameter of gonadotrophs (LH-immunopositive cells) were made at ×400 by projecting the image with a camera lucida drawing tube onto a calibrated surface (n = 100 cells per rat and group). Only cell profiles including the sectioned nucleus were analyzed. Numerical measurements were corrected by using the assumption-based method of Abercrombie (1946; see also Coggeshall and Lekan 1996).
Statistical analysis
Numerical data are expressed as mean ± SEM. A mean value was obtained from each animal, and four animals were used to calculate the mean and SEM. The statistical significances were assessed by analysis of variance (ANOVA) and Tukeys multiple comparison post-hoc tests for differences among groups.
Results
Our results show that, in the pituitary gland of both male and female adult rats, many cells are immunoreactive for nNOS, although their distribution pattern differs from each other.
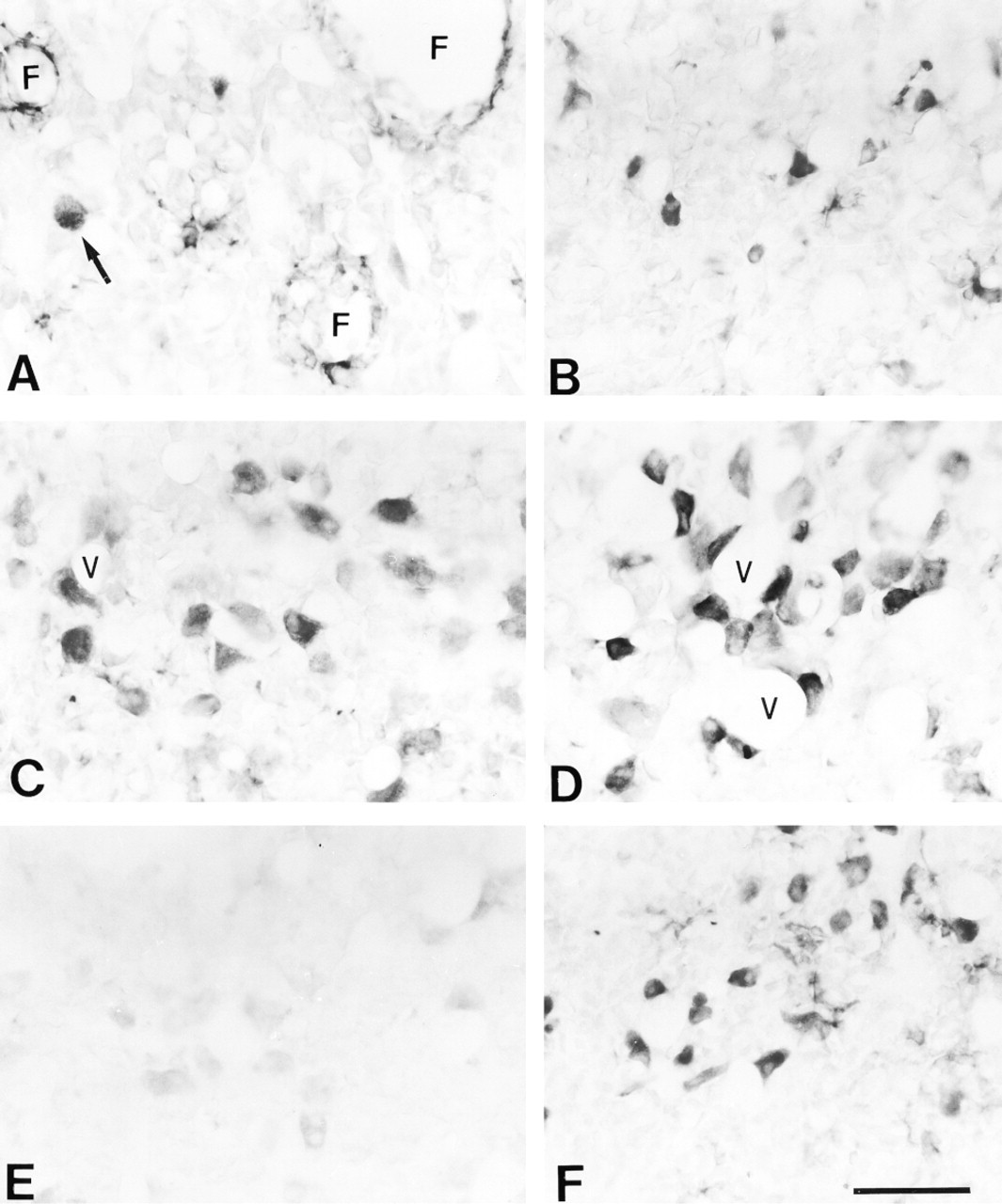
In males, most nNOS activity was detected in small cells with short processes found between larger immunonegative cells (Figure 1A and 2B). Small nNOS-positive cells were present everywhere in the gland. They were usually grouped in small clusters or forming “ring-like” structures with diameters ranging from 30 to 100 μm, which appeared to surround glandular cysts or follicles (F in Figures 1A and 2A–2C). Parallel sections immunostained for S-100 (Figure 2C), an FS cell marker, revealed a labeling pattern similar to that of nNOS. Immunopositive cells were small and often appeared in a “ring-like” arrangement. This suggests that, in male rats, nNOS is mainly expressed in FS cells. nNOS activity was also detected in a smaller number of large round or polygonal cells (Figure 1A, arrow) that, as confirmed by LH–nNOS double labeling, were gonadotrophs (Figure 2A and 2B, arrows). The quantitative analysis revealed a cell density of 42.0 ± 3.2 gonadotrophs/field (Figure 3A), with a diameter of 16.3 ± 0.9 μm (Figure 3B), although only a small percentage of them (4.4%; Figure 3A) expressed nNOS.
By contrast, in females, nNOS activity was virtually restricted to large cells (Figure 1B) which, as confirmed by LH–nNOS double labeling (Figure 2D and 2E), were gonadotrophs. nNOS-positive “ring-like” structures were not identified, and only a few dispersed small cells showed a faint immunoreaction. Therefore, we can state that there is a sexual dimorphism in the distribution pattern of nNOS in the pituitary gland: In males, nNOS is mainly found in FS cells, whereas in females it is found in endocrine cells. Quantitative study of the number and size of LH cells revealed that they were significantly sparser (22.9 ± 1.6 cells/field; p >0.01; Figure 3A) and smaller (diameter of 12.5 ± 0.1 μm; p >0.01; Figure 3B) in females than in males but, in contrast, most of them (89.7%) expressed nNOS.
Male and female rats were subjected to distinct endocrine manipulations in order to study the effect of different endocrine paradigms on the normal patterns of nNOS activity.
In the pituitary gland of castrated male rats, the distribution pattern of nNOS activity underwent a striking change (Figure 1C). FS cells became immunonegative or faintly stained, and the ring-like nNOS-positive structures were not observed. In contrast, LH cells became larger (23.6 ± 0.7 μm; p >0.001; Figure 3B) and most of them (85.2%; p >0.001) were intensely immunoreactive for nNOS (Figure 1C, 2F, and 2G). So, we can say that after castration, the pattern of nNOS activity turned into a “female-like” pattern, but with a reinforcement of normal female features.
After testosterone replacement, the pituitary gland of castrated rats partially recovered its normal nNOS activity pattern. Endocrine cells became immunonegative or weakly stained (Figure 1E), with a significant decrease in the proportion of both LH cells showing nNOS activity (70.1%; p >0.001; see Figure 3A) and their diameter (18.2 ± 0.3; p >0.001), although FS cells remained nNOS immunonegative, at least during the first 3 days of treatment.
In ovariectomized rats, the pattern of nNOS activity was very similar to that of castrated rats (compare Figure 1C and 1D). There was a reinforcement of the features of normal females, with a significant increase in the number of LH cells/field (53.4 ± 2.5; p >0.001), nNOS cells/field (54.9 ± 0.3; p >0.001), and LH–nNOS cells/field (49.6 ± 0.8; see also Figure 2H and 2I), as well as an increase in the size of LH cells (23.1 ± 0.2 μm; p >0.001; Figure 3B). FS cells were nNOS-immunonegative.
High-magnification photomicrographs showing the nNOS immunostaining pattern in intact male (
In lactating rats, there also was an increase in the number of nNOS-positive cells. The pattern of nNOS activity was similar to that of ovariectomized and castrated rats, with a large number of nNOS-positive cells that correspond in size, shape and distribution to endocrine cells (Figure 1F). Double immunofluorescence for LH and nNOS enabled us to confirm that, in a similar way to what occurs in gonadectomized rats, they were gonadotrophs (Figure 2J and 2K), although in this experimental group they were smaller (14.2 ± 0.2 μm; Figure 3B). Double immunofluorescence for LH and prolactin did not show co-expression.
An additional morphological finding worth mentioning concerns the distribution of endocrine cells. Although present throughout the entire anterior lobe of the pituitary gland, endocrine cells tend to organize in small clusters composed of four to six cells around holes with a diameter of 20–40 μm, resembling vascular sinusoids (Figures 2F–2I, arrows; see also Okere et al. 1998). These “rosette-like” images were found in every experimental group and were particularly conspicuous in castrated, ovariectomized, and lactating rats. They should be distinguished from those encircling presumed colloid-containing follicles formed by nNOS-positive FS cells.
Serum LH levels in the different experimental groups are shown in Figure 3C. As expected, basal LH levels in intact male and female rats were low, although slightly higher in females, 6.4 ± 0.0 ng/ml and 8.9 ± 1.4 ng/ml, respectively. Gonadectomy significantly increased these levels in both sexes, 21.8 ± 1.5 ng/ml (p >0.001) in males, and 19.9 ± 1.4 ng/ml (p >0.001) in females. Testosterone treatment significantly reduced castrated levels but not to basal levels (11.5 ± 1.6 ng/ml). Lactating females showed significantly lower LH levels than intact females (3.4 ± 0.4 ng/ml; p >0.017).
Discussion
Functional studies attribute an important role to NO in the control of gonadotropin secretion and therefore of reproductive functions. Several authors agree that, at the hypothalamic level, NO exerts a positive influence on the biosynthesis and secretion of LHRH (Bonavera et al. 1993; Moretto et al. 1993; Rettori et al. 1993; Wang et al. 1998). In contrast, data regarding its role at the pituitary level are contradictory. With the use of sodium nitroprusside as an NO generator, stimulatory (Pinilla et al. 1998), inhibitory (Ceccatelli et al. 1993), and no effects (Bonavera et al. 1993) of NO on basal or LHRH-induced LH release from hemipituitaries or anterior pituitary cell cultures have been reported. Although this discrepancy is probably due to methodological differences, our findings provide arguments supporting the possibility that pituitary NO can exert both effects (stimulatory and inhibitory), depending on the cell type that expresses NOS.
Confirming previous reports (Ceccatelli et al. 1993; Wang et al. 1997), double immunofluorescent labeling showed that nNOS is present in FS and LH cells in the anterior pituitary gland of male and female rats. However, our study also indicates the existence of sexual dimorphism in the distribution pattern of nNOS expression. In males, nNOS is mainly found in FS cells, whereas in females nNOS is mainly expressed in LH cells. These differences suggest that NO plays different roles in the male and the female pituitary gland.
FS cells have been defined as a type of non-hormone-secreting cell in the anterior pituitary gland, sharing functional and biochemical characteristics with macrophages (Vankelecom et al. 1992). Although their functional significance remains unclear, there are studies suggesting that FS cells exert a modulatory role, reducing both the stimulatory and the inhibitory effects of hypothalamic factors on endocrine cells, possibly via the release of paracrine molecules (Denef 1986; Baes et al. 1987). In this context, NO generated in FS cells might be one of the paracrine molecules that exert an inhibitory effect on pituitary endocrine cells and thus affect LH secretion. This hypothesis is supported in part by the fact that, in castrated rats, nNOS activity practically disappeared from FS cells when serum LH levels and LH cell size and density (number of cells/field) increase, indicating an inverse relationship between FS cell NOS expression and gonadotrophic activity. Contrasting with the findings in FS cells, in both male and female rats, gonadectomy produced nNOS upregulation in gonadotrophs. Furthermore, after testosterone replacement in orchidectomized rats, paralleling a further decrease in serum LH levels and LH cell size, LH cells became nNOS negative again. Therefore, gonadectomy, apart from eliminating gender differences in NOS expression, showed a direct relationship between LH cell nNOS expression and gonadotrophic activity in both sexes. Bearing in mind these findings, we can suggest a dual role for pituitary NO in the control of gonadotropin secretion: one that is paracrine inhibitory, associated with FS cells, and the other that is autocrine stimulatory, associated with gonadotrophs. The first may be involved in the negative feedback of gender-related steroids and the maintenance of basal (tonic) secretion of LH. The second, in addition to being responsible for the high levels of serum LH in gonadectomized animals, may be involved in phasic LH secretion. In support of this hypothesis, NO has been involved in pulsatile LH release (Rettori et al. 1993), in the LH surge of proestrus (Bonavera et al. 1994), and in afternoon LH surges in steroid-primed rats (Bonavera et al. 1993).
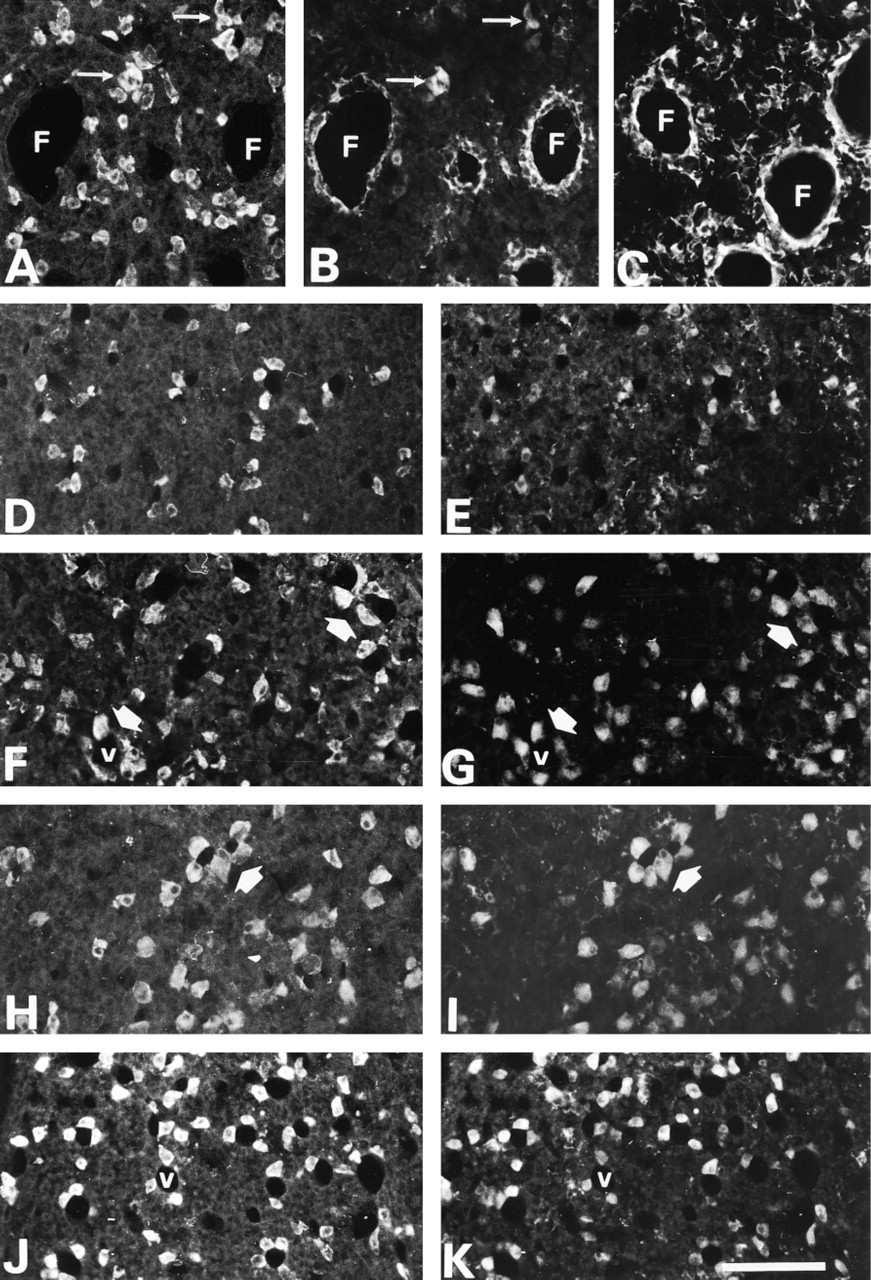
Double immunofluorescence for LH (
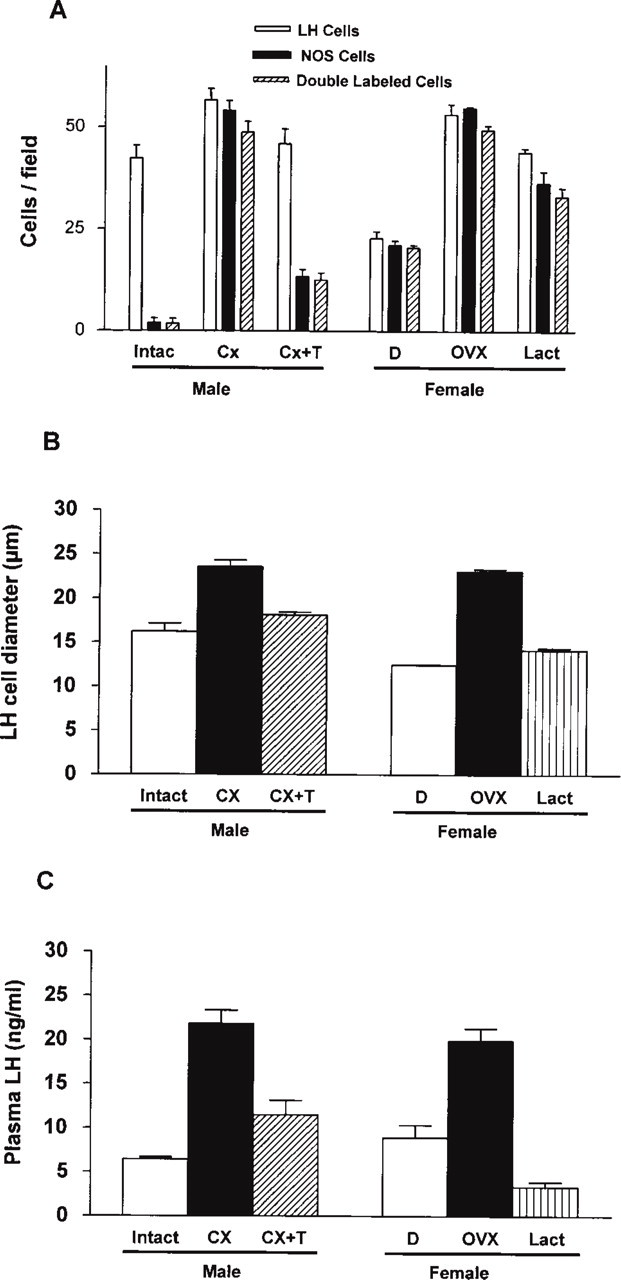
Graphs illustrating the quantitative study in the different experimental groups (intact, intact males; CX, castrated; CX + T, castrated with testosterone replacement; D, females in diestrus; OVX, ovariectomized; Lact, lactating): (
Several studies have shown that NO has a role in prolactin secretion and lactation. Intracerebroventricular injection of NO donors stimulates prolactin secretion by inhibiting tyrosine hydroxylase activity in the median eminence (González et al. 1996, 1998; González and Llorente 1998). It has been also reported that, during lactation, NOS activity increases in the supraoptic and paraventricular nuclei of the hypothalamus (Ceccatelli 1997). Taken together, these studies suggest that, at the suprapituitary level, NO exerts a stimulatory effect on prolactin release and lactation. When pituitary cells are studied in vitro, data concerning the direct effect of NO on prolactin secretion, like those for LH, are controversial. Some studies propose that it plays an inhibitory role (Duvilanski et al. 1995; Vankelecom et al. 1997), whereas others suggest a stimulatory role (Brunetti et al. 1995; González and Aguilar 1999). Interestingly, the inhibitory effect was particularly apparent in anterior pituitary cultures enriched with FS cells (Vankelecom et al. 1997). Therefore, it appears that NO released from FS cells exerts an inhibitory role on both LH and prolactin secretion. In lactation, prolactin secretion increases and gonadotrophin secretion decreases. Paracrine interactions between lactotrophs and gonadotrophs have been used as an argument to explain the impairment of gonadotropin secretion that frequently accompanies hyperprolactinemia (Catt and Dufau 1991), suggesting that lactotrophs exert an inhibitory action on gonadotrophs. In agreement with this assumption, we found two functional indices of gonadotroph hypofunction in lactating rats: low serum LH levels and small gonadotrophs that are just the opposite of those found in gonadectomized rats. In contrast, gonadotrophs overexpressed nNOS. Although our findings do not provide evidence about the functional meaning of this paradoxical pattern, some possibilities can be suggested. First, it is possible that, in an attempt to increase the reduced LH levels, gonadotrophs produce NO intended to act as an autocrine stimulatory agent, although it is insufficient against the inhibitory actions of lactotrophs. However, it is improbable that, under physiological conditions (lactation), an ineffective biological process is activated and that, in lactation, gonadotroph-released NO acts as an autocrine inhibitory agent, contributing to the inhibitory action of lactotrophs on gonadotrophs. However, it is also unlikely that the same cell type overexpresses NOS, resulting in two opposite effects on itself. Furthermore, for an inhibitory effect of NO, it would be more reasonable to activate FS cell NOS. A third possibility is that gonadotroph nNOS upregulation can carry out different endocrine functions from LH release, i.e., that gonadotroph-released NO exerts a paracrine stimulatory action on prolactin secretion. Although this hypothesis needs to be confirmed by further studies, our findings show this to be the most probable hypothesis. The absence of nNOS activity in endocrine cells other than gonadotrophs, together with gonadotroph nNOS upregulation in two endocrine situations characterized by opposite functional profiles, suggests that gonadotroph-released NO can play an stimulatory role on both LH (autocrine) and prolactin (paracrine) secretions.
In conclusion, bearing in mind this and previous studies, we propose that NO effects on pituitary hormone (LH and prolactin) secretion depend on the site of release and its cellular source. It exerts a stimulatory effect at a suprapituitary level. At a pituitary level, it also exerts a stimulatory effect when released from LH cells but its effect is inhibitory when released from FS cells.
Footnotes
Acknowledgements
Supported by a grant from Plan Nacional de I + D. Programa Sectorial de Promoción General del Conocimiento, Ministerio de Educación y Cultura (PM95–0060), and by a grant from Gobierno Autónomo de Canarias (PI 1998/008).
