Abstract
This study focused on expression of estradiol receptors (ER) during the estrous cycle. Labeling for ERα or β antigens and luteinizing hormone (LH) or follicle-stimulating hormone (FSH) β-subunits was done on freshly dispersed pituitary cells. The lowest expression of ERα and β was seen in estrus (23% and 12%, respectively). Expression increased to 42–54% of pituitary cells by diestrus. In males, cells with ERα or β were 37% or 20% of the population, respectively. ERα or β and gonadotropin antigens were in 6–9% of pituitary cells from male rats. Early in the cycle (estrus and metestrus), less than 5% of pituitary cells expressed ERα or β with gonadotropins. These values doubled to reach a peak of 10% during proestrus (just before ovulation). These data show that a rise in expression of both ERα and ERβ is a part of preovulatory differentiation of pituitary gonadotropes. (
E
Both ERα and ERβ are found in the pituitary of most species (Couse and Korach 1999). Most studies agree that gonadotropes are among the ER-expressing cells (Mitchner et al. 1998). Because the function of ERβ is unknown, this study was initiated to learn more about expression during the rat estrous cycle.
The stage of the cycle was determined by vaginal smears. The ACUC committee, University of Texas Medical Branch, approved the animal care and use protocol. Pituitaries were dispersed, cultured for 1–2 hr, fixed in 2% glutaraldehyde (30 min), and washed four times in phosphate buffer with 4% sucrose (Childs et al. 1994). Cells were pretreated with 3% hydrogen peroxide, washed twice in Tris-buffered saline (TBS), and treated with 0.3% Triton X-100 (10 min each), followed by a TBS wash. Nonspecific reactions were blocked with 1% bovine serum albumin and 1% nonfat dry milk in TBS (10 min). Dual immunolabeling involved detection of ERα or β with polyclonal rabbit anti-ERα (1:500) or goat anti-ERβ (1:500) (Santa Cruz Biotechnology; Santa Cruz, CA) and streptavidin peroxidase detection kits (DAKO; Carpinteria, CA) that used either a biotinylated anti-rabbit antibody (1:100, 30 min for the anti-ERα) or anti-goat antibody (1:100, 30 min for the anti-ERβ) as second antibody. Peroxidase was detected by nickel-intensified diaminobenzidine (Childs et al. 1994). After blocking with 1% normal goat serum, the cells were immunolabeled for luteinizing hormone (LH) or follicle-stimulating hormone (FSH) β-subunits with 1:30,000 anti-bovine LHβ or 1:10,000 anti-human FSHβ and DAKO streptavidin-peroxidase kits (amber DAB; Childs et al. 1994). ANOVA followed by Fisher's least significant differences test detected significant differences between percentages of each cell type.
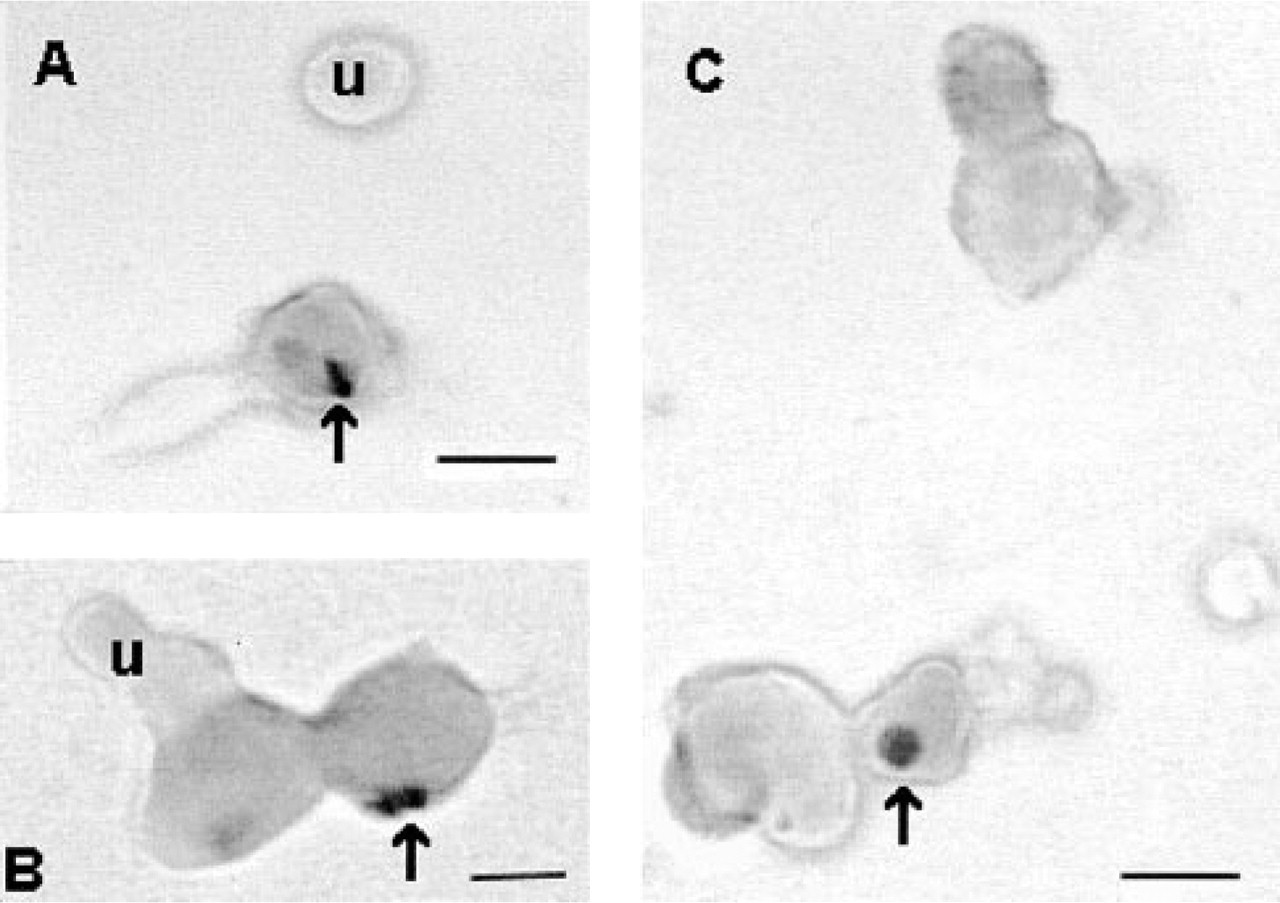
(
Estrous animals had only 12 ± 1% or 23 ± 1% cells with ERβ or α, respectively. A significant rise in expression to 41–42% of the population was seen on the morning of metestrus. Diestrous rats showed a continued rise in ERα-bearing cells to 54 ± 2%, but no further increase in cells with ERβ. Proestrous rats maintained relatively high percentages of ERα cells (48 ± 1%), whereas those with ERβ declined (36 ± 2%). Populations from male rats expressed 37 ± 5% or 20 ± 2% cells with ERα or β, respectively.
Pituitary cells with ERα or β and LH antigens increased from 4.7 ± 0.6% at estrus to 10 ± 1.6% at proestrus. Percentages of pituitary cells with ERα or β and FSH were lower at estrus (2.7 ± 0.2% or 1.3 ± 0.1%, respectively). After remaining below 5% during diestrus, cells with FSH and ERα or β increased to reach a peak of 9.5 ± 1% or 8 ± 0.3%, respectively, by proestrus. In male rats, percentages of pituitary cells with ERα or β and gonadotropins were similar to peak levels in proestrus. Figure 1 illustrates dual labeling for ERβ and FSH antigens in proestrous female rat pituitary cells. In most cells, the labeling is concentrated in the nucleus (see arrows in figure). However some cells exhibit either cytoplasmic or plasma membrane labeling (not shown).
Estrogen positive feedback on pituitary gonadotropes during diestrus promotes differentiation by stimulating the production of gonadotropin-releasing hormone receptors (Childs et al. 1994). This is vital for the preovulatory surge of LH and the rise in FSH. The peak expression of ERα and β during proestrus may facilitate the positive feedback, and the negative feedback after the surge activity. Our counts agree with previous studies that looked at ERα only (Kikuta et al. 1993). We also show a parallel increase in ERβ, suggesting that the two receptors may work cooperatively in the same cells. Studies by Mitchner et al. (1998) suggested that few pituitary cells expressed both types. ERβ is sometimes difficult to detect, and the pretreatment steps used in our protocol may better detect dual expression, especially during proestrus.
To summarize, although these studies have not detected a function for ERβ, they have shown that this new receptor type increases early in the cycle and that peak expression parallels that seen for ERα. This points to some cooperative potential for these receptors. The non-parallel expression seen in LH and FSH gonadotropes may reflect expression by monohormonal cells and/or unique functions for each of these receptors in different gonadotrope subtypes.
