Abstract
We mapped the cellular and subcellular distribution of the Na-K-Cl co-transporter (NKCC) in the adult gerbil inner ear by immunostaining with a monoclonal antibody (MAb T4) generated against human colon NKCC. Heavy immunolabeling was seen in the basolateral plasma membrane of marginal cells in the stria vascularis and dark cells in the vestibular system. Subpopulations of fibrocytes in the cochlear spiral ligament and limbus and underlying the vestibular neurosensory epithelium also stained with moderate to strong intensity, apparently along their entire plasmalemma. Because MAb T4 recognizes both the basolateral secretory (NKCC1) and the apical absorptive (NKCC2) isoforms of the co-transporter, we employed reverse transcription and the polymerase chain reaction (RT-PCR) to explore isoform diversity in inner ear tissues. Using NKCC1 and NKCC2 isoform-specific PCR primers based on mouse and human sequences, only transcripts for NKCC1 were detected in the gerbil inner ear. The presence of abundant NKCC1 in the basolateral plasmalemma of strial marginal and vestibular dark cells confirms conclusions drawn from pharmacological and physiological data. The co-expression of NKCC1 and Na,K-ATPase in highly specialized subpopulations of cochlear and vestibular fibrocytes provides further evidence for their role in recycling K+ leaked or effluxed through hair cells into perilymph back to endolymph, as postulated in current models of inner ear ion homeostasis.
I
Despite the large body of pharmacological and physiological data supporting its presence, the precise cellular distribution and isoform expression patterns of NKCC in the inner ear have yet to be firmly established. Immunocytochemical data presented in a recent unpublished abstract have provided preliminary evidence for the localization of NKCC in the basolateral membrane of strial marginal cells in the rabbit cochlea (Mizuta et al. 1996). Another very recent study using reverse transcription and the polymerase chain reaction (RT-PCR) has identified transcripts for NKCC in the rat cochlea (Hidaka et al. 1996). These reports have been limited to the lateral wall of the cochlea from two different species, and no comprehensive and definitive immunohistochemical or molecular biological studies of the inner ear NKCC have yet been published. Accordingly, the goals of the present investigation were to determine the cell type-specific and subcellular expression pattern of NKCC throughout the adult gerbil cochlea and vestibular system using an NKCC-specific MAb and to ascertain the presence of cochlear and vestibular NKCC isotypes using RT-PCR.
Materials and Methods
Animals and Tissue Processing
Inner ears were obtained from 14 Mongolian gerbils (Meriones unguiculatus) of both genders between 1 and 12 months of age. The gerbils were born and raised in a quiet vivarium with a mean noise level of approximately 40 dBA (Mills et al. 1990). The care and use of the animals was approved by The Medical University of South Carolina's Animal Care and Use Committee under NIH Grant DC00713.
Six gerbils between 6 and 12 months of age were anesthetized by IP injection of urethane (1.5 g/kg in dH2O) and then exsanguinated by transcardial perfusion with a warm 0.9% saline solution containing 0.1% sodium nitrite. For light microscopic (LM) studies, 50 ml of a fixative, consisting of 10% formalin, 0.9% NaCl, and 0.5% zinc dichromate was administered transcardially. The bullae were opened rapidly, the stapes removed, the round window perforated, and 0.5 ml of fixative was infused gently through the oval window. The inner ears were dissected free and immersed in fixative for a further 15 min, rinsed with saline, and decalcified by immersion in 0.12 M EDTA (pH 7.0) for 48–72 hr at room temperature (RT) with gentle stirring. The EDTA solution was changed daily. The decalcified inner ears were dehydrated in a graded series of ethanols (70% 2 hr; 80% 2 hr; 95% 2 hr; 100% three times for 1 hr), cleared in Histoclear (twice for 1 hr; National Diagnostics, Manville, NJ), and embedded in Paraplast Plus (twice for 1 hr at 58C; Curtin Matheson, Marietta, GA). Serial mid-modiolar sections were cut at 5 μm thickness and mounted on glass slides.
Four gerbils between 6 and 12 months of age were processed for electron microscopic (EM) studies according to the procedure described above, except that the fixative consisted of a 0.5% glutaraldehyde and 4% paraformaldehyde mixture in 0.1 M phosphate buffer at pH 7.2. After decalcification with EDTA as described above, the cochleas were sliced into half-turns and, along with pieces of vestibular tissue, were dehydrated through a graded series of 50, 70, 90, and 95% ethanols, and infiltrated into Lowicryl K4M at −20C as previously described (Sugiyama et al. 1992). The specimens were then embedded in Beem capsules containing K4M resin and polymerized under 360-nm wavelength UV light for 24 hr at −20C and 48 hr at RT.
Immunohistochemical Staining Procedures
An MAb (T4) raised against a fusion protein encompassing the carboxy-terminal 310 amino acids of the human colon NKCC was provided by Dr. Christian Lytle (Division of Biomedical Sciences, University of California, Riverside, CA). MAb T4 recognizes both the apical NKCC2 and basolateral NKCC1 isoforms of the Na-K-Cl co-transporter (Lytle et al. 1995).
For LM studies, immunoperoxidase staining was performed as described previously (Schulte and Adams 1989; Crouch and Schulte 1995). Rehydrated sections were incubated for 10 min in 0.3% hydrogen peroxide in absolute methanol to block endogenous peroxidases. Sections were then rinsed in 0.1 PBS at pH 7.2 and incubated for 1 hr in PBS containing 1% nonimmune horse serum (NHS). The sections were incubated overnight at 4C with MAb T4 diluted 1:80,000 with PBS-NHS. The slides were rinsed in PBS-NHS and flooded for 30 min with a 1:200 dilution of biotinylated horse anti-mouse IgG (Vector Laboratories; Burlingame, CA), rinsed in PBS, and incubated in Vectastain ABC reagent for 30 min. Sites of bound primary antibodies were visualized by monitoring the development of reaction product in 3,3′-diaminobenzidine-H2O2 substrate medium.
Nonimmune mouse serum diluted 1:80,000 was substituted for the primary antibody as a methods control. Sections from composite blocks containing a variety of tissues from a wide range of gerbil organs were processed in parallel with the inner ears as positive and negative tissue staining controls.
For EM studies, ultrathin sections of cochlear half-turns and the utricle and ampulla were cut and picked up on formvar-carbon-coated nickel grids (Ted Pella; Redding, CA). Immunogold cytochemistry was performed following procedures described in detail elsewhere (Nakazawa et al. 1995). The grids were incubated for 1 hr with 5% normal goat serum (NGS) in PBS and then reacted overnight at 4C with MAb T4 at dilutions ranging from 1:10,000 to 1:40,000 in PBS-NGS. Control sections were processed in parallel with substitution of similar dilutions of nonimmune mouse serum for primary antibody. The grids were rinsed in PBS-NGS and incubated for 2 hr at RT with a 1:20 dilution of goat anti-mouse IgG adsorbed to the surface of 10-nm colloidal gold particles (Sigma; St Louis, MO). After washes with PBS and distilled water, grids were stained with uranyl acetate for 15 min and lead citrate for 3 min and examined in a JEOL-100S electron microscope.
Reverse-transcription Polymerase Chain Reaction
Four 3-month-old Mongolian gerbils of either gender were anesthetized as described above and exsanguinated by transcardial perfusion with warm 0.9% saline solution. Inner ears and kidneys were rapidly removed, immersed in liquid nitrogen, and stored at −80C. Total RNA was obtained using acid guanidinium thiocyanate as described by Chomczynski and Sacchi (1987). cDNA was synthesized using random hexamers and Superscript reverse transcriptase (Life Technologies; Gaithersburg, MD) as described by the manufacturer. Primers specific for NKCC isoforms were designed based on published DNA sequences (Igarashi et al. 1995; Payne et al. 1995). PCR amplification was performed in a Perkin Elmer Cetus DNA Thermal Cycler (94C, 2 min; 52–60C, 30 sec; 72C, 30 sec) for 35 cycles. Products of the PCR were separated on 1% agarose gels and visualized by ethidium bromide staining. PCR products were cloned and sequenced as described previously (Crouch and Schulte 1996).
Results
Immunocytochemistry
In paraffin sections of adult gerbil inner ear, MAb T4 produced an intense immunostaining reaction in the stria vascularis and a less intense but still strong reaction in Type II fibrocytes in the inferior portion of the spiral ligament (Figure 1). Type V fibrocytes in the suprastrial region and a subpopulation of fibrocytes in the limbus were also immunoreactive (Figure 1). At higher magnification, immunostaining was clearly restricted to the basolateral plasmalemma of the strial marginal cells, whereas the entire plasma membrane of the nonpolarized fibrocytes appeared to stain (Figure 2). In the vestibular system, strong immunoreactivity was present in the basolateral plasmalemma of dark cells and along the cell membrane of select fibrocytes underlying the neurosensory epithelium (Figure 3).
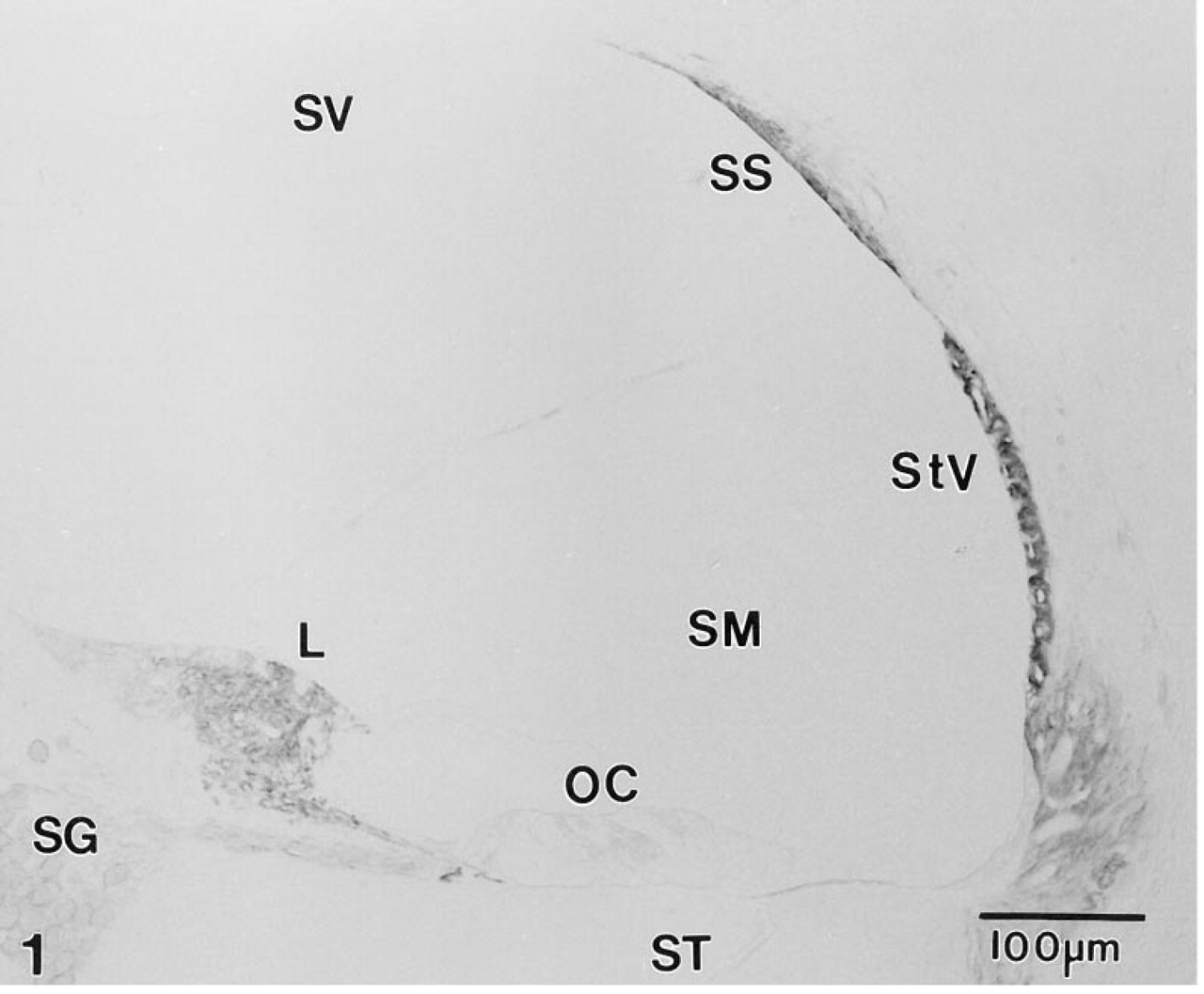
Mid-modiolar section through the lower middle turn of the gerbil cochlea immunostained with MAb T4. Immunoreactive NKCC is present in the stria vascularis (StV), and in select populations of fibrocytes in the inferior portion of the spiral ligament (SL), the suprastrial region (SS), and the spiral limbus (L). Satellite cells surrounding neurons in the spiral ganglion (SG) also show weak reactivity. Sensory hair cells and supporting cells in the organ of Corti (OC) are unreactive. SV, scala vestibuli; SM, scala media; ST, scala tympani.
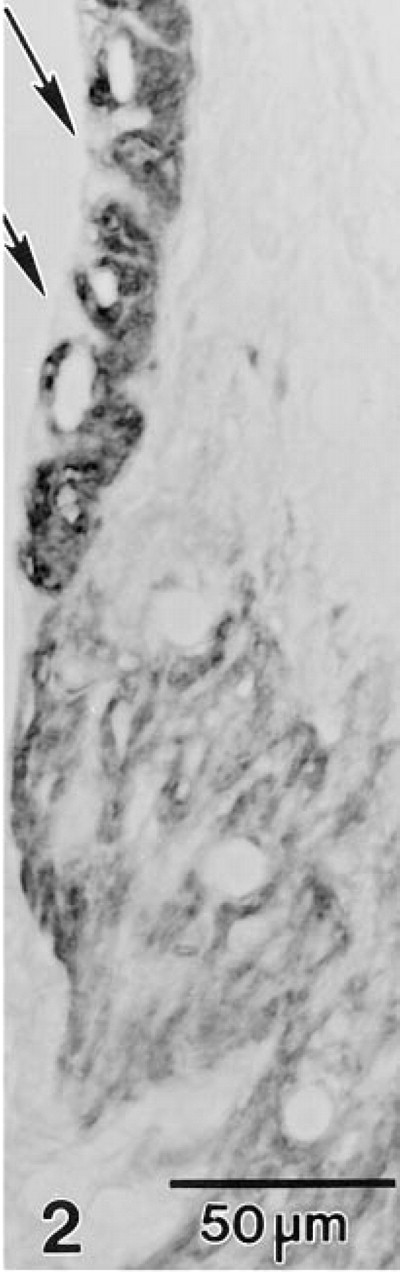
MAb T4 fails to react with the apical cell membrane (arrows) but reacts strongly with the basolateral plasma membrane of strial marginal cells. The plasma membrane of Type II fibrocytes also is heavily stained.
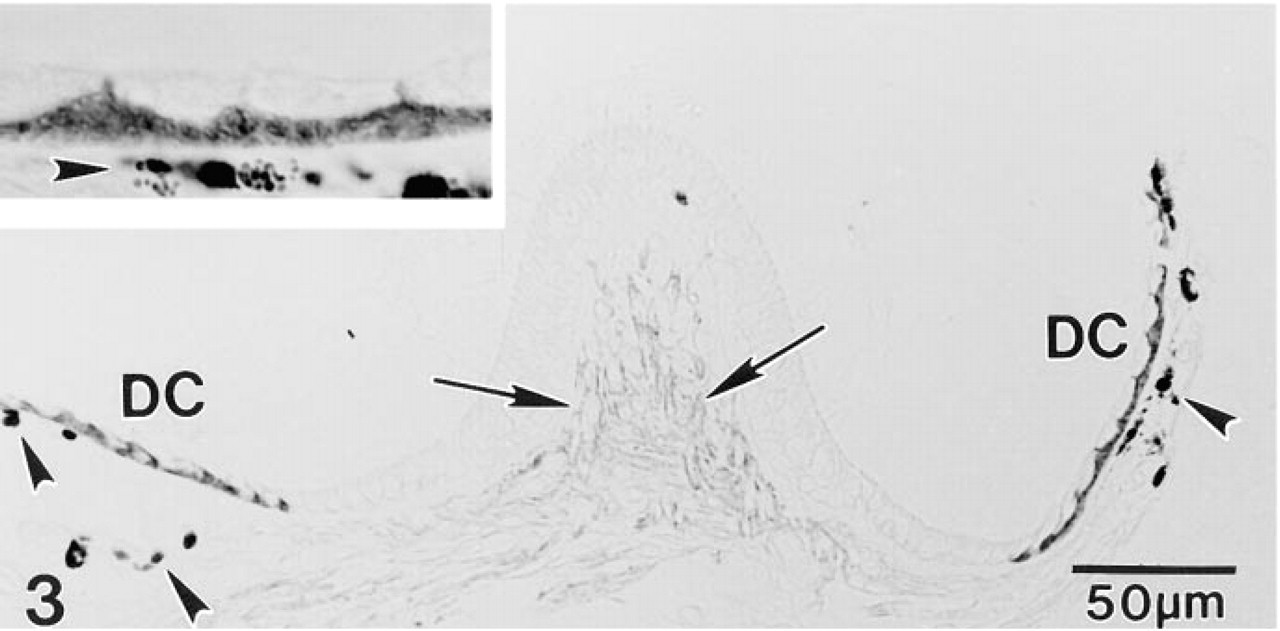
A section through the gerbil crista ampullaris illustrates abundant NKCC in the basolateral but not the apical plasma membrane of dark cells (DC) that overlie pigmented melanocytes (arrowheads). Specialized fibrocytes underlying the neurosensory epithelium are also immunopositive (arrows). Inset more clearly shows the absence of apical staining in dark cells.
For a positive tissue control, abundant NKCC was detected in the apical plasmalemma of cells lining the thick ascending limb segment of the gerbil nephron (Figure 4), in agreement with results in rabbit kidney (Lytle et al. 1995). No immunostaining was present in either the kidney control or gerbil inner ear sections when nonimmune mouse serum was substituted for MAb T4 (data not shown).
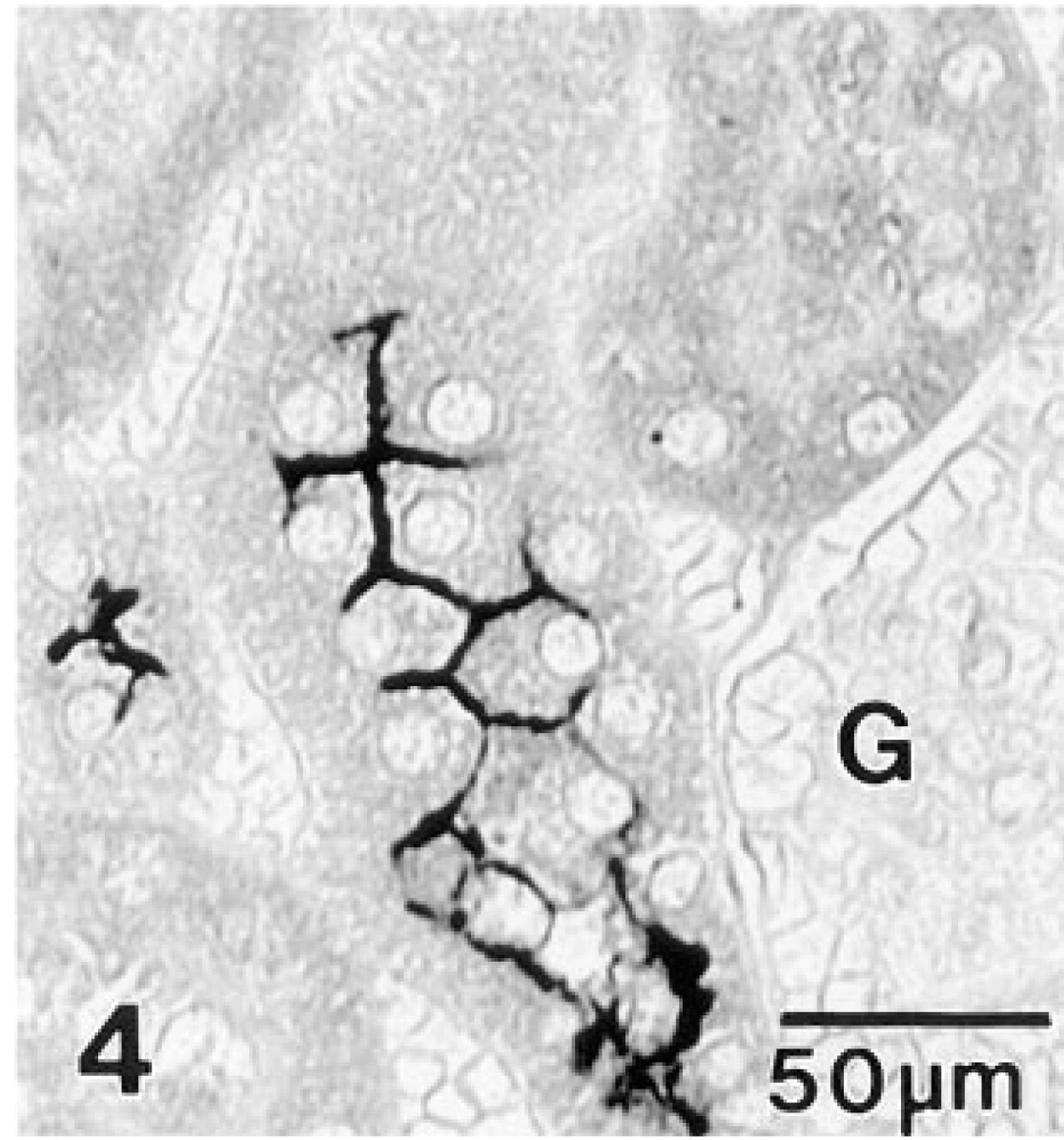
Gerbil kidney immunostained with MAb T4. Intense immunoreactivity is present along the apical plasma membrane of cells lining the ascending thick limb. Surrounding segments of the proximal convoluted tubule and a glomerulus (G) are unreactive.
At the ultrastructural level, gold spherules demonstrative of NKCC decorated the basolateral plasma membrane of strial marginal cells, whereas the apical plasmalemma was unreactive (Figure 5). Immunogold labeling of vestibular dark cells also was restricted to the basolateral plasma membrane (Figure 6).
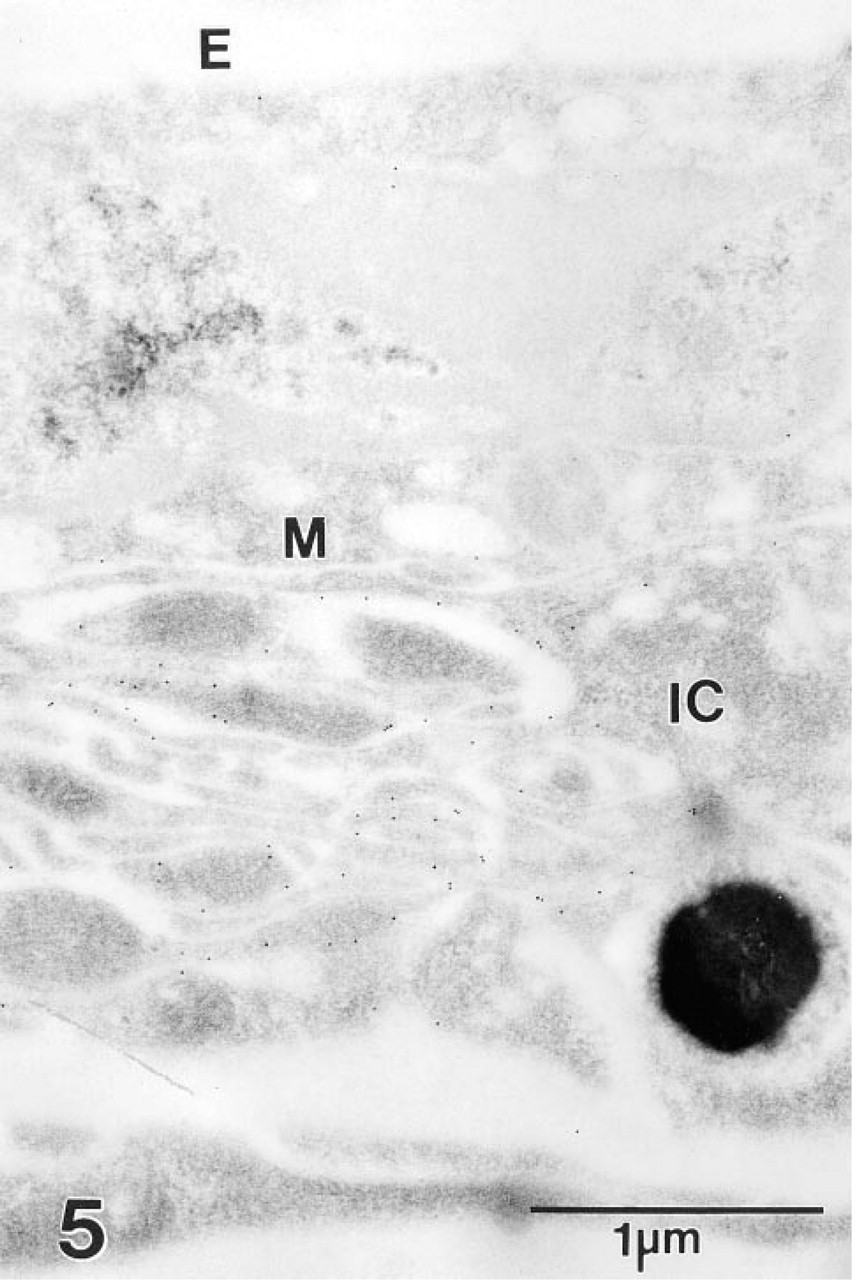
Section through the stria vascularis of the gerbil cochlea incubated with MAb T4 and second antibody adsorbed to colloidal gold. Gold particles overlie the basolateral plasma membrane of a strial marginal cell (M) but fail to decorate the cell's apical surface that borders endolymph (E). Adjacent processes of a pigment-containing intermediate cell (IC) also appear to be unreactive.
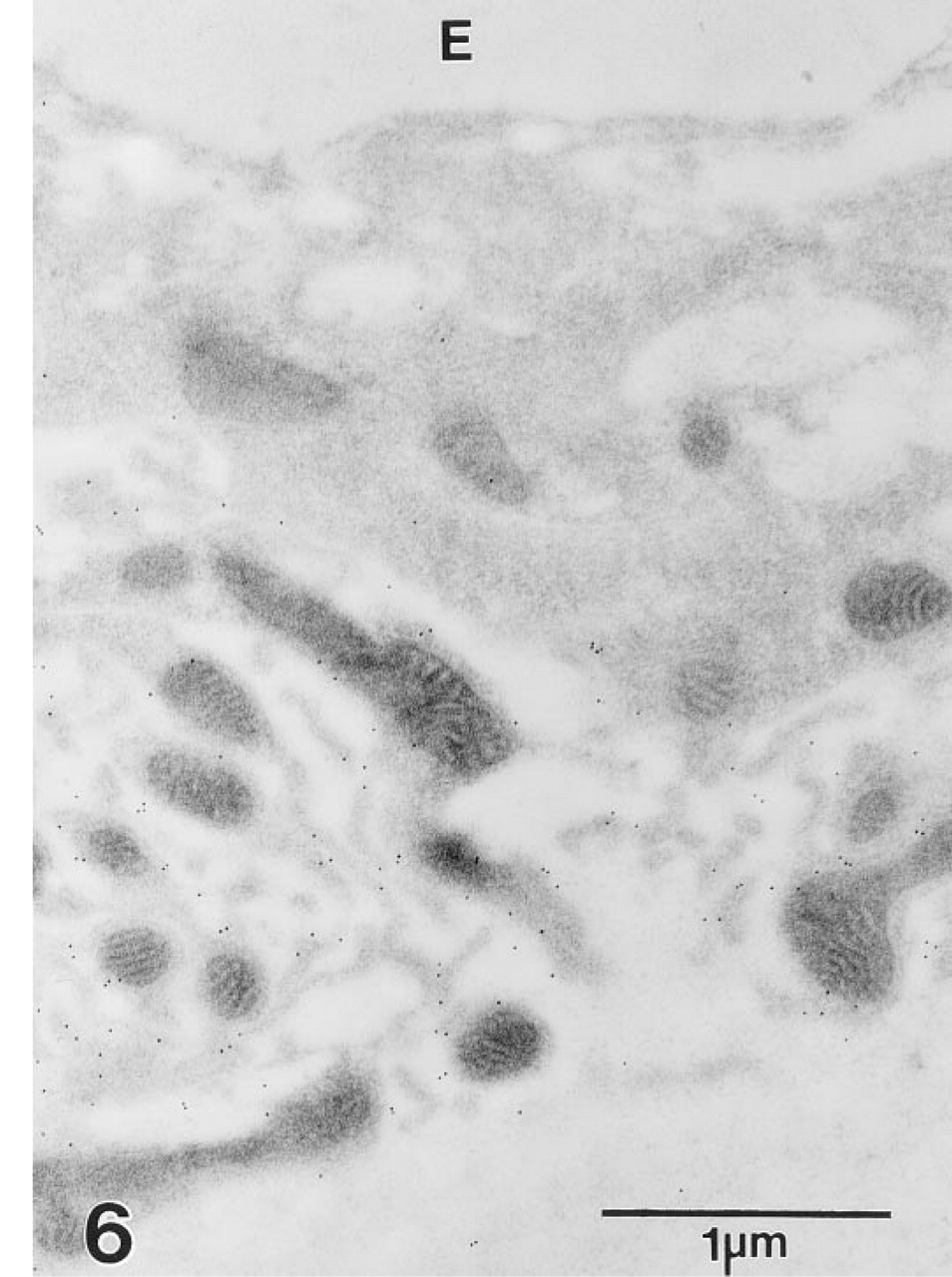
Many gold particles lie along the basolateral cell membrane of vestibular dark cells but fail to decorate the apical membrane bordering endolymph (E).
RT-PCR
Use of the NKCC1-specific PCR primers produced a 314-base pair (BP) PCR product in both the gerbil inner ear and kidney (Figure 7). The PCR products were cloned and confirmed by sequence analysis to be transcripts for NKCC1. In contrast, the NKCC2 specific PCR primers produced a 424-BP PCR product in gerbil kidney but not in the inner ear. This PCR product was cloned and identified by sequence analysis to be NKCC2.
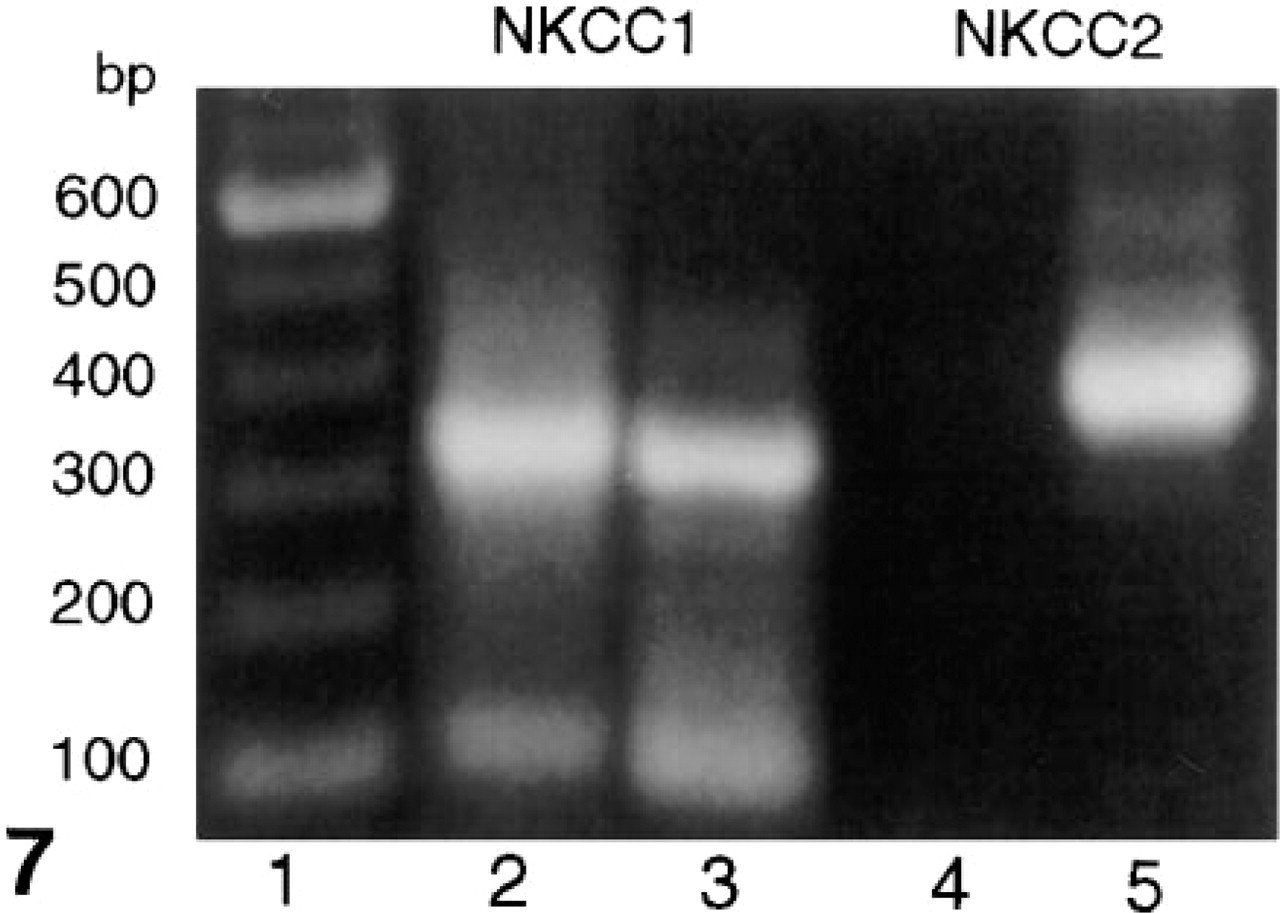
Separation of PCR products generated using NKCC1- and NKCC2-specific PCR primers on a 1% Agarose gel. Lane 1, DNA ladder; Lanes 2 and 4, gerbil inner ear; Lanes 3 and 5, gerbil kidney. bp, base pairs.
Discussion
The Na-K-Cl co-transporter (NKCC) is an integral membrane protein responsible for the coupled electro-neutral transport of 1Na:1K:2Cl across the plasma membrane in a variety of animal cells (see Haas 1994). Recent studies have identified two functionally distinct isoforms of NKCC. The basolaterally located “secretory” isoform (NKCC1) is expressed in several different tissues (Delpire et al. 1994; Xu et al. 1994; Lytle et al. 1995; Payne et al. 1995), whereas the apically expressed “adsorptive” isoforms (NKCC2) has been found only in the kidney (Gamba et al. 1994; Xu et al. 1994; Payne et al. 1995). Although MAb T4 recognizes both NKCC1 and NKCC2 (Lytle et al. 1995), the immunostaining results provided here presumably reflect the distribution of NKCC1 because only transcripts for this isoform could be detected by RT-PCR in inner ear tissues. This finding agrees with a previous RT-PCR study that failed to detect NKCC2 in the lateral wall of the rat cochlea (Hidaka et al. 1996).
The localization of the secretory isoform of NKCC to the basolateral plasma membrane of strial marginal and vestibular dark cells in the gerbil inner ear confirms previous pharmacological and physiological evidence indicating the presence of NKCC in these sites (Marcus et al. 1987, 1994; Wangemann et al. 1995). Our results also agree, in part, with unpublished immunocytochemical data localizing NKCC to the basolateral plasma membrane of strial marginal cells (Mizuta et al. 1996).
Previous biochemical, cytochemical, and pharmacological studies have demonstrated the expression of Na,K-ATPase in strial marginal and vestibular dark cells and mapped its distribution to the basolateral plasma membrane (see Salt and Thalmann 1988; Schulte and Adams 1989; Wangemann et al. 1995). The co-expression of large amounts of Na,K-ATPase and NKCC1 in this site provides a mechanism for maintaining high intracellular K+ levels for diffusion into endolymph via apically located K channels (Takeuchi et al. 1992). This activity, coupled with the recycling of Cl− via basolateral Cl channels (Marcus et al. 1993; Wangemann et al. 1995), provides an efficient mechanism for the generation of high K+ levels in endolymph necessary for mechano-electrical transduction by hair cells and for normal auditory and vestibular function.
An intriguing finding was the strong immunoreactivity observed in subpopulations of fibrocytes in the cochlea and vestibular system. A comparison of the immunostaining pattern for NKCC1 with previous data mapping the distribution of the Na,K-ATPase pump and its isoforms in inner ear tissues (McGuirt and Schulte 1994; Schulte and Steel 1994), indicates the co-localization of these two ion transport mediators in distinct subsets of inner ear fibrocytes. On the basis of their location and gap junctional connections with other cell types, these specialized fibrocytes comprise the initial segments of syncytial cellular networks believed to have evolved for resorbing K+ leaked or effluxed from endolymph through hair cell activity and releasing it near epithelial cells for recycling back to endolymph (Schulte and Steel 1994; Spicer and Schulte 1996). The demonstration of high levels of NKCC1 in these cells lends further support to a model whereby the continuous recirculation of K+ between endolymph and perilymph, at least partly through intracellular pathways, generates a standing current on which the microphonic current from acoustic stimulation is superimposed (Zidanic and Brownell 1994).
Also of interest are other cell types in the inner ear that have previously been shown to express moderate to abundant levels of immunoreactive Na,K-ATPase but do not appear to co-express the NKCC. The Na,K-ATPase-rich cell membrane of spiral ganglion neurons and their afferent nerve endings underlying the inner hair cells represent the most striking example of this non-co-expression. Other sites expressing Na,K-ATPase but failing to stain with MAb T4 include the basolateral plasma membrane of epithelial cells lining Reissner's membrane and the outer sulcus as well as that of interdental cells. Apparently these cell types express NKCC at levels below the detection limit of methods employed here or utilize an alternate mechanism(s) for generating the intracellular Na+ levels necessary to drive Na,K-ATPase activity.
Footnotes
Acknowledgements
Supported by research grants RO1 DC00713 and PO1 DC00422 from the National Institute on Deafness and Other Communication Disorders, National Institutes of Health and by American Heart Association Grant-In-Aid AHA 94015270. We thank Ms Leslie Harrelson and Ms Nancy Smythe for technical and editorial assistance.
