Abstract
SPARC is a matricellular Ca2+-binding glycoprotein that exhibits both counteradhesive and antiproliferative effects on cultured cells. It is secreted by cells of various tissues as a consequence of morphogenesis, response to injury, and cyclic renewal and/or repair. In an earlier study with Xenopus embryos we had shown a highly specific and regulated pattern of SPARC expression. We now show that ectopic expression of SPARC before its normal embryonic activation produces severe anomalies, some of which are consistent with the functions of SPARC proposed from studies in vitro. Microinjection of SPARC RNA, protein, and peptides into Xenopus embryos before endogenous embryonic expression generated different but overlapping phenotypes. (a) Injection of SPARC RNA into one cell of a two-cell embryo resulted in a range of unilateral defects. (b) Precocious exposure of embryos to SPARC by microinjection of protein into the blastocoel cavity was associated with certain axial defects comparable to those obtained with SPARC RNA. (c) SPARC peptides containing follistatin-like and copper-binding sequences were without obvious effect, whereas SPARC peptide 4.2, corresponding to a disulfide-bonded, Ca2+-binding domain, was associated with a reduction in axial structures that led eventually to complete ventralization of the embryos. Histological analysis of ventralized embryos indicated that the morphogenetic events associated with gastrulation might have been inhibited. Microinjection of other Ca2+-binding glycoproteins, such as osteopontin and bone sialoprotein, resulted in phenotypes that were unique. We probed further the structural correlates of this region of SPARC in the context of tissue development. Co-injection of peptide 4.2 with Ca2+ or EGTA, and injection of peptide 4.2K (containing a mutated consensus Ca2+-binding sequence), demonstrated that the developmental defects associated with peptide 4.2 were independent of Ca2+. However, the disulfide bridge in this region of SPARC was found to be critical, as injection of peptide 4.2AA, a mutant lacking the cystine, generated no axial defects. We have therefore shown for the first time in vivo that the temporally inappropriate presence of SPARC is associated with perturbations in tissue morphogenesis. Moreover, we have identified at least one bioactive region of SPARC as the C-terminal disulfide-bonded, Ca2+-binding loop that was previously shown to be both counteradhesive and growth-inhibitory.
E
Microinjection of anti-fibronectin antibodies into the blastocoel cavity of newt embryos inhibited gastrulation, and the use of Xenopus cells and RGD peptides in vitro confirmed that fibronectin was an adhesive substrate that promoted cell migration (Smith et al. 1990; Boucaut et al. 1984). Tenascin has also been found to be associated with mesodermal cell migration during amphibian gastrulation in vivo (Riou et al. 1990) and promotes neural crest cell migration in avian embryos (Akitaya and Bronner-Fraser 1992). Because both tenascin and thrombospondin inhibit cell adhesion to fibronectin substrates in vitro, these two large ECM glycoproteins might have counter-adhesive potential in vivo (Bornstein 1995; Chiquet-Ehrismann et al. 1988).
It has become apparent that proteins such as tenascin C, thrombospondin 1, and SPARC (secreted protein, acidic, and rich in cysteine) primarily modulate cell-matrix interactions and do not subserve structural roles in the ECM per se. This group of proteins has been termed “matricellular” and is typified by the Ca2+ and Cu2+-binding glycoprotein SPARC, a prominent component of remodeling tissues (Bornstein 1995; Lane and Sage 1994; Sage and Bornstein 1991).
We recently demonstrated that SPARC expression begins at Stage 13 (late gastrulation) in Xenopus embryos. Whole-mount in situ hybridization and immunohistochemical analysis demonstrated staining within the notochord and somites from the onset of their development (Damjanovski et al. 1994). As development progressed, SPARC RNA and protein underwent marked and rapid changes in expression during neurulation and early organogenesis. In particular, SPARC transcripts and protein were found to be enriched in cells known to undergo Ca2+-dependent adhesion and that were electrically coupled via gap junctions. The highly regulated pattern of expression indicates that SPARC has important functions in early embryonic development. This hypothesis is based in part on experiments in which microinjection of anti-SPARC antibodies into the blastocoel cavity led to major trunk defects (Purcell et al. 1993). Ectopic expression of SPARC produced comparable muscle defects in C. elegans (Schwarzbauer and Spencer 1993). Although the precise functions of SPARC during early embryonic development are not known, a variety of potential functions have been described for SPARC in vitro, e.g., inhibition of cell-substratum adhesion and cell spreading (Sage et al. 1989), binding to and/or modulation of growth factors (Hasselaar and Sage 1992; Raines et al. 1992), regulation of cell cycle progression (Funk and Sage 1991, 1993), and augmentation of interendothelial cell permeability (Goldblum et al. 1994; reviewed in Lane and Sage 1994). Furthermore, SPARC interacts with several ECM macromolecules in vitro (Tremble et al. 1993; Kelm and Mann 1991).
In this study we examined the developmental consequences of the ectopic expression of SPARC before its normal embryonic activation. We report that micro-injection of SPARC RNA, native protein, and synthetic peptides had distinct but nonexclusive developmental consequences. Our observations from a developmental system in vivo are consistent with some of the functions previously identified for SPARC in vitro.
Materials and Methods
Xenopus embryos were fertilized and were raised according to standard techniques (Wu and Gerhart 1991). Staging was according to Nieuwkoop and Faber (1956).
SPARC RNA and Protein
Full-length mouse SPARC cDNA (Mason et al. 1986) was subcloned, in both orientations, into the Pst 1 site of pGEM 4Z. Orientation was determined using restriction enzyme analysis. Both capped and poly(A)-tailed full-length sense and (control) anti-sense mouse RNAs were synthesized with Boehringer Mannheim cap-scribe and United States Biochemical poly(A) adenylation kits with the T7 plasmid promoter. Rat bone sialoprotein cDNA (a gift of Dr. Jaro Sodek, University of Toronto) was subcloned and transcribed similarly. Intact mouse SPARC protein was isolated from the conditioned medium of PYS-2 cells and was purified as previously described (Sage et al. 1989). This protein preparation was shown to disrupt barrier function in cultured endothelial cells, as described by Goldblum et al. (1994), and to inhibit endothelial and smooth muscle cell proliferation in vitro, according to Funk and Sage (1993). Porcine SPARC/osteonectin (Domenicucci et al. 1988) and porcine osteopontin (Zhang et al. 1990), obtained from Dr. Jaro Sodek, porcine bone sialoprotein (Goldberg et al. 1988), obtained from Dr. Harvey Goldberg (University of Western Ontario), bovine brain calmodulin (Calbiochem; La Jolla, CA), and bovine serum albumin (Pharmacia, fraction V; Uppsala, Sweden) were used as controls.
Immunoprecipitation
To confirm that SPARC RNA, injected 2 hr post fertilization (pf) into two-cell embryos, was translated, we co-injected [35S]-methionine. Embryos were lysed at 2 and 6 hr after injection [before the embryonic induction of SPARC at Stage 13 (Ringuette et al. 1992), 16 hr post fertilization], and protein was immunoprecipitated with a polyclonal anti-porcine SPARC antibody [previously demonstrated to bind to mouse and Xenopus SPARC (Ringuette et al. 1992; Bolander et al. 1989)] with a BRL (Gaithersburg, MD) Immunoprecipitin kit. Protein precipitates were fractionated by SDS-PAGE; the gels were subsequently fixed, dried, and exposed to X-ray film.
Synthetic Peptides
Peptides representing various regions of mouse SPARC (Figure 1), synthesized by the Howard Hughes Institute (University of Washington) or by Zymogenetics (Seattle, WA), were purified by high-performance liquid chromatography (Lane and Sage 1990). All peptides were assayed in vitro according to Lane and Sage (1990) and Funk and Sage (1993). In this study we injected peptide 2.1 [amino acids 54–73 (CQNHHCKHGKVCELDESNTP)] corresponding to the N-terminal end of the cysteine-rich Domain II; peptide 2.3 [amino acids 113-130 (TLEGTKKGHKLHLDYIGP)] representing the C-terminal end of Domain II, and peptide 4.2 [amino acids 254–273 (TCDLDNDKYIALEEWAGCFG)], representing the C-terminal EF-hand Domain IV of SPARC (Sage et al. 1995). Mutant peptide 4.2K (identical to peptide 4.2 except for an aspartic acid to lysine substitution at amino acid 258) and peptide 4.2AA (also identical to peptide 4.2 except for two cysteine to alanine substitutions at amino acids 255 and 271) were also microinjected into the embryos.
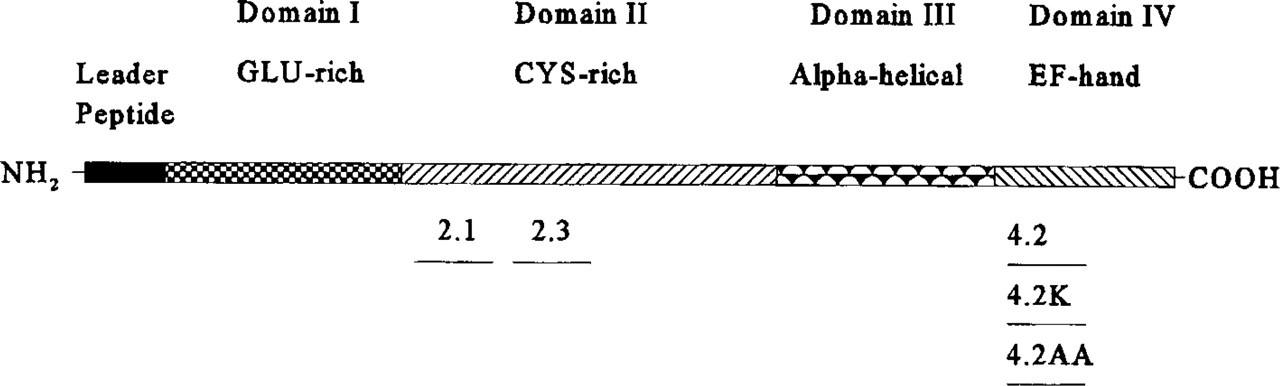
Diagrammatic representation of SPARC domains and position of synthetic peptides used for microinjection. SPARC can be subdivided into four distinct structural domains. The N-terminal glutamic acid-rich Domain I can bind with low affinity up to eight Ca2+ ions (Kd 10-3-10-5 M); SPARC is therefore likely to be sensitive to changes in ECM Ca2+ levels. Domain II contains a follistatin-like module with 10 conserved Cys residues and two Cu2+-binding sites. Domains III and IV, representing the C-terminal half, both contain a series of α-helical segments. In addition, Domain IV has a unique disulfide bridge and a Ca2+-binding EF hand, which can bind one Ca2+ with a Kd 10-7 M. Peptide 2.1 [amino acids 54-73 (CQNHHCKHGKVCELDESNTP)] corresponds to the N-terminal end of Domain II. Peptide 2.3 [amino acids 113-130 (TLEGTKKGHKLHLDYIGP)] is C-terminal to peptide 2.1. Peptide 4.2 [amino acids 254-273 (TCDLDNDKYIALEEWAGCFG)] represents the EF-hand of Domain IV. Mutant peptides 4.2K and 4.2AA are identical to peptide 4.2 except for an Asp Lys substitution at amino acid 258 and two Cys Ala substitutions at amino acids 255 and 271, respectively.
Microinjection
Embryos were injected with glass needles prepared with a Narishige glass needle puller and a Narishige forced-gas system. Stage 2 (RNA injections, 2 hr pf) and Stage 8 (protein and peptide injections, 8 hr pf) Xenopus embryos in 100% Steinberg's solution with 3% Ficoll were injected with 5–200 pg of RNA (in 5–50 nl water) into one cell of a 2-cell embryo. Between 10 and 1000 ng (in 10–300 nl PBS) of protein was injected into the blastocoel cavity of Stage 8 embryos. Embryos were healed for several minutes in 100% Steinberg's solution before transfer into 20% Steinberg's solution.
Whole-mount Immunohistochemistry
Embryos were immersed for 2 hr in Dent's fixative (25% dimethylsulfoxide in methanol), washed several times in PBS, and incubated with a neural-specific 2G9 antibody (1:250 dilution) overnight at 4C (Jones and Woodland 1989). After several washes with PBS, the neural tissue distribution was visualized with an anti-mouse alkaline phosphatase-conjugated secondary antibody reacted with NBT and BCIP. Embryos were immersed in Bouin's fixative, dehydrated in methanol, and cleared in benzyl benzoate and benzoic acid before photography.
Histology
Embryos were fixed for 2 hr in 4% formalin, washed several times in PBS, and dehydrated in ethanol. Dehydrated embryos were embedded in Paraplast, sectioned (10 μm), and placed on collagen-coated slides. Sections were stained with hematoxylin and eosin according to standard protocols (Kelly et al. 1991) and were mounted with Permount for photography.
Results
Generation of Unilateral Defects by Microinjection of SPARC RNA into Early Blastomeres
Microinjection of 5-25 ng of RNA had no effects on development, whereas microinjection of 200 ng of RNA resulted in death in greater than 75% of the embryos. Microinjection of 100 pg of capped and poly(A)-tailed mouse SPARC sense RNA into one cell of 2-cell embryos (Table 1) resulted in defects restricted to one side of some embryos (Figures 2A-2D). Co-injection of rhodamine dextran sulfate revealed that the affected region corresponded to the progeny of the injected blastomere (data not shown). Eyes were either small and deformed (arrow, Figure 2A) or absent, on the left or right side (Figure 2B vs 2C), coincident with the blastomere injected. Defects were also observed within the trunk. The absence of somites on one side resulted in embryos with severely curved axes (triangles, Figures 2A and 2D). Histological analysis revealed somite malformations that corresponded with the kinked axis (Figure 2E). More severe axial defects (Figure 2D) were associated with malformed axial features, such as notochords, identified histologically (Figure 2E). Control SPARC anti-sense RNA-injected embryos (Table 1) were normal, both morphologically (Figure 2F) and histologically (Figure 2G). Similarly, control BSP sense or anti-sense RNA-injected embryos were normal (Table 1).
To confirm that injected SPARC sense RNA was translated into SPARC protein, we immunoprecipitated labeled embryonic protein with SPARC antibody 2 and 6 hr after RNA injections and 4-8 hours before embryonic activation of SPARC RNA occurred. The Mr of SPARC on SDS-polyacrylamide gels is 43,000. [35S]-Methionine injected alone or with anti-sense RNA was not associated with a band of Mr 43,000 in embryos up to 6 hr after injection (Figure 3, Lanes A and B). After injection of [35S]-methionine with sense RNA, two bands were immunoprecipitated (at both 2 and 6 hr post injection) (Figure 3, Lanes C and D). The upper bands, with an estimated molecular weight of 43 kD, matched the molecular weight expected for SPARC. The lower molecular weight band (middle band; Figure 3, Lanes C and D) probably represents a degradation product of SPARC. An unidentified band present in all lanes corresponds to a nonspecific reactivity of the polyclonal SPARC antibody.
Summary of macromolecules injected and their phenotypic effects a
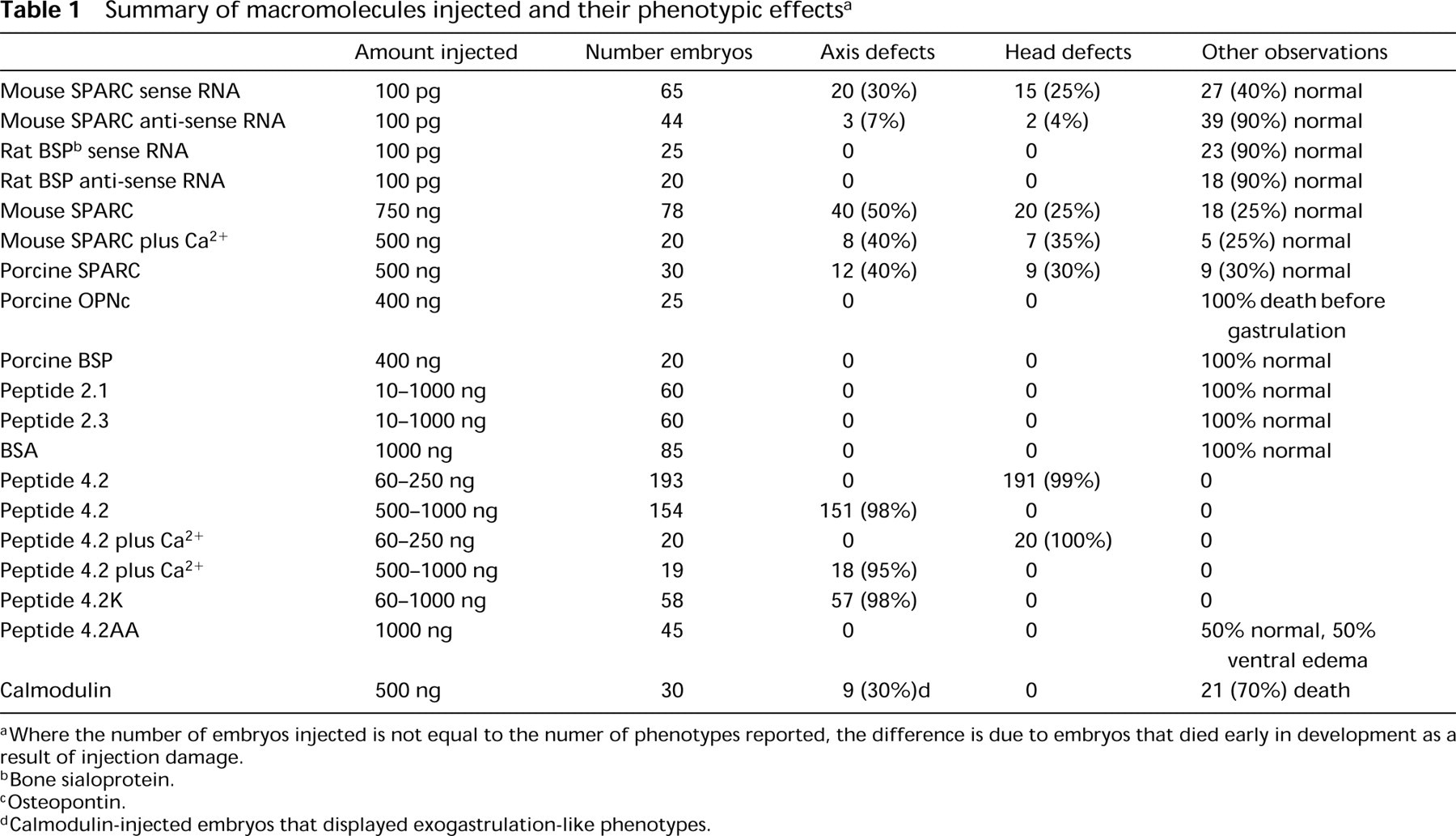
aWhere the number of embryos injected is not equal to the numer of phenotypes reported, the difference is due to embryos that died early in development as a result of injection damage.
bBone sialoprotein.
cOsteopontin.
dCalmodulin-injected embryos that displayed exogastrulation-like phenotypes.
Microinjection of Mouse SPARC into the Blastocoel Cavity Causes a Broad Spectrum of Defects
Control uninjected embryos and those injected with protein initiated gastrulation at the same time and appeared to progress through gastrulation and early neurulation in a similar manner, as judged by the morphological criteria of blastopore and neural tube formation. However, shortly after neurulation a range of defects was apparent for the embryos injected with SPARC: (a) approximately 25% of the embryos (20 of 78) had minor abnormalities in head structure and defective truncated axes by stage 28 (Figure 4A); (b) 50% (40 of 78) appeared to have a broad range of severe head and axial defects (Figure 4B); and (c) 25% of injected embryos appeared unaffected (see also Table 1). Head defects included a lack of eyes and reduced cement glands (arrows, Figures 4A and 4C). To confirm that observed effects were not unique to mouse SPARC, we also injected porcine SPARC. Amino acid analysis of SPARC, based on cDNA sequences, has demonstrated a 90% amino acid identity among mammals and 80% identity between Xenopus and mammals (Damjanovski et al. 1992). The effects of porcine SPARC (Figure 4C) were very similar to those of mouse SPARC (Table 1; Figures 3A and 3B). Sagittal sections of embryos with defective axes confirmed the absence of anterior structures and a lack of tissue differentiation, with the exception of a stacked columnar cell aggregate with cement gland-like appearance (arrow, Figures 4D and 4E). Within the trunk region of some of these embryos, a notochord-like structure could also be identified (Figure 4E).
SPARC has been demonstrated to bind to a variety of macromolecules (Lane and Sage 1994). Therefore, developmental defects generated by precocious over-expression of SPARC might have been due to nonspecific chelation of macromolecules that play critical roles in early embryonic development. To address this concern, we injected equivalent amounts of two structurally-related Ca2+-binding glycoproteins into the blastocoel cavity. Embryos injected with porcine osteopontin were arrested in development before gastrulation, whereas no developmental defects were observed after injection of porcine bone sialoprotein (Table 1; and M. Ringuette, unpublished experiments).
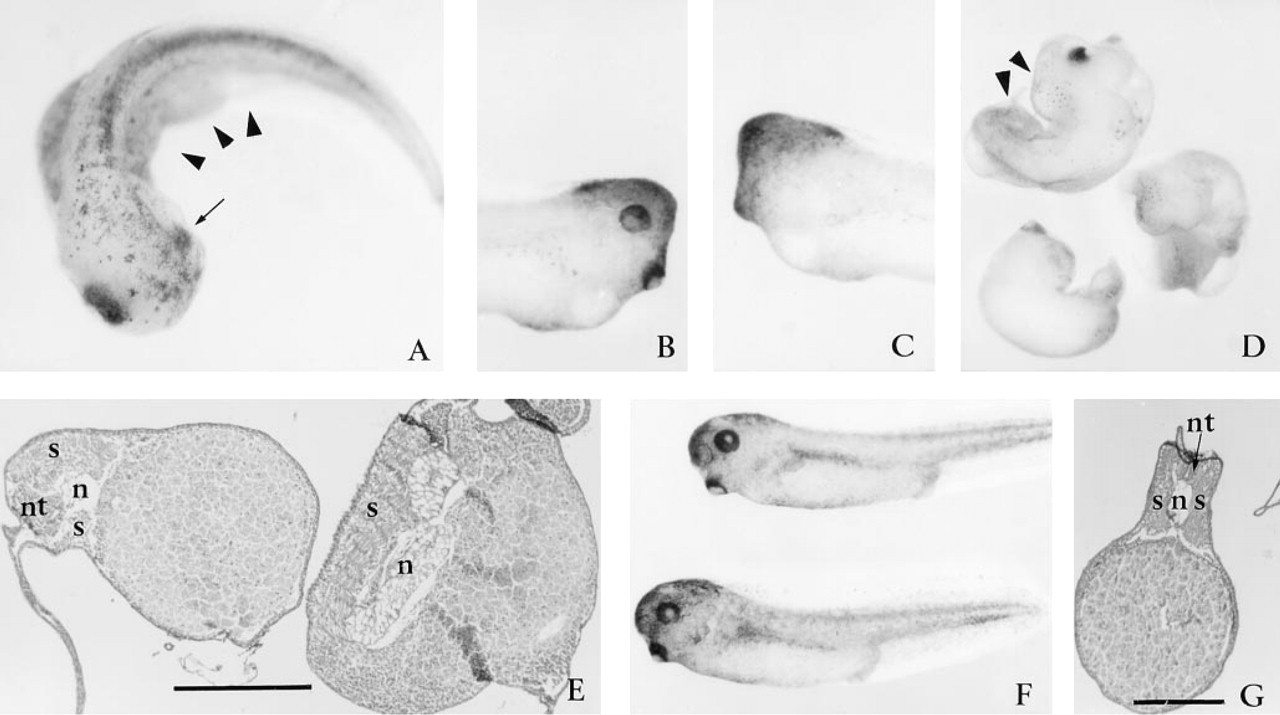
Injection of SPARC RNA into early blastomeres is associated with head and axis defects. Microinjection of 100 pg capped and poly(A)-tailed mouse SPARC sense RNA into one cell of a two-cell embryo resulted in the reduction of axial structures on the injected side. These defects included a reduced eye on one side of the embryo (
Because the N-terminal, glutamic acid-rich region of SPARC can bind up to 8 Ca2+ with Kd ranging from 10-3–10-5 M, we were also concerned that the effects shown in Figure 2 might have been due to the chelation of extracellular Ca2+ by SPARC; both inter-and intracellular Ca2+-mediated pathways would thus be compromised. However, pre-incubation and coinjection of SPARC with 0.3-10 mM CaCl2 did not alter the results (Table 1). Because the blastocoel cavity of Xenopus embryos is known to contain 1.5 mM free Ca2+, we also tested for Ca2+-dependent effects of SPARC by the injection of SPARC with EGTA. Co-injection with 0.3-3 mM EGTA did not interfere with the phenotypic effects of SPARC. However, further increases in EGTA concentration (with or without SPARC) led to embryonic death as a result of the dissociation of blastomeres, which depend on cadherins for their cohesion (Angres et al. 1991).
Because the conformation of SPARC is labile (even at – 70C), increasing doses of SPARC were required over time to obtain consistent results. Therefore, the data presented were generated by microinjection of 80-750 ng of SPARC. Similar results with respect to phenotypes were also obtained with 300 ng of porcine SPARC microinjected as described.
Microinjection of Synthetic Peptides
Synthetic peptides corresponding to Domains II and IV of SPARC (Figure 1) were found to mimic some of the major biological effects of native SPARC on tissue culture cells (Sage et al. 1995; Lane and Sage 1990). Therefore, we sought to determine whether these peptides could mimic the developmental changes associated with the microinjection of native SPARC.
Microinjection of Peptides 2.1 and 2.3 Does Not Interfere with Embryonic Development
Peptide 2.1, representing the N-terminal region of the cysteine-rich, follistatin-like Domain II (Figure 1), has been shown to arrest endothelial cells and fibroblasts at the G1 stage of the cell cycle (Lane and Sage 1994). Surprisingly, no effects on development were observed when 10–1000 ng of peptide 2.1 was injected into Xenopus embryos (Table 1; Figure 5C). Similar results (Figure 5D) were also obtained with peptide 2.3, a cationic peptide C-terminal to peptide 2.1 (Figure 1). Injection of up to 1000 ng of BSA (Figure 5A) or up to 300 nl of PBS (Figure 5B) had no apparent effects on development. Sections of control uninjected, PBS-, BSA-, peptide 2.1-, or peptide 2.3-injected embryos were indistinguishable (data not shown).
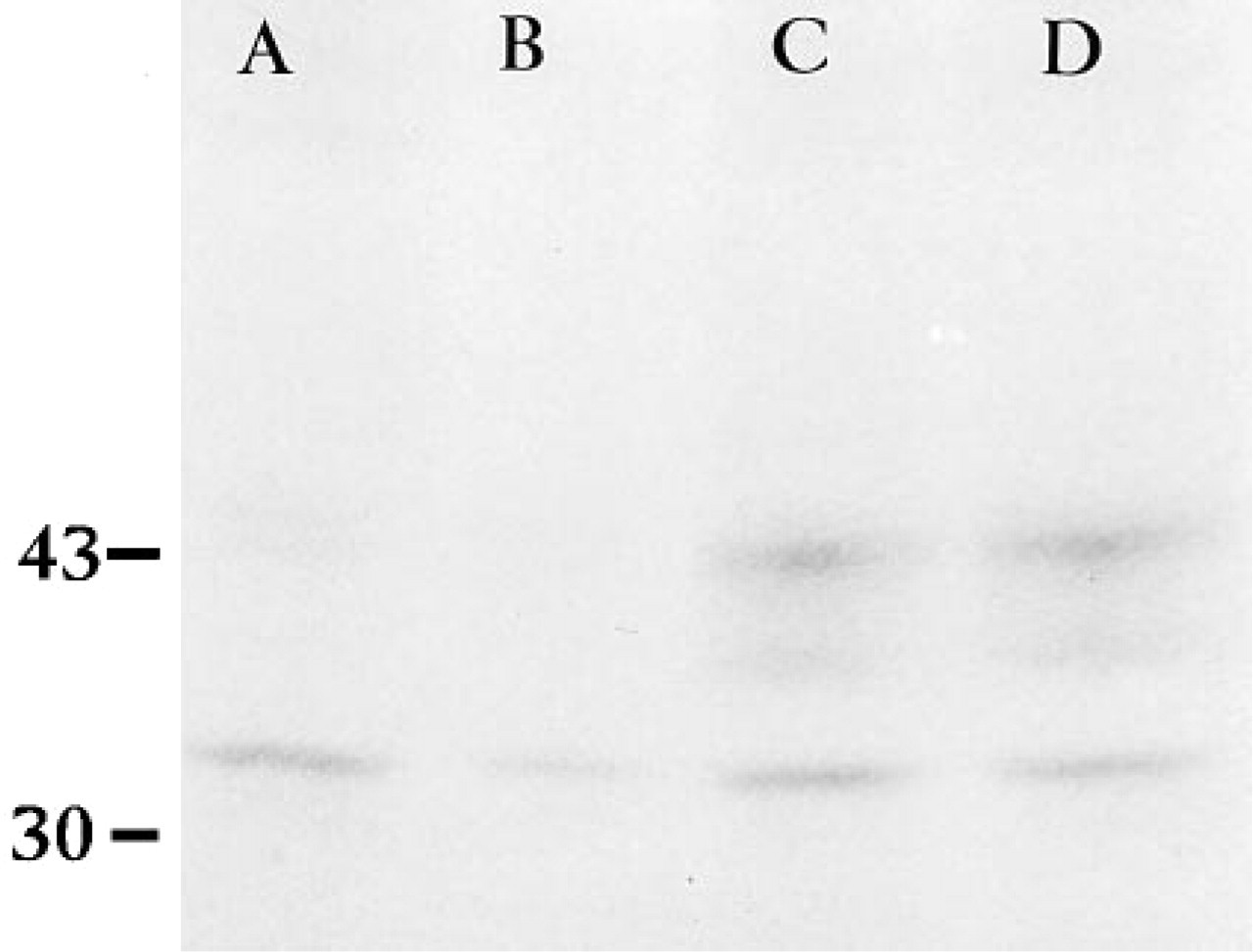
Translation of microinjected SPARC sense RNA. An auto-radiograph of [35S]-methionine-labeled protein extracts immunoprecipitated with anti-SPARC IgG and fractionated by SDS-PAGE. Lane A, control [35S]-methionine injected, and Lane B, [35S]-methionine, anti-sense SPARC RNA co-injected embryos did not express SPARC-like protein after 6 hr. In contrast, co-injection of [35S]-methionine with sense SPARC RNA resulted in the detection of a SPARC-like protein at 2 hr (Lane C) and 6 hr (Lane D), post injection. Molecular weight standards of 43 kD (ovalbumin) and 30 kD (carbonic anhydrase) are shown.
Abnormalities Associated with Injection of Peptide 4.2
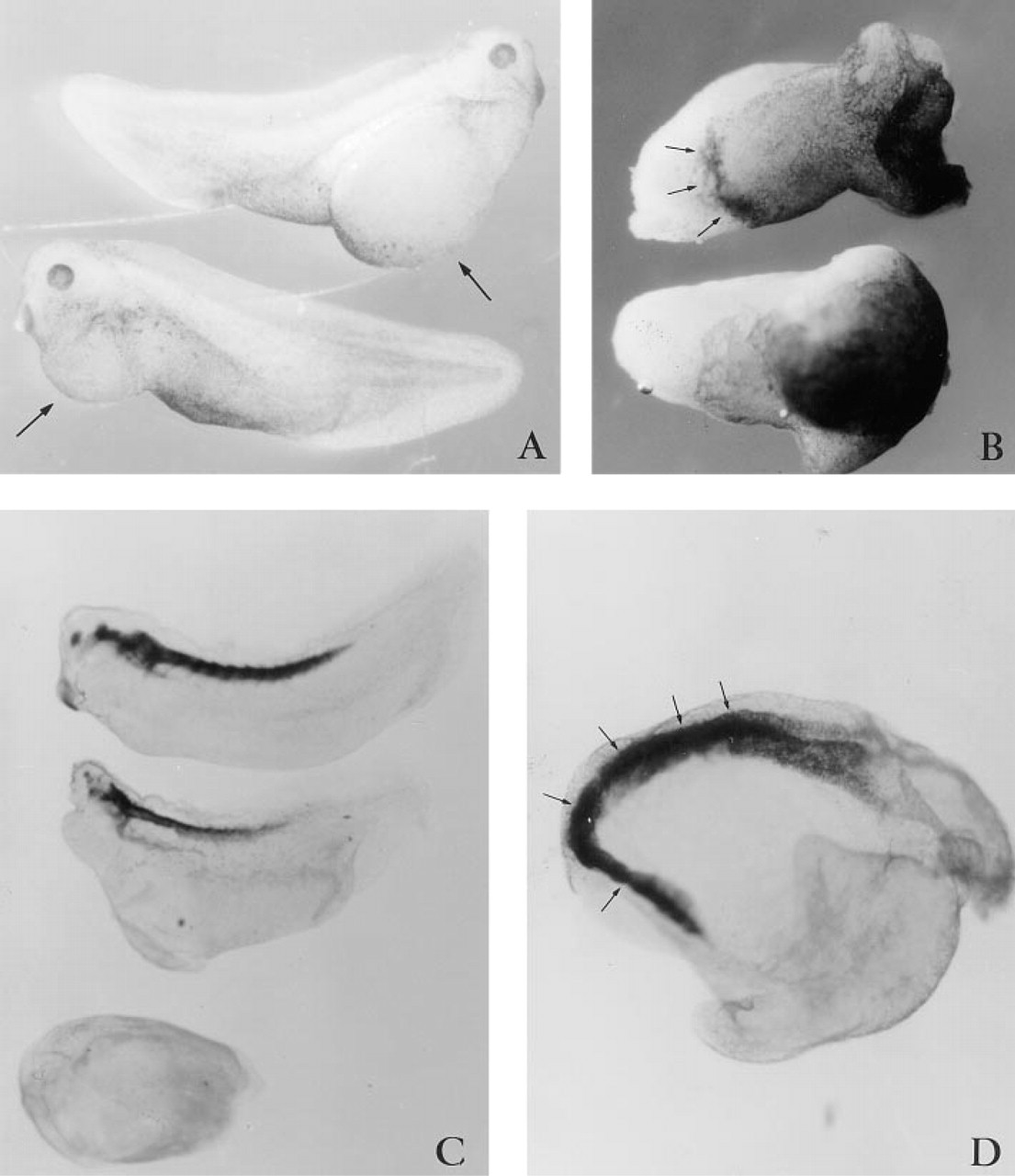
Microinjecting 60 ng (25 pM) of peptide 4.2, representing the Ca2+-binding EF-hand domain of SPARC (Figure 1), generated dorso-anterior defects. Virtually all embryos (63 of 65) had small heads, with abnormal eyes and cement glands by Stage 30 (Table 1; Figure 6A). Also visible were large ventral swellings, which sometimes collapsed to form highly convoluted surface folds. The trunk of the embryos appeared to have developed normally. Increasing the level of peptide 4.2 to 100 ng (40 pM) led to a complete absence of head development in 100% (58 out of 58) of the embryos (Table 1; Figure 6B). More than 90% of these embryos had relatively normal dorsal axes and continued to develop well into the late tailbud (Stage 50), and died shortly thereafter (Figure 6C). Histological sections revealed that otic vesicles (Figure 7D) were the first anterior tissues formed and thus confirm that anterior development was truncated (Figure 7A-7G). The neural tube and notochord were always present (Figure 7F), but differed in their anterior progression. Use of the neural 2G9 antibody, however, revealed that neural tissue did indeed reach the extreme anterior of most embryos (Figure 8C, middle embryo).
The above results indicated that there was a concentration-dependent decrease in anterior to posterior development associated with the microinjection of peptide 4.2. This observation was confirmed by microinjection of 250 ng (100 pM) peptide 4.2. All of the 70 injected embryos lacked heads and had significantly shorter dorsal axes (Table 1; Figure 6D). Histological analysis revealed that the otic vesicles were also missing from the anterior region (Figures 7H-7J). The somites, notochord, and neural tube were always present but often did not span the entire length of the dorsal axis as is characteristic of normal embryos. Frequently, transverse sections of the extreme anterior end of the axis revealed that the axial ridge was composed of only somite-like cells (Figure 7I). Posterior sections also revealed a loosely organized neural tubelike structure (Figures 7I and 7J). Similarly, the notochord frequently failed to extend into the anterior region (Figures 7H and 7I).
Increasing the concentration of microinjected peptide 4.2 from 250 ng to 500 ng (200 pM) or 1000 ng (400 pM) resulted in more severe axial defects (Figures 6E and 6F; Table 1). The increased volume required to microinject larger amounts (due to constraints on solubility) caused significant swelling of the blastocoel. For many embryos the swelling caused by injection was so significant that external signs of gastrulation could not be seen. However, by the time sibling control embryos had reached Stage 35 (tailbud), these embryos had formed an axial ridge (Figure 6F). The ridges spanned about one fifth of the circumference of the embryos. Embryonic death occurred shortly thereafter. Antibody 2G9 revealed no neural tissues within these embryos (bottom embryo, Figure 8C). As we had observed with native SPARC, pre-incubation of peptide 4.2 with 0.3-10 mM CaCl2 or 0.3-3.0 mM EGTA did not alter these results (Table 1).
Effect of Peptide 4.2 Mutants
To determine whether binding of Ca2+ to the EF-hand contributed to the effect of peptide 4.2 on development, we injected peptide 4.2K in which an aspartic acid was substituted by a lysine (Sage et al. 1995). Peptide 4.2K displays a significantly reduced affinity for Ca2+ (Sage et al. 1995). No differences between the effects of peptides 4.2 and 4.2K were observed in intact or sectioned embryos. Injection of 100-1000 ng (40-400 pM) of peptide 4.2K (Figure 6G and 6H) or wild-type peptide 4.2 (Figures 6B and 6E) generated near by identical dose-dependent effects (see also Table 1).
An unusual feature of the EF hand of SPARC is that it is stabilized by a disulfide bridge. To determine whether this disulfide bond contributed to the biological activity of peptide 4.2, we microinjected peptide 4.2AA, in which the two cysteine residues of peptide 4.2 had been replaced by alanines. Microinjection of up to 500 ng peptide 4.2AA did not interfere with development. Embryos injected with 1000 ng underwent normal development until the tailbud stage, at which time ventral swellings became prominent (Table 1; Figure 8A). The effect (or cause) of this phenotype is unclear, because these embryos were otherwise morphologically normal and had intact head and axial structures.
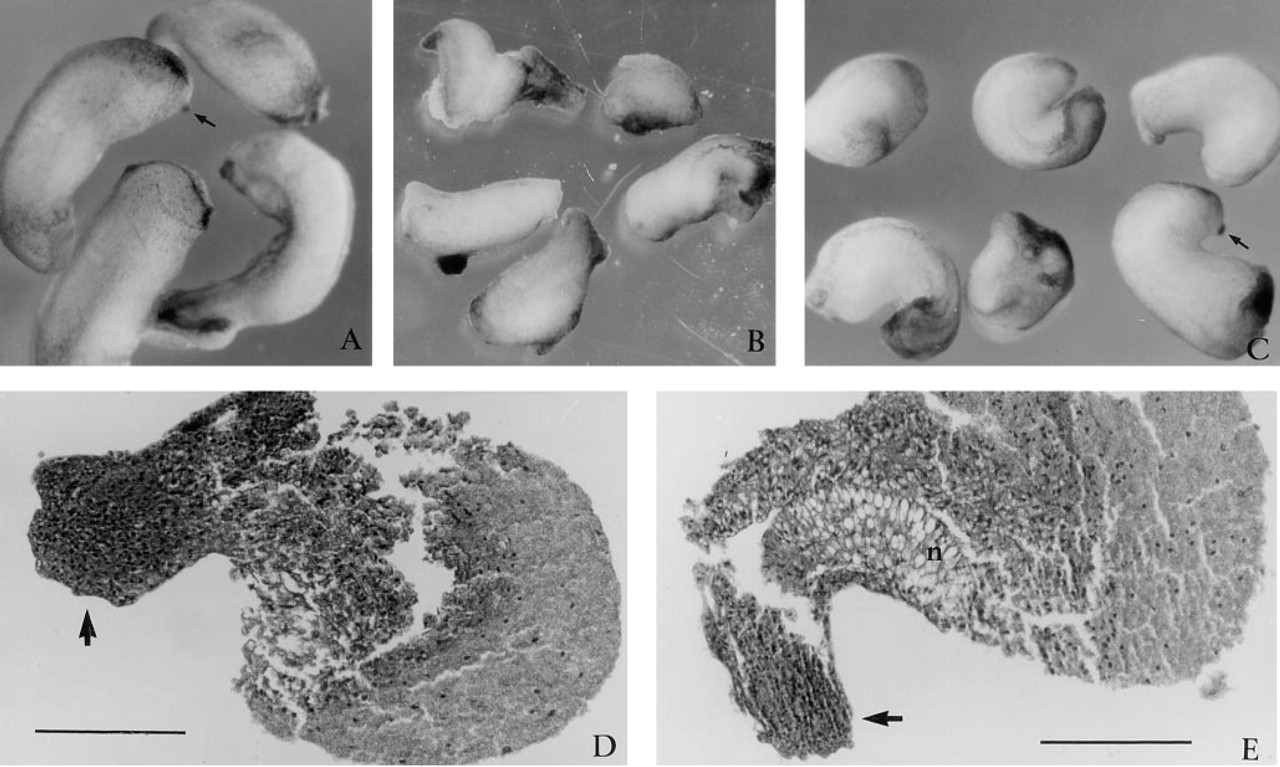
Microinjection of SPARC protein into the blastocoel cavity leads to severe axial defects. Injection of mouse SPARC into a Stage 8-9 blastocoel cavity resulted in reduction of head structures, including reduced cement glands (arrows in
As an additional control, a protein with four Ca2+-binding EF hands (bovine brain calmodulin) was also injected. Injection of up to 200 ng (10 pM) calmodulin did not interfere with development. Injection of 500-1000 ng (25-50 pM) led to embryonic death in 20 of 30 injected embryos by the time sibling control embryos reached late gastrulation. The remaining 10 injected embryos underwent an exogastrulation-like process (Table 1; Figure 8B), and appeared as endodermal masses that were capped by ectoderm (arrows, Figure 8B). Antibody 2G9 revealed that the ridge of the ectodermal cap was neural tissue (arrows, Figure 8D).
Discussion
Whole-mount in situ hybridization and immunohistochemical analysis demonstrated that SPARC was expressed by Xenopus embryos at Stage 13, from the onset of the formation of the notochord and somites (Damjanovski et al. 1994). Rapid changes occurred in the level and spatial distribution of SPARC throughout the neurulation and tailbud stages of development. In this study we show that precocious ectopic expression of SPARC affects Xenopus development. Microinjection of capped and poly(A)-tailed full-length mouse SPARC RNA into one blastomere of 2-cell embryos (Stage 2, 14 hr before the expression of endogenous SPARC occurred) often generated unilateral defects. For example, eyes were usually absent or defective only on the injected side. In addition, some embryos had both dorso–anterior and trunk defects only on one side. Microinjection of SPARC peptides resulted in a similar phenotype, although the effects were bilateral.
In Xenopus, eye development (the first anterior organ formed) is affected by a wide variety of treatments, including injection of anti-gap junction antibodies into blastomeres of 8-cell embryos (Warner et al. 1984) and injection of reagents that affect dorsal–anterior development (e.g., retinoic acid, Xgsk, Xwnt-8, trypan blue, heparin, suramin) (reviewed in Kao and Danilchuk 1991). Embryos with defective eyes or lacking eyes have also been generated by microinjection of anti-SPARC antibodies into the blastocoel cavity (Purcell et al 1993). Moreover, in situ and immunohistochemical studies have demonstrated that SPARC is a major component of developing retina in chicken, bovine, and monkey embryos (M. Ringuette, Q. Yan, H. Sage, A. Hendrickson, unpublished experiments). Although eye development in Xenopus is sensitive to a variety of experimental perturbations, several factors could account for the observations reported here. Lineage tracing, separation, and perturbation experiments indicate that the three body axes (anterior–posterior, dorsal–ventral, and left–right) are linked developmentally (reviewed in Almirantis 1995). It is therefore likely that their formation is in part influenced by common axis determinants. SPARC has been shown to modulate the activity of growth factors (Lane and Sage 1994; Raines et al. 1992). Therefore, binding of SPARC to inducing factors could interfere with the specification of embryonic tissues that would have occurred later in development. That the injection of other extracellular glycoproteins (osteopontin and bone sialoprotein) resulted in unique phenotypes indicates that nonspecific chelation of macromolecules by these acidic proteins was not a causal factor. However, it remains to be established if SPARC binds to and modulates the activity of inducing factors in early Xenopus embryos. We are now investigating whether or not SPARC has an affinity for, and/or interferes with, other known embryonic inducing factors.
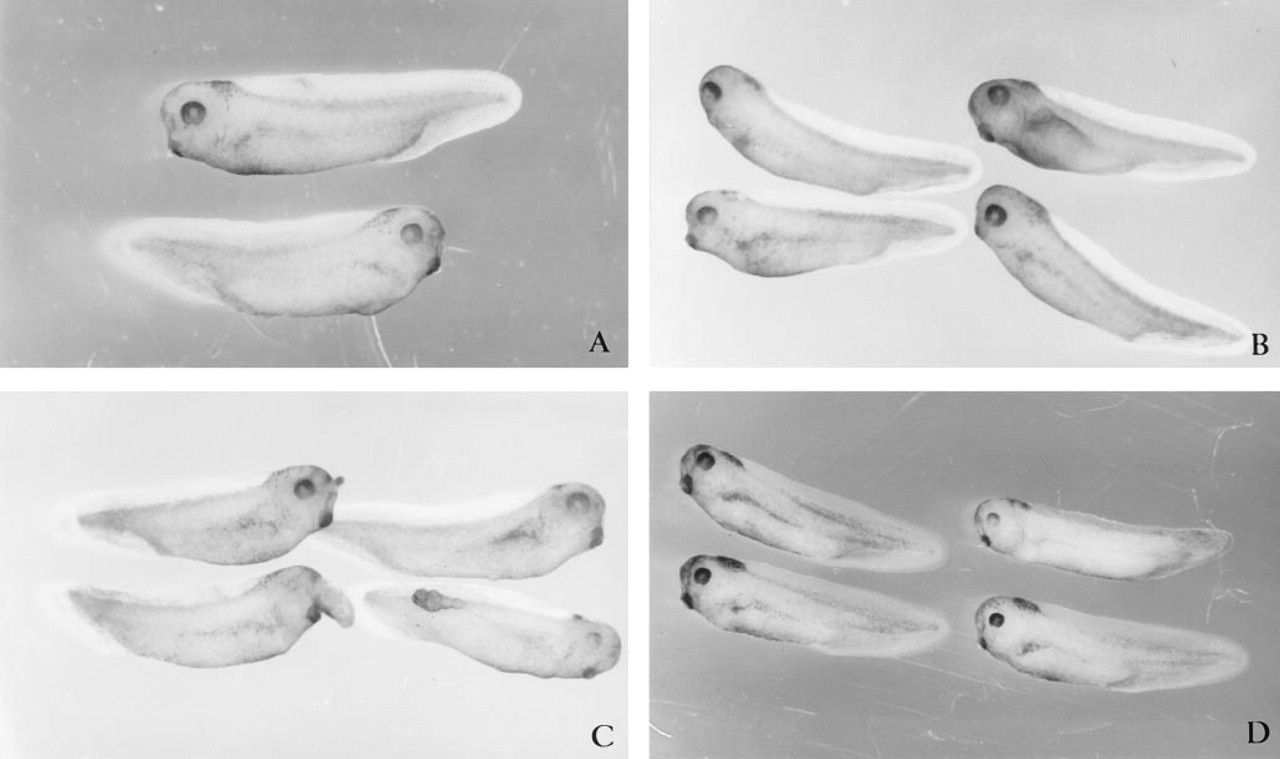
Microinjection of SPARC cysteine domain peptides does not affect development. Control microinjections of 300 nl PBS (
Microinjecting SPARC into the blastocoel cavity (a region into which proteins can be injected within a large ECM cavity) before gastrulation enabled us to assess the impact of the precocious presence of SPARC (a) just before the start of major morphogenetic movements, (b) while major cell-cell inductive events were still occurring, and (c) 8–9 hr before endogenous expression of SPARC began. As with the microinjection of SPARC RNA, a variety of head and tail defects were observed, e.g., defective cement gland and eyes, somite misalignment, axis bending, and ventral swelling. However, in contrast to the microinjection of RNA, bilateral defects were observed. A plausible explanation is that the injected SPARC was distributed evenly within the blastocoel cavity. Because Xenopus embryos do not begin to express SPARC until midgastrulation and SPARC does not accumulate to any significant levels until neurulation (Damjanovski et al. 1994), the defects generated here were probably due to the precocious presence of SPARC before its endogenous activation. Of concern was that the effects could have been due to sequences and structures unique to murine SPARC (amphibian SPARC has not been purified nor characterized functionally). Because SPARC is a highly conserved protein [90% amino acid identity among mammalian species and 80% between Xenopus and mammals (Damjanovski et al., 1992)], it is likely that common functions would be shared in different organisms. Indeed, when we microinjected porcine SPARC, the defects generated were very similar to those obtained with the murine protein.
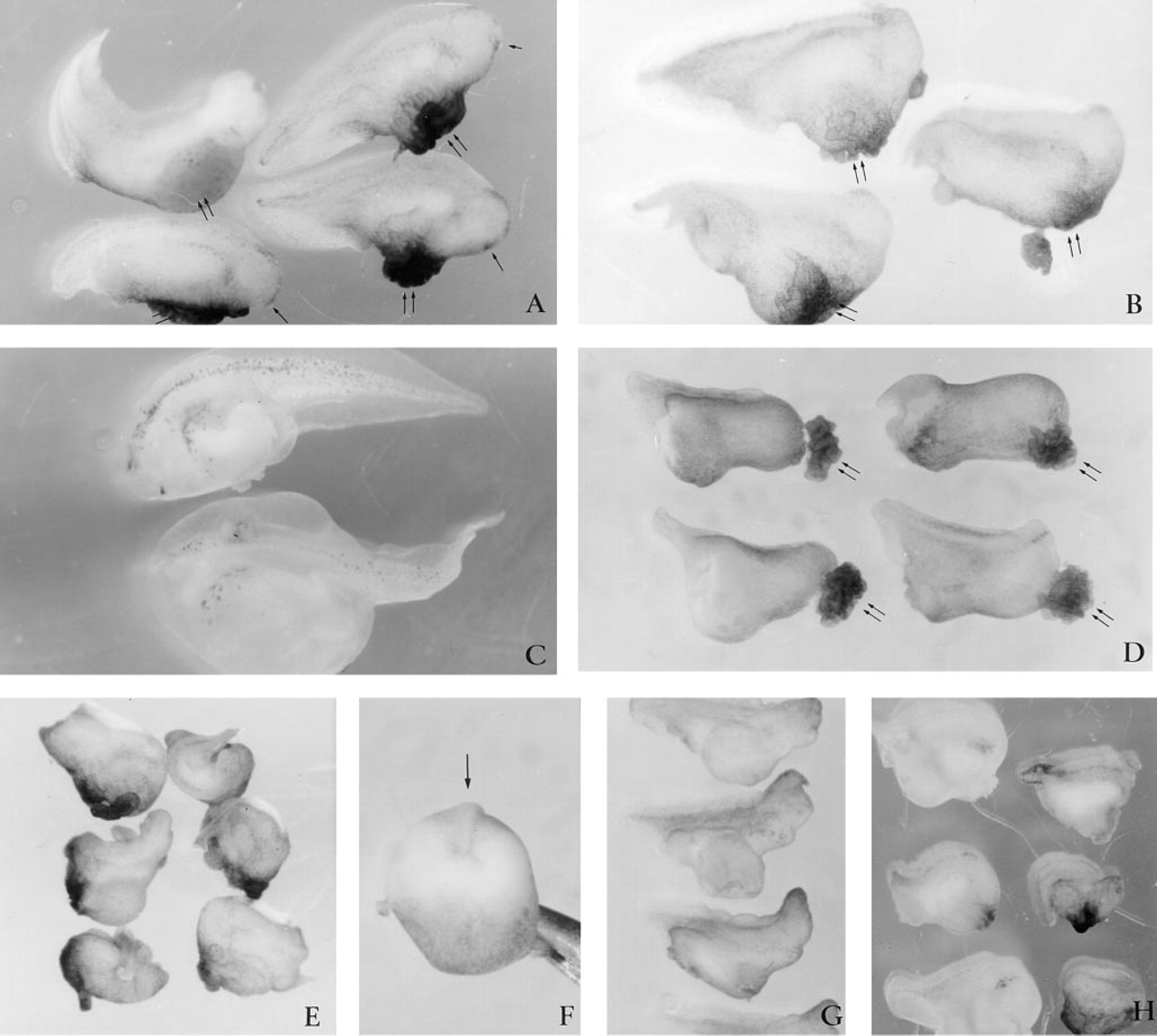
Abnormalities associated with microinjection of peptide 4.2. Microinjecting 60 ng of peptide 4.2 (
Studies in vitro on endothelial cells, smooth muscle cells, and fibroblasts have demonstrated that SPARC delayed cell cycle progression and reduced cell–substratum interactions (reviewed in Lane and Sage 1994). However, SPARC had no obvious morphological effect on early cell division and movement associated with gastrulation and early neurulation in Xenopus. Nevertheless, the effects observed, at a time when control sibling embryos had reached the tailbud stage, indicate that SPARC did interfere with gastrulation events and at high doses resulted in the ventralization of embryos. However, we have recently observed that SPARC did not interfere with the induction of mesoderm in Xenopus animal cap explants by activin or bFGF (S. Damjanovski and M. Ringuette, unpublished observations), two factors that play important inductive roles in early development. One potential target of SPARC could have been PDGF, because SPARC binds to PDGF-AB and -BB with high affinity. Interference with PDGF signaling in Xenopus has also been shown to generate some axis and anterior defects (Ataliotis et al. 1995) that are comparable to those observed with microinjected SPARC. However, PDGF is only one of several factors known to participate in early development. It is conceivable that the more severe defects observed with SPARC injection were due to its binding to multiple factors. Moreover, any potential effects of SPARC on cell division and adhesion might have been subtle and therefore not obvious until later in development. Interestingly, the microinjection of two other acidic Ca2+-binding glycoproteins, bone sialoprotein and osteopontin, resulted in developmental effects that were distinct from each other and from those induced by SPARC; these results indicate that the phenotypes generated were protein-specific.
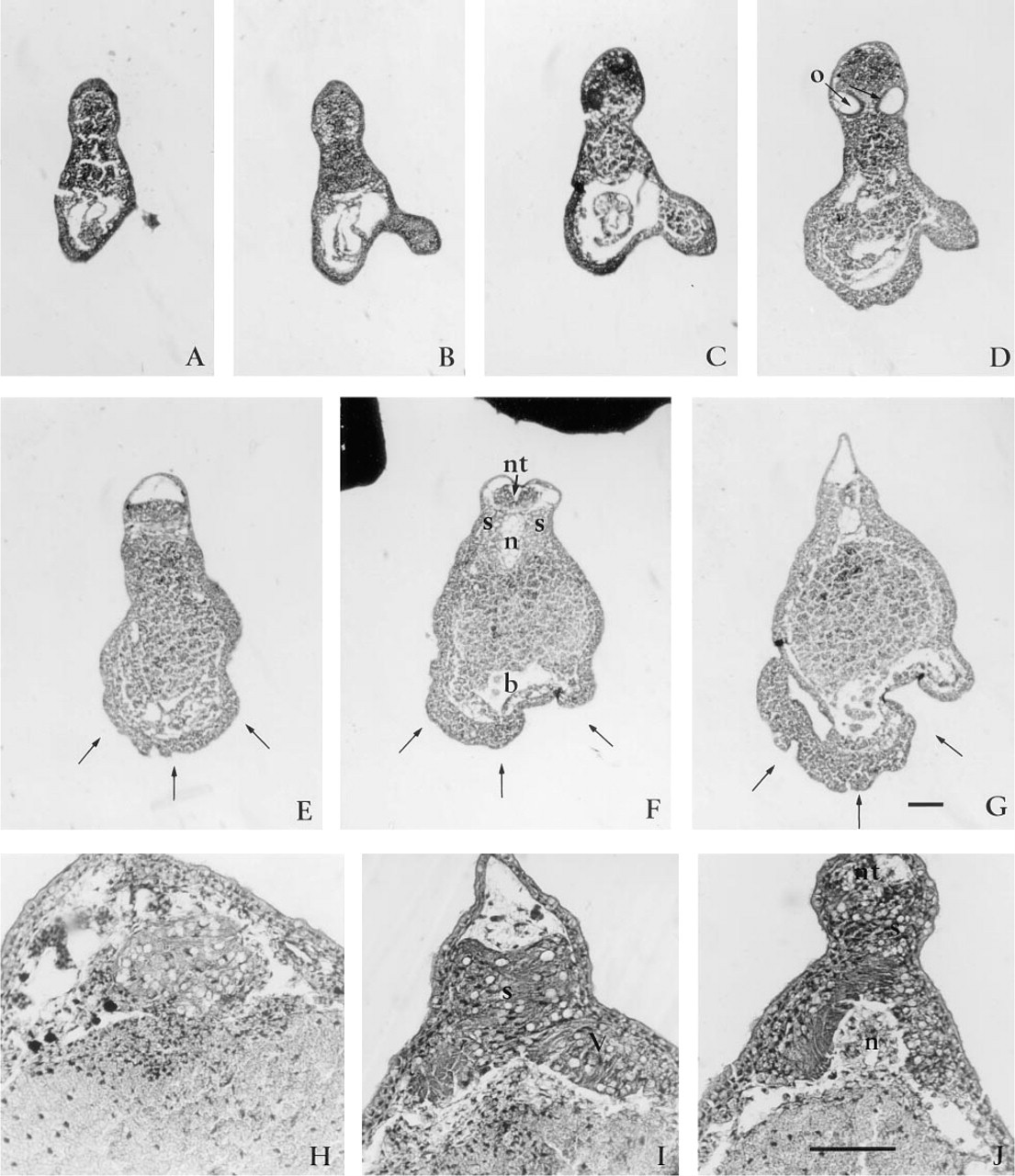
Histological analysis of embryos injected with SPARC peptide 4.2. Transverse serial histological sections (anterior to posterior series,
Injection of a mutant SPARC Domain IV peptide results in different phenotypes. Microinjection of 1000 ng peptide 4.2AA resulted in embryos which underwent normal development until the tailbud stage, at which point ventral swelling became prominent (
The effects of native SPARC on tissue culture cells have been recapitulated by synthetic peptides corresponding to discrete domains of SPARC. For example, peptides from the cysteine-rich, follistatin-like domain (2.1) and the C-terminal EF hand (4.2) inhibit cell cycle progression in mid-G1 (Funk and Sage 1993). In addition to inhibiting cell cycle progression, peptides 2.1 and 4.2 were found to compete for the binding of SPARC to collagen Types I and III and to endothelial cells. No effects on Xenopus development were observed when 60-1000 ng of peptide 2.1 or 2.3 was microinjected into the blastocoel cavity. However, injection of 60 ng of peptide 4.2 generated dorso-anterior defects. When the concentration of peptide 4.2 was increased to 100 ng, 100% of the embryos were headless. Further increases were associated with concentration-dependent defects in an anterior to posterior direction. The substantial effects of peptide 4.2 were surprising because, as discussed above, peptides 2.1 and 4.2 have similar effects on cultured cells in vitro. A potential explanation is that peptide 4.2 is a more potent inhibitor of cell cycle progression than is peptide 2.1 (4- to 100-fold) (Sage et al. 1995). It also inhibits cell spreading in vitro. The combination of cell-cycle modulation, cell surface receptor and collagen binding, and counteradhesion may account for the interference of peptide 4.2 with Xenopus development. Whereas gastrulation did not appear to be affected by peptide 4.2, as judged by closure of the blastopore, the absence of the dorso-anterior axis and structures indicates that it is very likely that interference with cell movement and/or inductive events occurred.
The inhibition of endothelial cell proliferation by peptide 4.2 was abrogated by two mutant peptides, 4.2AA and 4.2K. Peptide 4.2 K was associated with apparently the same developmental defects as were seen with peptide 4.2. In contrast, embryos injected with peptide 4.2AA appeared normal until the tailbud stage, with the exception of a subset that had ventral swelling. The effects in vivo of peptide 4.2 are therefore in part dependent on the formation of the disulfide bridge but are tolerant of a disruption of the Ca2+-binding consensus sequence. The potentially Ca2+-independent effect was confirmed by the observation that the co-injection of peptides 4.2, 4.2AA, or 4.2K with Ca2+ or EGTA did not alter the results seen with the peptide alone. Although direct comparisons between the effects observed in tissue culture vs whole embryos are difficult, the Ca2+-independent effects of peptide 4.2 in vivo might be consistent with data showing that SPARC does not act as a chelator of Ca2+ in the extracellular space, despite its ability to modulate endothelial cell shape (Sage 1992). The requirements of a disulfide bridge (an unusual feature of the EF-hand of SPARC) indicates that the secondary structure (and/or stability) of this region is critical. That microinjection of calmodulin (a protein containing four prototypic, nondisulfide-bonded EF hands) generated substantially different results indicated that the phenotypic effects of peptide 4.2 were particular to SPARC.
We have shown that precocious expression of SPARC led to a variety of developmental defects in Xenopus embryos. As a matricellular, multifunctional protein, SPARC has been shown to affect a variety of cell behaviors, including counteradhesion and regulation of the cell cycle. Although, the molecular basis of the phenotypes generated by precocious expression of SPARC, in particular with peptide 4.2, remains to be determined, the data indicate that cell migration and cell proliferation were probably affected during Xenopus gastrulation. Therefore, the counteradhesion and inhibition of cell cycle observed in vitro appear to be augmented in vivo by precocious overexpression of SPARC at a time during which rapid cell migration and proliferation are occurring.
Acknowledgments
Supported in part by a Natural Sciences and Engineering Research Council grant (MJR) and by a National Institutes of Health grant GM40711 (EHS).
We wish to thank Drs Jaro Sodek and Harvey Goldberg for their kind gifts of reagents.
