Abstract
During a spectroscopic study to identify biochemical changes in cervical tissue with the onset of carcinogenesis, residual paraffin wax contributions were observed on almost all dewaxed formalin-fixed paraffin-processed (FFPP) tissue sections examined. Subsequently, the present study was formulated to evaluate the efficacy of current dewaxing agents using Raman spectroscopy. Three cervical FFPP sections were subjected to each of the protocols. Sections were dewaxed using four common dewaxing protocols, namely, xylene, Histoclear, heat-mediated antigen retrieval (HMAR) using xylene and citrate buffer, and Trilogy (combined deparaffinization and unmasking of antigens). The potential for hexane as a dewaxing agent was also evaluated. Sections were dewaxed in multiple dewaxing cycles using xylene, Histoclear, and hexane. Residual paraffin wax contributions remained at 1062 cm−1, 1296 cm−1, and 1441 cm−1. HMAR using xylene and citrate buffer, and HMAR using Trilogy, showed a similar efficacy, resulting in incomplete removal of wax. Hexane was shown to be the most effective dewaxing agent, resulting in almost complete removal of wax. Immunohistochemistry was carried out on dewaxed slides, and those dewaxed with hexane displayed a stronger positivity (≍28%). Implications for histopathology and immunohistochemistry are considered, as well as problems that residual wax poses for spectroscopic evaluation of dewaxed FFPP sections with a view to disease diagnosis.
M
Formalin-fixed paraffin-processed (FFPP) tissues are used extensively for immunohistochemical detection of normal and tumor cell markers. A major step forward was made in the 1990s with the discovery that some antigens previously unreactive in FFPP tissue, even after protease treatment, could be “retrieved” by heating sections in a microwave oven. Originally, this was carried out in a solution of rather toxic heavy metal salts (Shi et al. 1991). Subsequently, these salts were replaced with buffers such as citrate buffer at (pH 6.0) (Cattoretti et al. 1993). It was shown that heat, rather than microwaves per se, is important in the retrieval process. Boiling the section in a pressure cooker (Norton et al. 1994) or autoclaving (Bankfalvi et al. 1994) in the buffer solution achieved the same effect (Polak and Van Noorden 1997). A more recent dewaxing protocol involves the application of a reagent for simultaneous dewaxing and antigen unmasking, namely, Declere and Trilogy (
Raman microspectroscopy is an analytical, nondestructive technique that provides information about the molecular structure of the investigated sample (Long 2002). The Raman effect arises when the incident light excites molecules in the sample, which subsequently scatter the light. While most of this scattered light is at the same wavelength as the incident light (λ1), some is scattered at a different wavelength (λ2). This inelastically scattered light (λ2) is called Raman scatter and results from the interaction of the incident light with the molecular motions or vibrations (Long 2002). The positions, intensities, and linewidths of the Raman lines, corresponding to vibrational energy levels, yield information on the composition, secondary structure, and interaction of molecules, including the chemical microenvironment of molecular subgroups. Many molecules are Raman active with fingerprint spectra, providing molecular-specific information that can be used as a marker of cellular damage (Conroy et al. 2003; Ó Faoláin et al. 2003). The molecular structure of nucleic acids, proteins, and lipids differ between normal and tumor tissues and, therefore, Raman spectroscopy has been considered promising for the molecular characterization of cancer cells (Liu et al. 1992).
Raman spectroscopy has been employed in the examination of a variety of common cancers, including cervical precancers (Mahadevan-Jansen et al. 1998), gastrointestinal lesions (Dacosta et al. 2002), benign and malignant skin lesions (Gniadecka et al. 1997), breast cancer (Frank et al. 1995; (Shafer-Peltier et al. 2002), colon polyps (Molckovsky et al. 2003), prostate cancer, and bladder cancer (Crow et al. 2003a,b).
To examine the biochemical structure of the FFPP tissue using Raman spectroscopy, the sample must be as close to its original/in vivo state as possible. This requires the removal of the paraffin wax and rehydration to the aqueous phase. In this present study multiple methods of dewaxing were examined and the efficacy of each assessed using Raman spectroscopy.
This study compared the effectiveness of the most commonly used dewaxing agents, namely, xylene, and Histoclear, as well as heat-mediated antigen retrieval (HMAR) using xylene followed by a citrate buffer and HMAR using Trilogy alone. The effectiveness of hexane as a dewaxing agent was also examined, due to its industrial use as a cleaning and degreasing solvent.
Materials and Methods
Raman Spectroscopy
An Instruments S.A. Labram Raman spectroscopic confocal microscope was used [Jobin Yvon (formerly Instruments SA); Stanmore, Middlesex, UK], with an argon ion laser operating at a wavelength of 514.5 nm. The Labram imaging system is a fully confocal Raman microscope system with a motorized XY sample stage for automated Raman imaging. The system includes a stigmatic spectrometer with two motorized gratings, of which the 1800 grooves/mm grating was used. The slit width was 920 μm. The resolution of the system operating with the 1800 grooves/mm was 1.65 cm−1/pixel. Detection of the scattered light was performed using a Peltier cooled, 16-bit dynamic range CCD detector with 1024 × 256 pixels. A × 50 objective lens was used. The laser power at the sample was measured and found to be ~~6.5 ± 0.05 mW, focused to a spot size of 2 μm at the tissue surface. The scattered Raman signal was integrated for 60 to 150 sec and measured over a spectral range of 400 to 1900 cm−1 with respect to the excitation frequency. Once acquired, each spectrum was baseline corrected, noise subtracted, and lightly filtered using a third-order linear model to improve clarity.
Sample Preparation
All cervical FFPP sections were obtained from the National Maternity Hospital, Holles St., Dublin. All specimens were automatically processed to wax as follows: (a) vacuum fixed in 10% buffered formal saline histo-grade, pH 6.8–7.2 (J.T. Baker; Deventer, The Netherlands) heated to 35C, (b) vacuum dehydration in industrial methylated spirit IMS T100 (Lennox; Dublin, Ireland) heated to 35C, (c) vacuum clearing in xylene (Serosep; Limerick, Ireland) heated to 35C, and (d) vacuum impregnation with Tissue Tek III Embedding Wax with polymer added (Sakura, Zoeterwoude, The Netherlands) and heated to 59C.
Three parallel FFPP cervical sections were dewaxed using each of the five protocols outlined below.
Xylene. After the wax impregnation, tissue was embedded and sliced into 5-μm sections using a microtome, mounted on glass slides, and dried. The unstained samples were immersed in a series of baths consisting of two baths of xylene (BDH; Dorset, UK) for 5 min and 4 min, respectively, two baths of Ethanol Absolut (Merck; Dorset, UK) for 3 min and 2 min, respectively, and a final bath of Industrial Methylated Spirits 95% (Lennox) for 1 min.
Histoclear. The same procedure was used for dewaxing using General Purpose Grade Histoclear (Fisher Scientific; Loughborough, UK), where xylene was substituted with Histoclear in protocol A.
Hexane. Again, the same procedure was used for dewaxing using Hexane (BDH), whereas xylene was substituted with hexane in protocol A.
Each of the sections examined was put through the dewaxing procedure outlined four successive times. Finally, each of the specimens was left sitting in a bath of xylene, Histoclear, and hexane for 18 hr.
Spectra were recorded between each of the successive cycles and after immersion in reagents for 18 hr. Raman spectra were taken from 10 random points from each of the three sections. All spectra were recorded from normal ectocervical squamous epithelial cells.
Xylene Dewaxing and HMAR in Citrate Buffer. To examine the effect of the HMAR technique using xylene and a citrate buffer on the wax content, sections were processed according to the following HMAR protocol. Sections (mounted on positively charged slides) were passed through two changes of xylene for 6 min each and three changes of spirit for 3 min each. Slides were then submerged in 600 ml of 0.1 M citrate buffer (pH 6) and placed in a pressure cooker for 20 min.
Simultaneous Dewaxing and HMAR in Trilogy. Two staining dishes were filled with ≍200 ml of Trilogy (Cell Marque Corporation; Hot Springs, AR), and the sections (mounted on positively charged slides) were submerged in the first staining dish. Both dishes were placed in a pressure cooker for 8 min on the high-pressure setting. After 8 min the pressure was released, and the slides were transferred to the second dish (hot rinse) to soak in Trilogy for an additional 10 min. The slides were agitated and rinsed in deionized water.
Raman spectra were recorded from 10 random points of normal ectocervical squamous epithelial cells from the HMAR-treated sections.
Raman spectra were also recorded from paraffin wax sections, tissue sections prior to deparaffinization, and frozen sections not embedded in paraffin wax. All sections were air dried and examined spectroscopically.
Immunohistochemistry
Sections were dewaxed for 18 hr at room temperature in hexane or xylene. Sections were rehydrated to water through Ethanol Absolut (Merck) and Industrial Methylated Spirits 95% (Lennox). The sections were placed in 0.1 mM citrate buffer (pH 6) and microwaved for 12 min at 750 W. They were kept in the hot solution for an additional 20 min, washed in water, treated with 0.3% hydrogen peroxide in methanol for 10 min, and transferred to phosphate buffered saline (pH 7.2). The Vector Elite Avidin-Biotin immunoperoxidase kit (Vector Laboratories; Peterborough, UK) was employed for the immunoperoxidase method. Slides were treated with normal horse serum 1:50 for 10 min, and then the monoclonal antibody to cytokeratin MNF 116 (Dakocytomation; Glostrup, Denmark) was applied at a dilution of 1:100 in PBS for 60 min at room temperature. Sections were washed in PBS for 10 min, and biotinylated secondary antibody (1:200) was applied for 15 min. Following a wash in PBS for 10 min, the avidin-biotin complex solution (1:50) was applied for 15 min, sections were washed in PBS for a final 10 min, and incubated in the chromogenic DAB substrate for 5 min (0.003% hydrogen peroxide and 0.06 g/ml diaminobenzidene in PBS). A nuclear counterstain with hematoxylin was applied, the slides were dehydrated through alcohols, placed in xylene, and cover slipped with DPX resin medium.
Results
The spectrum of paraffin wax clearly shows distinctive Raman contributions at 1063 cm−1, 1133 cm−1, 1296 cm−1, and 1441 cm−1 (Figure 1). These are attributed to C-C (carbon-carbon) stretching and CH2 and CH3 deformation (Barry et al. 1992; Lakshmi et al. 2002), as would be expected given the straight-chain hydrocarbon structure of wax (Figure 2). A small contribution at 1004 cm−1 (C-C aromatic ring) is attributed to the small percentage of cycloparaffins contained within the wax.
Figure 3 compares a Raman spectrum of A, paraffin wax; B, an ineffectively dewaxed histological section (using xylene); and C, a frozen tissue section that has not been embedded in wax. It demonstrates that the contributions from the paraffin wax seen at 1062 cm−1, 1296 cm−1, and 1441 cm−1 are visible in the dewaxed FFPP section (Figure 3B), and that they are not present in unprocessed tissue (Figure 3C). The C-C stretch band at 1133 cm−1 appears in both paraffin wax and biological samples and for this reason has not been highlighted as an exclusive paraffin contribution. Many additional peaks are present in both the spectra of the dewaxed FFPP section and the frozen section. These peaks correspond to vibrations of bonds emanating from the biological material itself. The spectra are dominated by peaks in the regions of 1330 cm−1, 1450 cm−1, and 1659 cm−1, corresponding to CH2 and CH3 twisting and wagging of collagen, CH2 bending mode of proteins, and the amide I band (C=O) stretching of proteins, respectively. Other peaks include 622 cm−1 (CS stretch), 644 cm−1 (CS stretch; Amide IV), 725 cm−1 (= CH in plane bending), 829 cm−1 (CCH aliphatic deformation), 853 cm−1 (CCH aromatic deformation), 1172 cm−1 (CC stretch), 1583 cm−1 (C = C bending), 1601 cm−1 (C = C in plane bending), and 1620 cm−1 (C = C stretching) (Barry et al. 1992; Stone et al. 2000).
Because the units associated with spectroscopic intensity are arbitrary units (a.u.), it is only the relative intensity to the baseline of each spectrum that is relevant. Hence, the spectra have been offset to facilitate clarity. This method of display has been used throughout.
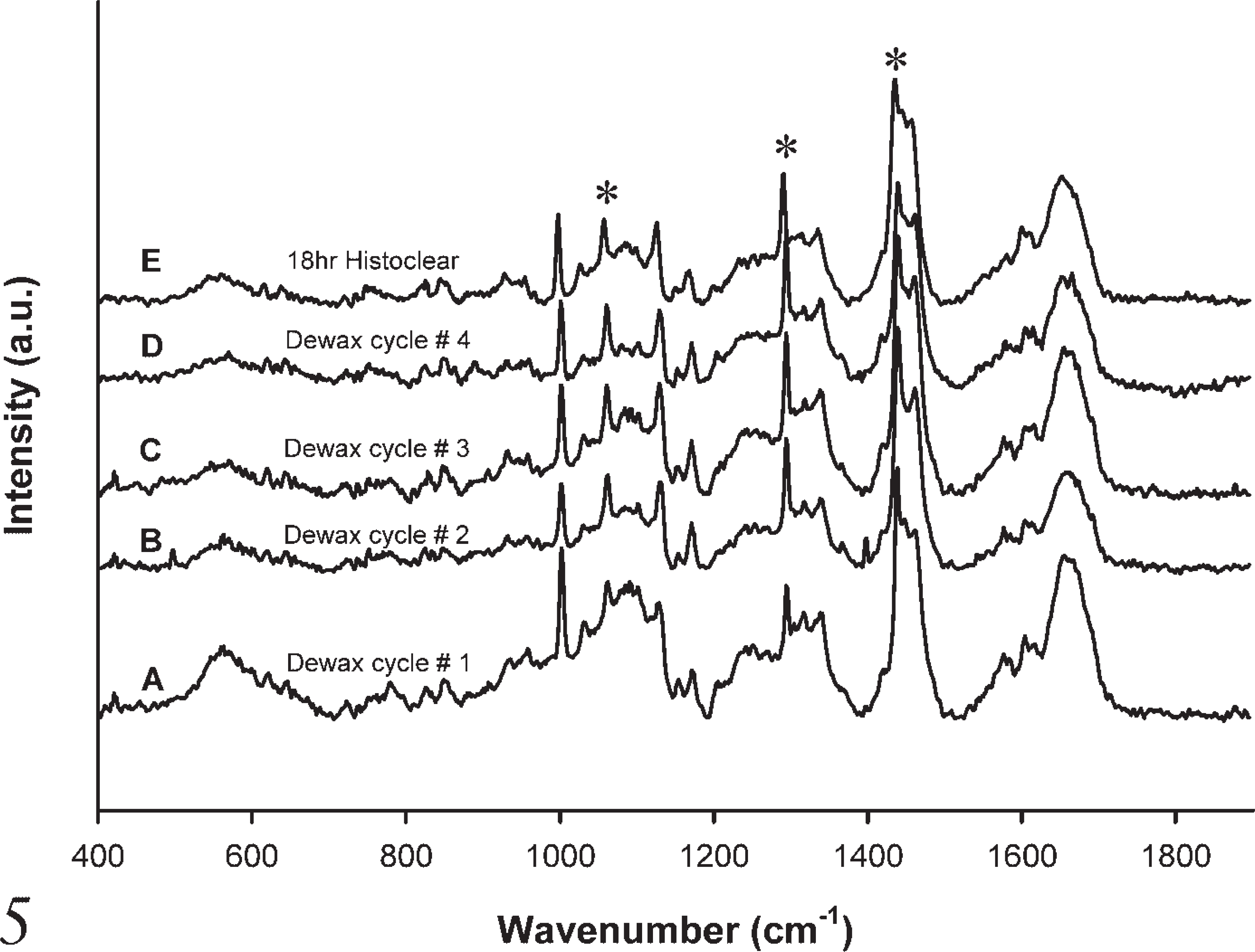
The effectiveness of xylene (Figure 4) and Histoclear (Figure 5) on the wax content was examined and found to be ineffective at complete removal of wax. The effect of a single dewaxing cycle (Figure 4A) and subsequent dewaxing cycles (Figures 4B-4D) left the signature wax contributions at 1062 cm−1, 1296 cm−1, and 1441 cm−1. These contributions were not eliminated even after immersing slides for 18 hr in xylene (Figure 4E). The same residual wax contributions were present after a single dewaxing cycle (Figure 5A), subsequent dewaxing cycles (Figures 5B-5D), and immersion in Histoclear for 18 hr (Figure 5E).
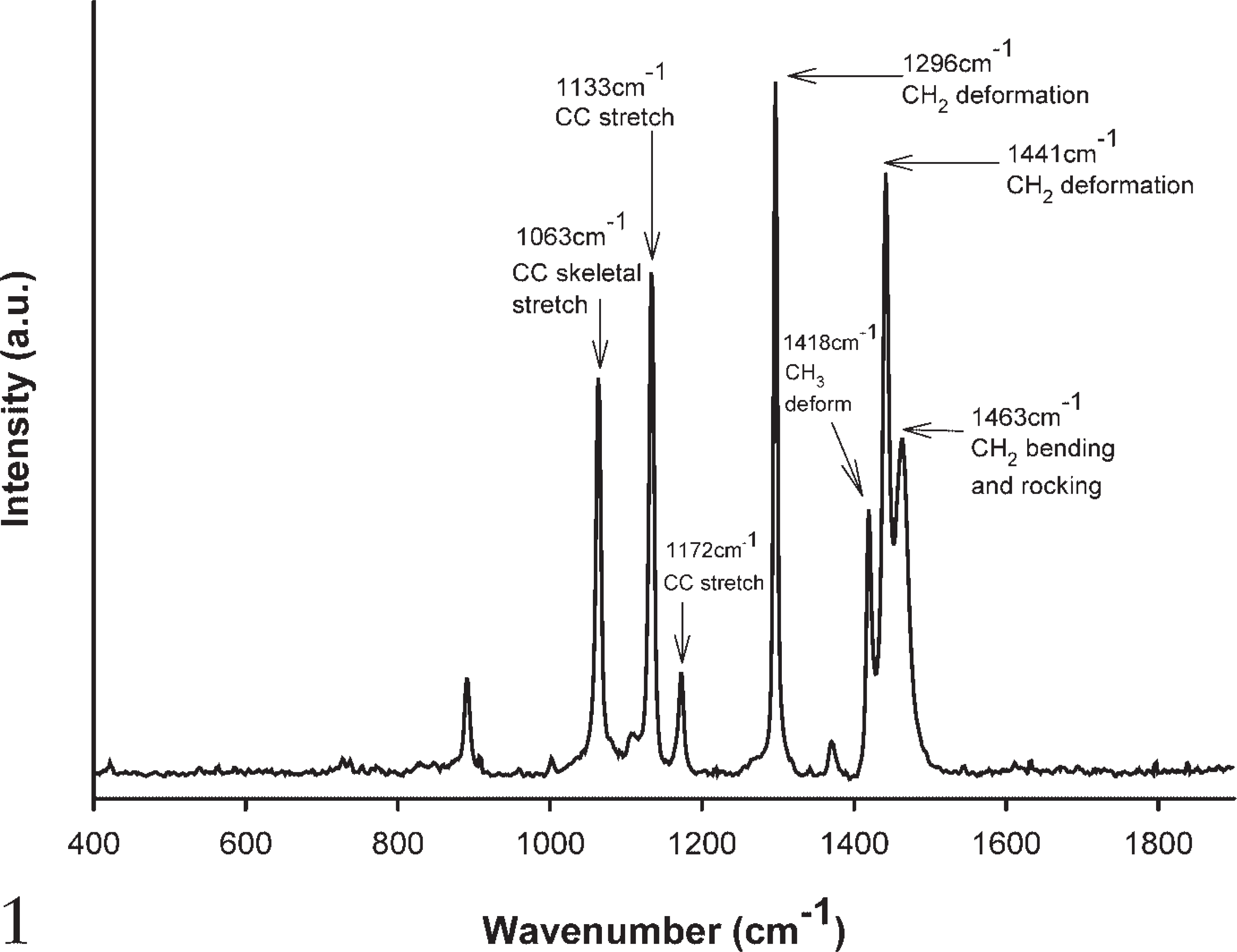
Raman spectrum of paraffin wax.
Chemical structure of paraffin wax.
Hexane, however (Figure 6), proved to be much more effective at wax removal than either xylene or Histoclear. The wax content reduced after each subsequent cycle (Figures 6A-6D) and had almost been completely removed after immersion in hexane for 18 hr (Figure 6E).
Following dewaxing with xylene and HMAR using citrate buffer, residual wax contributions were observed at 1062 cm−1, 1296 cm−1, and 1441 cm−1 (Figure 7A). Following simultaneous dewaxing and HMAR with Trilogy, the same residual wax contributions were also observed (Figure 7B). Due to the high pressure and temperature involved in the pressure cooker technique, the tissue begins to degrade after multiple cycles. This tissue degradation is evident in the Raman spectra in Figure 7, where an overall deterioration in signal intensity compared with the signals measured in Figures 3–6 is easily seen. However, multiple-cycle investigations were carried out and residual wax remained (results not shown).
The impact of wax removal on immunostaining was evaluated by directly comparing parallel sections dewaxed using xylene and hexane after immersion for 18 hr. The immunostaining protocol was applied to both slides, where brown staining indicated immunopositivity (antigen/antibody binding). Subsequently, images were recorded from the same areas of both slides and were compared. Figure 8 shows images taken from both slides after dewaxing with xylene and hexane (Figures 8A and 8B, respectively).
Qualitatively, it is clear from Figure 8 that there is a stronger positivity in Figure 8B (dewaxed using hexane). To quantify this increase in positivity, both images were converted to greyscale, and the number of gray pixels and hence the overall intensity of the stained region was calculated using the Image J analysis program (National Institutes of Health, Bethesda, MD). The overall intensity after dewaxing using xylene was 124.99 ± 5.03 a.u., whereas the value measured from the slide dewaxed using hexane was 97.46 ± 1.52 a.u. This reduction in intensity quantifies the increase in positivity, which has been improved by 28% when dewaxed using hexane as opposed to xylene.
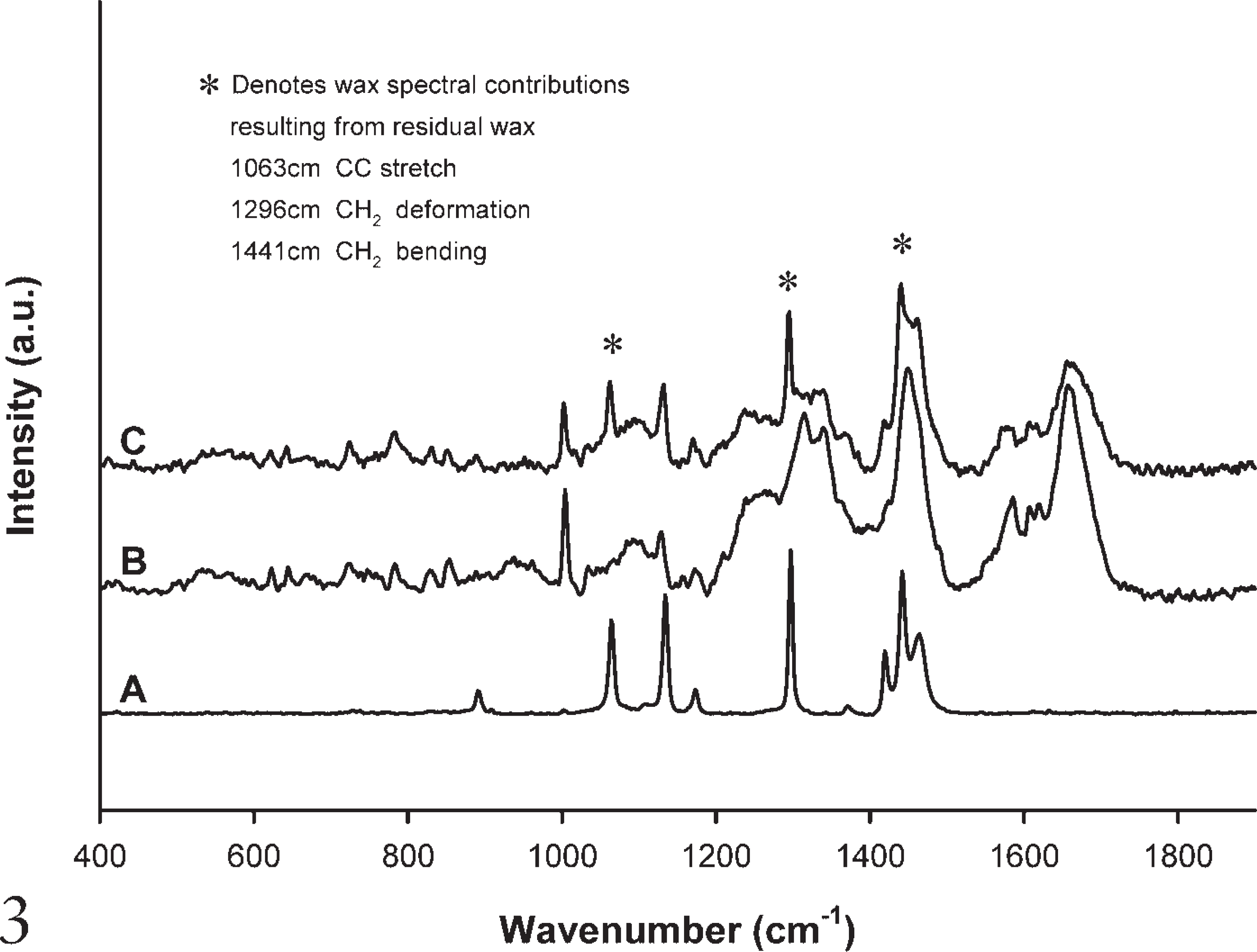
Raman spectrum comparing paraffin wax, dewaxed FFPP section, and frozen tissue.
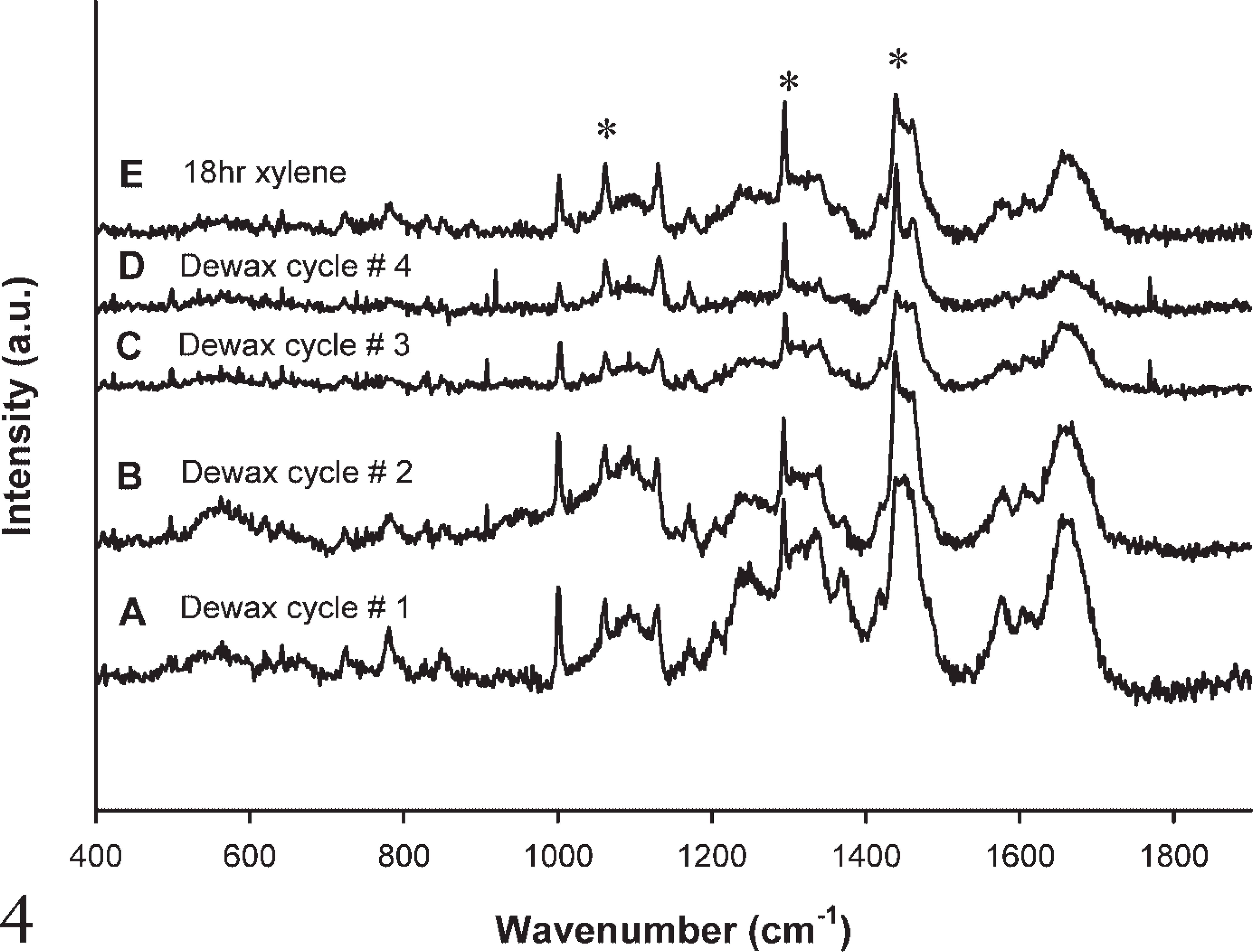
Raman spectra after each subsequent dewaxing cycle using xylene.
Raman spectra after each subsequent dewaxing cycle using Histoclear.
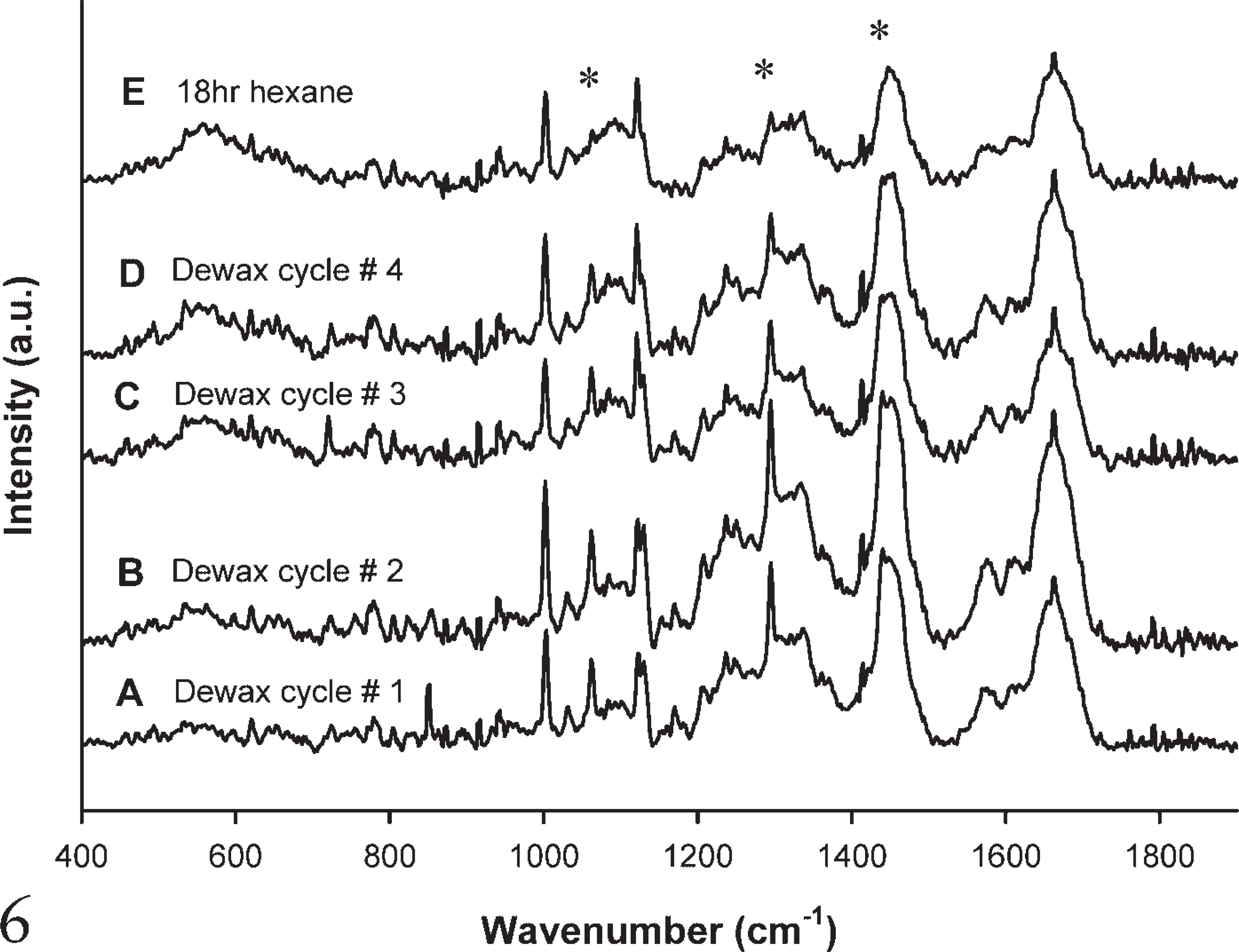
Raman spectra after each subsequent dewaxing cycle using hexane.
Discussion
The present study investigated five clearing agents [xylene, Histoclear, hexane, HMAR (using xylene and citrate buffer) and Trilogy]. Raman spectroscopy clearly demonstrated that peaks corresponding to the presence of wax were present in all samples examined, and that wax is not currently being removed with existing protocols. Hexane was shown to be more effective at wax removal than all other agents examined. This increased level of wax removal resulted in an approximate 28% improvement in immunohistochemical staining compared with a parallel section dewaxed using xylene.
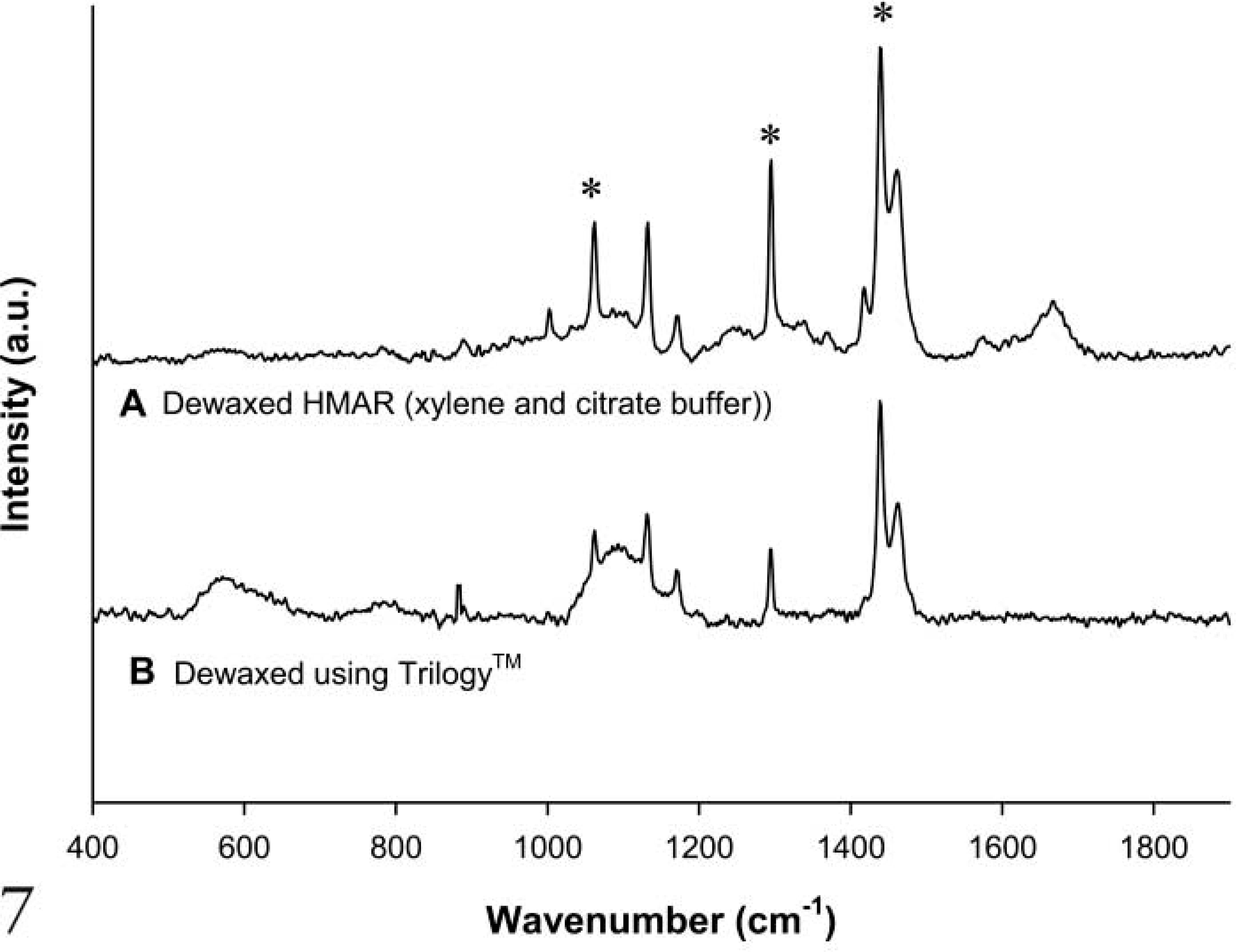
Raman spectra comparing dewaxing HMAR (using xylene and citrate buffer) and Trilogy.
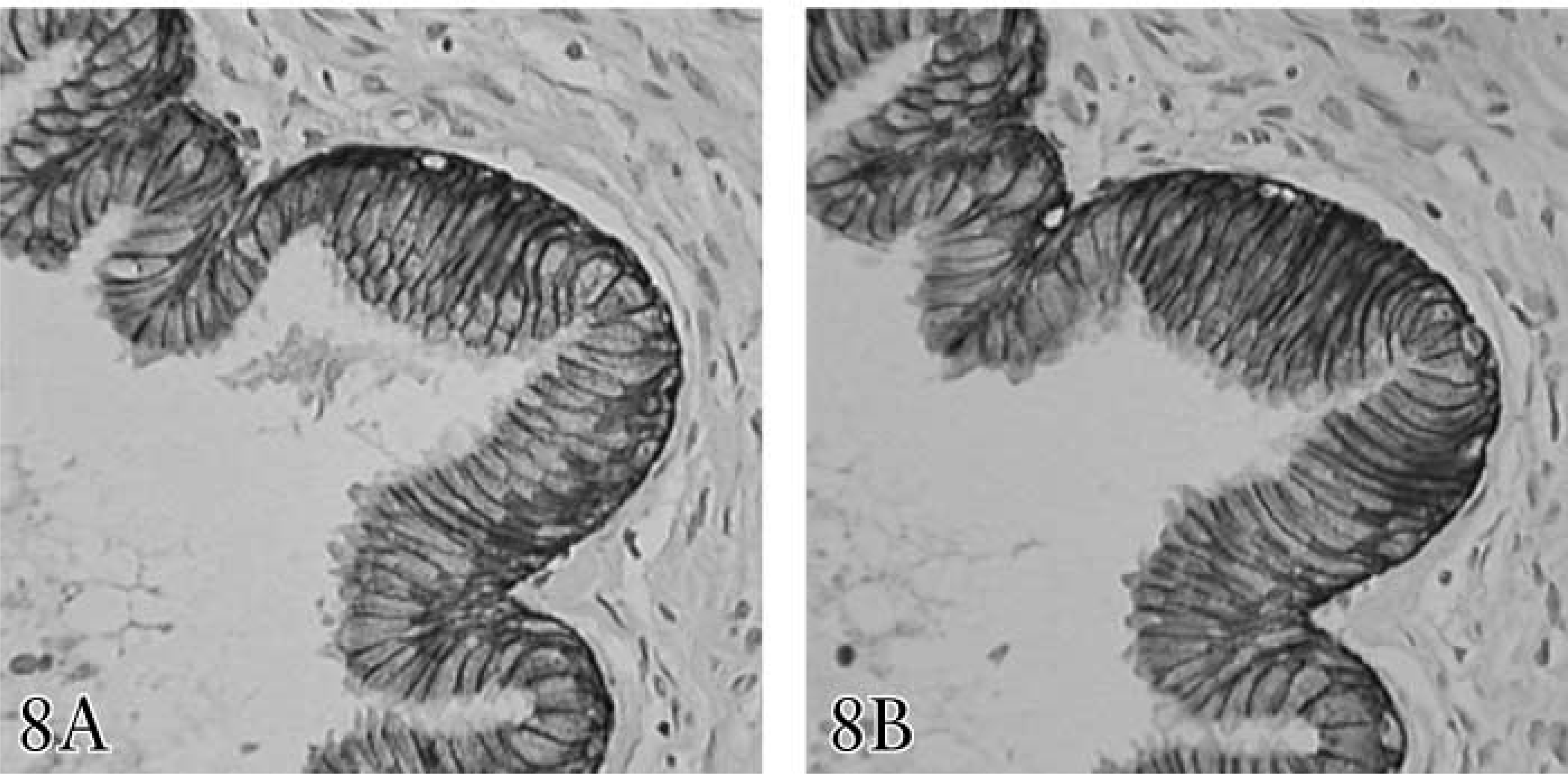
Images of samples after immunostaining.
Xylene is the most commonly used solvent, due to its rapid and supposedly efficient dewaxing for common histological, histochemical, and immunohistochemical procedures. It is a highly toxic, flammable substance and must be handled with care. Histoclear, on the other hand, is non-toxic, non-flammable, and biodegradable but is a less-efficient clearing agent and potentially causes the hematoxylin stain to fade (Culling et al. 1985). The HMAR technique uses xylene as a clearing agent and reverses formaldehyde fixation effects using citrate buffer in a pressure cooker (Mac-Intyre 2001). Trilogy, on the other hand, is a novel product that combines deparaffinization, rehydration, and unmasking of antigens during pressure cooking. However, Trilogy is under evaluation for application
in diagnostic immunohistochemistry, and this is the first report comparing the dewaxing efficacy of Trilogy to the more standard reagents using Raman spectroscopy. Hexane is a colorless liquid with a slight odor and, like many solvents, is highly flammable. It should be handled with care and used only in a fume hood. The US Department of Health and Human Services, International Agency for Research on Cancer, and the US Environmental Protection Agency have not classified hexane for carcinogenicity. This paper is also the first to examine the dewaxing properties of hexane and to directly compare it to all commonly used dewaxing agents.
During investigations into the biochemical changes in tissue with the onset of carcinogenesis (Ó Faoláin et al. 2003), it was noticed that the standard dewaxing procedure used on FFPP tissue did not completely remove the wax as previously supposed. A quantity of residual wax remained. From a spectroscopic perspective, with a view to detection of biochemical changes in the composition of FFPP sections, residual wax is problematic. Its spectral contributions add a substantial degree of variation to the overall spectrum. Contributions at 1062 cm−1, 1296 cm−1, and 1441 cm−1, corresponding to CC skeletal stretch, CH2 deformation, and CH2 bending, respectively, mask contributions from these molecules emanating from the tissue. Thus, potential biochemical changes in these regions are rendered useless. Also, the application of an automated analysis technique is not possible while there are random variables in the datasets.
Ineffective removal of wax from sections can cause birefringence of cell nuclei (Nedzel 1951). In addition, failure to completely remove the wax from the sections will result in impairment of staining. This can result in the Pink Disease artifact (Vlachos 1968). The artifact, which is most noticeable in lymphoid and epithelial tissue, is an extremely patchy distribution of stains and results in the loss of distinction of nuclear margins (Drury and Wallington 1980). There are also implications for antigen demonstration by immuno-histochemistry. It is only in recent times that immuno-histochemistry has been carried out on paraffin-fixed sections. It is clear from the results of both HMAR and Trilogy investigations that all the wax is not being removed.
This research is the first study to report that a low level of wax is residual in solvent-treated paraffin wax sections. The majority of histochemical and immunohistochemical methods employed in histopathology laboratories utilize these solvents as their dewaxing agents. Many dyes stain more intensely on cryostat sections, and some do not work well on paraffin sections, particularly metachromatic stains for carbohydrates (H. Lambkin, unpublished data); thus, the residual wax may be a factor contributing to these differences. Immunostaining has been optimized for paraffin sections with hundreds of antigens now detectable in these preparations. Heat-based antigen unmasking has been introduced since the 1990s and has contributed to a lowering of the antigen detection threshold in paraffin sections (Shi et al. 1991); however, the heat effects are considered to be related to reversing of the effects of formaldehyde cross-linking of proteins, rather than removal of wax.
In this study it has been demonstrated that current dewaxing procedures are not completely effective. Hexane has been identified as a superior dewaxing solvent to xylene as well as to Histoclear and Trilogy. It has also been demonstrated that increased wax removal using hexane results in better antigen/antibody binding and, hence, a stronger positivity. In addition, care should be taken when using FFPP sections for spectroscopic investigation of diseased tissue. This study recommends dewaxing using hexane to minimize wax contributions.
Footnotes
Acknowledgements
FOCAS is funded under the Irish National Development Plan 2000 to 2006 with assistance from the European Regional Development Fund. Primary author is funded by the Technological Sector Research, Postgraduate Research, and Development Skills Programme.
The authors would like to express thanks to all the staff in the Histology Department in the NMH Holles St. for their generous cooperation. We also thank Stephen M. Hewitt for his advice and encouragement.
