Abstract
We present a systematic analysis of the sensitivity and specificity of immunohistochemical stainings for components of the plasminogen activation system, i.e., uPA, tPA, PAI-1, PAI-2, and uPAR, on routinely processed (formalin-fixed, paraffin-embedded) tissues. Five to nine antibodies per component were tested and the influence of different antigen retrieval regimens on immunoreactivity was investigated. We studied six different microwave-mediated pretreatments and two pretreatments by proteolytic digestion. First, positive and negative control tissues were stained. Then, frozen and paraffin sections from the same cancer lesions were stained after specific modes of pretreatment and with selected antibodies. For each component, one or a few of the tested Abs gave optimal staining on paraffin sections when combined with a particular tissue pretreatment. For PAI-1, and to a lesser degree also for tPA, an underrepresentation of stromal cell staining in paraffin material was found, whereas tumor cells showed good staining. For uPA, PAI-2, and uPAR, consistent staining results were obtained on paraffin sections.
Keywords
I
In addition to quantitative measurement by ELISA, immunohistochemistry (IH) is a good approach to investigate the status of the PA system. IH provides information on the quality of expression by the different cell types, e.g., tumor cells and/or stromal cells, and on its intensity and distribution. When IH yields reliable results on paraffin sections, it also allows retrospective studies using archival material.
Antigen retrieval is a technique that re-exposes epitopes in paraffin sections and makes them detectable by IH. Both enzymatic retrieval by proteases (Cattoretti et al. 1993; Dookhan et al. 1993) and microwave (MW)-based retrieval (Taylor et al. 1996; Shi et al. 1997) have been shown to improve IH staining with various Abs. With regard to the components of the PA system, several methods of pretreating paraffin sections to enhance epitope accessibility have been reported (Pyke et al. 1993; Bianchi et al. 1994,1995; Christensen et al. 1996). Remarkably, many groups do not report the performance of any antigen unmasking method (Sier et al. 1991; Takeuchi et al. 1993; Kobayashi et al. 1994; Muchahy et al. 1994; Nagayama et al. 1994; Heiss et al. 1995; Hsu et al. 1995; Constantini et al. 1996).
To the best of our knowledge there are only very few reports describing a quality-controlled staining procedure for the components of the PA system in formalin-fixed paraffin sections (Bianchi et al. 1994, 1995; Christensen et al. 1996). Therefore, in this study we systematically evaluated the results of IH on paraffin sections for all five components of the PA system.
Materials and Methods
Study Design
First we selected primary Abs that gave good results on acetone-fixed cryostat sections of positive and negative control tissues. These Abs were further tested for use on paraffin sections obtained from the same control tissues. To compare omission of tissue pretreatment, proteolytic enzyme digestion, and MW heating, different pretreatments were applied on consecutive paraffin sections of control tissue blocks. Abs that performed well on paraffin control sections were subsequently used on human malignant lesions. From these lesions, both frozen sections and formalin-fixed sections were stained. Paraffin sections underwent the treatments found successful in the former stage of this investigation. Staining patterns were compared, and those Abs that gave similar staining on frozen and on paraffin sections were considered to be suitable for use on paraffin-embedded material.
To test the benefit of antigen retrieval in case of prolonged formalin fixation, we stained control tissues that had been fixed overnight, 3 days, 1 week, and 3 weeks before embedding in paraffin. The samples for the different fixation times were obtained from the same tissue specimen. Pretreatment regimens and Abs found successful were applied to the sections and staining results on tissue sections fixed for prolonged times were compared with the results on tissue sections that had been fixed overnight.
Tissue Controls
As controls for uPA and PAI-1, we used xenograft tumors from the human melanoma cell lines BLM and IF6. The cell line BLM produces uPA and PAI-1 in large quantities (Quax et al. 1991). The cell line IF6 produces no or low levels of uPA and PAI-1 (unpublished results; not shown). Rat and mouse xenograft tumor lesions were grown as described previously (van Muijen et al. 1991). Mouse xenografts were used only to test polyclonal antibodies (PAbs), whereas rat xenografts were used to test both mouse monoclonal antibodies (MAbs) and PAbs. One part of each tumor was freshly frozen in liquid nitrogen and the other parts were fixed overnight in 4% buffered formaldehyde and subsequently embedded in paraffin.
For uPAR, the strongly uPAR-positive human melanoma cell line M24met (Stahl and Mueller 1994) and the uPAR-negative human breast carcinoma cell line BT474 (American Type Culture Collection; Rockville, MD) (Bianchi et al. 1994) were used as controls. M24met cells are strongly tumorigenic in rats, allowing establishment of rat xenografts. Because BT474 cells were not tumorigenic in nude rats, cytospins were prepared which either were stored at -80C until use or were formalin-fixed and subsequently processed in a tissue processor to mimic the procedure for paraffin-embedded tissue.
Normal uterine cervix was used as control tissue for tPA because this tissue is densely vascularized [tPA expression in most normal tissues is confined to the endothelial cells of the small blood vessels (Levin and del Zoppo 1994), whereas other structures are negative].
Normal human placenta was used as control tissue for PAI-2 [syncytiotrophoblasts of the chorionic villi express PAI-2, whereas the villous stroma is negative (Åstedt et al. 1986)].
Human Malignant Tumors
Tumor specimens of diverse origin (breast, cervix, colon, lung, and melanoma) were obtained from the tissue bank of the Department of Pathology (University Hospital Nijmegen, The Netherlands). Parts of the tumors had been freshly frozen in liquid nitrogen and stored at -80C. The largest part had been routinely fixed in formalin and embedded in paraffin. On the basis of staining patterns observed on cryostat sections, lesions were selected for further staining of the corresponding paraffin sections. For each component, a minimum of 15 lesions were selected so that both positive and negative lesions as well as various staining profiles, such as tumor cell positivity, stromal cell positivity, extracellular matrix positivity, and combinations of these, were included.
Antigen Retrieval
For enzymatic pretreatment, dewaxed and rehydrated sections were incubated with (a) 0.1% (w/v) pronase E (protease type XIV; Sigma, St Louis, MO) in PBS or (b) 0.1% (w/v) trypsin (protease type III; Sigma) in PBS. Incubations were performed at 37C and digestion times were varied (10, 15, 20, or 25 min).
For MW pretreatment, six retrieval solutions were tested: distilled water, 0.05 M sodium citrate buffer (pH 2.5, pH 4.5, and pH 6.0) (Evers and Uylings 1994), 3 M urea, and 0.5 M Tris-HCl (pH 10.0). Dewaxed and rehydrated sections were placed in 100-ml beakers containing retrieval solution. A maximum of six slides were placed in each beaker and a total of four beakers were placed in an MW oven with a rotating plateau (Miele Supratronic M752). The fluid was heated to boiling at a power setting of 850 W; then the power was turned down to 150 W. With these adjustments the temperature was maintained at 97C. Heating times of 10 and 20 min were tested. After heating, the slides were allowed to cool in the retrieval solution for 45 min before further processing for IH.
Immunoreagents
For each of the five PA components, a number of primary Abs were tested, as listed in Table 1. Working dilutions were first determined on frozen sections of the control tissues; these concentrations were used throughout the study.
Biotinylated secondary Abs used were goat anti-rabbit immunoglobulin (Vector Laboratories; Burlingame, CA), donkey anti-mouse immunoglobulin (Jackson; West Grove, PA), and donkey anti-goat immunoglobulin (Jackson). A three-step staining procedure with avidin–biotin complex (ABC) from Vector Laboratories was performed.
Abs tested and their staining results on paraffin sections after antigen retrieval
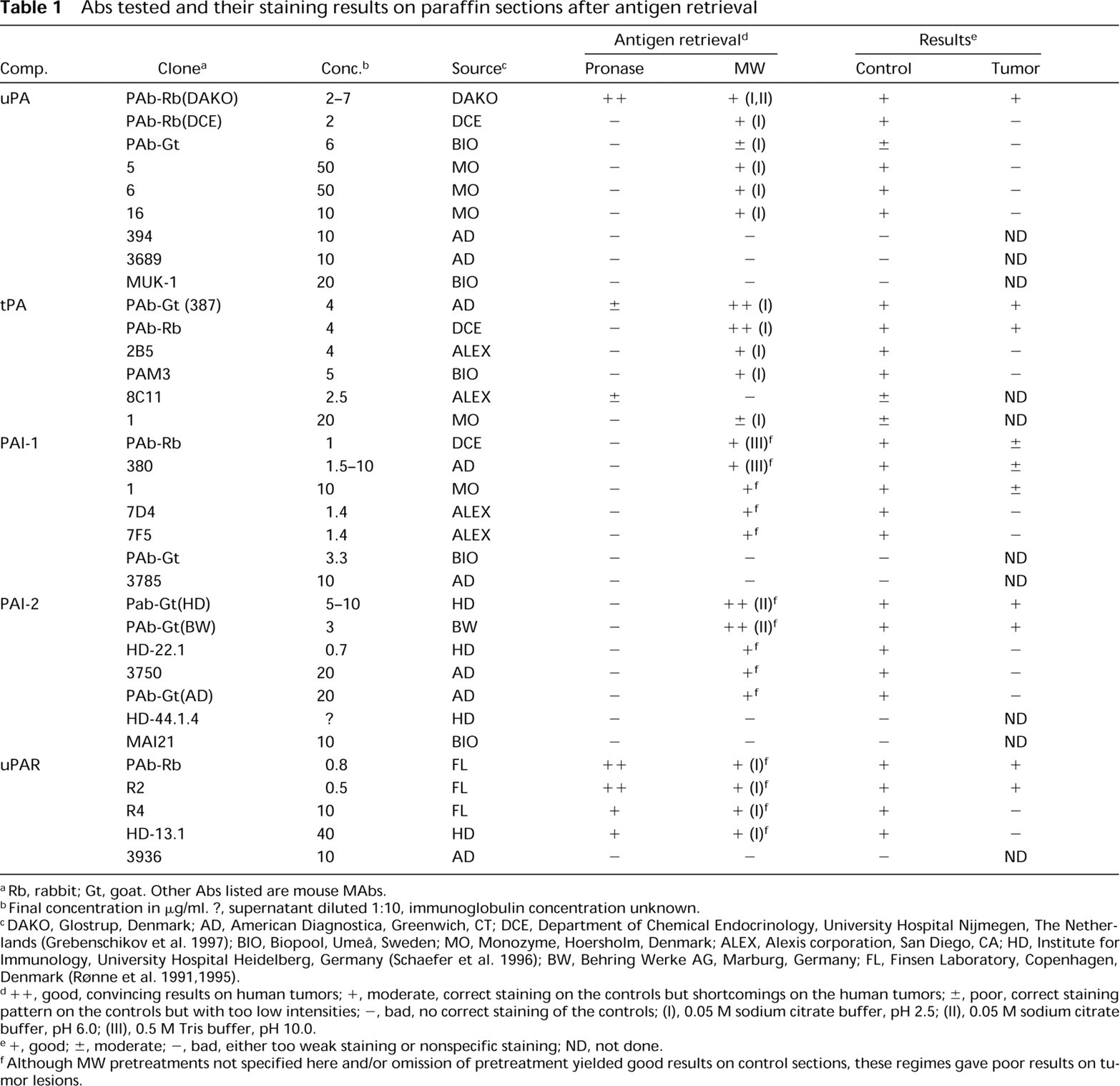
Rb, rabbit; Gt, goat. Other Abs listed are mouse MAbs.
Final concentration in μg/ml. ?, supernatant diluted 1:10, immunoglobulin concentration unknown.
DAKO, Glostrup, Denmark; AD, American Diagnostica, Greenwich, CT; DCE, Department of Chemical Endocrinology, University Hospital Nijmegen, The Netherlands (Grebenschikov et al. 1997); BIO, Biopool, Umeå, Sweden; MO, Monozyme, Hoersholm, Denmark; ALEX, Alexis corporation, San Diego, CA; HD, Institute for Immunology, University Hospital Heidelberg, Germany (Schaefer et al. 1996); BW, Behring Werke AG, Marburg, Germany; FL, Finsen Laboratory, Copenhagen, Denmark (Rønne et al. 1991,1995).
+ +, good, convincing results on human tumors; +, moderate, correct staining on the controls but shortcomings on the human tumors; ±, poor, correct staining pattern on the controls but with too low intensities; -, bad, no correct staining of the controls; (I), 0.05 M sodium citrate buffer, pH 2.5; (II), 0.05 M sodium citrate buffer, pH 6.0; (III), 0.5 M Tris buffer, pH 10.0.
+, good; ±, moderate; -, bad, either too weak staining or nonspecific staining; ND, not done.
Although MW pretreatments not specified here and/or omission of pretreatment yielded good results on control sections, these regimes gave poor results on tumor lesions.
To determine the specificity of the PAbs, Western blotting analysis was performed using lysates of BLM xenograft tumor, hemangioma, lung carcinoma with an ELISA-determined high PAI-1 concentration, normal placenta, and M24met xenograft tumor for uPA, tPA, PAI-1, PAI-2, and uPAR respectively. For each component, a purified standard was run in parallel with the lysates. The procedure was performed essentially as described by Verbeek et al. (1997).
Specificity of the first Abs was ascertained by the following negative controls. MAbs were replaced with isotype-matched irrelevant Ab (DAKGO1; DAKO, Glostrup, Denmark). PAbs were replaced with (a) a nonimmune IgG preparation from the same species and (b) a primary Ab dilution that was affinity-absorbed with purified antigen coupled to CNBr–Sepharose beads (Pharmacia; Uppsala, Sweden).
Immunohistochemistry
Frozen sections were fixed for 10 min in acetone at room temperature (RT). For Abs R2 and 3936, an acid pretreatment as described by Carriero et al. (1994) was performed before staining. Paraffin sections were mounted on Super-frost microscope slides (Menzel–Gläser; Braunschweig, Germany). After dewaxing and rehydration, the consecutive sections from a tissue specimen underwent different pretreatments (enzymatic or MW-mediated) or omission of pretreatment. The further procedure was basically the same for frozen and for paraffin sections. Tissue sections were incubated with 10% normal horse serum (Vector Laboratories) for 10 min and then incubated with primary Ab for 2 hr. Then the sections were incubated with a biotinylated secondary Ab for 30 min and with ABC for 45 min. For frozen sections, peroxidase-conjugated ABC was used in combination with the chromogen diaminobenzidine (Sigma); the staining signal was intensified by a wash in 0.5% CuSO4/PBS. For paraffin sections, we used alkaline phosphatase-conjugated ABC in combination with the Vector Red substrate kit. Normal horse serum, primary Abs, secondary Abs, and ABC were diluted in PBS containing 1% bovine serum albumin (Organon Teknika; Boxtel, The Netherlands). All incubations were performed in a dark moist chamber at RT. Sections were counterstained with Mayers’ hematoxylin (Merck; Darmstadt, Germany) and mounted (Imsol mounting medium; Klinipath, Duiven, The Netherlands).
Results
General Aspects
Selected primary Abs directed against the same PA component showed comparable staining profiles on frozen sections from human malignant tumors, although intensities varied. All PAbs analyzed on Western blot showed a band at the same molecular weight as the purified standard. For PAI-1, an additional band was seen at the front of the gel, most probably due to some degradation. For PAI-2 an additional band was found that corresponded to the dimerized form.
For staining on paraffin sections, we found that without pretreatment, no convincing staining could be obtained for uPA, tPA, PAI-1, and uPAR. For two of the tested Abs against PAI-2, pretreatment was less essential, although it markedly reduced the background staining.
Pronase and trypsin showed comparable unmasking effects, but pronase pretreatment yielded a somewhat stronger staining with less background. In most cases a 15-min digestion time was optimal.
For MW pretreatments, a heating time of 20 min gave better results than a heating time of 10 min only for PAI-1.
After the optimal pretreatment had been found for each component, substitution of the primary Ab by an Ab of irrelevant specificity or by nonimmune serum, as well as staining with affinity-absorbed primary Ab dilution (not done for PAI-2) yielded negative staining results.
After prolonged tissue fixation, good staining was achieved for tPA (Figure 1A), PAI-1, PAI-2, and uPAR up to 3 weeks of formalin fixation. For uPA, fixation longer than 3 days led to a marked decrease in the staining signal.
Specific Aspects
Important data are summarized in Table 1. Examples of stainings are shown in Figures 1 and 2.
uPA. Only the rabbit PAb (DAKO) yielded good results. Eighteen tumor lesions were stained after pronase pretreatment, and in 17 of 18 cases the staining patterns of the paraffin sections were equivalent to those of the frozen sections. In one lesion, stromal cell positivity was underrepresented. Five Abs performed well on tissue controls but were disappointing on tumor lesions because of the absence of stromal cell staining or because of high levels of nonspecific staining.
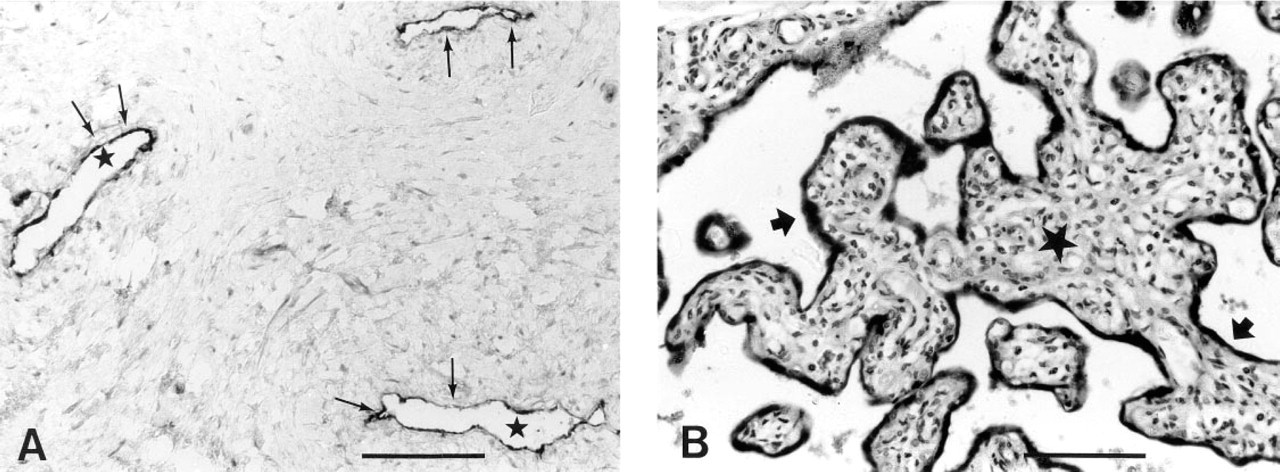
Staining of paraffin sections from control tissues. (
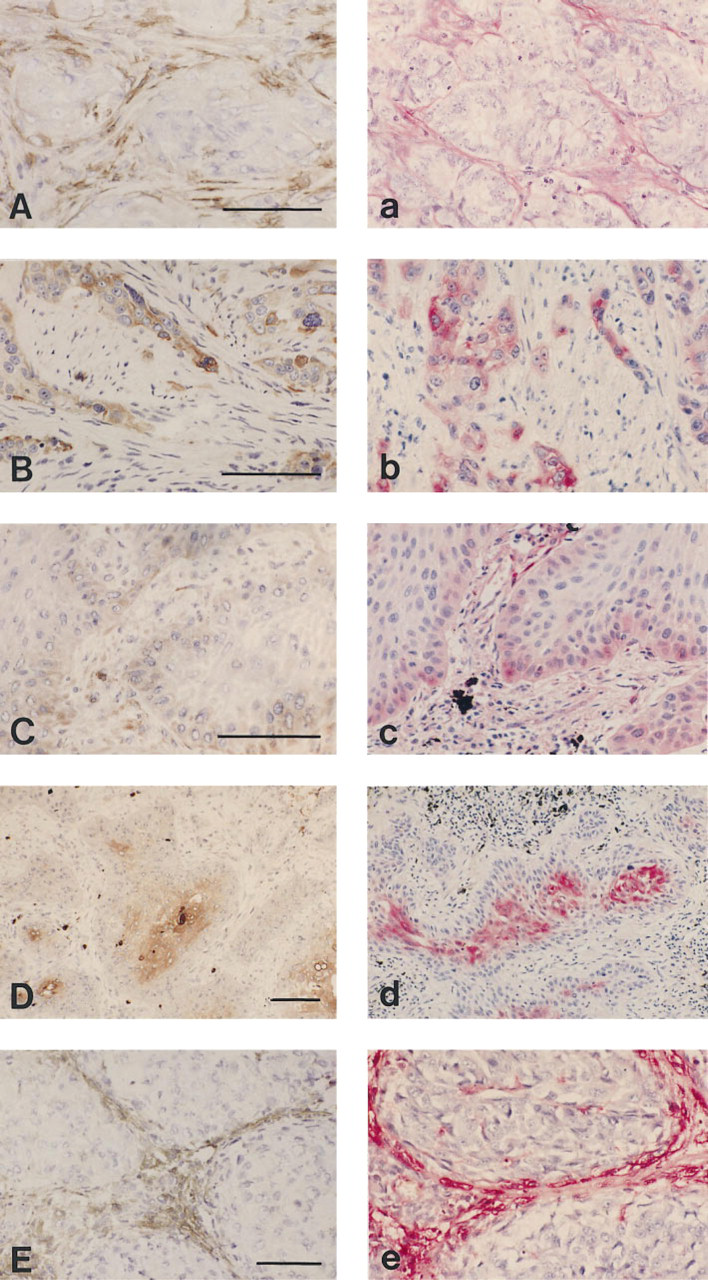
Staining for uPA (
tPA. Consistent staining results were obtained with the goat PAb (387) and the rabbit PAb. Fifteen tumor lesions were stained with both PAbs and showed concordance of staining pattern in frozen and in paraffin sections in 11 [goat PAb (387)] and 12 (rabbit PAb) cases, respectively. Discordancies were due to under-representation of stromal cell positivity in the paraffin sections, and in one case [with goat PAb (387)] to some nonspecific tumor cell staining.
PAI-1. Optimal staining results were obtained with the rabbit PAb and with the MAbs 380 and clone 1 (these two MAbs recognize different epitopes). Although these Abs stained tumor cells correctly, they all gave underrepresentation of stromal cell positivity, which was least severe for the PAb. Because a large number of tumor lesions with stromal cell staining in the frozen sections were included, concordance of staining in frozen and in paraffin sections was found in only 12 (PAb), 8 (clone 380), and 9 (clone 1) of 26 cases.
PAI-2. Good results were found with two goat PAbs (HD and BW). Twenty tumor lesions were stained with these goat PAbs, and completely satisfactory staining patterns were seen in all cases.
uPAR. Convincing staining results on paraffin sections were obtained with rabbit PAb and R2. Fifteen lesions were stained with these Abs after pronase pretreatment, and results were consistent in all cases.
Discussion
Antigen unmasking for the components of the PA system in paraffin sections yielded retrieved immunoreactivity for uPA, tPA, and PAI-1, markedly enhanced immunoreactivity for uPAR, and reduced background staining for PAI-2 compared with untreated sections. We were able to restore immunoreactivity in tissues that had been formalin-fixed for up to 3 weeks, except for uPA. Our results support the view that antigen unmasking by protease digestion and by MW cooking complement each other (Cattoretti et al. 1993; Dookhan et al. 1993). We also found that the effects of MW retrieval for the different components are highly pH-dependent, as described earlier by Evers and Uylings (1994) and by Shi et al. (1997). Whereas for PAI-2 a retrieval solution with pH 6.0 gave the best results, stainings for tPA and PAI-1 were successful only after MW retrieval in solutions with a more extreme pH of 2.5 and 10.0, respectively.
Whereas tumor cells exhibited a good response to unmasking procedures for all components, this was different for stromal cells. For PAI-1 in particular, and also to some extent for tPA, we found an underrepresentation of fibroblast-like cell positivity compared with the corresponding frozen section. Dookhan et al. (1993) demonstrated for vimentin how different levels of detection sensitivity may be needed to reveal the various cell types, and they caution that without optimal antigen retrieval a false cell type selectivity or selection bias towards cells with high antigen content will be introduced. Mighell et al. (1995) showed for fibronectin that two techniques of antigen retrieval revealed different but consistently restricted patterns of the total immunoreactivity seen on frozen sections. They suggest that some antigen isoforms may differ in their response to retrieval, or that various tissue constituents react differently on tissue processing and antigen unmasking. These considerations may also apply to PAI-1 (and to a lesser degree to tPA) in our test system.
For uPA, tPA, and PAI-2, good results could be obtained only with PAbs. For PAI-1, two MAbs (clone 1 and 380) performed almost as well as the rabbit PAb. For uPAR, the MAb R2 and the rabbit PAb performed equally well. In general, however, PAbs were more often successful on paraffin sections than MAbs, as found earlier by Åstedt et al. (1986) for PAI-2. This could be explained by the fact that, in contrast to MAbs, PAbs recognize a set of epitopes, which enhances the chance of epitope recognition.
Surprisingly, one mode of pretreatment proved optimal for different Abs directed against the same component. A plausible explanation might be that Abs found suitable on paraffin sections have common epitope reactivity. To address this question, we performed subsequent incubations on pretreated paraffin sections with two primary Abs directed against the same component but from different species (results not shown). For tPA, such a blocking incubation decreased the binding of the primary Ab incubated second. However, for PAI-1 and uPAR there was no noticeable decrease in signal. Therefore, common epitope reactivity does not appear to be the only reason for the fact that Abs of the same specificity require the same pretreatment. Because not all Abs that yield good results on frozen sections are suitable for paraffin sections, it holds that only a subset of the epitopes seems re-exposable. It is conceivable that the epitopes of such a subset are located close to each other on an easily unmaskable domain of the antigen. Cattoretti et al. (1993) also found that Abs directed against different epitopes of the same protein benefit by the same unmasking system. These authors noted that Abs with the same specificity for one epitope are not necessarily equally effective on paraffin sections.
Abs may react nonspecifically on paraffin sections. Indeed, with some Abs we encountered false reactivity on the tissue controls or positivity that deviated strongly from the patterns seen on frozen sections. In particular, false positivity of the tumor cells was occasionally observed in paraffin sections. Positivity, with or without antigen retrieval, is not always specific for the component of interest.
The major conclusions to be drawn on the basis of the present work are that (a) for each Ab used after a particular procedure of antigen retrieval, sensitivity and specificity on paraffin sections should be ascertained by use of control tissues and by comparison of staining profiles on frozen and paraffin sections, and (b) a carefully designed pre-test based on three different pH values (low, middle, and high) of antigen retrieval solution, a “test battery” as previously reported (Shi et al. 1997), is helpful to determine the optimal protocol of antigen retrieval for a certain Ab.
Footnotes
Acknowledgements
Supported by the Dutch Cancer Society (project number NKB-KWF 94–772).
We gratefully acknowledge Drs B.M. Mueller (The Scripps Research Institute, La Jolla, CA) for the M24met cell line, J. Askaa (DAKO, Glostrup, Denmark) for the rabbit PAb against uPA, E. Schüler (Behring Werke AG, Marburg, Germany) for the goat PAb against PAI-2, N. Brünner (Fin-sen Laboratory, Copenhagen, Denmark) for the Abs against uPAR, and J.H. Verheyen (Gaubius Laboratory IVVO-TNO, Leiden, The Netherlands) for antigen-coupled Sepharose beads.
Part of this work was performed in collaboration with the Concerted European Action BIOMED-1 research group on “Clinical Relevance of Proteases in Tumor Invasion and Metastasis.”
