Abstract
Brown spider (Loxosceles genus) venom causes necrotic lesions often accompanied by fever, hemolysis, thrombocytopenia, and acute renal failure. Using mice exposed to Loxosceles intermedia venom, we aimed to show whether the venom directly induces renal damage. The experimental groups were composed of 50 mice as controls and 50 mice that received the venom. Light microscopic analysis of renal biopsy specimens showed alterations including hyalinization of proximal and distal tubules, erythrocytes in Bowman's space, glomerular collapse, tubule epithelial cell blebs and vacuoles, interstitial edema, and deposition of eosinophilic material in the tubule lumen. Electron microscopic findings indicated changes including glomerular epithelial and endothelial cell cytotoxicity as well as disorders of the basement membrane. Tubule alterations include epithelial cell cytotoxicity with cytoplasmic membrane blebs, mitochondrial changes, increase in smooth endoplasmic reticulum, presence of autophagosomes, and deposits of amorphous material in the tubules. We also found that the venom caused azotemia with elevation of blood urea levels but did not decrease C3 complement concentration or cause hemolysis in vivo. Confocal microscopy with antibodies against venom proteins showed direct binding of toxins to renal structures, confirmed by competition assays. Double-staining immunofluorescence reactions with antibodies against type IV collagen or laminin, antibodies to venom toxins, and fluorescent cytochemistry with DAPI revealed deposition of toxins in glomerular and tubule epithelial cells and in renal basement membranes. Two-dimensional electrophoresis showed venom rich in low molecular mass and cationic toxins. By immunoblotting with antibodies to venom toxins on renal extracts from venom-treated mice, we detected a renal binding toxin at 30 kD. The data provide experimental evidence that L. intermedia venom is directly involved in nephrotoxicity.
L
A number of enzymes and biologically active molecules that might contribute to the deleterious effects of the venom have been identified and biochemically characterized. A sphingomyelinase D of 32-35 kD isolated from brown spider venom can induce dermonecrosis, platelet aggregation, and experimental hemolysis (Futrell 1992). Metalloproteases of 32-35 kD and 20-28 kD identified in the venom with gelatinolytic, fibronectinolytic, and fibrinogenolytic activities can also play a role in hemorrhage evoked by envenomation, such as hemorrhage into the dermis, injury of blood vessels, imperfect platelet adhesion, and the defective wound healing observed in some cases (Feitosa et al. 1998; Veiga et al. 2001a,b; da Silveira et al. 2002; Zanetti et al. 2002). A hyaluronidase with electrophoretic mobilities at regions of 33 kD and 63 kD is likely to contribute to the gravitational spread of dermonecrotic lesions, a hallmark of brown spider bites (Wright et al. 1973; Futrell 1992). A number of other molecules and activities have been identified in the venom, including a lipase, alkaline phosphatase, and proteolytic activities on entactin, basement membranes, and the protein core of a heparan sulfate proteoglycan from endothelial cells (Futrell 1992; Veiga et al. 2000,2001b). The mechanism underlying the involvement of these individual venom constituents or activities in the noxious effects of the venom on cell tissue has not been fully determined.
Some reports have indicated the participation of blood cells and molecular components in the noxious effects of the venom. The serum amyloid P component appears to be a target for platelet activation and ischemic effects and is likely to play a role in the necrosis caused by the venom (Gates and Rees 1990). Leukocytes, and especially polymorphonuclear cells (PMNs) such as neutrophils and eosinophils, appear to play a role in the dermonecrotic lesion evoked by the venom, because histopathological findings have revealed a massive infiltration of these cells into the dermis and related structures in the dermonecrotic regions induced by the venom (Elston et al. 2000; Ospedal et al. 2002). Depletion of leukocytes in the blood results in reduction of clinical signs in the skin injected with venom (Smith and Micks 1970). The complement system in the plasma also appears to participate in the deleterious activities of the venom, especially on erythrocytes, evoking a complement-dependent hemolysis (Futrell 1992).
Although some clinical signs of loxoscelism have been well described, and putative molecules in the venom and physiopathological events involved in cell destruction have been characterized as described above, data about renal disorders evoked by brown spiders have been limited to earlier reports describing clinical data from victims (Futrell 1992; Lung and Mallory 2000). We report here the effect of L. intermedia venom on kidney structures. Mice were used because these animals do not develop dermonecrotic lesions induced by Loxosceles venom, so that the occurrence of nephrotoxicity secondary to complications of dermonecrotic lesions can be ruled out. We hope to bring some insight into loxoscelism that could be useful to physicians who diagnose and treat the victims.
Materials and Methods
Reagents
Polyclonal antibodies to L. intermedia venom toxins were produced in a rabbit using crude venom as antigen and complete Freund's adjuvant (Sigma; St Louis, MO) for the first primary injection at one SC point and two IM points (20 μg of proteins as total antigens were used divided into three parts). As a booster for the second, third, and fourth injections, incomplete Freund's adjuvant (Sigma) was similarly injected at 2-week intervals. Twelve days after the last immunization, blood was collected and the production of specific antibodies analyzed by ELISA and Western blotting (Harlow and Lane 1988). Hyperimmune IgGs were purified from serum using protein-A Sepharose (Amersham Biosciences; Piscataway, NJ) as recommended by the manufacturer. Polyclonal antibodies that recognize type IV collagen were purchased from Chemicon International (Temecula, CA) and monoclonal antibodies against laminin were purchased from DAKO (Carpinteria, CA). Fluorescein- and rhodamine-conjugated anti-IgG antibodies were purchased from Chemicon. DAPI (4′,6-diamidino-2-phenylindole HCl) nucleic acid stain was purchased from Molecular Probes (Eugene, OR).
Spider Venom Extraction
The venom was extracted from spiders captured from the wild and kept for a week without any food but with water ad libitum. The venom was extracted from the fangs of spiders by electrostimulation (15 V) applied to the cephalothorax and was collected with a micropipette, dried under vacuum, and frozen at −85C until use. Pools of venom collected from 100 to 200 spiders in different batches were used during all the experiments, involving approximately 1000 spiders (Feitosa et al. 1998). Protein content was determined by the Coomassie Blue method (Bradford 1976).
Animals
Adult Swiss mice weighing approximately 25 g from the Central Animal House of the Federal University of Paraná were used for in vivo experiments with the venom. All experimental protocols using animals were performed according to the “Principles of Laboratory Animal Care” (NIH Publication 85-23, revised 1985) and Brazilian federal laws.
Venom Administration
Pooled crude venom and mouse samples of 1 mg of protein/kg were diluted in PBS (pH 7.3). These samples were injected IP in a volume of 100 μl in each mouse. The animals were divided into two groups, a control (C) group and a test (T) group. The control group consisted of five animals receiving only PBS and the test group consisted of five animals receiving L. intermedia venom. During the experimental procedures, the envenomation of animals was repeated at least 10 times, completing a number of 50 animals as controls and 50 animals that received the venom. All animals were kept under the same experimental conditions. All kidney and blood samples were collected from living animals.
Kidney and Blood Sample Collections and Laboratory Analysis
Kidney and blood (directly from the heart) samples were obtained from mice anesthetized with ketamine (Agribands; Paulinia, SP, Brazil) and acepromazin (Univet; São Paulo, SP, Brazil). Blood was anticoagulated with EDTA-K3 and used for red cell, hemoglobin, hematocrit, leukocyte, and platelet counts that were determined with an automated CELL-DYN 1,400 blood counter (Abbott Laboratories; Chicago, IL). Urea and C3 complement were determined in serum. Assays were performed using standardized techniques and reagents as described by Kaplan and Pesce (1996) and Henry (2001).
Statistical Analysis
The Student's t-test for unpaired observations was used to detect statistically significant differences between control and test groups for red cell concentration (106 ml/blood), hemoglobin (g/dl blood), hematocrit (%), platelets (103 ml/blood), leukocytes (103 ml/blood), serum urea (mg/dl), and C3 complement (mg/dl). The threshold level for significance was P = 0.05. All statistical calculations were done with the GraphPad InStat program version 3.00 for Windows 95. Morphometric analysis of sections stained with hematoxylin and eosin or from electronmicrographs were measured by use of software Jandel Sigma Scan Pro.
Gel Electrophoresis
Lysed renal cells were obtained by treatment of kidneys with lysis buffer (50 mM Tris-HCl, pH 7.3, 1% Triton X-100, 50 mM NaCl, 1 mM CaCl2,1 mM phenylmethanesulfonyl fluoride, and 2 μg/ml aprotinin) for 15 min at 4C. The extract was clarified by centrifugation for 10 min at 13,000 × g. Renal extracts or crude venom (normalized for their protein contents) were submitted to electrophoresis under non-reducing conditions. Linear gradient 3-20% or 8-18% SDS-PAGE was performed as described by Laemmli (1970). For protein detection, gels were stained with Coomassie Blue. For immunoblotting, proteins were transferred to nitrocellulose filters overnight as described by Towbin et al. (1979). The molecular mass markers used were from Sigma. A two-dimensional gel was run with some modifications as previously described by the manufacturers using Immobiline Dry Strip Gel, pH range 3-10 (Amersham Biosciences). Crude venom 100 μg collected in water was diluted in rehydration solution (6 M urea, 2 M thiourea, 2% w/v CHAPS, 1% IPG buffer, and a trace of bromophenol blue) and applied to an IEF strip. The second dimension was carried out using 8-18% linear gradient SDS-PAGE under non-reducing conditions. Gel was stained with the silver method as described by Heukeshoven and Dernick (1986).
Histological Methods for Light Microscopy
Kidneys were fixed in modified Carnoy's fixative (5% acetic acid instead of 10% as originally proposed) for 3 hr. After fixation, tissues were processed for histology, embedded in paraffin, and cut into 4-μm sections. The sections were stained with hematoxylin and eosin, acid-Schiff (PAS), and silver and by the method of Rosenfeld (Culling et al. 1985; Beautler et al. 1995).
Transmission Electron Microscopy
Kidneys were fixed with modified Karnovsky's fixative (without calcium chloride and with glutaraldehyde 2.5%) (Karnovsky 1965) for 2 hr, washed in 0.1 M cacodylic acid buffer, pH 7.3, postfixed in 1% OsO4 in 0.1 M cacodylic acid buffer, pH 7.3, for 1 hr, dehydrated with ethanol and propylene oxide, embedded in Epon 812, contrasted with uranyl acetate and lead citrate, and examined with a JEOL-JEM 1200 EX II transmission electron microscope at an accelerating voltage of 80 kV (Peabody, MA).
Immunofluorescence and Fluorescence Cytochemistry
For immunofluorescence microscopy, kidney tissues were fixed with 2% formaldehyde in PBS for 30 min at 4C, incubated with 0.1 M glycine for 3 min, and blocked with PBS containing 1% BSA for 1 hr at room temperature (RT). Histological sections were incubated for 1hr with specific antibodies raised against laminin (0.33 μg/ml), type IV collagen (1:40), and venom toxins (2.0 μg/ml) as described above. The sections were washed three times with PBS, blocked with PBS containing 1% BSA for 30 min at RT, and incubated with fluorescein- or rhodamine-conjugated anti-IgG secondary antibodies (Chemicon) at RT for 40 min. After washing with PBS, samples were observed under a confocal fluorescence microscope (Confocal Radiance 2,100; BioRad, Hercules, CA) coupled to a Nikon-Eclipse E800 with Plan-Apochromatic objectives (Sciences and Technologies Group Instruments Division; Melville, NY). For nuclear fluorescence cytochemistry, samples of renal tissue were incubated with DAPI (O.5 μg/mL diluted in PBS) for 5 min (Molecular Probes). The samples were washed and observed under a confocal microscope as above. For antigen competition assay, the immunofluorescence protocol was the same as described above except that the hyperimmune serum to venom toxins was incubated previously for 1 hr with 50 μg/ml of crude venom diluted in PBS. Then the mixture was incubated with renal biopsies identically as above.
Results
Histopathological Findings in Kidneys from Mice That Received L. intermedia Venom
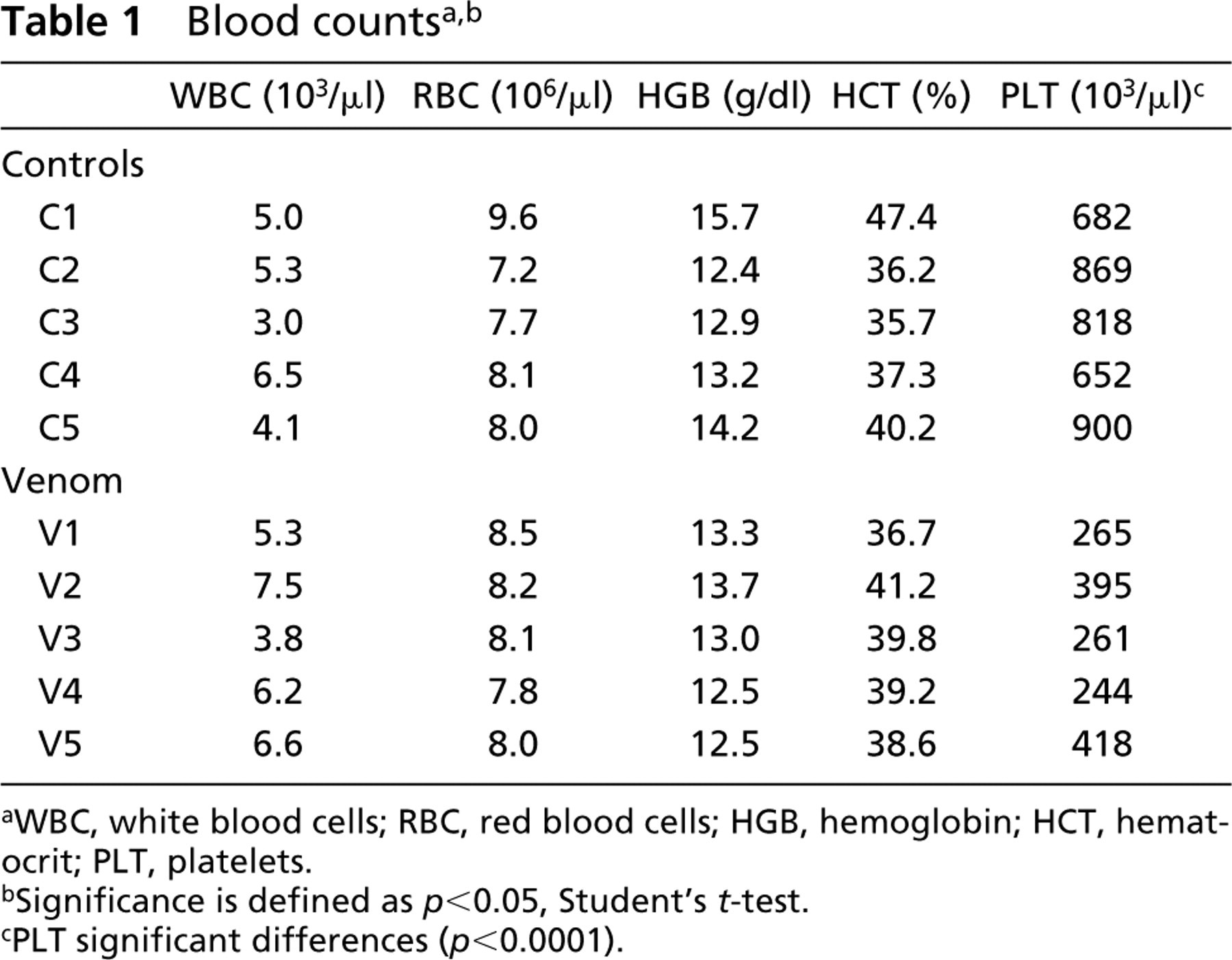
To obtain information about the degree of renal damage caused by Loxosceles venom, mice were exposed IP to L. intermedia venom for 4 hr. As shown in Figure 1, the effects of the venom on the kidney revealed a complex pattern of nephrotoxicity. Light microscopy and histochemistry techniques revealed glomerular alterations consisting of collapse of basement membranes and deposition of hyaline and eosinophilic masses in the glomeruli (hyalinization), as well as focal intraglomerular erythrocytes. Tubule alterations were detected by accumulation of eosinophilic material in the lumen of cortical and medullary tubules, interstitial edema with extravascular red blood cells, and vacuolar degeneration of proximal and distal tubules. Morphometric analysis (Figure 1A) showed that 75-80% of the kidney area was affected by the venom. No glomerular or tubule leukocyte infiltration was detected.
Light microscopic analysis of kidneys from venom-treated mice. Sections of kidneys from mice treated with L. intermedia venom were stained with a combination of dyes and analyzed by light microscopy. (
Ultrastructural Evidence of Renal Injuries
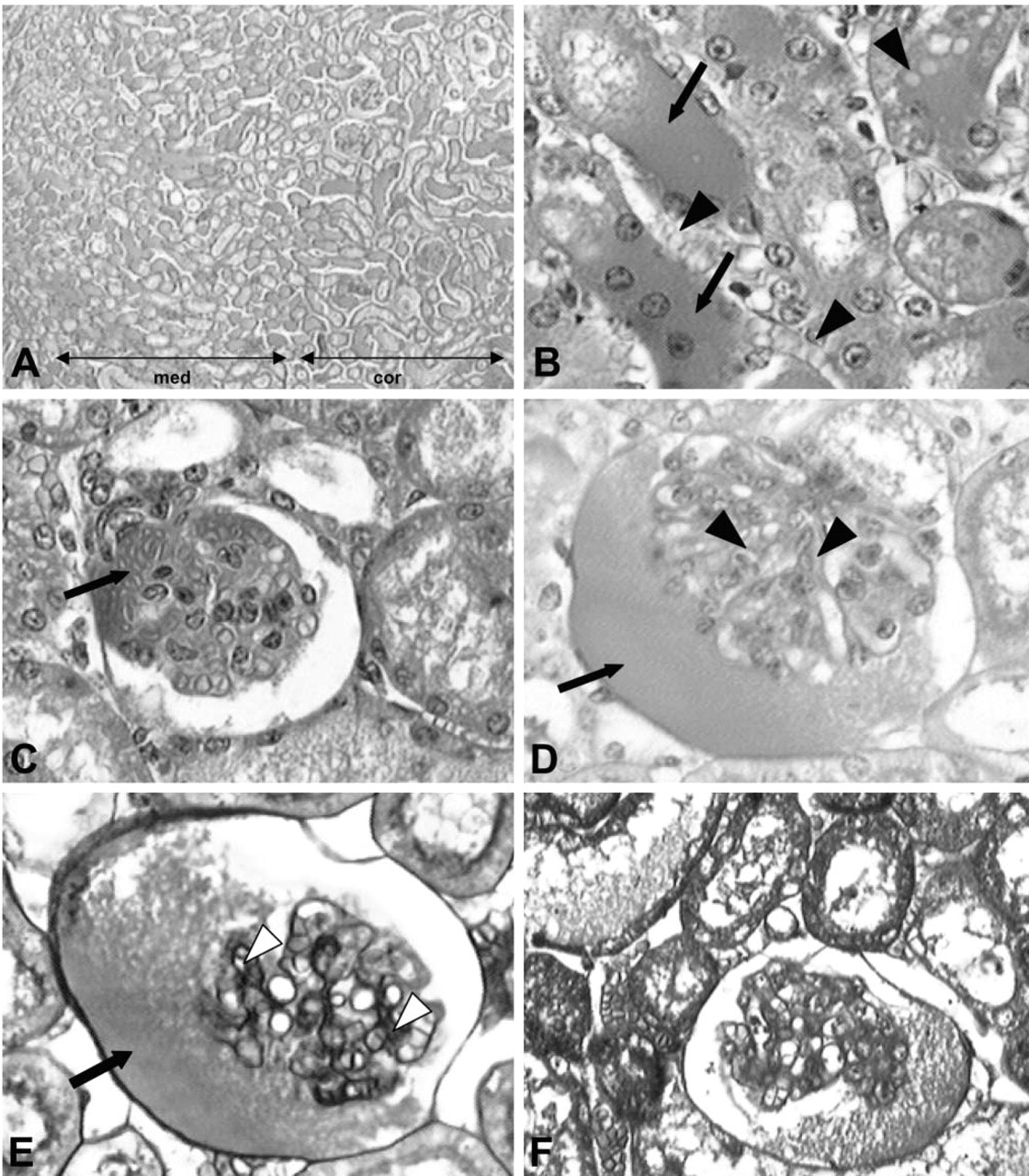
To improve the evidence of the activity of brown spider venom toxin on kidney structures and to better understand these alterations, biopsies from venom-treated mice were studied by transmission electron microscopy. As shown in Figure 2, additional evidence of glomerular damage was provided by structural signs of podocyte cytotoxicity as shown by many disturbed foot processes (pedicels) and filtration slits. At the capillary level, fenestrated endothelial cells also showed signs of alterations, such as detachment from subendothelial basement membrane and structural signs of disorganization of the fenestra. The basement membrane was decreased compared to the control group. Morphometric analysis of glomerular basement membranes showed a general reduction of approximately 24%. Tubule injuries included deposition of amorphous electron-dense material in the distal tubule lumen. Necrotic destruction of tubule epithelial cells can be visualized by vacuolization and blebs on the cytoplasmic membrane, diffuse destruction of mitochondria, prominent smooth endoplasmic reticulum, and autophagosomes indicating necrotic cell destruction.
Ultrastructural findings of kidneys from venom-treated mice. Transmission electron micrographs of cross-sectioned kidneys exposed to the venom. (
Laboratory Investigations After Administration of Brown Spider Venom
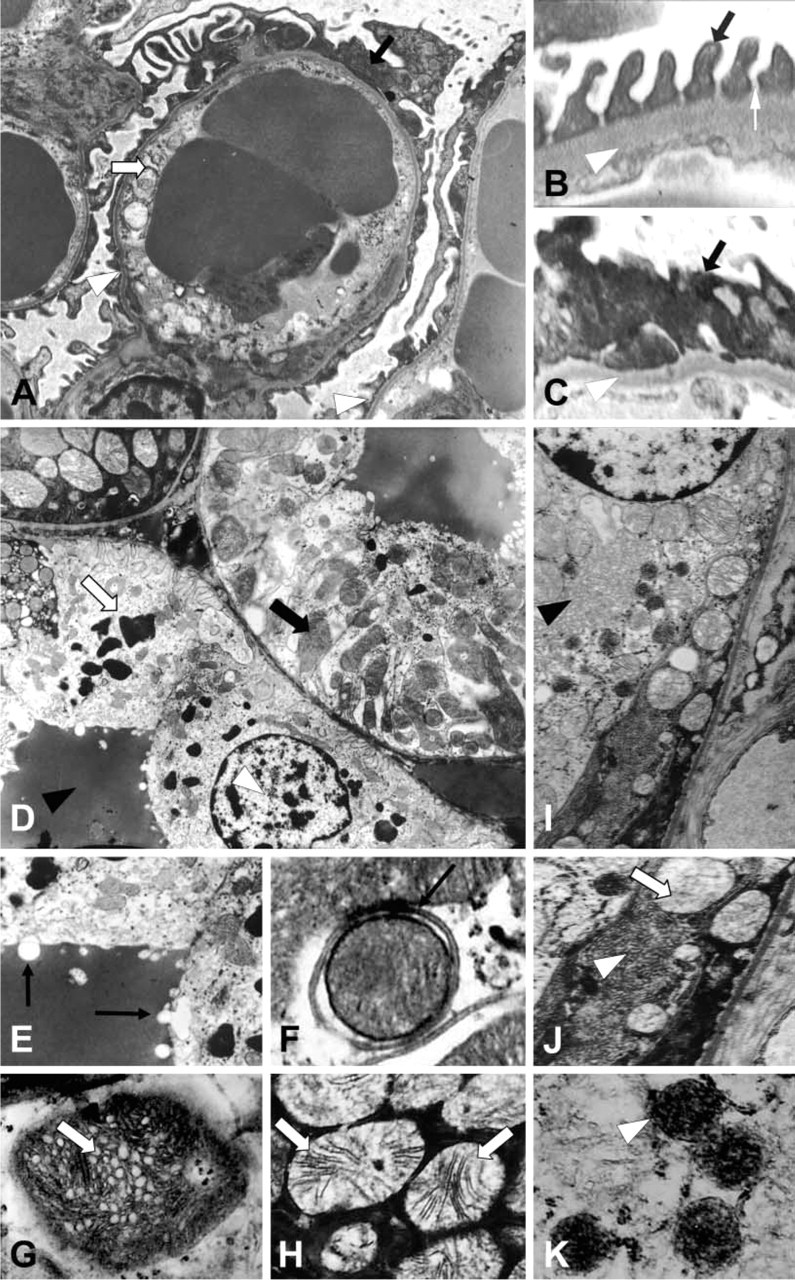
Our initial approach to study the involvement of venom-induced hemolysis in renal pathogenesis in the experimental model used was to determine blood cell counts, urinalysis, C3 complement, and serum urea and to compare venom-treated mice with a control group. As shown in Table 1, neither red blood cell count, hemoglobin, or hematocrit values showed significant differences between venom-treated and control groups, suggesting that the venom was unable to lyse mouse erythrocytes in vivo under the conditions assayed. In addition, no significant evidence of hemoglobinuria or of the presence of red blood cells in urine was observed in venom-treated mice (Table 2), indicating that hemolysis was not taking place. Table 3 shows no significant differences in C3 complement component between venom-treated and control mice, demonstrating that the venom was unable to decrease this serum molecule in vivo under the conditions used. On the other hand, as shown in Table 4, serum urea was significantly increased in venom-treated mice compared to control group. Finally, renal biopsies from mice treated with the brown spider venom were negative for iron when submitted to histochemistry by the Prussian blue method (Figure 3), supporting the idea that venom-induced hemolysis was not involved in nephrotoxicity in the model used.
Sections from venom-treated kidneys stained with Prussian blue. L. intermedia venom-treated sections from mouse kidneys were stained with Prussian blue plus hematoxylin to detect iron (hemoglobin) deposition along the renal structures. Although deposition of proteinaceous materials in the tubule lumen (arrows) (
WBC, white blood cells; RBC, red blood cells; HGB, hemoglobin; HCT, hematocrit; PLT, platelets.
Significance is defined as p<0.05, Student's t-test.
PLT significant differences (p<0.0001).
Urinalysis a

−, negative; +, trace.
Complement (C3)
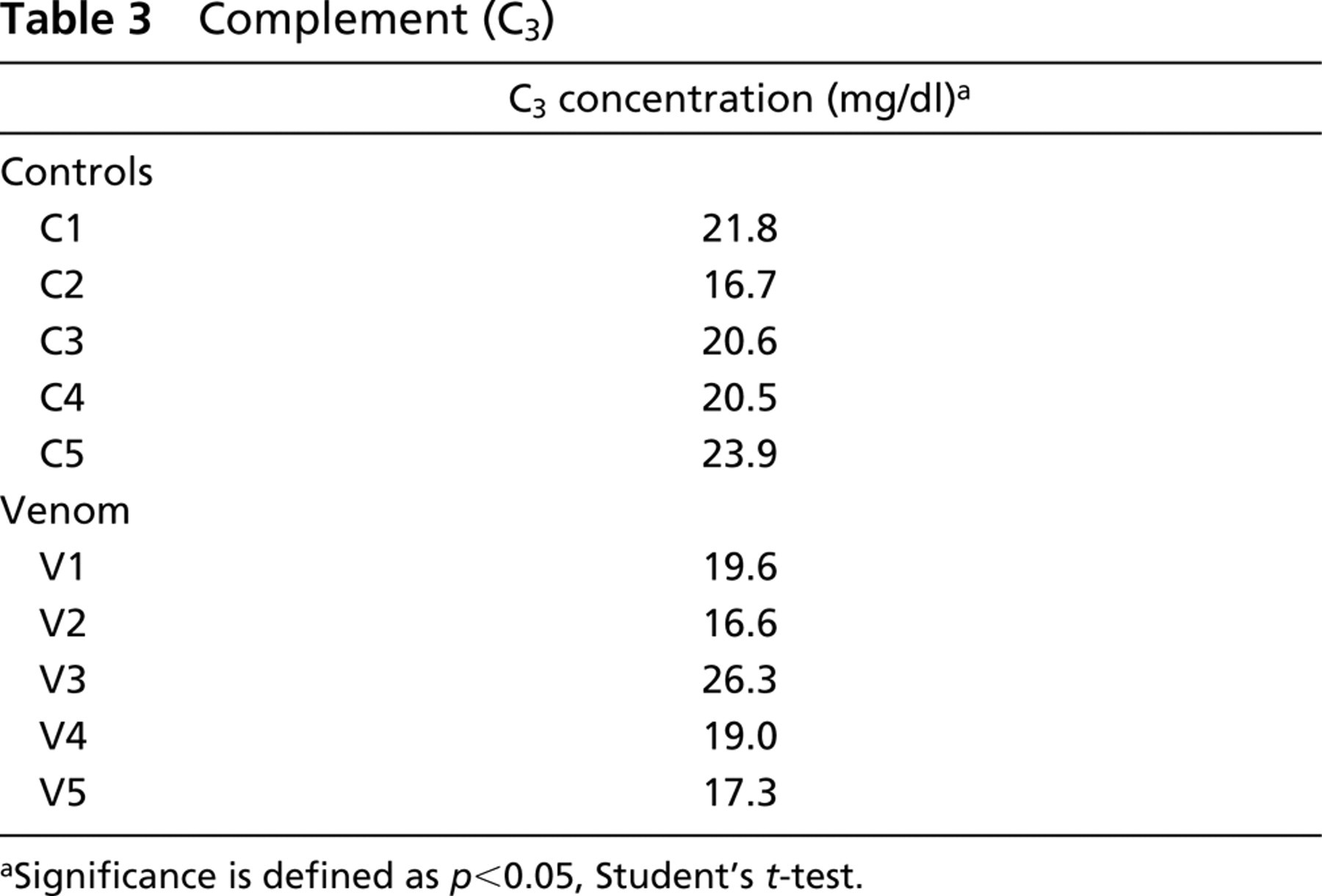
Significance is defined as p<0.05, Student's t-test.
Urea a
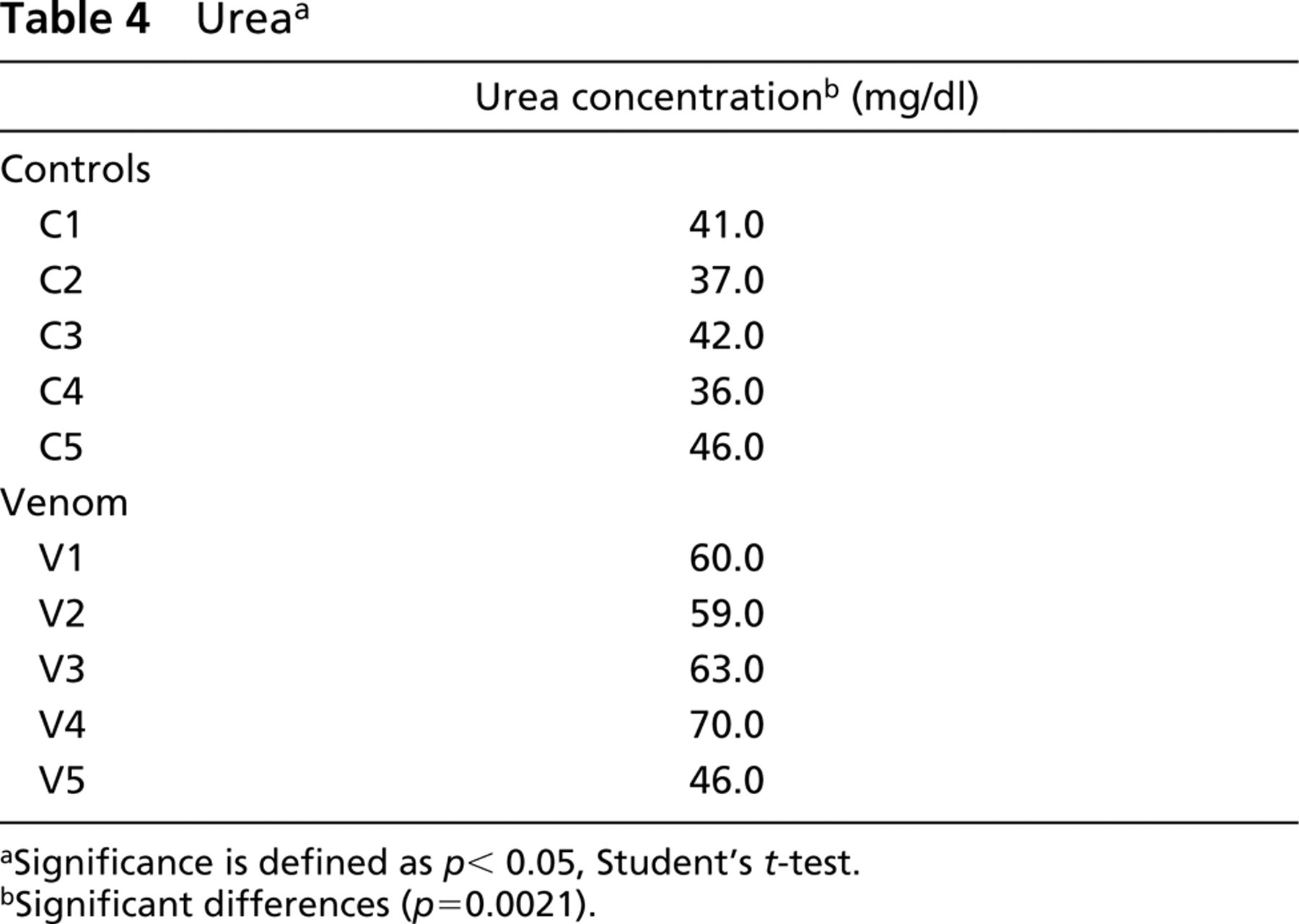
Significance is defined as p < 0.05, Student's t-test.
Significant differences (p=0.0021).
Evidence That Loxosceles Venom Toxins Bind Directly to Kidney Structures
To study the molecular pathways of venom pathogenesis in the kidney, we investigated the possibility of a direct interaction of venom toxins with kidney structures by submitting renal biopsies from venom-treated and control mice to immunofluorescence using affinity-purified hyperimmune IgG that reacts with venom toxins (Montero 2003). As shown in Figure 4, we found that hyperimmune IgG produced a positive reaction in renal biopsies from venom-treated mice but did not react with biopsies from normal mice by immunofluorescence. In addition, confirm the antibody-specific interactions of venom toxins with the kidney, we repeated the same immunofluorescence approach, this time incubating hyperimmune IgG with a medium containing venom in solution and then exposing renal biopsies from venom-treated mice to this mixture (antigen competition assay). We found that soluble venom effectively inhibited binding of antibodies to the kidney, supporting the evidence of venom toxins as “planted antigens” bound to renal structures.
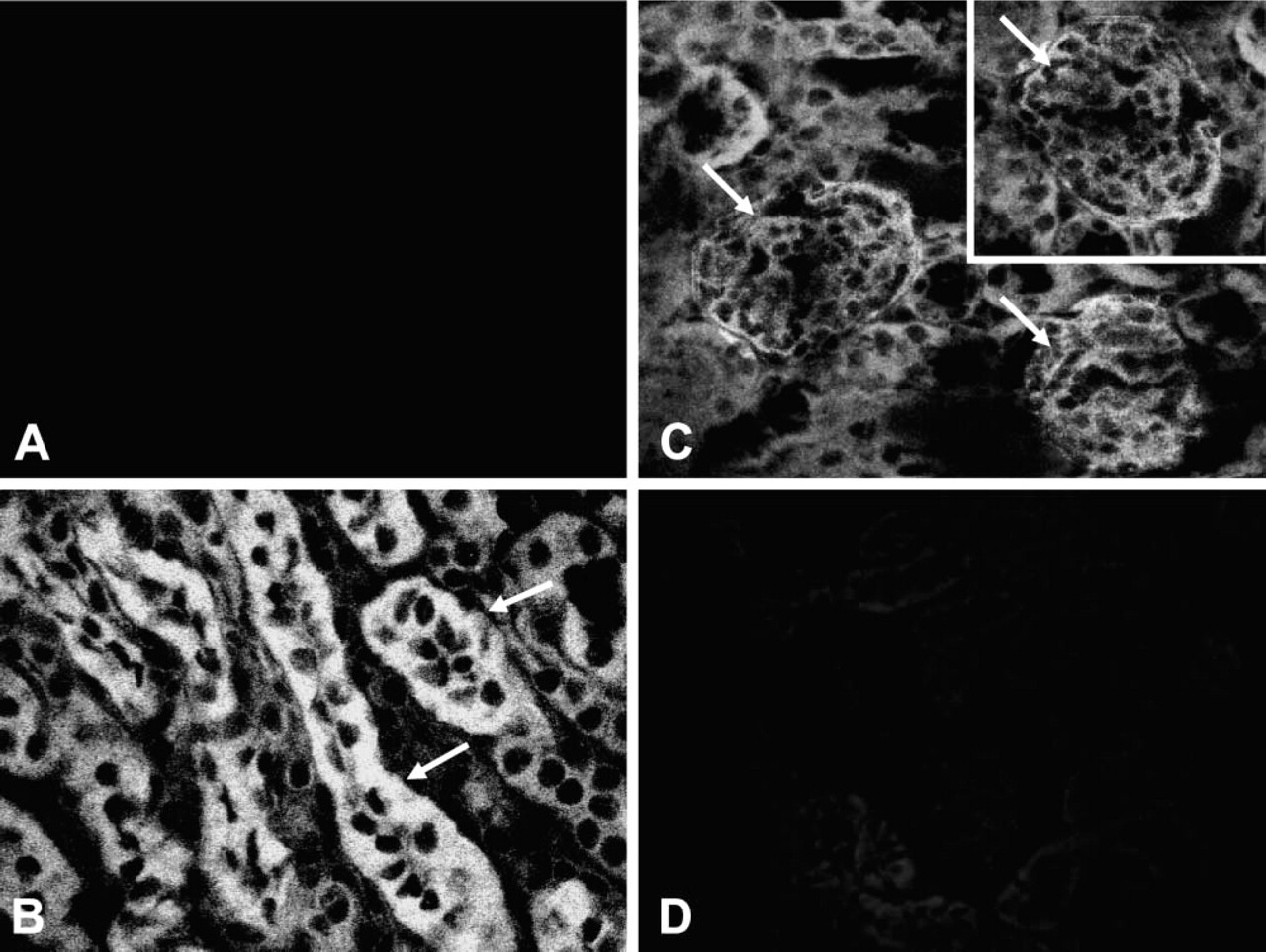
Confocal immunofluorescence microscopy analysis of kidney sections from venom-treated mice. Cross-sectioned kidneys immunolabeled with purified antibodies against L. intermedia venom toxins. (
Renal Injuries by Loxosceles Venom Resulting from Binding of Toxins to Glomerular and Tubule Cells and Basement Membrane Structures
After confirming the direct binding of venom toxins to the kidney, we examined the possibility that intrinsic kidney components were targets of the toxins. We speculated that toxins can act as “planted antigens” and bind to renal structures as previously reported for several other antigens such as viral or bacterial products and drugs (Barnes 1989; Kerjaschki and Neale 1996; Cotran et al. 1999). We performed double staining and confocal microscopy immunofluorescence reactions on renal biopsies from venom-treated mice using antibodies that react with type IV collagen and laminin (two molecular constituents of basement membranes; see Courtoy et al. 1982 and Rohrbach and Timpl 1993) and antibodies that react with venom toxins as well as fluorescent cytochemistry for chromosomes with DAPI. As shown in Figure 5, there was overlapping fluorescence of reactions using antibodies to laminin or type IV collagen and antibodies to venom toxins. Fluorescence staining cytochemistry of nuclei with the blue fluorescent dye DAPI revealed no overlapping with anti-venom fluorescence pattern. These data support diffuse binding and deposition of venom toxins along the glomerular and tubule cells, with prominent staining mainly in tubule cells and along renal basement membranes.
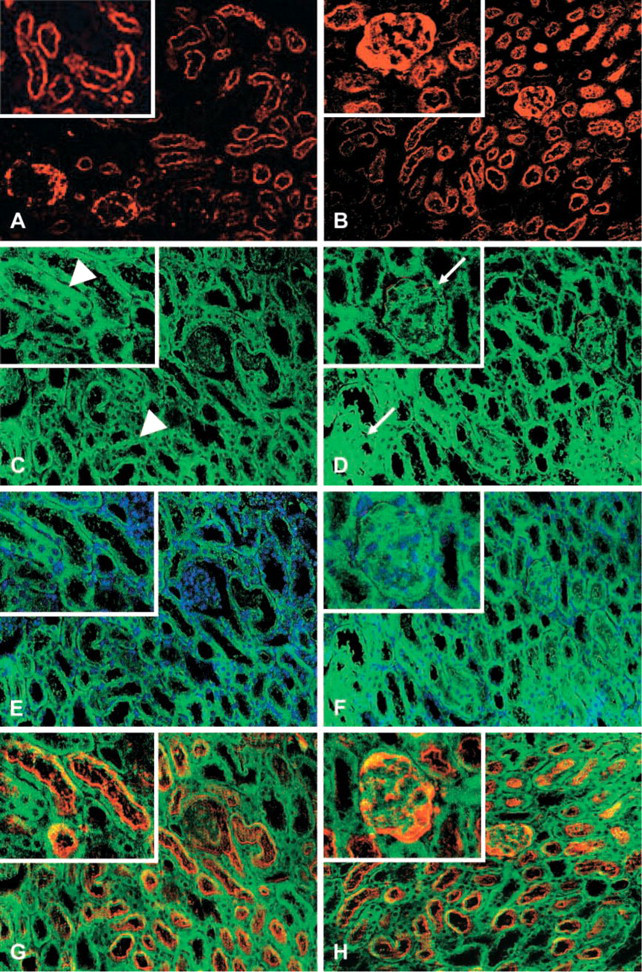
Venom toxin deposition along the glomerular and tubule cells and renal basement membranes detected by confocal double-staining immunofluorescence microscopy or fluorescence cytochemistry. Sections of kidneys from venom-treated mice were incubated with antibodies against basement membrane constituents such as laminin (
L. intermedia Venom Is Enriched in Cationic and Low Molecular Mass Proteins with Toxin(s) of Approximately 30 kD Binding to Intrinsic Renal Components
To corroborate the findings described above, we looked for the molecules in the venom involved in noxious influences on renal structures. For this purpose and to study venom toxins' physicochemical properties such as their charge and size, crude venom was submitted to a two-dimensional electrophoresis (isoelectric focusing and gradient SDS-PAGE) that was stained by the highly sensitive monochromatric silver method (Figure 6A). The venom was enriched in basic toxins at physiological pH, with low molecular mass in the range of 35-20 kD. Such cationic and low molecular mass properties might explain the localization of venom toxins in the kidney by interacting with components of the nephron. To strengthen such evidence, renal lysates from venom-treated mice were electrophoresed and immunoblotted with hyperimmune serum to venom toxins (see Materials and Methods). Antigen competition assays were also performed to substantiate the specificity of immune reactions. As shown in Figure 6B, we detected a band at 30 kD, in the venom-treated lysate, which specifically reacted with antibodies against venom toxins, confirming the above results and identifying toxin(s) from L. intermedia venom as a direct ligand on renal structures.
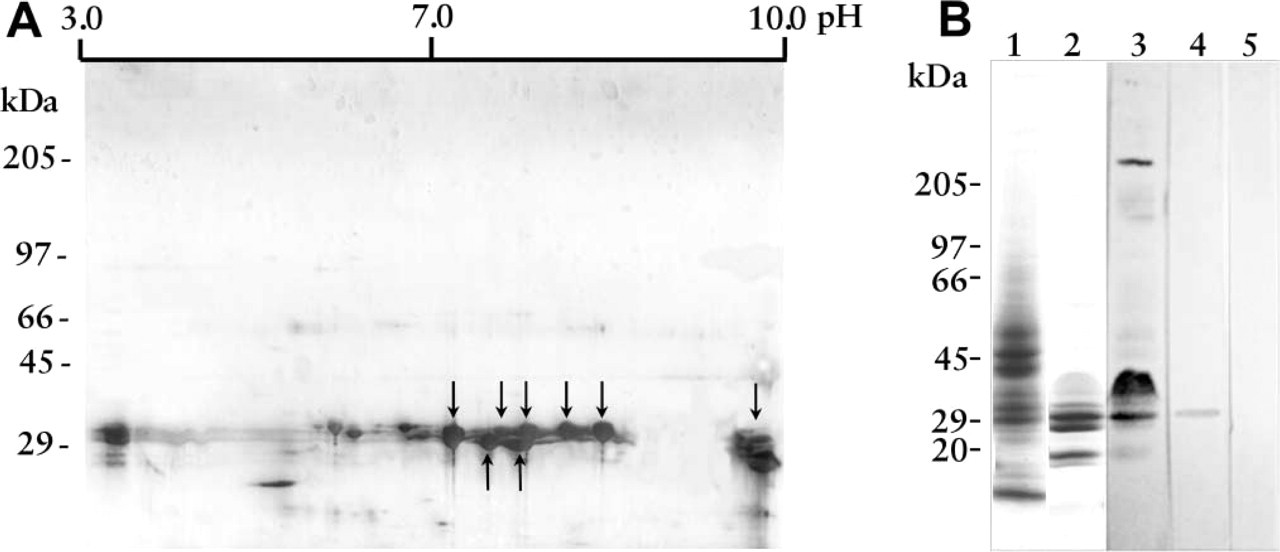
Identification of 30-kD venom toxin(s) as direct ligands of renal structures. Crude L. intermedia venom was separated by two-dimensional electrophoresis (isoelectric focusing and linear gradient 8-18% SDS-PAGE) under non-reducing conditions. The gel was stained by the silver method. Molecular mass protein standard positions are shown at left and pH gradient at the top. Arrows point to cationic proteins. (
Discussion
Clinical data have shown that envenomation by brown spiders can have a severe nephrotoxic effect (Futrell 1992; Lung and Mallory 2000). This venom nephrotoxicity, although less common than necrotic lesions and gravitational spreading (which are the hallmark signals of poisoning), is generally involved in the complications that occur after spider bites (Futrell 1992; Lung and Mallory 2000). The clinical and laboratory features observed in victims may include hemoglobinuria and proteinuria (Futrell 1992; Williams et al. 1995; Lung and Mallory 2000). According to some specialists, the pathological processes in the kidney may reflect hematologic disturbances such as intravascular hemolysis and disseminated intravascular coagulation caused by envenomation, which may lead to renal failure (Futrell 1992; Williams et al. 1995; Lung and Mallory 2000). However, there is no direct experimental evidence confirming such a hypothesis. On the other hand, renal disorders may also be the consequence of toxic components of the venom that act directly on renal cells or structures such as glomerular and tubule basement membranes, as demonstrated for several venoms (Bjarnason and Fox 1995; Veiga et al. 2000). Experimentally, it was demonstrated that the venom is cytotoxic to rabbit blood vessel endothelial cells (CLPs) (Veiga et al. 2001b) and human umbilical vein endothelial cells (HUVECs) (Patel et al. 1994) in culture and also to endothelial cells from skin blood vessel walls of rabbits intradermally injected with the venom (Zanetti et al. 2002). The venom also acts on EHS (Engelbreth-Holm-Swarm tumor) basement membrane integrity and on purified basement membrane molecules such as entactin, fibronectin, and heparan sulfate proteoglycan (Feitosa et al. 1998; Veiga et al. 2000,2001b) but, again, there is no direct experimental evidence for venom cytotoxicity to renal cell lines or renal basement membrane structures in vivo.
To obtain additional information about the extent of renal damage and the mechanisms of the disorders induced by L. intermedia venom, we presented here laboratory and biochemical data and morphological data from examinations of kidney structures from mice exposed to the venom. Histological studies showed that envenomation induced a complex pattern of nephrotoxicity. A general view of renal tissue revealed alterations at the level of glomerular and tubule structures, supporting the nephrotoxic activity of venom toxins. The glomerular damage becomes apparent by the presence of extravascular red blood cells around the glomerular capillaries in Bowman's space. A protein-rich exudate inside Bowman's space confirms some damage at the glomerular level, suggesting loss of vascular integrity. In addition, tubule injuries are supported by the presence of proteinaceous material in their lumen in addition to tubule edema, causing the tubules to be susceptible to ischemic damage caused by occlusion. Ultrastructural features of nephrotoxicity were further supported by glomerular and tubule disorders. The azotemia detected by the increase in serum urea strengthened this evidence.
The mechanism by which Loxosceles venom induces nephrotoxic lesions has remained elusive and is currently unknown. In contrast to cutaneous lesions evoked by the venom, in which leukocytes (neutrophils) play an essential role in pathogenesis (Smith and Micks 1970; Futrell 1992; Patel et al. 1994; Ospedal et al. 2002), in the model studied here there was no leukocyte infiltration in the presence of the renal injuries, which were not associated with inflammatory disease.
On the other hand, several case reports have described intravascular hemolysis associated with brown spider envenomation (Murray and Seger 1994; Williams et al. 1995), and many studies have indicated a direct hemolytic activity of Loxosceles venom on erythrocytes (Futrell 1992). Such hematological disturbances could be related to renal injuries as secondary agents (Futrell 1992; Williams et al. 1995; Lung and Mallory 2000). We found that, in the model and under the conditions used, despite the renal injuries caused by the venom there was neither a direct hemolytic effect in vivo nor hemoglobin deposition in the kidney structures. These results agree with those described by Futrell (1992), who pointed out the susceptibility of some animal species to venom-induced hemolysis. Human and swine erythrocytes are more susceptible to hemolysis than rabbit and guinea pig erythrocytes. We cannot rule out the involvement of venom hemolysis in the human renal failure evoked by the venom. Nevertheless, because the venom did not induce dermonecrosis (data not shown) or hemolysis in mice and was extremely active against renal integrity, we can speculate about a direct and primary activity of the venom on kidney structures.
We confirmed this possibility by confocal immunofluorescence microscopy using antibodies to venom toxins. We were able to detect toxins as “planted antigens” deposited along the kidney structures of animals exposed to the venom. A competition assay using crude venom toxins in solution blocked the immunofluorescence positivity in the kidney of venom-treated animals, further supporting this evidence and the idea of “planted toxins.” The present results agree with several reports indicating the binding of exogenous molecules, such as bacterial products, viral antigens, and drugs, to intrinsic components of renal structures as etiological agents of renal injuries (Kerjaschki and Neale 1996; Cotran et al. 1999). In the model used here, we did not find a role for immune mechanisms in renal injuries. Because the biopsies were collected just 4 hr after exposure to the venom, there was no alteration in C3 complement levels in the serum of venom-treated animals compared to controls, and an immunofluorescence assay with an anti-mouse IgG was negative (data not shown), excluding the possibility of renal deposits of immunoglobulins. Taken together, the above results support the notion that venom toxins act as direct ligands in renal structures and as primary agents, playing a role in renal disorders.
In addition, our data also showed that venom toxins bind to glomerular and tubule cells and basement membranes but do not bind to kidney cell nuclei. This conclusion was based on double-staining immunofluorescence reactions that demonstrated co-localization of venom toxins with basement membrane constituents such as type IV collagen and laminin, but no co-localization along the chromosomes revealed by DAPI. The basement membranes are specialized extracellular matrices involved in several physiological events, especially those dependent on cell adhesion. In the kidney these structures play a role in glomerular filtration during urine formation in addition to organizing podocyte, endothelial, and epithelial cell adhesion (Courtoy et al. 1982; Rohrbach and Timpl 1993; Cotran et al. 1999). The deposition of venom toxins along the renal basement membranes can explain glomerular epithelial cell injury, fenestrated endothelial cell cytotoxicity, hyalinosis, and proteinuria, as well as tubule cell damage. Because some L. intermedia venom toxins are proteases, with the ability to degrade basement membrane constituents (as discussed above), such venom injuries can result from loss of renal basement membrane integrity with consequent cytotoxicity to epithelial and endothelial cells and detachment and loss of the glomerular basement membrane charge barrier.
Using two-dimensional electrophoresis, we observed that L. intermedia venom is enriched in basic proteins with molecular masses ranging from 35 to 20 kD. Immunoblotting analysis using antibodies to venom proteins identified venom toxin(s) at 30 kD as direct ligands of renal structures. Physicochemical properties such as the molecular charge and size of “planted antigens” along the kidney are very important factors that affect the interaction of these antigens along the renal basement membranes, especially with glomerular structures (Cotran et al. 1999). Highly cationic molecules (as is the case for Loxosceles venom toxins) tend to bind to glomerular basement membrane anionic sites (proteoglycans) (Cotran et al. 1999). In addition, the glomerular barrier function is dependent on the molecular mass of proteins. Molecules with mass larger than 70 kD are less permeable than low molecular mass proteins (as is the case for a large number of Loxosceles venom toxins) (Cotran et al. 1999). This charge and size properties of venom toxins can account for their binding to glomerular basement membranes in a first step and later to tubule structures accumulated in tubule epithelial cells.
On the basis of the above findings, we have identified a possible cellular and molecular mechanism for the nephrotoxicity that occurs after envenomation by Loxosceles spiders. Although the renal injuries occurring after envenomation can be increased by dermonecrotic products and hemolysis, we conclude that Loxosceles venom toxins are direct and potentially nephrotoxic agents.
We hope that this report will bring some insight into loxoscelism, opening the possibility for a rational basis for therapy after brown spider bites.
Footnotes
Acknowledgements
Supported by grants from CNPq, CAPES, Fundação Araucária-PR, FUNPAR-UFPR, and Parana Tecnologia.
We acknowledge LIPAPE for spider capture and venom extraction and Prof Marco A. F. Randi (Department of Cell Biology, Federal University of Paraná), who helped us during morphometic analysis.