Abstract
We have previously cloned the full-length cDNA (approximately 28 Kb) and established the complete genomic organization (25 exons/introns over 100 kb) of the human MUC4 mucin. This large molecule is predicted to protrude over 2 μm above the cell surface, in which MUC4α is an extracellular mucin-type glycoprotein subunit and MUC4β is the transmembrane subunit. Over two thirds of the encoded protein sequence consists of 16-amino-acid tandem repeats (TR), which are flanked by unique sequences. In this study we generated and characterized monoclonal antibodies (MAbs) directed against the TR region of MUC4. Mice were immunized with a KLH-conjugated MUC4 TR peptide, STGDT-TPLPVTDTSSV. Several clones were purified by three rounds of limited dilutions and stable clones presenting a sustained antibody production were selected for subsequent characterization. Antibodies were tested for their reactivity and specificity to recognize the MUC4 peptide and further screened by enzyme-linked immunosorbent assay (ELISA) and Western blotting analyses. One of the MAbs (8G7) was strongly reactive against the MUC4 peptide and with native MUC4 from human tissues or pancreatic cancer cells in Western blotting, immunohistochemistry, and confocal analysis. Anti-MUC4 MAb may represent a powerful tool for the study of MUC4 function under normal and pathological conditions and for diagnosis of solid tumors including those in the breast, pancreas, lungs, and ovaries.
MUC4 is a high molecular weight
The MUC4 mucin is the homologue of rat sialomucin complex (SMC, rat Muc4) owing to similarities in structural organization. However, rat Muc4 lacks the central domain containing the 16-amino-acid TR units found in human MUC4 (Sheng et al. 1992; Wu et al. 1994). Expression of MUC4 is detected in various epithelial tissues, including the trachea, colon, stomach, cervix, and lung (Audie et al. 1993, 1995). MUC4 is detected in goblet, ciliated, and absorptive cells in adults but also in poorly differentiated cells in embryos and fetuses (Buisine et al. 1998, 1999). The primitive gut expresses MUC4 at an early stage, before respiratory and digestive epithelial cells have acquired their tissue and cell specificity. However, an abnormal expression of MUC4 has been reported in various carcinomas of the colon, pancreas, breast, and ovaries (Andrianifahanana et al. 2001; Ogata et al. 1992). For example, MUC4 is highly expressed in the vast majority of human pancreatic tumors and pancreatic tumor cell lines but is not detected in the normal pancreas or in chronic pancreatitis (Balague et al. 1994; Hollingsworth et al. 1994; Choudhury et al. 2000a; Andrianifahanana et al. 2001). Likewise, MUC4 is expressed by metasplastic ducts and its expression increases with a higher grade in pancreatic intra-epithelial neoplasias (PanINs) (Swart et al. 2002; Park et al. 2003).
The rat Muc4 has been shown to act as a ligand for the receptor tyrosine kinase ErbB2/HER2/neu (Carraway et al. 1999), and its expression in human melanoma cells resulted in lung metastasis in a mouse model (Komatsu et al. 2000). Because of the MUC4 homology to rat Muc4 and its unique pattern of expression in human fetal and tumor tissues, it is believed that MUC4 plays a crucial role in development and tumorigenesis. However, no direct evidence is available to validate these hypotheses. One of the main reasons for this poor understanding is the lack of a specific antibody recognizing the human MUC4 molecule.
To generate unique molecular tracers of human MUC4, mice were immunized by repeated IP injections of a KLH-conjugated MUC4 peptide. Splenocytes were fused with NS-1 myeloma cells to generate hybridomas. Antibodies were screened against MUC4 peptides and native protein by enzyme-linked immunosorbent assay (ELISA) and immunoblotting. One of the MAbs, 8G7, was strongly reactive against the MUC4 peptide and was used to investigate native MUC4 expression in tissue samples and cell lines by Western blotting, immuhistochemistry (IHC), and confocal analysis. MAb 8G7 recognized a very high molecular weight protein, well over 500 kD, which was the expected size for native MUC4 (Moniaux et al. 1999; Choudhury et al. 2000a). Moreover, the pattern of expression of MUC4 in 15 pancreatic adenocarcinoma cell lines, as detected by 8G7, paralleled MUC4 expression as revealed by RT-PCR and Northern blotting analyses of cell lines (Andrianifahanana et al. 2001; Choudhury et al. 2000a). The size of the MUC4 protein was cell line-specific and exhibited a direct correlation with the MUC4 allelic variation previously reported using Southern blotting (Choudhury et al. 2000a). MAb 8G7 was also able to immunoprecipitate MUC4 and to recognize MUC4 by IHC and confocal analyses. In the latter case, MUC4 was expressed on the surface membrane of the cells.
Altogether, this work presents the generation and characterization of important MAbs that specifically target the complete human high molecular weight MUC4 mucin and could be a powerful tool for study of MUC4 function and for diagnosis of adenocarcinomas.
Materials and Methods
Materials
The peptides used in this study were designed according to the deduced amino acid sequence from the clone JER64, isolated and characterized earlier with accession number M64594 (Porchet et al. 1991b). Two peptides were synthesized (Invitrogen; Carlsbad, CA), a 16-amino-acid peptide STGDTTPLPVTDTSSV, called 16TR, and an 8-amino-acid peptide GDTTPLPV, called 8TR.
Mouse Immunization
The immunization and selection of MAbs were carried out using established procedures at the UNMC Hybridoma Core Facility. Briefly, mice were immunized by repeated IP injections of KLH-conjugated MUC4 peptide (16TR) at 2-week intervals. Sera from these mice were evaluated in direct binding assays for antibody reactivity with MUC4 peptide and protein lysate from the MUC4-expressing CD18/HPAF cell line. KLH was used as a negative control. Once an appropriate antibody response was noted, as determined by reciprocal 50% end point titers in excess of 5000 vs antigen, the animals were given a final booster injection 3 or 4 days before splenectomy and exsanguination. Splenocytes were isolated and fused with NS-1 and/or Sp2/0 myeloma cells. Hybridomas producing the antibodies of interest were selected by screening for specific antibody binding to the immunogen of interest and lack of binding to irrelevant control antigens.
Screening of the MUC4-positive Hybridomas
Supernatants from different hybridomas were screened by ELISA using KLH- and MUC4 peptide-coated 96-well plates. Cells from the positive wells were further cloned three additional times. For the ELISA, plates were coated with 100 μl/well of MUC4 peptide or KLH at a concentration of 10 μg/ml in freshly prepared bicarbonate buffer by incubation for 2 hr at 37C. After several washes in PBS, 100 μl of the test supernatant was incubated for 6 hr at room temperature (RT), which was followed by several washes in PBS. Plates were further incubated with a 1:12,000 dilution of anti-mouse Ig conjugated to alkaline phosphatase for 45 min at 37C. At the end, plates were washed and bound antibody was detected using (
For the competitive ELISA, MAbs were pre-incubated overnight at 4C with 1 μg/ml of MUC4 peptide 16TR or 8TR. PBS was used as a negative control. After the incubations, the reactivity of the MAbs was checked by ELISA on 16TR-coated plates as described above.
Isotyping of the purified MAbs was performed using the mouse MAb isotyping kit code RPN 29 (Amersham Pharmacia Biotech; Piscataway, NJ).
Preparation of Cell Lysates and Immunoblotting
Cell lysates were prepared in lysis buffer (50 mM Tris-HCl, pH 7.4, 150 mM NaCl, 1% NP-40, 0.25% Na-deoxycholate, 1 mM EDTA, 1 mM PMSF, 1 μg/ml of aprotinin and leupeptin, 1 mM Na3VO4, and 1 mM NaF). The protein content of the lysates was determined by the Bio-Rad DC protein assay (Bio-Rad; Hercules, CA) as recommended by the supplier.
For immunoprecipitation, MAb 8G7 was used at a concentration of 4 μg/ml with 500 μl of cell lysate (prepared from one confluent T75 flask of cells in 1 ml of RIPA buffer). After an overnight incubation at 4C on a shaker, 40 μl (1:1 v/v in lysis buffer) of protein G-sepharose beads (Sigma; St Louis, MO) was added to the mixture. The beads were pelleted by centrifugation, washed twice with a buffer of 50 mM Tris containing 500 mM NaCl, followed by a final wash in 50 mM Tris, and boiled in sample loading buffer (2% SDS, 62.5 mM Tris-HCl, pH 6.8, 5% glycerol, 2% mercaptoethanol, and 0.01% bromophenol blue) for loading on SDS-Agarose gels and Western analysis.
Cell lysates or immunoprecipitates were resolved on a 0.1% sodium dodecyl sulfate (SDS)-2% agarose gel electrophoresis (AGE), transferred to polyvinylidene difluoride (PVDF) membranes, and the membrane were blocked in 5% non-fat dry milk in PBS for 1 hr. Membranes were incubated for 4 hr at RT with the test or control antibodies diluted at 1 μg/ml in PBS, followed by six 10-min washes in TBST (50 mM Tris-HCl, pH 7.4, 150 mM NaCl, and 0.05% Tween-20). Horseradish peroxidase-conjugated secondary antibodies were diluted at 1:2000 in PBS and incubated for 1 hr at RT. Three washes in TBST followed secondary antibody incubation. Membranes were developed using ECL Western blotting detection reagents (Amersham Life Science) and blots were exposed to ECL-sensitive film (Eastman Kodak; Rochester, NY).
Phosphoglycerate kinase (PGK) was used as a protein-loading control for Western blotting analysis. The immunoblotting procedures were carried out according to the conventional SDS-PAGE Western blotting method (Laemmli 1970).
Confocal Analysis
Cells were grown at a low density on sterilized coverslips for 20 hr. Cells were washed with 0.1 M HEPES containing Hanks buffer and fixed in ice-cold methanol at −20C for 2 min. Methanol-fixed cells were blocked in 10% goat serum containing 0.05% Tween-20 for 30 min at RT before incubation with 8G7 primary antibody diluted (1:100) in 10% goat serum containing PBS for 60 min at RT. Cells were washed four times for 5 min with PBS containing 0.05% Tween-20 and then incubated with FITC-conjugated goat anti-mouse secondary antibodies (Jackson Immuno Research laboratories; West Grove, PA) for 60 min at RT. Cells were again washed with PBS-Tween five times for 5 min and once in water before mounting on a glass slide in anti-fade VECTASHIELD mounting medium (Vector Laboratories; Burlingame, CA). Immunostaining was observed under a Zeiss Laser Scan confocal microscope (Zeiss; Oberkochen, Germany).
Immunohistochemical Analysis
Tissues were fixed in 10% buffered formalin and embedded in paraffin. Sections (5 μm) were assayed for MUC4 apomucin by using a modification of the previously described ABC IHC method (Pour et al. 1993; Batra et al. 1995). Briefly, tissue sections were deparaffinized in xylene, rehydrated in graded ethanol, and treated for 20 min with 0.3% H2O2/methanol to block endogenous peroxidase. The sections were blocked with normal goat serum for 1 hr followed by incubation at 4C overnight with 8G7 MAb or pre-immune rabbit serum as a negative control. The immunoreactivity and the specificity of the antibodies generated against the tandem repeat peptide was evaluated for the immunocytochemical procedure as previously described (Choudhury et al. 2000b).
Results
After cell fusion, screening, and cloning procedures, six clones secreting the anti-MUC4 antibody were selected, and one clone secreting the anti-KLH antibody was used as a negative control (Table 1). None of the six MUC4-specific MAbs presented any reactivity against KLH. Two of the six MAbs, 13F12 and 8G7, showed strong reactivity to the MUC4 peptide. Mabs 9H8, 12B8, IG2, and 11A8 MAbs presented mild reactivity, whereas the 1G2 MAb showed low reactivity. Table 1 summarizes the respective isotypes of these antibodies, IgG2b(k) or IgG1(k).
The six anti-MUC4 MAbs were further tested against the linear concentration of synthetic MUC4 peptide (0.5–10 μg/ml per well). As shown in Figure 1A, the six antibodies differed in their respective titers to the 16TR peptide. The 13F12 and 8G7 MAbs presented the best reactivity compared to 1G2 and 11A8. The K2G6 anti-KLH MAb did not show any reactivity.
Six clones secreting anti-MUC4 and one anti-KLH were isolated by ELISA screening against MUC4 16TR peptide- and KLH-coated plates (in OD)
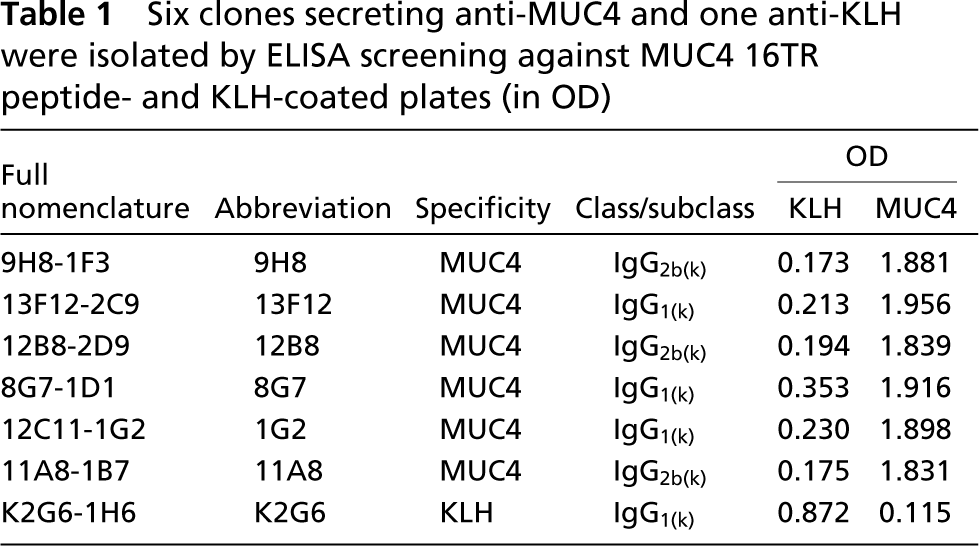
Anti-MUC4 MAbs and epitope mapping. (
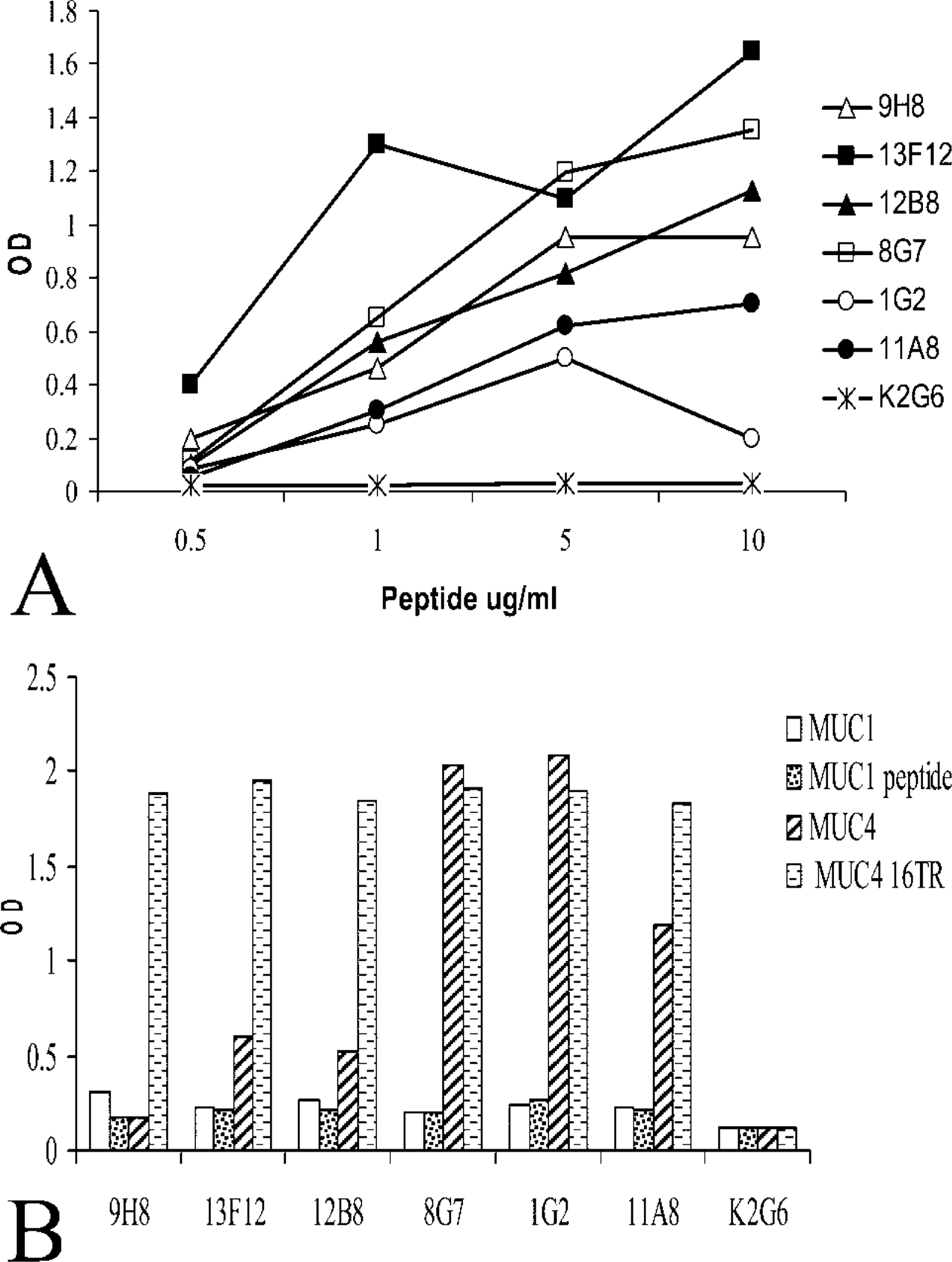
ELISA was performed to check the specificity of each antibody for different mucins. Mucins share the common property of being rich in serine and threonine residues, which are mainly concentrated in the central domain of the molecules. Specifically, the central domain of mucins is composed of tandemly repeated units, and the number can reach up to 400 in the case of MUC4. To check the specificity of MUC4 MAbs and to investigate any potential crossreactivity with other mucins, anti-MUC4 MAbs were analyzed against the 16TR MUC4 peptide, a 100-amino-acid MUC1 peptide (Hiltbold et al. 1998) and purified MUC1 and MUC4, as described previously (Khorrami et al. 2002). The results are shown in Figure 1B. The anti-KLH MAb was used as a negative isotype control. As previously described, six anti-MUC4 MAbs reacted against the 16TR peptide. None of the MUC4 MAbs reacted against the MUC1 peptide or the native MUC1 protein. MAbs had a distinct reactivity toward MUC4 native protein, however, with MAb 8G7 and 1G2 exhibiting the strongest binding, whereas MAb 11A8 showed mild reactivity and MAbs 12B8, 13F12, and 9H8 showed low reactivity. Despite the strong recognition of the 16TR, some MAbs (11A8, 12B8, 13F12, 9H8) reacted weakly with the native protein in ELISA. Therefore, 8G7 and 1G2 appeared promising in their ability to recognize the native MUC4.
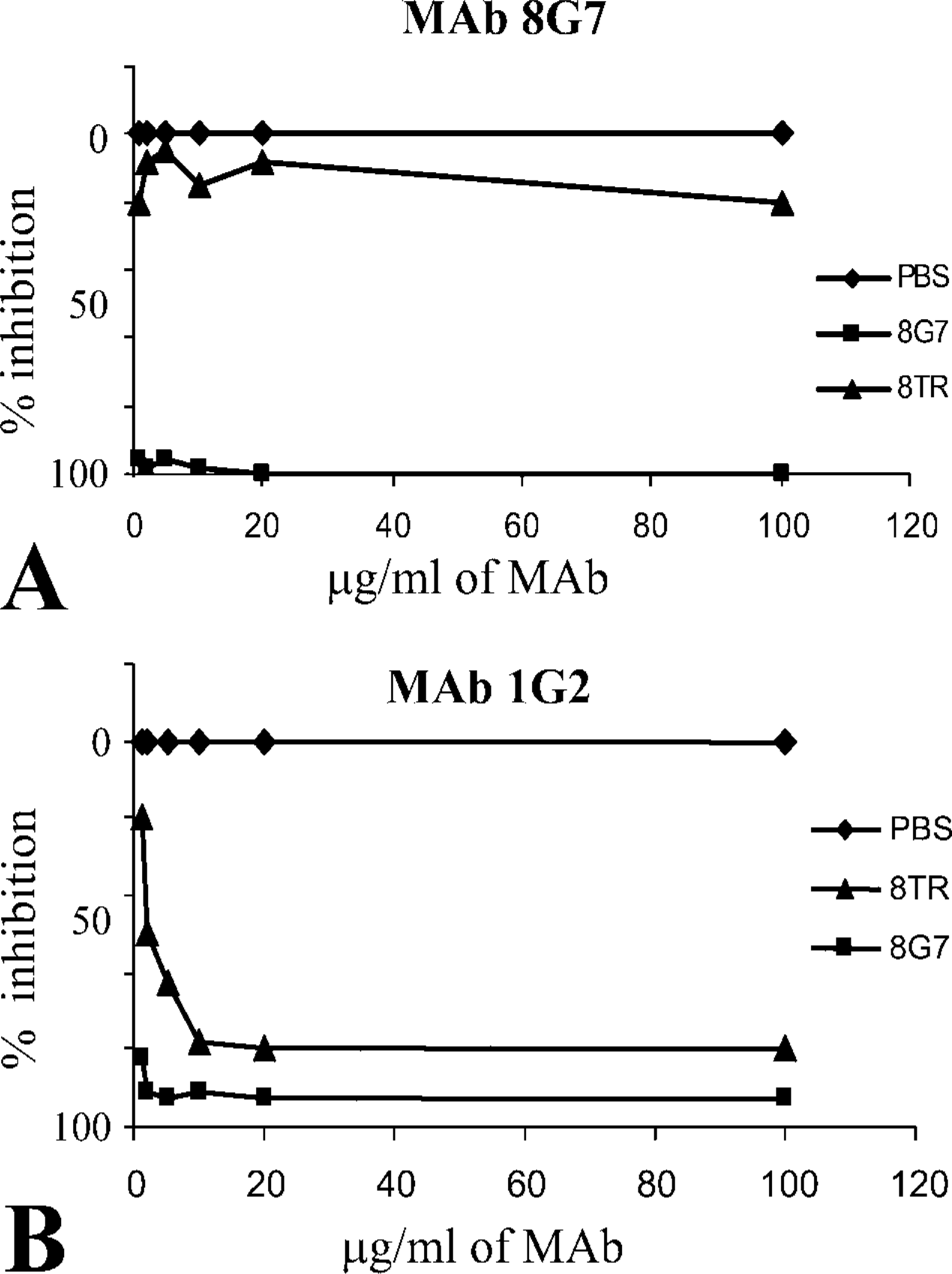
Competitive ELISA performed with 16TR and 8TR. (
To determine if the MAbs were recognizing the same epitope within the 16TR, each MAb was tested by competitive ELISA, using the 16TR and the 8TR peptides (Figure 2). The 8TR was designed within the 16TR and defined as the sequence less likely to be
Because antibodies can present a distinct reactivity between ELISA and Western blotting, six MAbs were also tested for their ability to recognize the MUC4 protein in Western blotting. Mucins are high molecular weight proteins that cannot be resolved by traditional SDS-PAGE. For example, MUC1, which is one of the smallest mucins, varies in size between 250 and 1000 kD, depending on the allele and its level of glycosylation (Lan et al. 1987; Shimizu and Yamauchi 1982). MUC4, which is the largest human mucin fully characterized thus far, has an expected size, without any glycosylation, of 930 kD (Moniaux et al. 1999; Choudhury et al. 2000a). To resolve such high molecular weight proteins, the strategy of 2% AGE was chosen. Protein lysates (30 μg of protein/well) from the MUC4-expressing human pancreatic tumor cell line (CD18/HPAF) were resolved on an agarose gel. Under these conditions, the 250-kD size marker protein (myosin) (BioRad) migrated at 1 cm from the bottom of a 15-cm (6-inch) gel. Western blotting analysis of various anti-MUC4 antibodies is shown in Figure 3. MAb 8G7, at a concentration of 1 μg/ml, presented the highest reactivity, with a high molecular weight band resolved at the top of the gel. The 1G2 detected native MUC4 with 50% less intensity. The MAbs 13F12 and 11A8 presented a weak signal. Other MAbs, 9H8 and 12B8, reacted in Western blotting with a very strong background. Increasing the antibody concentration to 10 μg/ml did not have any effect on the signal (data not shown). Therefore, the concentration of 1 μg/ml of MAb was used for further investigations. To solve the background problem associated with 9H8 and 12B8, all MAbs were assayed using a nitrocellulose membrane, with different conditions of membrane blocking and primary MAb incubation buffers. All the conditions tested gave similar results (data not shown).
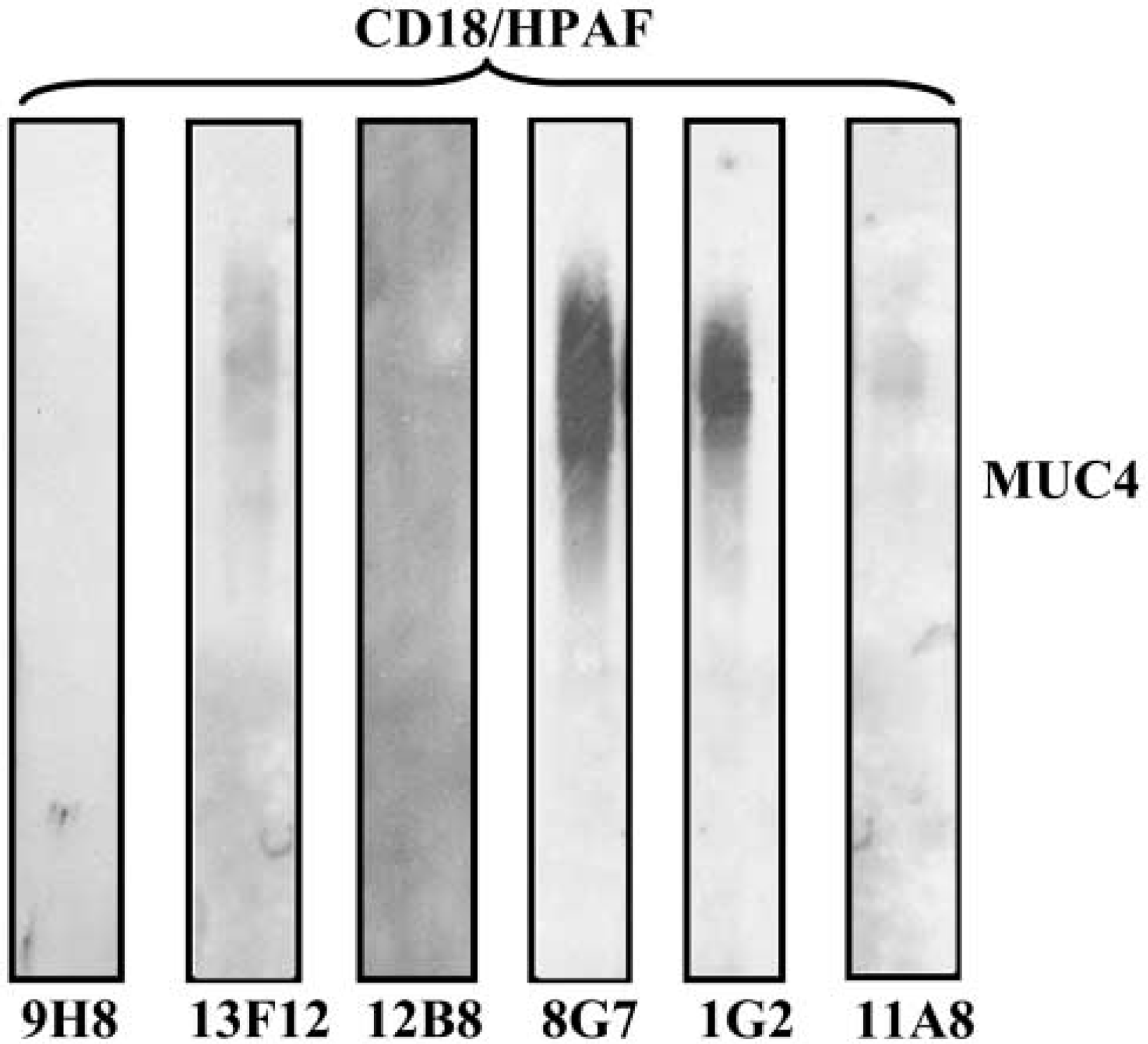
Western blotting analysis of CD18/HPAF protein lysate resolved on a 2% agarose/SDS gel of the six MUC4-specific MAbs. Resolved proteins were passively transferred overnight on PVDF membrane using Tris/glycine/methanol buffer. One μg/ml of each MAb was incubated for 2 hr at RT in PBS. The 8G7 MAb showed the strongest signal, with a band detected in the high molecular weight range (far over 500 kD).
Overall, 8G7 was the best MAb specifically recognizing the MUC4 peptide as well as the MUC4 native protein in ELISA and Western blotting analyses. This MAb was used to investigate MUC4 protein expression in 15 different pancreatic cell lines (Figure 4A1). PGK was used as a control to check the quality of the protein lysates. Once again, high molecular weight bands were detected in the upper part of the gel (over 500 kD). No band was detected for five cell lines, including MiaPaCa and Panc 1, that are known to be MUC4 non-expressing by RT-PCR (Figure 4A2) and Northern blotting (Andrianifahanana et al. 2001), as well as AsPC1, Hs766T, and BxPC3 that express low levels of the MUC4 transcript (Andrianifahanana et al. 2001). One or two bands were detected for the other cell lines. The size of the bands detected showed a perfect correlation with the allelic VNTR polymorphism as previously detected by Southern blotting in the same cell lines (Choudhury et al. 2000a). For comparative purposes, the pattern of MUC4 expression investigated by RT-PCR on the same 15 pancreatic adenocarcinoma cell lines is presented in Figure 4A2. In addition, 8G7 was able to recognize native MUC4, as evidenced by data from Western blotting performed on tissue samples. MUC4 was detected in tracheal and pancreatic adenocarcinoma tissue samples, whereas the normal pancreas sample appeared negative for MUC4 expression (Figure 4B). In a similar way, 8G7 (at 4 μg/ml) was also able to immunoprecipitate the MUC4 protein from the CD18/HPAF lysate (Figure 4C).
IHC and confocal analyses were carried out to test the ability of 8G7 to recognize MUC4 using these techniques. Under a normal physiological situation, the main site of MUC4 expression is the respiratory tract (Audie et al. 1993; Copin et al. 2000, 2001). MUC4 is expressed by the trachea, the bronchus, throughout the bronchiole, and with a decreasing gradient of intensity from the trachea to the bronchiole. Figure 5A shows MUC4 expression by airway epithelium using 8G7 MAb. Cells from the surface epithelium of the bronchiole appeared positive for MUC4 expression, whereas the surrounding pneumocytes were negative. In a similar way, Figure 5B shows MUC4 expression by a lung adenocarcinoma tissue sample, a tissue known to be MUC4-positive (Copin et al. 2001). A high level of staining was seen in a cancerous area in the pancreas (Figure 5C). The staining appeared concentrated in the cytoplasm of the cells, which is consistent with the secretory granules of that accumulate in the cytoplasm to be released in the mucus. The ratio of the membrane-bound:secreted form varies for MUC4 from tissue to tissue (Carraway et al. 2003).
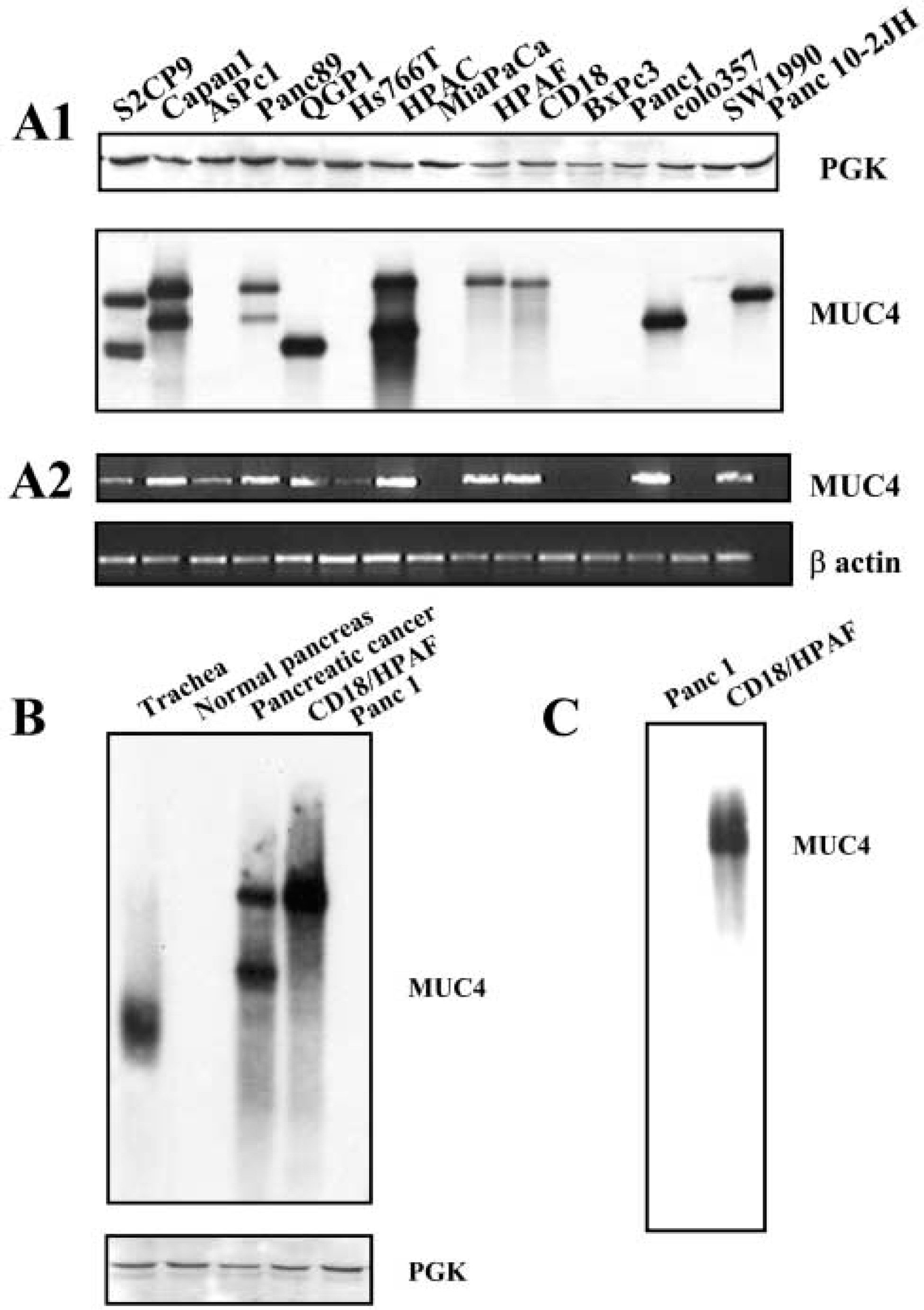
Biochemical, molecular, and immunological characterization of 8G7. (
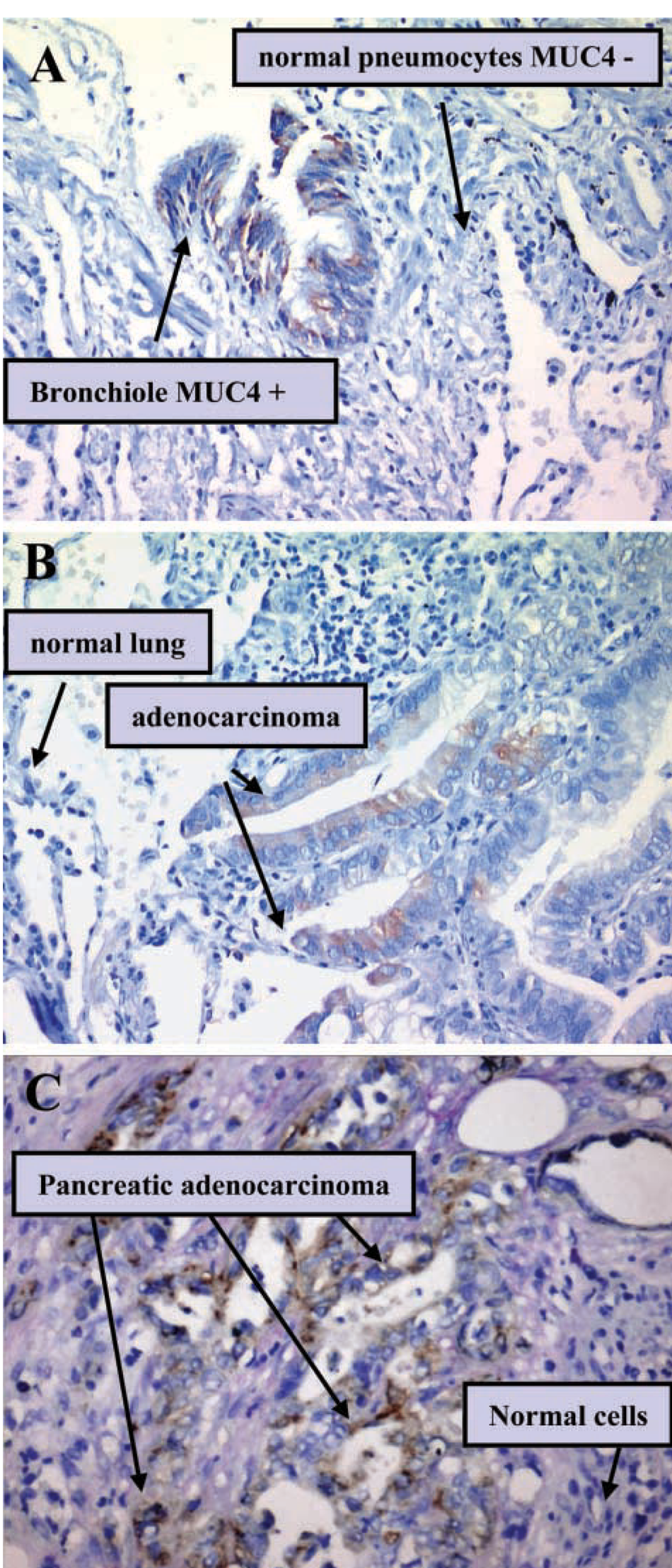
IHC analysis of MUC4 using 8G7 MAb. Normal airway epithelium cells from the surface of the bronchiole appeared positive for MUC4 expression, whereas the surrounding pneumocytes were negative (
Confocal microscopic analysis was performed on the MUC4-expressing CD18/HPAF cell line. In confluent monolayer cells, as predicted by the MUC4 structural organization, 8G7 MAb immunoreacted predominantly with the plasma membrane of CD18/HPAF cells and moderately with the cytoplasmic compartment (Figure 6). The profile of expression was directly related to the membrane-bound expression of MUC4.
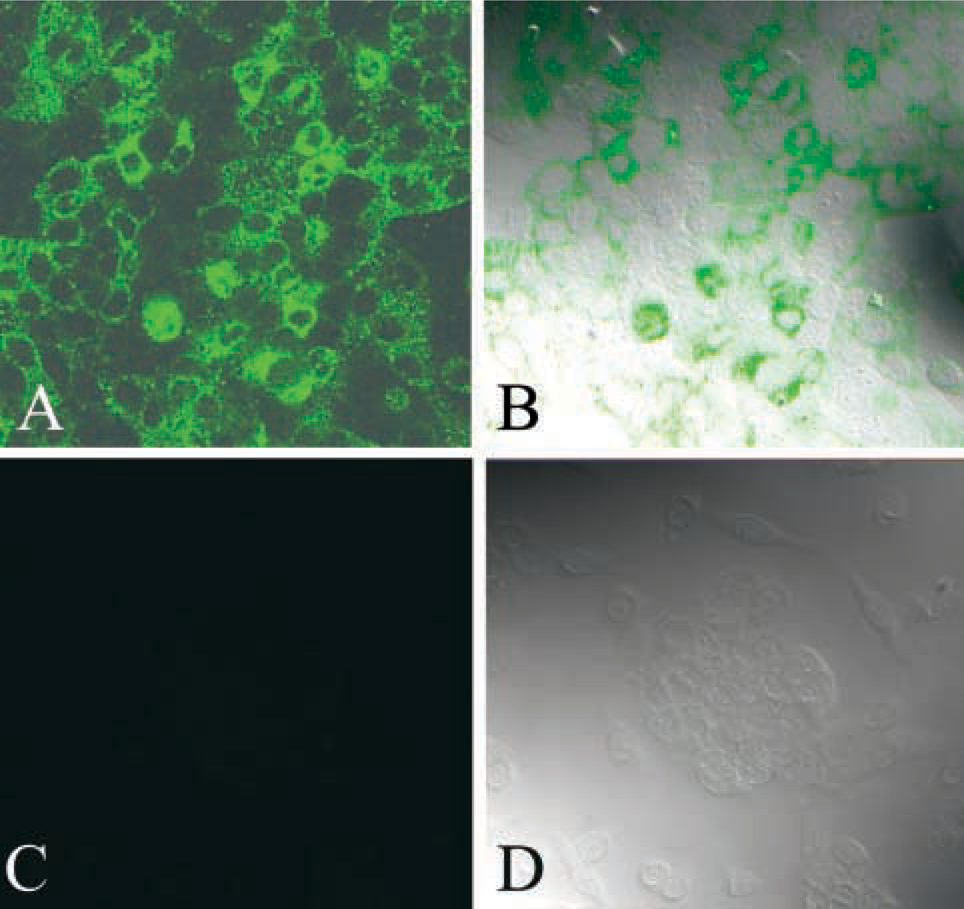
Confocal photomicrograph demonstrating expression pattern of MUC4 in methanol-fixed CD18/HPAF human pancreatic tumor cells. (
Discussion
MUC4 is a promising target for both diagnosis and therapeutic purposes in several tumor situations, most importantly pancreatic cancer (Andrianifahanana et al. 2001). The lack of specific anti-MUC4 antibodies has limited our progress in many areas of MUC4 research, including an early diagnosis and targeted therapy. MUC4 belongs to the membrane-anchored mucins and is the largest member of this family (Moniaux et al. 1999, 2001). It is a modular molecule composed of two non-covalently linked subunits, a large mucin-type subunit, MUC4α, and a membrane-bound growth factor-like subunit, MUC4β. The two subunits result from cleavage at a GDPH site of a unique 930-kD precursor.
The problem in obtaining antibodies to specifically target mucin proteins is related to their structure. Mucins are high molecular weigh
The reactivity of the anti-MUC4 MAbs listed in Table 1 was established by multiple approaches but most critically by ELISA, competitive ELISA, purified native MUC4 protein, immunoprecipitation, and blotting of lysates from the MUC4-expressing pancreatic tumor lines. The lack of reactivity to MUC1 and other MUC4-non-expressing cell lines established the specificity of these MAbs for MUC4. Among the six clones isolated, the 8G7 MAb was reactive against the MUC4 peptide as well as the native protein. In Western blotting, 8G7 detected MUC4 expression with a pattern similar to that observed in RT-PCR analysis (Figures 4A1 and 4A2). MAb 8G7 binds to the α-sub-unit of MUC4, which has a size of 850 kD. The fact that 8G7 recognized a very high molecular weight protein (well over 500 kD) in Western blotting under the conditions used (2% agarose/SDS gel) confirmed the specificity of binding to MUC4. Only mucins are reported to present such a molecular weight in epithelial cells. Moreover, the 2% agarose/SDS Western blotting technique enabled us to detect the MUC4 VNTR polymorphic allele with very good resolution. The size of MUC4 detected for each cell line was in full agreement with the VNTR of MUC4 gene previously reported (Choudhury et al. 2000a).
Importantly, the reactivity of 8G7 for the MUC4 native protein appeared to not be dependent on the level of glycosylation. The fact that 8G7 recognized the MUC4 protein in the tumor situation in Western blotting as well as in IHC analysis was anticipated because mucins are reported to be under-glycosylated in cancer. In confocal analysis, MUC4 was found expressed at the membrane surface of the cell, which is also consistent with the MUC4 structure. It should be noted, however, that 8G7 also recognized the MUC4 protein in normal tissue, such as the trachea in Western blotting (Figure 4B) or the normal bronchiole in IHC analysis.
In summary, six specific anti-MUC4 MAbs have been generated and two exhibit high avidity for a high molecular weight MUC4 native protein. One of the anti-MUC4 MAbs, 8G7, showed high specificity and avidity in ELISA, Western blotting, immunoprecipitation, and in IHC and confocal analyses for the MUC4 protein. Rat SMC (rMuc4), a homologue of human MUC4 because of its similar structural organization, is a heterodimeric glycoprotein complex composed of mucin (ascites sialoglycoprotein 1, ASGP-1) and transmembrane ASGP-2 subunits. ASGP-2 potentiates HER2 signaling and possesses a crossroad position between triggering the cells towards differentiation or proliferation via p27kip (for review see Carraway et al. 2002). Therefore, the MAb 8G7 would be an important tool for diagnostic applications and for understanding MUC4 interactions in human normal and cancer tissues.
Footnotes
Acknowledgements
Supported by an RO1 grant CA78590 and a SPORE grant PA566A 72712 from the National Institutes of Health.
We thank Erik Moore for technical support and the Molecular Biology Core Facility, UNMC, for oligonucleotide synthesis and DNA sequencing, the Monoclonal Antibody Core facility, and Kristi L.W. Berger (Eppley Institute) for editorial assistance.
