Abstract
Astrocytosis is a hallmark of damage that frequently occurs during aging in human brain. Astrocytes proliferate in elderly subjects, becoming hypertrophic and highly immunoreactive for glial fibrillary acidic protein (GFAP). These cells are one type that actively responds in the repair and reorganization of damage to the neural parenchyma and are a source of several peptides and growth factors. One of these biomolecules is apolipo-protein D (apo D), a member of the lipocalin family implicated in the transport of small hydrophobic molecules. Although the role of apo D is unknown, increments in brain apo D expression have been observed in association with aging and with some types of neuropathology. We have found an overexpression of apo D mRNA in reactive astrocytes by in situ hybridization in combination with immunohistochemistry for apo D in normal aged human brains. The number of double-labeled cells varied according to the cerebral area and the gliosis grade. The possible significance of this increased synthesis of apo D in reactive astrocytes is discussed in relation to the role of apo D in aging and in glial function.
D
Apolipoprotein D (apo D) is a lipocalin superfamily member, which binds and transports small hydrophobic ligands. Apo D is a component of high-density plasma lipoproteins (HDLs) and it is also present in breast cyst fluid, lachrimal secretions, apocrine axillary secretions, cerebrospinal fluid (CSF), and urine (Rassart et al. 2000). Apo D is widely expressed, mainly by fibroblasts, in a wide variety of mammalian tissues (Boyles et al. 1990; Provost et al. 1990; Smith et al. 1990; Séguin et al. 1994). In the peripheral nervous system (PNS), apo D is synthesized by endoneurial fibroblasts, whereas in the CNS apo D expression is observed in neurons, oligodendrocytes, astrocytes, and perivascular cells (Boyles et al. 1990; Smith et al. 1990; Patel et al. 1995; Navarro et al. 1998). Increments of apo D in PNS may be related to the repair process after a lesion. It has been suggested that apo D probably has a role in binding and transporting cholesterol and its esters during regeneration in association with other apolipoproteins, such as apo E and A-I (Spreyer et al. 1990). Moreover, synthesis and secretion of apo D by primary astrocyte cultures of mouse brain have been described (Patel et al. 1995). Apo D is upregulated by progesterone and oxysterol in cultured astrocytes. These results suggest a potential role as a ligand carrier in astrocytes that might be important in maintaining cholesterol homeostasis during reinnervation and regeneration.
The increased apo D expression, compared with apo E in some neuropathologies (Suresh et al. 1998; Terrisse et al. 1998; Kalman et al. 2000; Belloir et al. 2001) and with experimental brain lesions in animals (Ong et al. 1997; Franz et al. 1999; Montpied et al. 1999; Terrisse et al. 1999), suggests that apo D could be a good marker of pathology. Apo D has been shown to accumulate mainly in astrocytes and neurons after injury, as well as in the elderly brain (Kalman et al. 2000; Belloir et al. 2001). Increases in astrocyte apo D mRNA expression were only proved in brain from aged PDAPP and clozapine-treated mice (Thomas et al. 2001a,c).
The aim of this work was to detect the location and synthesis of apo D in reactive astrocytes of human cerebral cortex during normal aging by double immuno-cytochemistry (ICC) and in situ hybridization (ISH). Apo D synthesis by astrocytes has not been previously shown in human brain.
Materials and Methods
Human tissues were provided by the Pathologic Anatomy Department at General Hospital of Asturias. Twelve elderly subjects (between 82 and 88 years) and eight young subjects (between 30 and 44 years) without neurological, psychological, or neuropathological disorders were used in this study. Pieces of human cerebral cortex from different areas were fixed by immersion for about 4 hr in 4% paraformaldehyde in PBS (pH 7.4). After fixation, pieces were dehydrated, cleared in butylacetate, and embedded in paraffin. Double immuno-hybridocytochemical staining for apo D protein and its mRNA was carried out according to the following protocol. Sections were dehydrated, dried at room temperature for 5 min, digested with 2 μg/μl proteinase K in 0.001 M PBS at 37C for 5 min, and rinsed in 0.001 M PBS. Twenty μl of ISH solution (Sigma Hybridization Solution, H-7782; St Louis, MO) was applied on each section for 10 min. Twenty μl of digoxigenin-labeled RNA probe for apo D (obtained from Dr. Carlos López-Otin, Dpto. Bioquímica y Biología Molecular, Universidad de Oviedo; see Díez-Itza et al. 1994) was applied on each section and sections were incubated in a moist chamber for 16 hr at 55C. The slides were rinsed in PBS and the hybridized probe was detected with an alkaline phosphatase-coupled anti-DIG antibody (Roche 1093274, Mannheim, Germany) diluted 1:1000 for 60 min at room temperature. The slides were rinsed with PBS and incubated in Sigma Fast BCIP/NBT (Sigma B5655) solution (14 hr at RT). Then slides were rinsed in distilled water and processed for ICC detection of apo D. Endogenous peroxidase and nonspecific binding were blocked by sequential incubation in 3% hydrogen peroxide solution and normal serum. Incubation with a specific antibody against apo D (1:2000 dilution) was carried out overnight at 4C [provided by Dr. C. López-Otín, Departamento de Bioquímica y Biología Molecular, Universidad de Oviedo; see López-Boado et al. (1994) and Navarro et al. (1998)]. The immunoreactivity was detected using the Extravidin-biotin-peroxidase staining kit (Sigma Extra-3). Peroxidase activity was shown by incubation with Sigma Fast DAB (Sigma D 4168). Double immunostaining for apo D and GFAP was carried out according to the following protocol. Immunodetection of apo D was achieved as previously described and the immunoreactivity was detected using the Extravidin-biotin-alkaline phosphatase staining kit (Sigma Extra-3). Enzymatic activity was shown by incubation with Vector-blue substrate (Vector SK-5300; Burlingame, CA). Slides were rinsed in PBS, placed in a plastic coplin jar filled with 0.01 M sodium citrate buffer (pH 6), and incubated in a household microwave. Microwave treatment involves completely blocking contaminating staining in the double-labeling technique, using primary antibodies from the same species (Lan et al. 1995). Incubation with a specific antibody against GFAP (Sigma G-9269, 1:1000 dilution) was carried out overnight at 4C. The immunoreactivity was detected using the Extravidin-biotin-peroxidase staining kit (Sigma Extra-3). After several washes, peroxidase was visualized by a red AEC reaction (Sigma A-6926) (0.5 mg AEC, 50 μl dimethylformamide, 10 μl H2O2 (3%) in 940 μl acetate buffer). The sections were counterstained with thionine and mounted in aqueous mounting medium. The usual specificity control tests were carried out.
Results
In addition to confirming the presence of astrogliosis by its morphological changes, consisting of swelling of the nucleus that appears in a eccentric position and/or hypertrophy of the cytoplasm with vigorous processes, we performed a double IHC technique for GFAP (red) and apo D (blue) (Figures 1A–1C). As expected, an increment of GFAP was found in aging (Figures 1A and 1B). The number of apo D-immunoreactive astrocytes was greater in old brains (Figure 1B) with respect to young brains (Figure 1A). The reactive astrocytes showed a strong positive signal for GFAP and strong staining for apo D (Figure 1C). When the double immuno–hybridocytochemical staining for apo D was achieved on sections, the cells identified as reactive astroglial cells exhibited the presence of apo D protein (brown) and its mRNA (dark blue) (Figures 1D–1H). On the other hand, we observed that increases of apo D immunoreactivity during aging appeared to be due to astrocytes, mainly reactive ones. The deposits of apo D appeared principally in the cell somata in peri-nuclear position at first, and they could occupy the cytoplasm and even the initial tree of processes when apo D staining increased (Figures 1D–1H). By ISH, apo D mRNA was clearly observed in many astroglial cells in all cases. This signal seems to be more intense in the astrogliotic areas present in the cerebral cortex of aged subjects (Figures 1D–1H). In addition to the apo D location, the increment was linked to reactive astrocytes (Figures 1D–1H). The mRNA signaling appeared to be principally located in a perinuclear position, but some positive granules were also observed in other cytoplasm regions (Figure 1G and 1H). Apo D mRNA signal principally adopted a crescent form near the nucleus, while the apo D protein showed a more uniform localization in cytoplasm (Figures 1F and 1H).
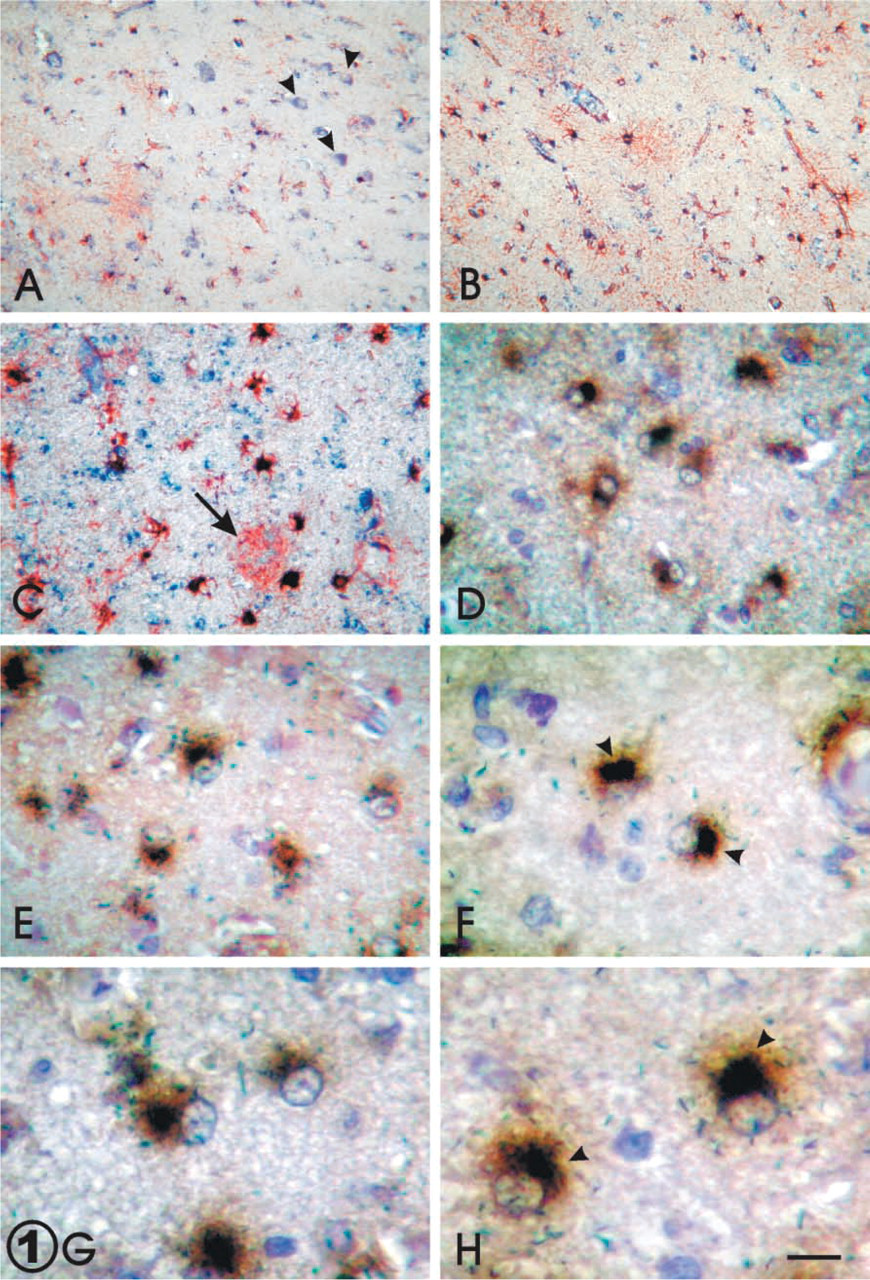
(
We have found that there are increments of apo D protein and apo D mRNA in aged cerebral cortex, probably due to the increased number of astrocytes and the higher levels of expression of apo D in reactive glia.
Discussion
Several observations have shown that the aging process causes dramatic changes in many neuronal markers (Morgan and May 1990). Nevertheless, understanding the aging brain implies knowledge of the glial modifications. The predominant change that occurs in glia during aging is glial activation, which can progress to reactive gliosis in response to neurodegeneration (Nichols 1999). It is known that in the aged rat brain there is an increment of GFAP levels as compared to young animals (Topp et al. 1989). Therefore, the degree of glial activation appears to be related to aging in rodent brain. Furthermore, activated glial cells are able to express a multitude of inflammatory mediators such as cytokines, adhesion molecules, chemokines, metalloproteinases (Podor et al. 1992; Hopkins and Rothwell 1995; Ghirnikar et al. 1998; Yong et al. 1998), and other reactive molecules in response to brain injury. We have observed that, during aging, reactivite glial progression appears to be accompanied by an increase in apo D presence and expression. Moreover, this increment is much higher in astrogliotic areas, probably produced after neuron loss. In the present work, we show by ISH that human astrocytes are able to synthesize apo D in vivo and that its expression might be enhanced by the glial reactivation pathway and compensatory mechanisms. The physiological function of apo D is yet unknown. A possible role in the transport of small hydrophobic molecules has been suggested (Rassart et al. 2000). GFAP responds dynamically to aging and neurodegenerative lesions, and changes in its expression can occur at both transcriptional and translational levels. Apo D could be also part of the glial activation response to injury. GFAP expression can be modulated by cytokines, growth factors, and steroids (Laping et al. 1994), and steroids have been demonstrated to regulate apo D expression (Simard et al. 1991). During glial activation, many different primary mediators are expressed, resulting in propagation and enhancement of the inflammatory response. Astrocytes and microglial cells then express additional inflammatory mediator, such as different kinds of interleukins (Giulian et al. 1988), which are also able to regulate apo D expression (Blais et al. 1995).
Astroglial apo D synthesis and secretion in cultured astrocytes were reported by Patel et al. (1995). These authors studied apo D expression in primary astrocyte cultures derived from neonatal mouse brain and observed that apo D is constitutively secreted by astrocytes, with little intracellular storage. They also observed that some substances, such as 25-hydroxy-cholesterol, which is a potent regulator of cellular cholesterol homeostasis in mammalian cells, were able to stimulate apo D secretion. These results suggest a physiological role for apo D in cholesterol metabolism in the nervous system. Kalman et al. (2000) observed using IHC methods that apo D was present in normal human astrocytes. Immunoreactivity was found in the cytoplasmic region but not in the cell membrane or the nucleus. These authors also reported increased apo D-immunopositive signal in old subjects compared to young ones. We have found that apo D protein in reactive astrocytes is localized all along the cytoplasm, whereas its mRNA is mainly in the cytoplasm perinuclear area. This may be because apo D is a glycosylated protein and its synthesis takes place in the ERR. Performance of ISH techniques enabled us to determine that there is also an increment in apo D mRNA, leading to the conclusion that the enhanced IHC signal we observed with aging is due to the endogenous production of apo D by the astrocytes. Similar findings have been described in PDAPP transgenic mice by ISH (Thomas et al. 2001c). These authors have shown that apo D mRNA is increased in brains of aged PDAPP mice compared to aged controls. They hypothesized that, although increases in apo D expression are a normal feature of brain aging, extreme increases may represent a glial cell compensatory response to β-amyloid deposition in Alzheimer' disease. The increased apo D expression in some neuropathologies (Ghiso et al. 1994; Dandoy-Dron et al. 1998; Suresh et al. 1998; Terrisse et al. 1998; Navarro et al. 2001; Thomas et al. 2001b) and in experimental brain lesions and treatments in animals (Ong et al. 1997; Montpied et al. 1999; Terrisse et al. 1999; Thomas et al. 2001a) suggests that apo D could be a good marker of pathology induced as soon as any type of injury occurs, and enables us to speculate that apo D in the CNS might be involved in repair and regeneration after neurodegeneration or in removal of neurotoxic molecules after cell death.
Footnotes
Acknowledgements
Supported by grants from the Fondo de Investigación Sanitaria Española (PI020324 and 03-RED-C03/06).
