Abstract
Membrane-associated carbonic anhydrase (CA) has a crucial role in renal HCO3 − absorption. CA activity has been localized to both luminal and basolateral membranes of the tubule epithelial cells. CA XII is a transmembrane isoenzyme that has been demonstrated in the basolateral plasma membrane of human renal, intestinal, and reproductive epithelia. The present study was designed to demonstrate the distribution of CA XII expression in the rodent kidney. A new polyclonal antibody to recombinant mouse CA XII was used in both Western blotting and immunohistochemistry. Western blotting analysis revealed a 40–45-kD polypeptide in CA XII-expressing CHO cells and isolated membranes of mouse and rat kidney. Immunofluorescence staining localized CA XII in the basolateral plasma membranes of S1 and S2 proximal tubule segments. Abundant basolateral staining of CA XII was seen in a subpopulation of cells in both cortical and medullary collecting ducts. Double immunofluorescence staining identified these cells as H+-secreting type A intercalated cells. The localization of CA XII in the peritubular space of proximal tubules suggests that it may play a role in renal HCO3 − absorption, whereas the function of CA XII in the type A intercalated cells needs further investigation.
Keywords
C
Membrane-associated renal CA activity has earlier been attributed to CA IV, which is expressed in proximal tubules and the thick ascending limb of rat nephrons (Brown et al. 1990), and also in intercalated cells of rabbit collecting ducts (Schwartz et al. 2000). Recently, two novel membrane-associated CAs, CA XII and XIV, were discovered and found to be expressed also in kidney (Ivanov et al. 1998; Türeci et al. 1998; Mori et al. 1999). Using polyclonal antibodies and immunofluorescence, we recently localized CA XIV to luminal and basolateral membranes of proximal tubule and thin descending limb of Henle in mouse and rat nephron (Kaunisto et al. 2002). CA XIV was therefore partly co-expressed in the nephron segments where CA IV was thought to be involved in HCO3 − absorption (Brown et al. 1990; Alpern 2000).
CA XII was originally identified as a tumor-associated gene expressed in renal and lung cancer cells (Ivanov et al. 1998; Türeci et al. 1998). However, its mRNA was found to be expressed in some non-malignant tissues, including kidney (Ivanov et al. 1998; Türeci et al. 1998). Antibodies to human CA XII protein localized it to the basolateral plasma membranes of the epithelia in reproductive and intestinal tracts (Karhumaa et al. 2000,2001; Kivelä et al. 2000). We recently characterized the immunohistochemical (IHC) localization of CA XII in the human kidney, where it was found in the basolateral plasma membranes of the proximal and distal tubules and the collecting duct principal cells (Parkkila et al. 2000).
Marked species differences in CA distribution along the nephron have been reported (Dobyan et al. 1982; Brown et al. 1990; Alpern 2000; Schwartz et al. 2000). Histochemical studies in mouse and rat kidney have demonstrated membrane-associated CA activity in distal tubule and intercalated cells of the collecting ducts, suggesting that these tubule segments express thus far unidentified membrane-associated CA isoenzymes (Lönnerholm and Ridderstråle 1980; Dobyan et al. 1982; Ridderstråle et al. 1992). To compare the localization of CA XII with CA IV and XIV along the mouse and rat nephron and with the localization of CA XII in human kidney, we raised specific polyclonal antibodies to recombinant mouse CA XII. These antibodies were used to define the distribution of CA XII expression in mouse and rat kidney.
Materials and Methods
Antibodies
Polyclonal rabbit antiserum to mouse CA XII was used in Western blotting and IHC. This antibody was produced as follows. Chinese hamster ovary (CHO) cells were transfected with a gene construct containing a truncated secretory form of mouse CA XII according to earlier procedures (Okuyama et al. 1995; Karhumaa et al. 2000). Recombinant mouse CA XII was purified from the secretion of CHO clones using a
In double-labeling studies, rat anti-mouse CA XIV (Kaunisto et al. 2002), a goat anti-Tamm–Horsfall glycoprotein (Cappel ICN; Costa Mesa, CA), a goat anti-calbindin D28K (Santa Cruz Biotechnology; Santa Cruz, CA), a goat anti-aquaporin-2 (AQP2) antibody (Santa Cruz), and a monoclonal antibody, IVF12, to AE1 anion exchanger (Jennings et al. 1986) (provided by Dr. Michael Jennings) were used. Secondary antibodies used in immunofluorescence were purchased from Molecular Probes (Eugene, Oregon).
Animals and Tissue Preparation
Adult male Sprague–Dawley rats and Balb/c mice were sacrificed by CO2 asphyxiation followed by decapitation. For Western blotting, kidneys were removed and rapidly frozen in liquid nitrogen. For IHC, kidneys were perfused in situ through the abdominal aorta with 3% paraformaldehyde in PBS, removed, and cut into coronal slices. The slices were further immersion-fixed for 2–3 hr at room temperature, cryoprotected overnight in 20% sucrose–PBS and rapidly frozen in liquid nitrogen-chilled isopentane.
Western Blotting
CHO cells expressing truncated mouse CA XII or full-length mouse CA XIV cDNA (Parkkila et al. 2001) were lysed in 20 mM Tris-HCl, pH 7.5, plus 100 mM NaCl plus PMSF, EDTA,
Cell lysates or renal membrane proteins were subjected to standard SDS-PAGE under reducing conditions. After electrophoresis, the polypeptides were transferred to Immobilon-P membranes (Millipore; Bedford, MA). The membranes were immunostained using rabbit anti-mouse CA XII, anti-mouse CA XIV, or preimmune serum as primary antibodies and peroxidase-conjugated goat or donkey anti-rabbit IgG as secondary antibody, as described previously (Kaunisto et al. 2002). The polypeptides were visualized by enhanced chemiluminescence (ECL; Amersham, Little Chalfont, UK).
Immunohistochemistry
Sections were cut at 5 μm using a Microm Cryo-Star microtome (Microm; Walldorf, Germany), dried onto Superfrost Plus microscope slides (Menzel; Braunschweig, Germany), and incubated with PBS containing 20% cow colostral whey for 30 min. The sections were then incubated for 2 hr (mice) or overnight (rats) with polyclonal rabbit anti-CA XII or preimmune serum, diluted 1:100–1:300 in 1% BSA–PBS, washed three times for 5 min in PBS, and incubated for 1 hr with Alexa 488- or 568-coupled goat anti-rabbit IgG, diluted 1:300 in PBS. After four 5-min washes in PBS, slides were mounted in Immu-mount (Shandon; Pittsburgh, PA).
For double immunofluorescence staining, the rabbit anti-CA XII was detected with Alexa 592-coupled donkey anti-rabbit IgG (1:200) and the goat antibodies against calbindin D28K (1:100), AQP2 (diluted 5 μg/100 μl), or Tamm–Horsfall glycoprotein (1:100) with Alexa 488-coupled donkey anti-goat IgG (1:200). In the double-labeling experiments, the rabbit anti-mouse CA XII antibodies were detected with Alexa 568-coupled goat anti-rabbit IgG (1:300), rat anti-mouse CA XIV (1:20) with Alexa 488-coupled goat anti-rat IgG (1:300), and mouse monoclonal anti-human AE1 (1:10) with Alexa 488-coupled goat anti-mouse IgG (1:300). The sections were first examined with a conventional epifluorescence microscope (Nikon; Tokyo, Japan) and the final images were obtained with a confocal laser scanning microscope (Zeiss; Göttingen, Germany).
Results
Antibody Characterization and CA XII Expression in the Kidney
The specificity of rabbit anti-mouse CA XII antiserum was tested by SDS-PAGE and Western blotting of lysates from CHO cells transfected with cDNAs for truncated mouse CA XII or full-length mouse CA XIV. Polypeptides of 42–44 kD for truncated mouse CA XII and 50–55 kD for full-length mouse CA XIV reacted with their respective antisera (Figure 1). There was no crossreaction between mouse CA XII and XIV antisera. No reaction was seen with preimmune serum.
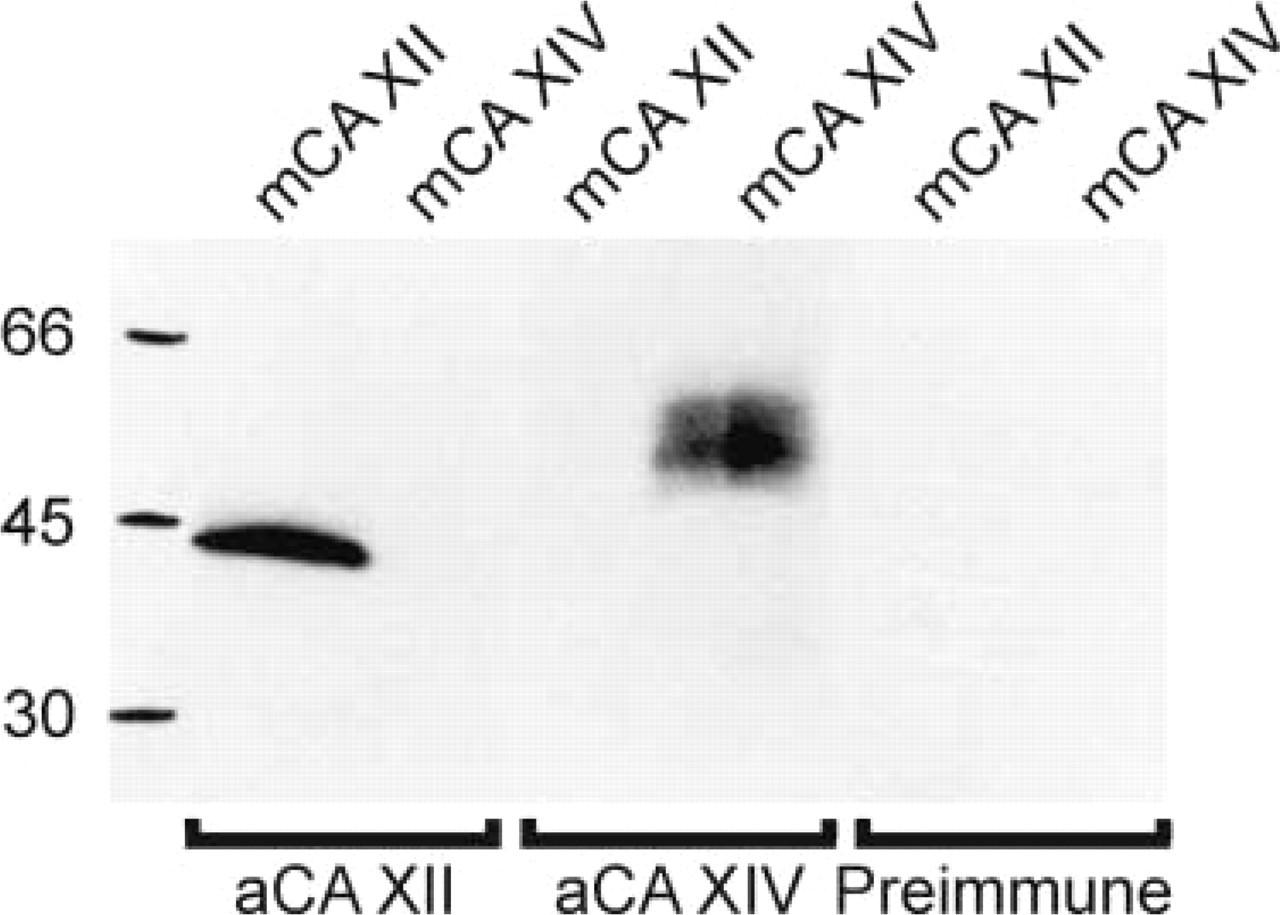
Immunoreaction of the anti-mouse CA XII and anti-mouse CA XIV antisera with the secretory recombinant mouse CA XII and the membrane-associated mouse CA XIV. The cell lysates containing 10 μg protein from CHO cells transfected with truncated mouse CA XII or full-length mouse CA XIV cDNA were analyzed by SDS-PAGE followed by Western blotting analysis. Polypeptides of 42–44 kD for truncated mouse CA XII and 50–55 kD for full-length mouse CA XIV reacted with their respective antisera. There was no crossreaction between mouse CA XII and mouse CA XIV antisera. No reaction was seen with preimmune serum.
Western blotting analysis revealed a diffuse 40–45 kD polypeptide band in both mouse and rat kidney membrane samples (Figure 2). In addition, anti-CA XII antibody recognized a faint unidentified band of 65 kD in both species. When specific antiserum was replaced by preimmune serum, no signal was detected. The observed size of the CA XII-specific band is in agreement with the size of the glycosylated human CA XII (Türeci et al. 1998).
IHC Localization of CA XII in the Kidney
The distribution of CA XII along the rodent nephron and its subcellular localization were studied using indirect immunofluorescence. Low-magnification images of mouse kidney cortex demonstrate CA XII immunostaining in proximal tubules and cortical collecting ducts (Figure 3A). In the medulla of mouse kidney, strong immunostaining was seen in tubules, which represented medullary collecting ducts (Figure 3C). CA XII-positive cells were seen in the cortical and outer medullary collecting ducts but not in the inner medulla (not shown). In proximal tubules, CA XII immunostaining was intense in the S1 segment and decreased towards the S2 segment (Figures 3A and 3B), whereas S3 proximal tubules were negative (not shown). The labeling was restricted to the basolateral plasma membrane, while the luminal brush border membrane was negative (Figures 3A and 3B). In the cortical (Figure 3A) and medullary collecting ducts (Figure 3C), the labeling was present in the basolateral plasma membrane of an epithelial cell subpopulation. In rat kidney, the staining pattern was similar, although the signal was weaker in proximal tubules where it was clearly visible after longer incubations with the primary antibody (Figure 3D). Collecting ducts in the rat kidney cortex and outer medulla showed strong basolateral immunostaining in a subpopulation of epithelial cells (Figures 3E and 3F). The control immunostaining with preimmune serum remained negative (Figure 3G).
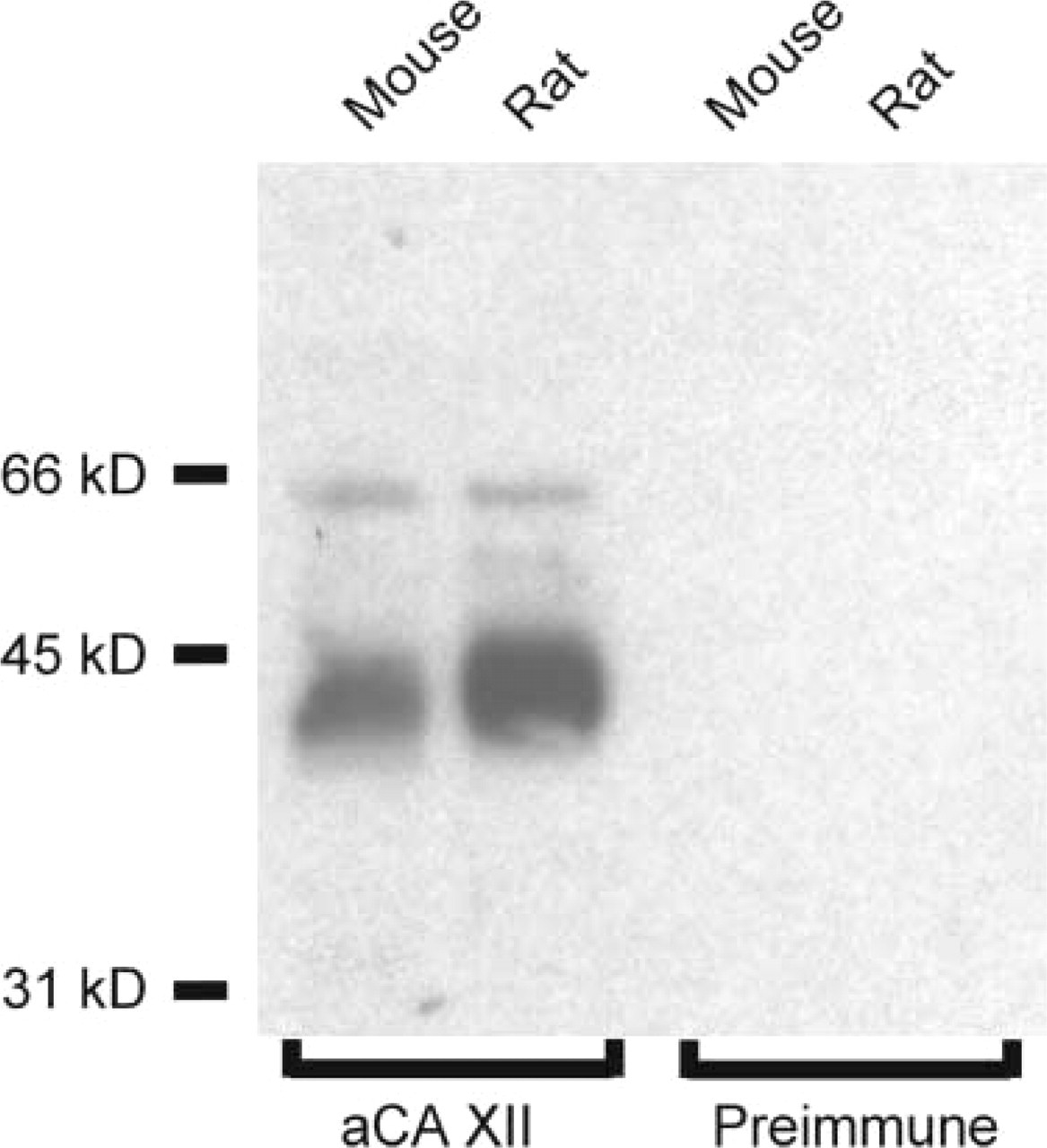
Western blotting analysis of CA XII in 50 μg of membranes from mouse and rat kidney. Anti-CA XII antibody indentifies a 40–45-kD polypeptide in renal membranes from both species. An additional band of 65 kD can also be seen in both samples. Preimmune serum shows no reaction.
We have recently reported that another membrane-associated CA, CA XIV, was localized in the S1 and S2 proximal tubule segments and in the initial segment of the thin descending limb mouse and rat kidney (Kaunisto et al. 2002). In double immunofluorescence staining, CA XII immunostaining was observed in the basolateral plasma membrane of proximal tubules, which also showed CA XIV in the brush border membrane (Figure 4A). It should be noted that rat anti-mouse CA XIV antibody used in the double immunostaining appeared to label only luminal membranes, even though another antibody (rabbit anti-mouse CA XIV) recently showed positive CA XIV immunoreactivity also in the basolateral membranes (Kaunisto et al. 2002). This slight difference in the staining reactivity is probably due to the low titer of our rat anti-mouse CA XIV antibody. The collecting ducts with intense CA XII immunostaining were negative for CA XIV in both cortex (Figure 4A) and medulla (not shown). CA XII was absent from thick ascending limbs, which were identified using the antibody against Tamm–Horsfall glycoprotein (Hoyer et al. 1979) (Figure 4B). Distal convoluted tubules did not express CA XII, which was confirmed by double immunostaining with the calbindin D28K antibody (Figure 4C). This calcium-binding protein has been localized to mouse distal convoluted tubules, connecting segments, and cortical collecting ducts (Campean et al. 2001; Loffing et al. 2001). AQP2 was used as a marker protein for collecting duct principal cells (Nielsen et al. 1993; Breton et al. 1995). Figure 4D shows that CA XII-positive cells were distinct from adjacent AQP2-positive principal cells, identifying CA XII-positive cells as intercalated cells. CA XII signal was localized to the basolateral plasma membrane also in these cells. Different subtypes of intercalated cells can be distinguished based on the polarized expression of H+ and HCO3 − transport proteins: Type A intercalated cells are H+-secreting cells, whereas type B intercalated cells secrete HCO3 − (Alper et al. 1989; Schuster 1993). To identify the intercalated cell subtype, we subjected the sections to double immunofluorescence staining for CA XII and an anion exchanger protein AE1, which is a marker of the acid-secreting type A intercalated cells (Drenckhahn et al. 1985; Alper et al. 1989). Figures 4E and 4F show that CA XII and AE1 were co-localized in the basolateral membrane of type A intercalated cells in rat kidney collecting duct.
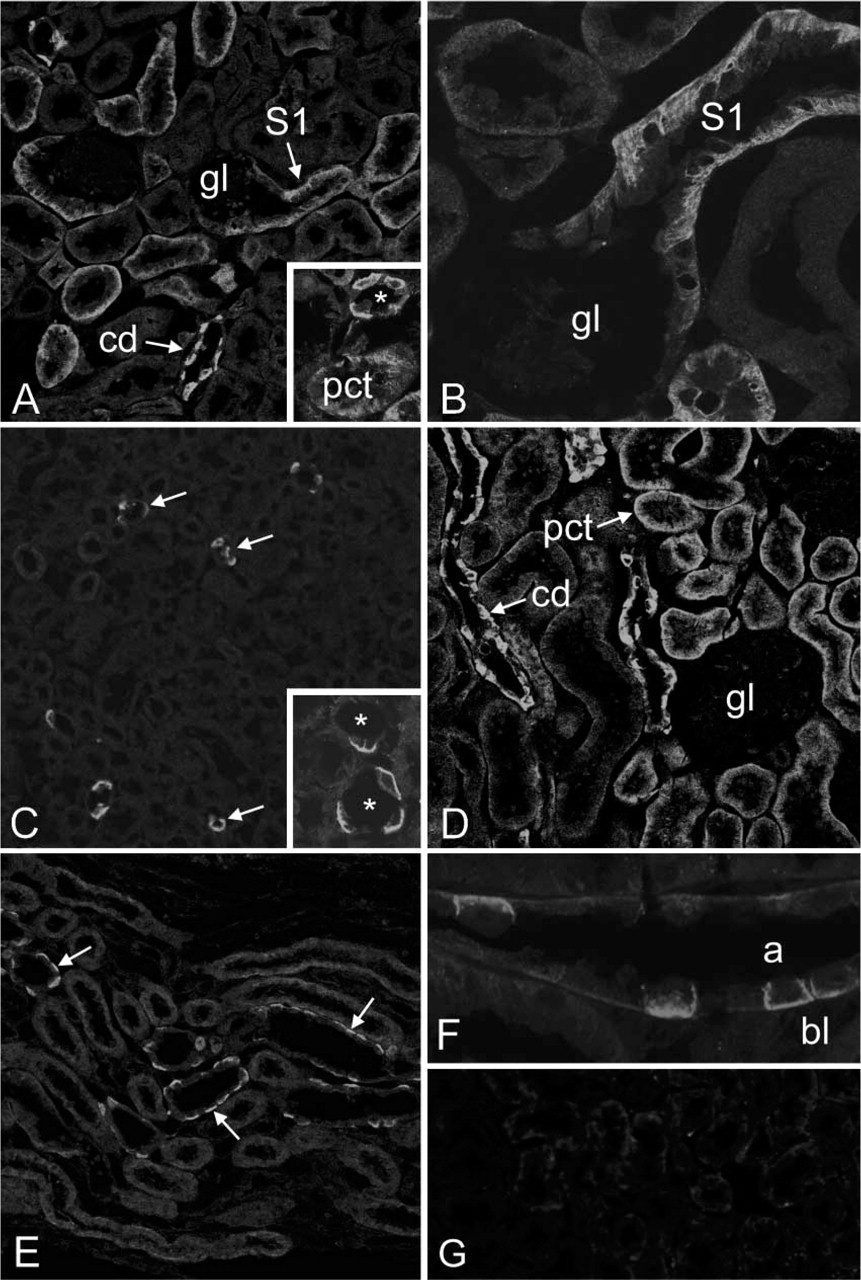
Immunofluorescence staining of CA XII (
Discussion
Membrane-associated CA activity plays an important role in renal acidification even though it comprises only 5% of total renal CA activity (McKinley and Whitney 1976; Wistrand and Kinne 1977; Alpern 2000). Luminal CA in the proximal tubule and thick ascending limb of Henle is needed to catalyze the formation of CO2 from urinary HCO3 − and H+ ions secreted by the apical Na+/H+ exchanger, thereby preventing the development of acid disequilibrium pH (Alpern 2000). Until recently, only CA IV was believed to be responsible for this activity (Brown et al. 1990). However, our recent finding that CA XIV is also localized to the brush border membrane of S1 and S2 proximal tubule segments suggested that CA XIV is another isoenzyme that may have an important role in renal acidification (Kaunisto et al. 2002). The respective roles of these luminal CA isoenzymes await further investigations with isoenzyme-specific inhibitors and knockout animal models.
CA activity has been identified in basolateral membranes, in addition to renal luminal membranes, by biochemical (McKinley and Whitney 1976; Wistrand and Kinne 1977; Brechue et al. 1991) and histochemical (Lönnerholm and Ridderstråle 1980; Dobyan et al. 1982; Lönnerholm and Wistrand 1984; Ridderstråle et al. 1992) methods. IHC studies have revealed CA IV in basolateral membranes of proximal tubules of rat and rabbit kidney (Brown et al. 1990; Schwartz et al. 2000). In our recent IHC study, CA XIV protein was also present in basolateral membranes of the proximal tubules and in the initial portion of the thin descending limb of Henle (Kaunisto et al. 2002).
In the present study we used polyclonal antibodies and IHC to investigate the distribution and cellular localization of CA XII in mouse and rat nephron. Basolateral localization of CA XII in the proximal tubule indicates that at least three isoenzymes, CA IV, XIV, and XII, may contribute to CA activity in that membrane domain. The importance of basolateral CA in proximal tubule to HCO3 − and fluid absorption has been clearly defined by Tsuruoka and colleagues (2001), who found that inhibition of basolateral CA activity decreased HCO3 − absorption by 60% and fluid absorption by 30%. The Na+–HCO3 − co-transporter is able to transport CO3 2- with Na+ and HCO3 − across the basolateral membrane (Soleimani and Aronson 1989; Soleimani and Burnham 2001). CA activity in the peritubular space is believed to supply H+ ions for conversion of CO3 2- to HCO3 −, thus preventing the accumulation of CO3 2- which would inhibit the function of the co-transporter (Seki et al. 1996; Tsuruoka et al. 2001).
We found intense immunostaining in the basolateral membrane of intercalated cells in cortical and outer medullary collecting ducts. The finding of basolateral CA in intercalated cells is in agreement with earlier histochemical studies on mouse kidney (Dobyan et al. 1982; Ridderstråle et al. 1992). Co-localization of CA XII with AE1 indicated that at least most of these cells were of the A subtype, which are involved in H+ secretion to urine (Schuster 1993). Type A intercalated cells exchange interstitial chloride ions with intracellular HCO3 − and possess a basolateral Na+/H+ exchanger (Breyer and Jacobson 1989; Hays and Alpern 1990). It is not known whether CA activity in intercellular space facilitates the function of these ion tranporters. However, in his mathematical model of the type A intercalated cell, Weinstein (2000) argues that peritubular CA catalysis may not have significant impact on luminal acid secretion but would increase NH3 secretion by removing excess H+ ions from the intercellular space.
The localization of mouse and rat renal CA XII has similarities but also some differences compared to the human enzyme (Parkkila et al. 2000). In all three species, the epithelial cells of the proximal tubule expressed CA XII. In the human kidney, the thick ascending limb of Henle and distal convoluted tubule (DCT) also expressed CA XII, whereas in rodents both segments were devoid of immunostaining. Conflicting results have been reported concerning the basolateral CA activity in DCT using histochemical methods. Rat DCT has been shown to exhibit CA activity in basolateral membranes (Lönnerholm and Ridderstråle 1980). In contrast, Dobyan and colleagues (1982) found no CA activity in mouse DCT, while Ridderstråle and colleagues (1992) reported basolateral CA activity in mouse late DCT. The present finding that CA XII is not expressed in mouse DCT may result from different strains used or may indicate the existence of another CA isoenzyme in that nephron segment. The collecting ducts represented the third tubule region in which we observed a clear discrepancy in CA XII immunoreactivity between human and rodent tissues. In the rodent collecting ducts, CA XII was expressed in the intercalated cells, whereas in the human kidney, CA XII has been localized to the principal cells (Parkkila et al. 2000). Our present finding is compatible with histochemical studies that have detected basolateral CA activity in the mouse intercalated cells (Dobyan et al. 1982; Ridderstråle et al. 1992). It is notable that enzyme histochemistry has revealed CA activity in the basolateral membrane of the principal cells in human collecting duct (Lönnerholm and Wistrand 1984), which is in line with our previous IHC results in the human kidney. During the final preparation of this manuscript, Schwartz and colleagues (2003) reported the expression of CA XII mRNA in proximal convoluted and straight tubules, cortical and medullary collecting ducts, and papillary epithelium of rabbit kidney. Hence, despite slight differences in the distribution of CA XII along the rabbit and rodent nephron, those results are in line with our present findings.
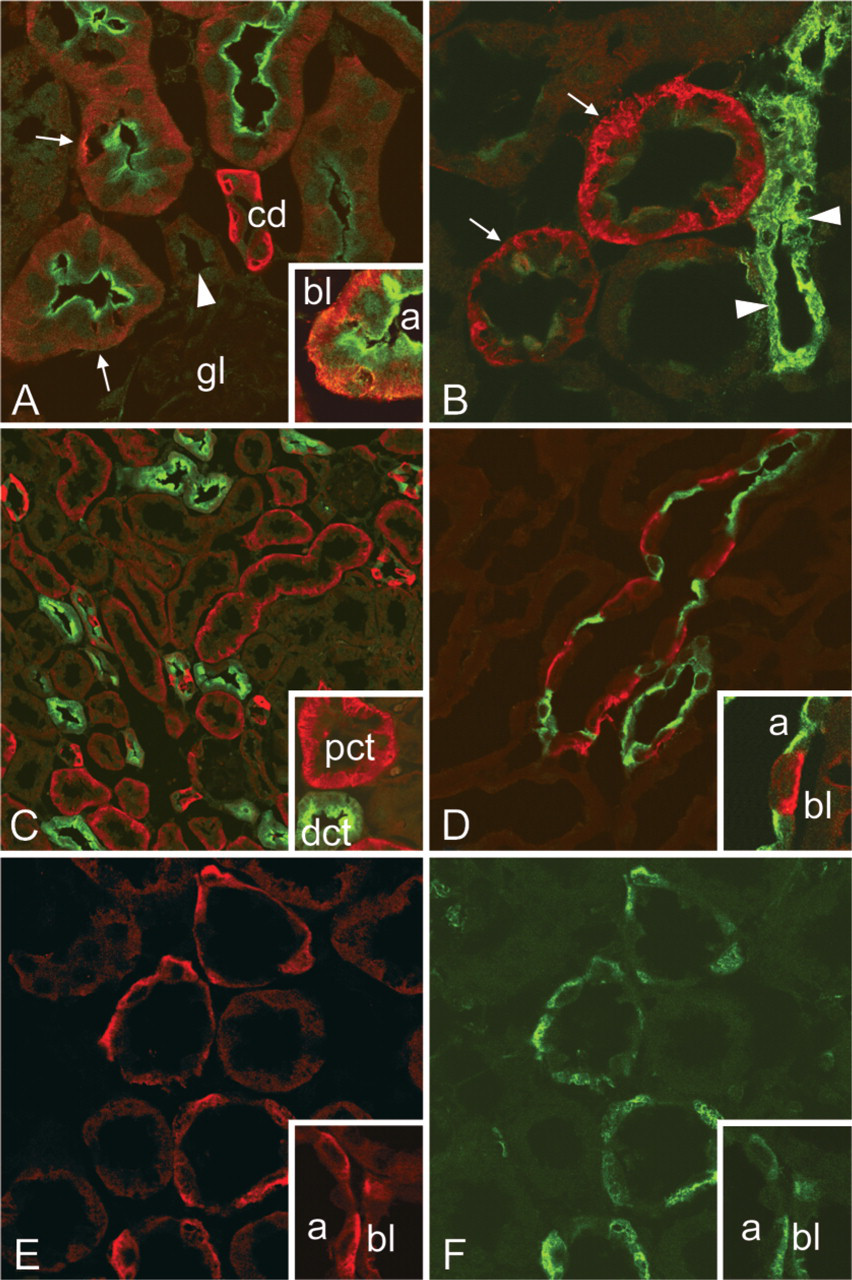
Double immunofluorescence staining of CA XII (red) with CA XIV (green) (
In summary, we have studied the localization of CA XII in the rodent kidney using a new antibody raised against the recombinant mouse enzyme. CA XII was localized to the basolateral plasma membrane of proximal tubules and type A intercalated cells of the collecting ducts. Therefore, CA XII is one of the three CA isoenzymes responsible for the intercellular CA activity needed for efficient HCO3 − and fluid absorption in the proximal tubule. The possible role of CA XII in the basolateral membrane of type A intercalated cells needs further studies using selective inhibition of that activity.
Footnotes
Acknowledgements
Supported by a grant from the Sigrid Juselius Foundation to S.P. and by grants DK53405, DK40163, and GM34182 from the National Institutes of Health to W.S.S.
We thank Dr Michael Jennings for providing the AE1 antibody and Dr Maritta Pietilä for helping in confocal microscopy. The technical assistance of Ms Sirpa Kellokumpu and Ms Lissu Hukkanen is gratefully acknowledged.
