Abstract
Proteins of the presynaptic exocytic machinery have been found associated with the acrosome of male germ cells, suggesting that the sperm acrosome reaction and neurotransmission at chemical synapses may share some common mechanisms. To substantiate this hypothesis, we studied the expression and ultrastructural localization of prominent pre- and postsynaptic protein components in rat testis. The presynaptic membrane trafficking proteins SV2 and complexin, the vesicular amino acid transporters VGLUT and VIAAT, the postsynaptic scaffolding protein ProSAP/Shank, and the postsynaptic calcium-sensor protein caldendrin, could be identified in germ line cells. Immunogold electron microscopy revealed an association of these proteins with the acrosome. In addition, evidence was obtained for the expression of the plasmalemmal glutamate transporters GLT1 and GLAST in rat sperm. The novel finding that not only presynaptic proteins, which are believed to be involved in membrane fusion processes, but also postsynaptic elements are present at the acrosome sheds new light on its structural organization. Moreover, our data point to a possible role for neuroactive amino acids in reproductive physiology.
Keywords
T
Interestingly, other synaptic protein constituents, not implicated in membrane fusion, may also play important roles in male germ cells. For example, testicular germ cells appear to be equipped with distinct neurotransmitter receptor subtypes for the neuroactive amino acids glutamate, GABA, and glycine (Wistrom and Meizel 1993; Melendrez and Meizel 1996; Storto et al. 2001). Although there is evidence that the activation of GABA and glycine receptors is involved in the acrosome reaction (Meizel 1997), much remains to be learned about the functional role of neuroactive amino acids during spermatogenesis and fertilization.
The study of synaptic proteins in the testis is likely to provide novel insights into the molecular architecture and physiology of the male reproductive system. This prompted us to examine the expression and localization of further synaptic proteins in male rat germ cells, using immunohistochemical (IHC) and immunochemical (ICC) techniques. In our study we have included vesicle trafficking proteins of the presynaptic nerve terminal, scaffolding and calcium-binding proteins of the postsynaptic density (PSD) of the central nervous system (CNS), and transporter proteins for amino acid neurotransmitters.
Materials and Methods
Animals and Tissue Preparation for IHC (Light Microscopy)
Adult Wistar rats kept under controlled laboratory conditions were used for the investigation. Principles of laboratory animal care and specific national laws were followed. Rats were sacrificed by decapitation under CO2 anesthesia, and small pieces of the dissected testes were fixed by immersion for 48 hr at 4C in Bouin's solution. In addition, anesthetized animals were transcardially perfused with pre-wash and fixative solution (a mixture of phosphate-buffered 0.5% picric acid and 4% paraformaldehyde) as detailed elsewhere (Redecker and Bargsten 1993), followed by dissection of the testes. Tissue specimens were then dehydrated and embedded in paraffin. Alternatively, some fixed testicular samples were frozen in liquid nitrogen and cryostat sections were cut at a thickness of 5–8 μm. For analysis of serial semithin sections, rats were sacrificed by decapitation under CO2 anesthesia. Thereafter, small testicular specimens were quenched in isopentane precooled with liquid nitrogen, freeze-dried for 48 hr, and fixed by vapor-phase paraformaldehyde. After embedding of the specimens in epoxy resin (Araldite), serial semithin sections were cut at a thickness of 0.5 μm. For the IHC analysis of spermatozoa, caudal epididymal spermatozoa were collected in Dulbecco's PBS on ice containing 10 mM iodoacetamide, 0.5% aprotinin, and 0.1% PMSF. Spermatozoa were purified by Percoll step gradient centrifugation (20%, 30%, 40% in PBS; Syntin and Cornwall 1999) at 3000 × g for 10 min at 4C. After washing of the pelleted spermatozoa in PBS and centrifugation at 500 × g for 10 min at 4C, spermatozoa were resuspended in PBS and smeared on glass coverslips. The cells were then postfixed with phosphate-buffered 0.5% picric acid and 4% paraformaldehyde for 15 min and processed for immunolabeling as described below.
Tissue Preparation for Immunoelectron Microscopy
Small testicular specimens were fixed by immersion in solutions containing either phosphate-buffered (0.1 M, pH 7.3) 1% glutaraldehyde and 0.25% picric acid or phosphate-buffered 4% paraformaldehyde and 0.25% picric acid for 4 hr at 4C, followed by embedding of the unosmicated tissue in LR White (London Resin; Hampshire, UK) as described previously (Redecker 1993; Redecker and Bargsten 1993).
Immunohistochemistry
IHC Protocol for Light Microscopy
Sections were processed either for the avidin–biotin–peroxidase complex (ABC) technique or for immunofluorescence staining as outlined previously (Redecker and Bargsten 1993; Pabst and Redecker 1999). Before immunostaining, paraffin sections were subjected to microwave heating in 0.01 M sodium citrate buffer (pH 6.0) for 10 min at a power setting of 900 W, and semithin sections were treated as described elsewhere (Redecker and Bargsten 1993; Redecker 1996). Briefly, the ABC method included incubation of cryostat, deparaffinized, or semithin sections with primary antibodies in PBS containing 0.1% Triton X-100 for 24 hr at 4C, followed by incubation for 30 min at room temperature (RT) with the second antibody, biotin-labeled goat anti-rabbit, goat anti-mouse, or donkey anti-guinea pig IgG (Jackson ImmunoResearch; West Grove, PA). The sections were then incubated for 30 min with a pre-formed complex of biotin– peroxidase–streptavidin (Jackson ImmunoResearch), and peroxidase activity was revealed using 0.02% diaminobenzidine hydrochloride with 0.3% nickel sulfate as chromogen. Some sections were counterstained with nuclear Fast Red. Immunofluorescence staining comprised the use of indocarbocyanine (Cy3)- or carbocyanine (Cy2)-conjugated secondary antibodies (Jackson ImmunoResearch). Mounted sections were examined and photographed with a Leitz Orthoplan photomicroscope using brightfield optics, phase-contrast optics, interference contrast optics, or fluorescence filters for the detection of Cy2 or Cy3 fluorescence. To improve the prints of micrographs, images were digitized and their contrast and brightness were adjusted using Adobe Photoshop software.
Method controls consisted of the omission of single steps in the IHC protocol, the application of primary antibodies of unrelated specificities, the use of ascending dilutions of the first antibody or the use of high-molar (0.5 M) PBS as a rinsing solution between the various steps of the IHC protocol.
Immunogold Staining for Electron Microscopy
On-grid labeling of thin sections was performed essentially as outlined elsewhere (Redecker and Bargsten 1993; Redecker 1996) without pretreatment with etching or oxidizing agents. For detection of antigen–antibody binding sites, we used goat anti-rabbit, goat anti-mouse, or goat anti-guinea pig immunoglobulins conjugated with 10-nm colloidal gold (British BioCell; Cardiff, UK).
Immunoblotting
Electrophoresis and immunoblotting of total tissue lysates were performed essentially as reported previously (Redecker et al. 1996; Pabst and Redecker 1999). SDS-PAGE was carried out on 4%, 10%, or 14% vertical mini-slab gels (30–50 μg protein/lane). Detection of antigen–antibody complexes bound to polyvinylidene fluoride (PVDF)-based membranes (Pall; Dreieich, Germany) was accomplished by enhanced chemiluminescence (Amersham, Braunschweig, Germany; Pierce, Rockford, IL).
Results
Light Microscopic IHC
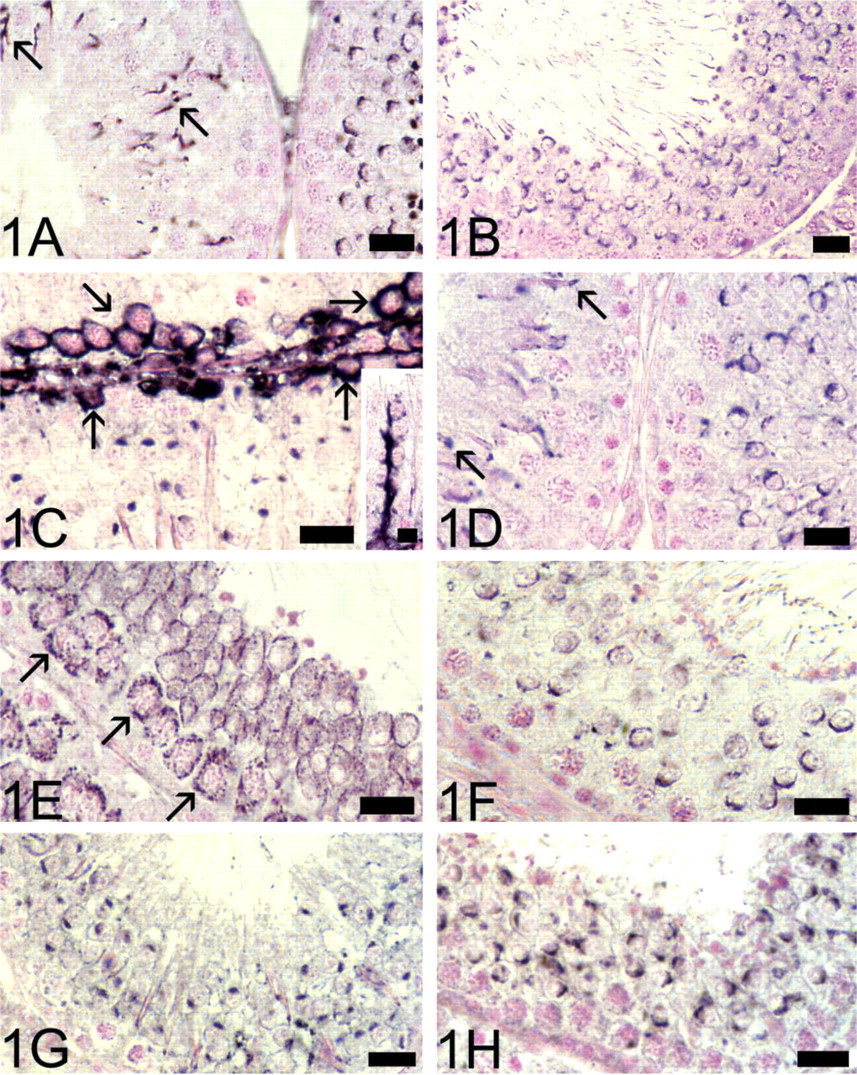
The analysis of testicular immunostaining was focused on the epithelium of the seminiferous tubules. In extension of previous findings on the expression of presynaptic vesicle trafficking proteins in male germ cells, we could identify complexin and SV2 immunoreactivity in these cells. The antibodies directed against complexin1/2 and SV2B labeled the acrosome in round and elongated spermatids (Figures 1A and 1B). Elongated spermatids were only very weakly stained by the SV2A antiserum. In agreement with earlier studies, positive immunoreactions in germ cells were observed with antibodies directed against syntaxin1, synaptotagmin1/2, synaptobrevin1, and synaptobrevin2 (not shown).
Most interestingly, immunostaining revealed the presence of ProSAP/Shank proteins, core components of the PSD, within seminiferous tubules. Therefore, in addition to weak cytoplasmic immunolabel, which was visible in spermatocytes, immunoreactivity for both ProSAP1/Shank2 (Figure 1C) and ProSAP2/ Shank3 was associated with the acrosomes of round and elongated spermatids. We also found discrete ProSAP1/Shank2 immunolabeling in Sertoli cells (Figure 1C) and in the cell cortex of spermatogonia/young spermatocytes (Figure 1C). Acrosomal labeling of spermatids was also detected with the caldendrin antiserum (Figure 1D).
Next, we assessed whether transporter proteins of the neurotransmitters glutamate and GABA are present in rat testis. Both vesicular and plasmalemmal glutamate and GABA transporter proteins appear to be expressed in testicular germ cells. Immunoreactivity for the plasmalemmal glutamate transporter protein GLT1 was detected in the cytoplasm of spermatocytes and was most prominent at the cell surface of round spermatids (Figure 1E). Moreover, the tail of elongated spermatids was clearly labeled by the GLT1 antiserum (not shown). In contrast to strong immunoreactivity for GLAST in Leydig cells, spermatids in the seminiferous epithelium were only moderately immunostained by the GLAST antiserum (not shown). The vesicular glutamate transporter proteins VGLUT1 (Figure 1F) and VGLUT2 and the vesicular GABA transporter VIAAT (Figure 1G) could all be localized in early and mature spermatids, in which the acrosome appeared prominently immunostained. It is noteworthy that immunostainings using an antibody directed against the GABA-synthesizing enzyme GAD67 also led to reproducible staining of acrosomes (Figure 1H).
These results suggest that most of the investigated pre- and postsynaptic proteins may be co-localized in the acrosomal region of spermatids. Immunostaining of serial semithin sections revealed extensive co-localization in the acrosomal region as demonstrated for VGLUT1, VGLUT2, VIAAT, ProSAP1/Shank2, ProSAP2/Shank3, and caldendrin (Figure 2).
Finally, acrosomal labeling was found in smears of epididymal spermatozoa when they were incubated with antibodies against caldendrin (Figure 3A), ProSAP1/ Shank2, ProSAP2/Shank3 (Figure 3C), VGLUT1, VGLUT2, VIAAT (Figure 3E), and GAD67. The acrosomes of epididymal spermatozoa were also weakly immunopositive for SV2B and complexin1/2. Moreover, we noted variable degrees of ProSAP/ Shank immunofluorescence in the tails of spermatozoa (Figure 3C), which were also immunopositive for GAD67. Immunoreactivity for GLT1 and GLAST (Figure 3G) was mainly present in the tails of spermatozoa.
Immunogold Electron Microscopy
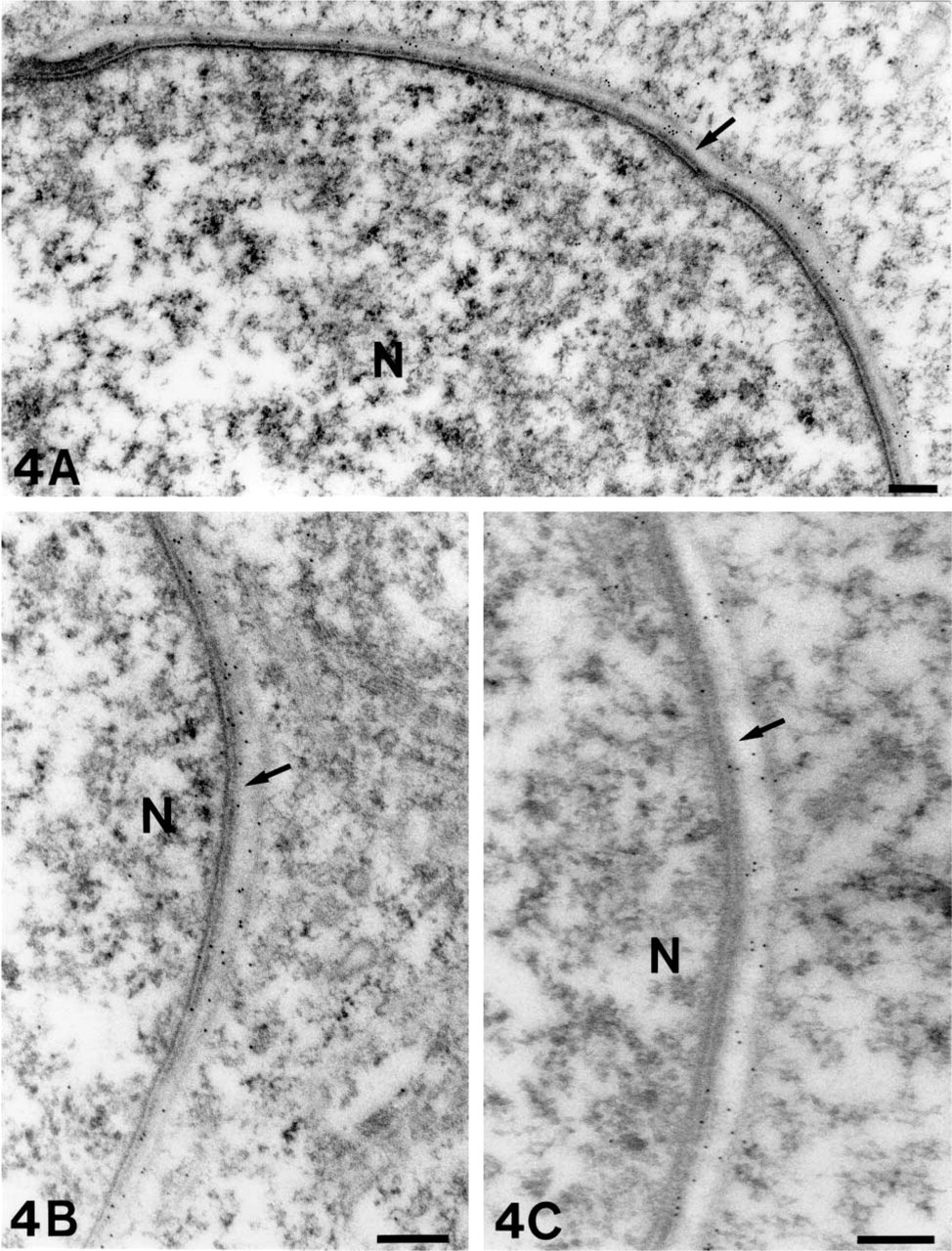
To verify the acrosomal immunolabeling observed by light microscopy, we carried out an ultrastructural analysis of LR White-embedded testicular tissue. Immunogold electron microscopy confirmed the tight association of complexin1/2, SV2B, ProSAP1/ Shank2, ProSAP2/Shank3, Caldendrin, VGLUT1, VGLUT2, VIAAT, and GAD67 with the acrosome. Gold particles indicating exposed epitopes of the respective synaptic proteins were consistently distributed along the outer and inner acrosomal membrane. This labeling pattern was observed in both round and elongated spermatids. Some examples are depicted in Figure 4.
Immunoreactivity for complexin1/2 (
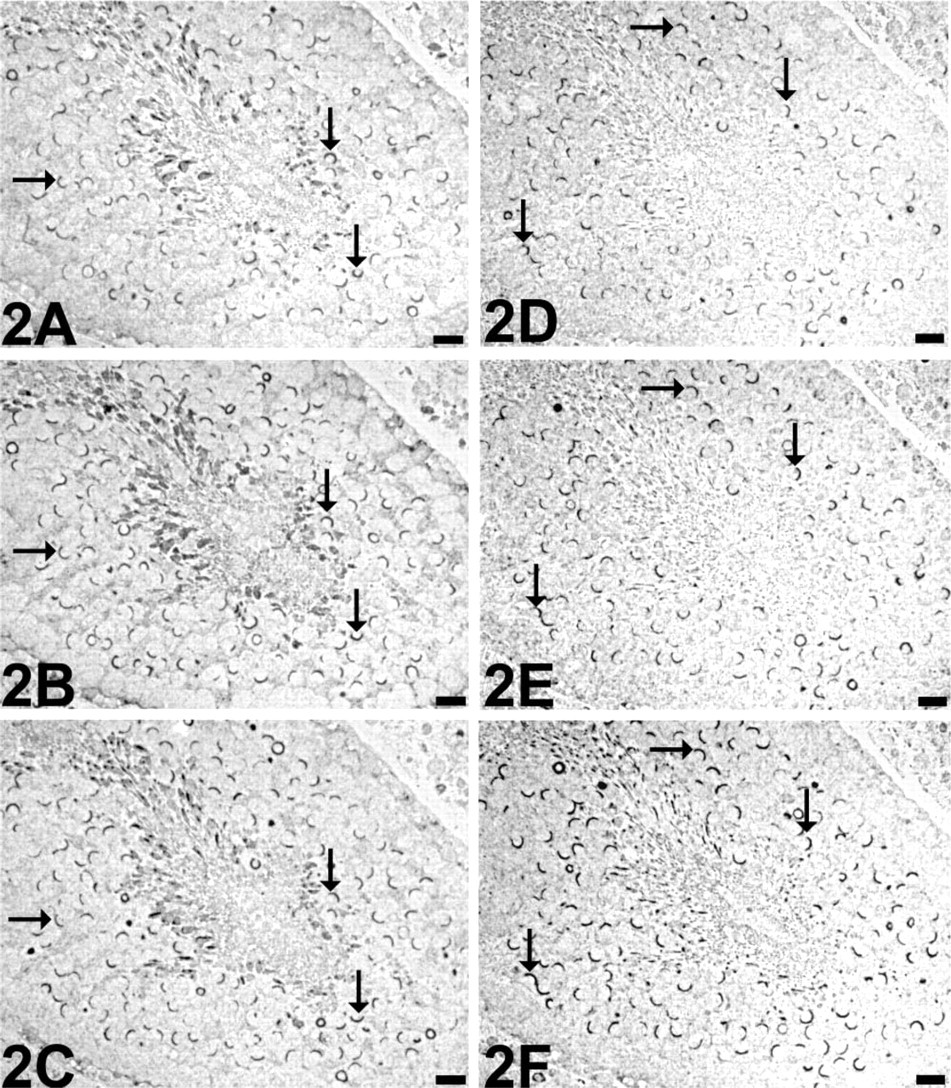
Adjacent semithin sections through a seminiferous tubule immunostained for VGLUT1 (

Immunofluorescence labeling of epididymal spermatozoa in smears that were incubated with antibodies against caldendrin (
Subcellular localization of ProSAP1/Shank2 (
Immunoblotting
Western blotting analyses of testis protein homogenates (Figure 5) showed that all antibodies employed in this study detect immunoreactive bands of molecular weights that were also seen in brain homogenates (not shown). Therefore, it is unlikely that a hitherto not characterized crossreactivity of the employed antisera with proteins exclusively expressed in testis has obscured the staining results. Compared to testis homogenates, we noted additional strongly stained bands above 200 kD apparent molecular weight in immunoblotting analyses of ProSAP/Shank proteins in brain homogenates. This observation probably reflects the differential expression of splice variants and/or degradation of these proteins (Sheng and Kim 2000). A band around 100 kD apparent molecular weight was more prominent in SV2B immunoblots of brain homogenates than of testis homogenates, which may be due to different degrees of glycosylation of SV2 (Jakobsen et al. 2002).
Discussion
A steadily growing set of data speak in favor of an important role of presynaptic vesicle trafficking proteins during spermiogenesis and fertilization. Various members of the corresponding protein families have already been implicated in the biogenesis of the acrosome and in the exocytic acrosome reaction (Ward et al. 1999; Kierszenbaum 2000; Michaut et al. 2001; Ramalho-Santos et al. 2001; Hutt et al. 2002; Tomes et al. 2002). In the present study we have documented the association of two further presynaptic vesicle proteins with the acrosome, i.e., complexin and SV2. The two closely related complexin proteins are known to date to interact with components of the basic membrane fusion machine and to modify the kinetics of neurotransmitter release (Archer et al. 2002; Pabst et al. 2002). Recent experimental data indicate that SV2 regulates exocytosis at a step upstream of the final events of membrane fusion (Xu and Bajjalieh 2001). Therefore, it is possible that, in male germ cells, complexins and members of the SV2 gene family may also regulate the fusion apparatus that operates during the acrosome reaction.
The most remarkable finding of the present study is that, in the acrosomal region, presynaptic vesicle proteins are co-localized with proteins that are concentrated at the PSD of CNS synapses (Seidenbecher et al. 1998; Boeckers et al. 1999,2002; Lim et al. 1999). Within this electron-dense thickening, the ProSAPs/ Shanks act as master scaffolding proteins that link clustered neurotransmitter receptors and cell adhesion molecules to the actin-based cytoskeleton via interactions with cortactin and α-fodrin (Du et al. 1998; Naisbitt et al. 1999; Tu et al. 1999; Boeckers et al. 2001,2002). We have previously reported that ProSAP1/Shank2 is also present in various non-neuronal cells, in which it may serve similar scaffolding functions as in neurons (Redecker et al. 2001a). In male germ cells, both ProSAP1/Shank2 and ProSAP2/ Shank3 were found to be associated with the acrosome. Immunoelectron microscopic analysis has shown that ProSAPs/Shanks are localized close to the acrosomal inner and outer membranes. The tight association of the PSD proteins with the acrosomal membrane points to the possible presence of binding partners for ProSAP/Shank in these membranes. It remains to be evaluated whether testicular ProSAP/Shank proteins are also connected to transmitter receptors via binding partners such as Homer or GKAP/SAPAPs and membrane-associated guanylate kinases (Boeckers et al. 2002). In this context, it is worth noting that the expression of the metabotropic glutamate receptor subtypes mGluR1α and mGluR5 in cells of the germinal line in rat testis has recently been reported, although no details concerning the ultrastructural localization of the receptors were given (Storto et al. 2001). Our own studies have disclosed a significant immunoreactivity for mGluR5 in the acrosomal region of rat spermatids (unpublished observations). Moreover, it can be speculated that akin to the PSD, ProSAPs/ Shanks may link the actin-based cytoskeleton of germ cells to acrosomal membranes. Actin is prominently present at the subacrosomal space of spermatids and in the head of spermatozoa (Aumüller and Seitz 1988; Paranko et al. 1994) where it plays an important role during the acrosome reaction (Liu et al. 2002). In addition to ProSAP/Shank, our investigation has revealed a close spatial relationship between caldendrin and the acrosome. Caldendrin is a neuronal calcium sensor protein, which is localized in the subplasmalemmal cortex of neuronal somata and dendrites and which is highly enriched in the PSD (Seidenbecher et al. 1998; Laube et al. 2002). Therefore, it is tempting to speculate that caldendrin is involved in calcium signaling at the acrosomal cytoskeleton in sperm, given that calcium ions are known to regulate the acrosome reaction in male germ cells (Wassarman 1999).
Interestingly, for the first time we also provide evidence for the concomitant presence of plasmalemmal and vesicular amino acid neurotransmitter transporter proteins in testis. Functional roles for the neuroactive amino acids glutamate, GABA, and glycine in germ line cells are just beginning to emerge. For example, it has been shown that receptors for GABA and glycine are expressed in mammalian sperm (Sato et al. 2000; Hu et al. 2002). Furthermore, both amino acids are believed to be important for the acrosome reaction, probably because of their impact on the plasma membrane potential of sperm cells (Meizel 1997; Bray et al. 2002). It is also worth noting that glutamate and glycine are among the predominant amino acids in seminiferous tubule fluid (Hinton 1990). According to our IHC stainings, rat germ cells are equipped with the high-affinity plasma membrane glutamate transporters GLT1 and GLAST, which are mainly expressed by glial cells in the CNS (Rothstein et al. 1994,1996; Lehre et al. 1995). These transporters are necessary for signal termination during glutamatergic neurotransmission and for preventing glutamate neurotoxicity (Rothstein et al. 1996). Although the exact function of these proteins in mammalian sperm remains to be elucidated, it should be noted that there is also some evidence for the presence of plasmalemmal high-affinity GABA transporters in human and rodent spermatozoa (Aanesen et al. 1996; Hu et al. 2000). Moreover, the detection of GAT1 in the testis of wildtype and GAT1-overexpressing transgenic mice suggests that this plasmalemmal GABA transporter subtype is involved in testicular function (Hua et al. 2000).
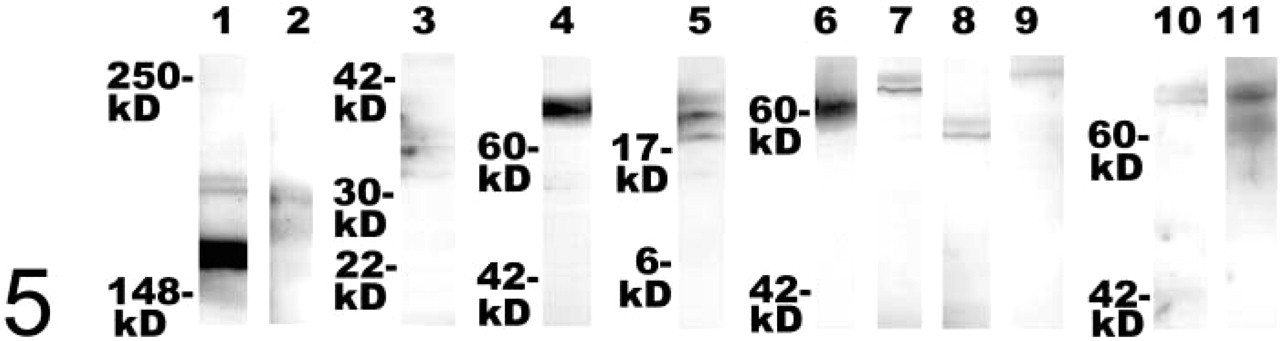
Immunoblots of SDS-PAGE-separated proteins from testis homogenates after probing with antibodies against ProSAP1/Shank2 (Lane 1), ProSAP2/Shank3 (Lane 2), caldendrin (Lane 3), SV2B (Lane 4), complexin1/2 (Lane 5), VGLUT1 (Lane 6), VGLUT2 (Lane 7), VIAAT (Lane 8), GAD67 (Lane 9), GLT1 (Lane 10), and GLAST (Lane 11). The following apparent molecular weights were observed: several bands of ∼160–200 kD (ProSAP1/Shank2, ProSAP2/Shank3), a doublet of bands of ∼33/36 kD (caldendrin), a band of ∼70 kD (SV2B), a doublet of bands of ∼18/19 kD (complexin1/2), a band of ∼60 kD (VGLUT1), a band of ∼65 kD (VGLUT2), a band of ∼57 kD (VIAAT), a band slightly above ∼67 kD (GAD67), a band of ∼70 kD (GLT1), and bands of ∼65–70 kD (GLAST).
Remarkably, the results of our study strongly suggest that the acrosome is endowed with the vesicular amino acid transporters VGLUT and VIAAT. VIAAT is a neuronal transporter protein that mediates the uptake of GABA, and probably also of glycine, into synaptic vesicles (Chaudhry et al. 1998; Dumoulin et al. 1999). On the other hand, VGLUT1 and VGLUT2 have been identified as two of three currently known vesicular proteins responsible for transporting glutamate in glutamatergic neurons (Bellocchio et al. 2000; Takamori et al. 2000,2001; Herzog et al. 2001). The association of immunoreactivity for these vesicular transporters with the acrosome suggests an acrosomal uptake and storage of the respective amino acids, which may eventually be released during the acrosome reaction. It remains to be shown whether the recent characterization of a testis-specific vacuolar type ATP-dependent proton pump in the acrosome (Sun-Wada et al. 2002) is also relevant for the transport of neuroactive amino acids into this organelle, taking into account that in neurons the respective vesicular transporter proteins are driven by an electrochemical proton gradient across the vesicle membrane. The expression of the GABA-synthesizing enzyme glutamate decarboxylase (GAD) in spermatocytes and spermatids of different mammals (Persson et al. 1990; Tillakaratne et al. 1992; and this study) supports the view that a GABA/glycine transporter might be situated in the acrosomal membrane.
Finally, it must be stressed that the co-localization of VIAAT, VGLUT1, and VGLUT2 in a single vesicular organelle, as shown here for the acrosome, is a unique feature, taking into account that in neural tissue (a) the VGLUT transporters reside on vesicle populations distinct from GABA-containing synaptic vesicles (Fujiyama et al. 2001; Takamori et al. 2001), and (b) the expression of VGLUT1 and VGLUT2 is highly complementary (Fremeau et al. 2001; Herzog et al. 2001; Takamori et al. 2001).
In summary, the present study shows that not only pre- but also postsynaptic proteins are assembled in the acrosomal region of male germ cells. This observation should be helpful to provide further new insights into the molecular architecture of sperm cells by addressing the known functional interactions of these proteins that have been found in neurons. It is tempting to speculate that a highly specialized cytoskeleton is attached to acrosomal membranes that closely resembles that of the pre-and postsynaptic cytomatrix. The possibility that the organization of this cytoskeleton is similar to that of the synapse clearly awaits further investigation. The extent of this similarity could provide important insights into basic mechanisms of membrane specialization. Moreover, our results show that neuroactive amino acids may play a more significant role in the physiology of the reproductive system than was previously believed. Finally, the presence of vesicular neurotransmitter transporters in the acrosome supports the view that the acrosome has emerged as a modified secretory granule rather than as a lysosome-like organelle (Martínez-Menárguez et al. 1996; Ramalho-Santos et al. 2001).
Footnotes
Acknowledgements
Supported by grants from the Deutsche Forschungsgemeinschaft DFG BO 1718/2-1 to TMB, and KR 1879/2-1, 2-2 to MRK and EDG, and the Fonds der Chemischen Industrie to EDG.
We gratefully acknowledge the skillful technical assistance of H. Böning and D. von Mayersbach. We thank R. Jahn (Göttingen) for his gifts of antibodies against synaptic vesicle-associated proteins.
